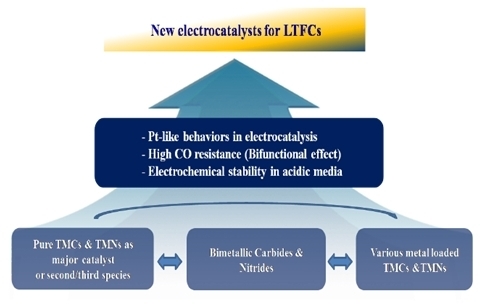
Transition Metal Carbides and Nitrides as Electrode Materials for Low Temperature Fuel Cells
Abstract
:1. Introduction

2. Transition Metal Carbides (TMCs)
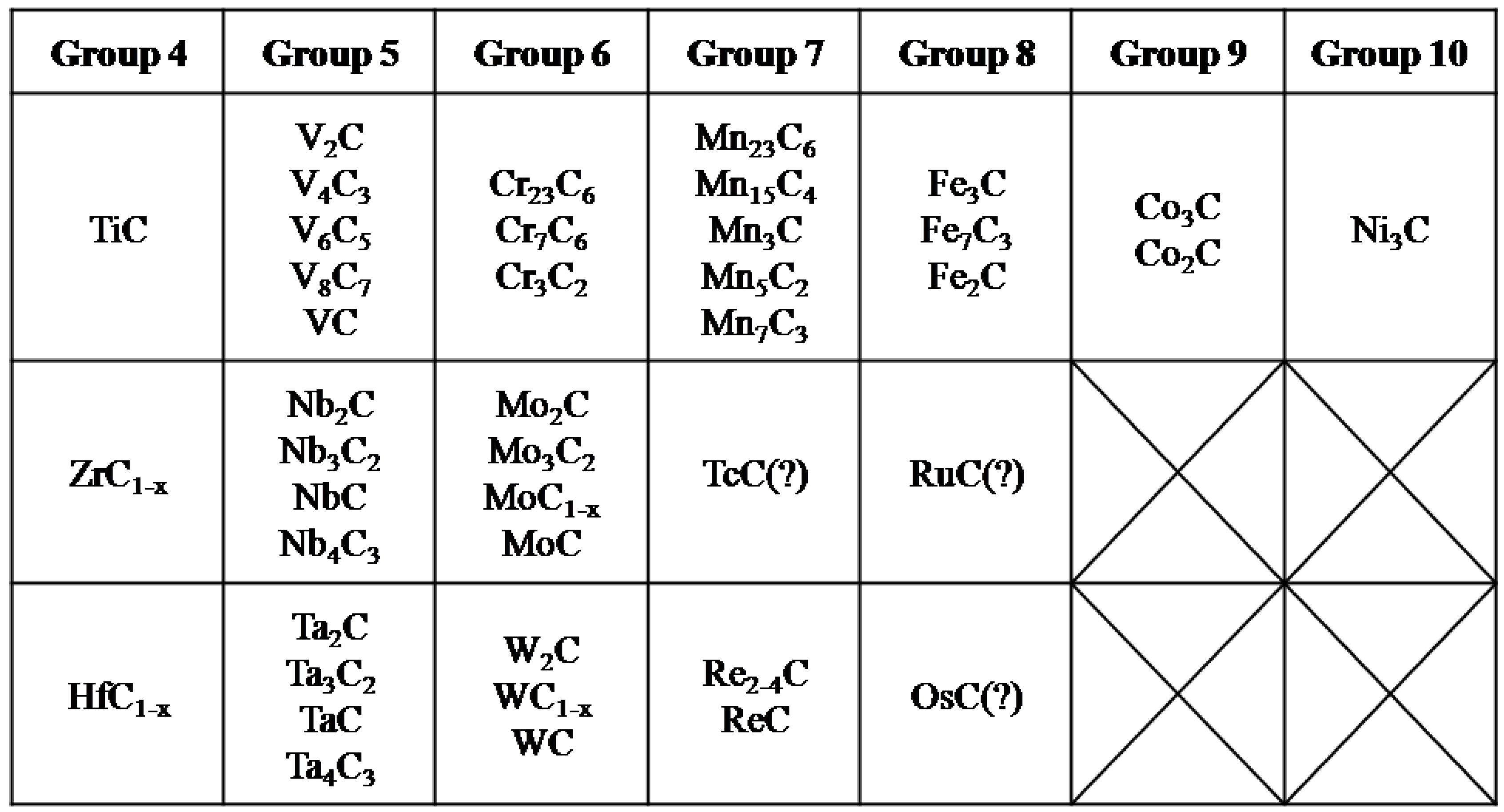
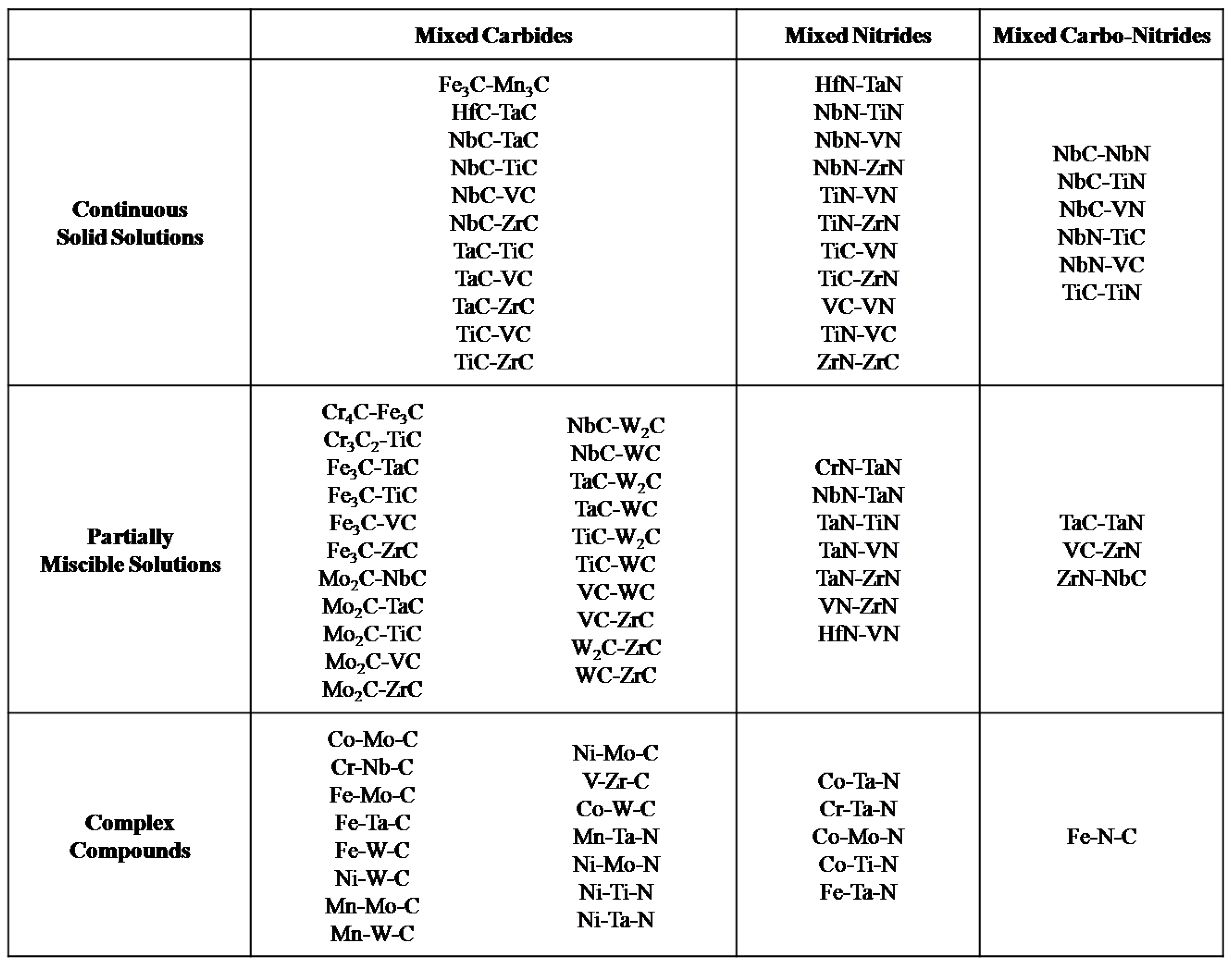
2.1. Electrochemical Stability of TMCs


2.2. CO Oxidation on TMCs
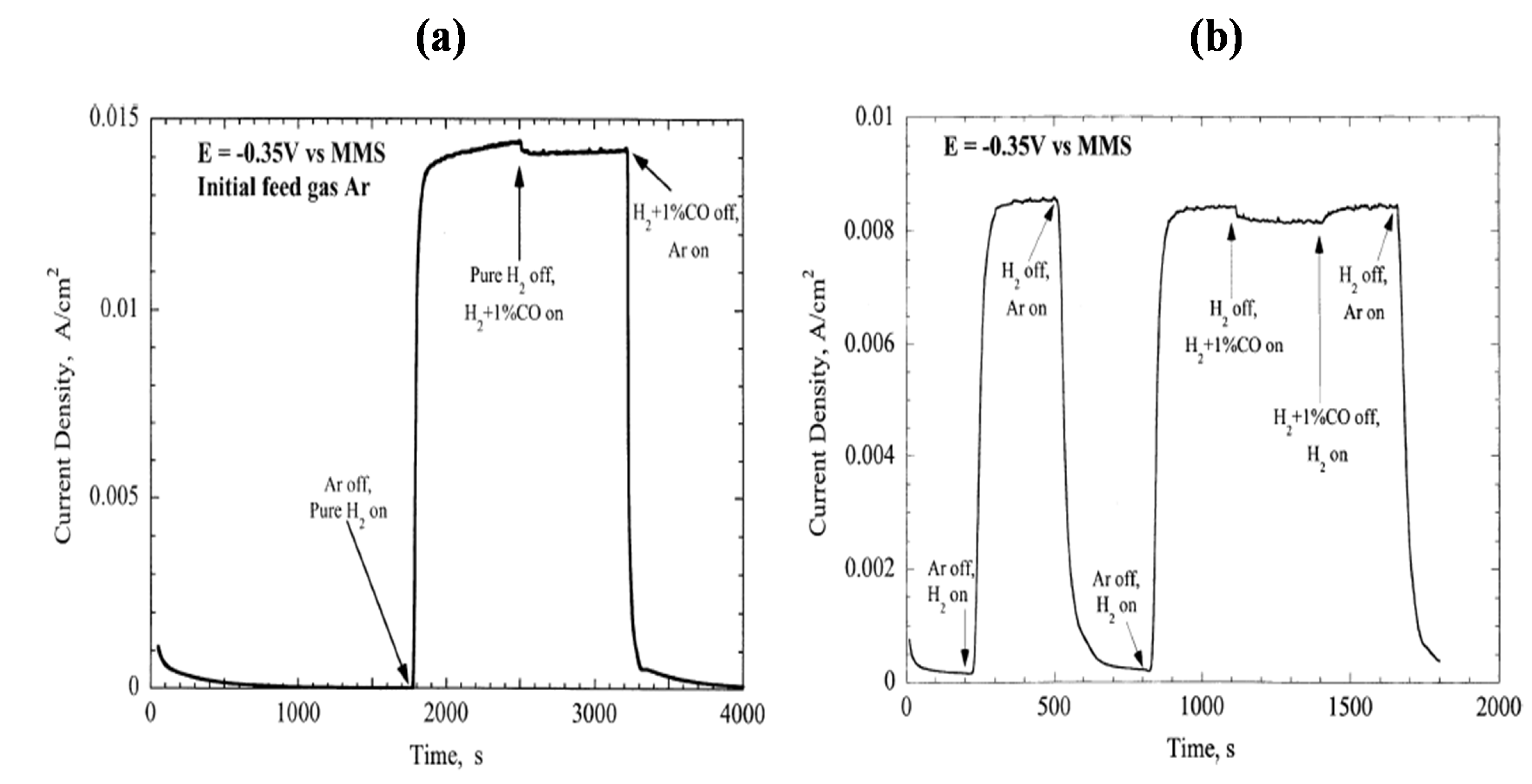

2.3. Hydrogen Oxidation on TMCs
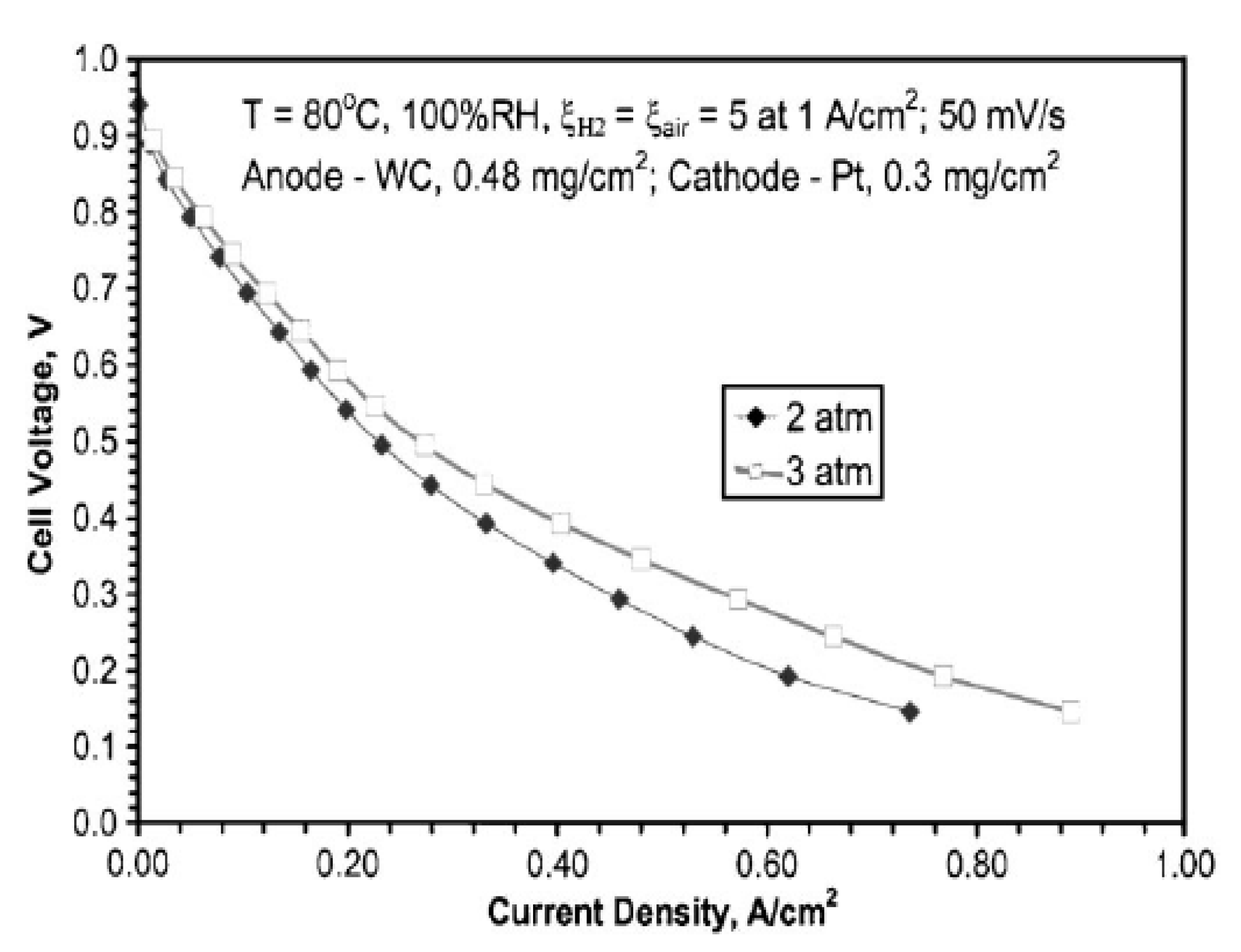
2.4. Oxidation of Methanol and Ethanol on TMCs
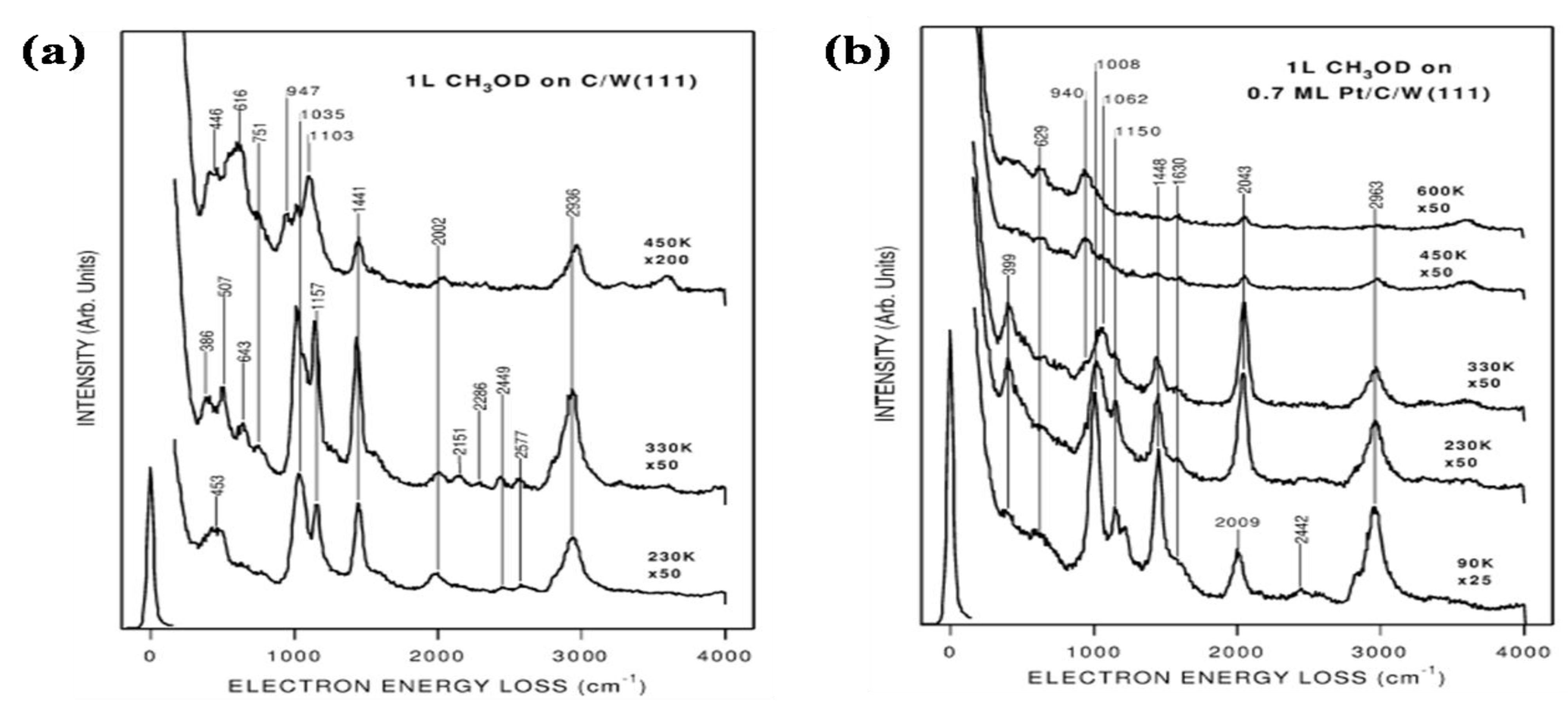
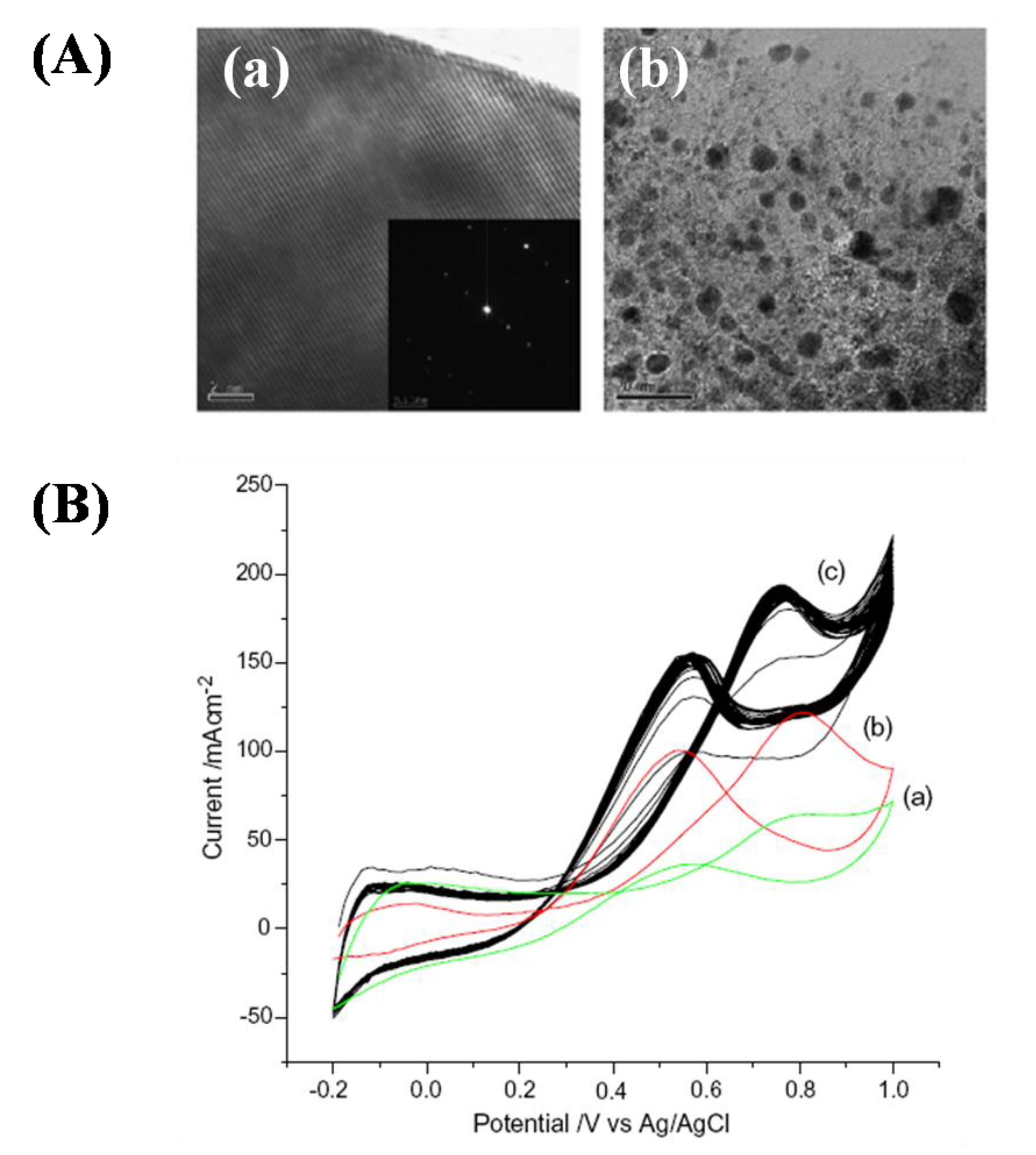

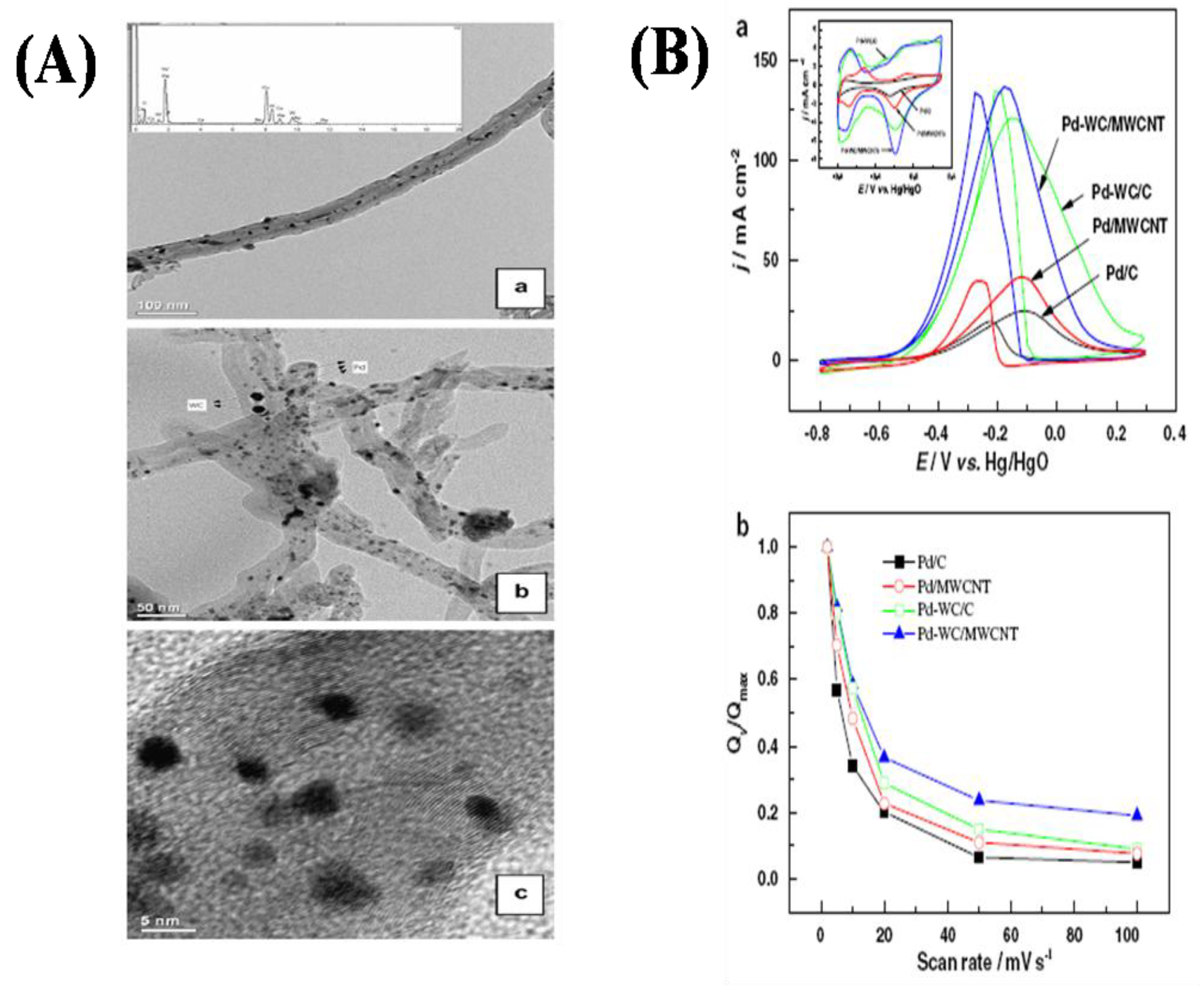
2.5. Oxygen Reduction on TMCs

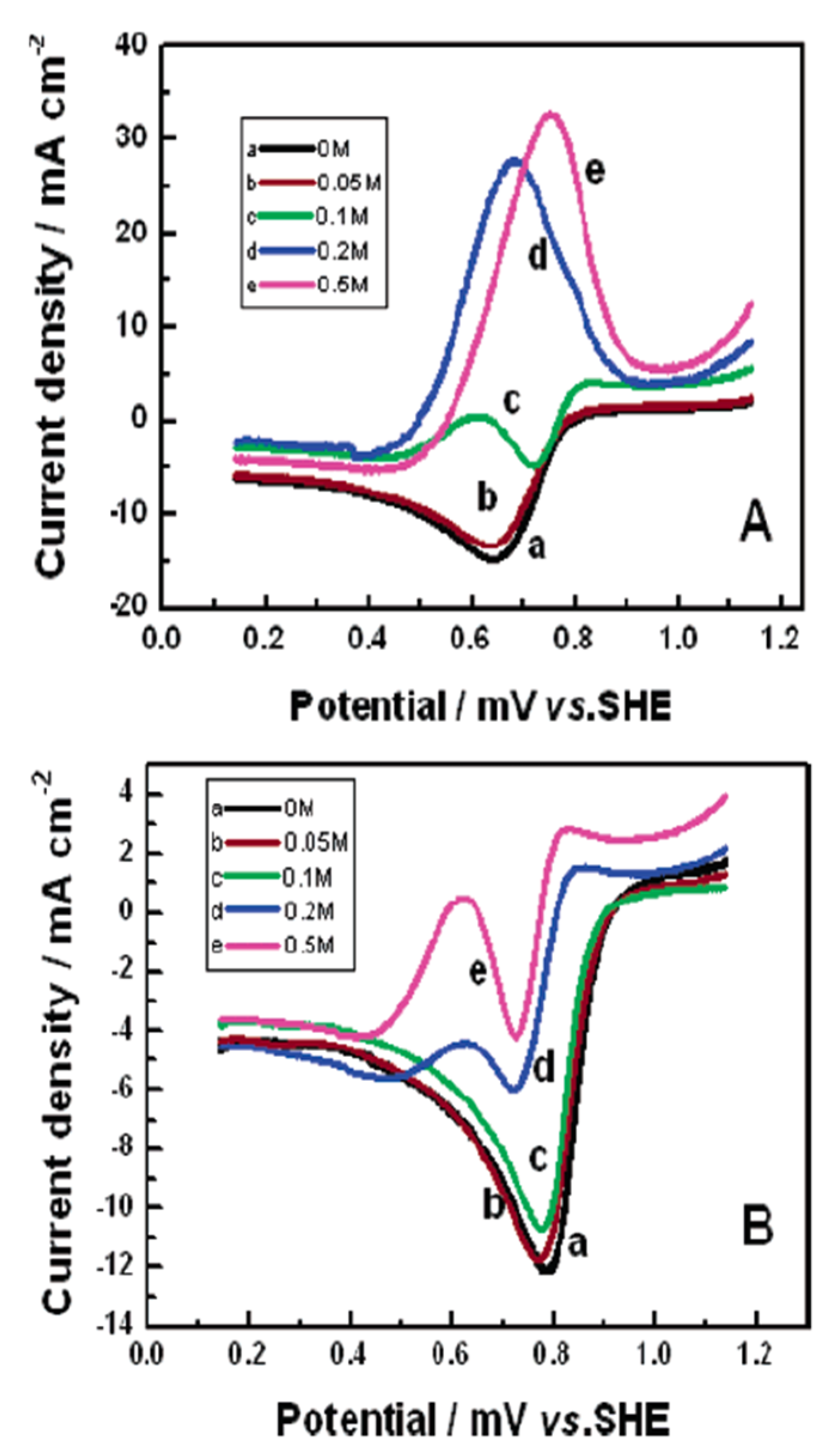
3. Transition Metal nitrides (TMNs)
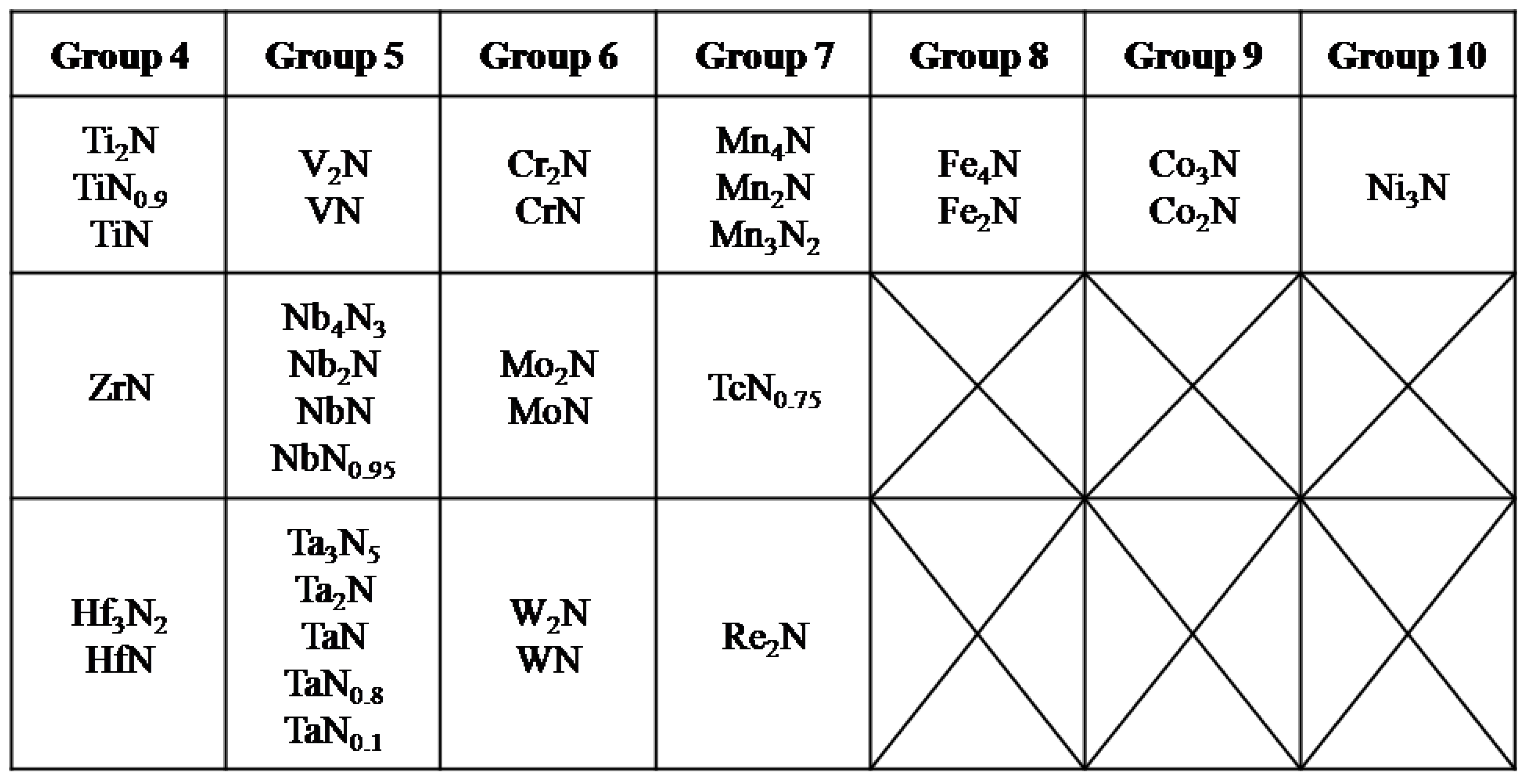
3.1. Electrochemical Stability of TMNs
3.2. Methanol Oxidation on TMNs

3.3. Oxygen Reduction on TMNs

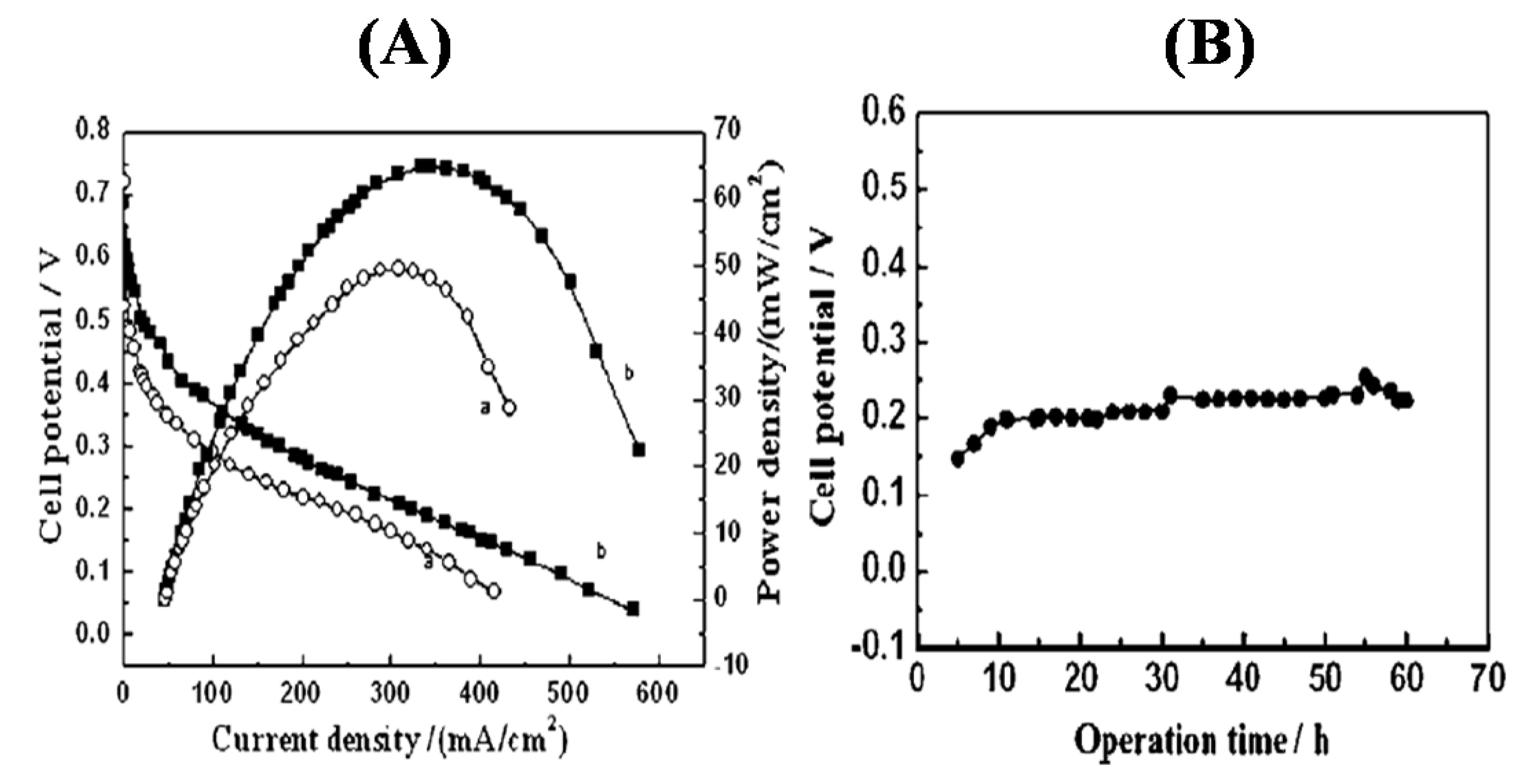
4. Conclusions
Acknowledgements
References and Notes
- Yu, J.; Matsuura, T.; Yoshikawa, Y.; Islam, M.N.; Hori, M. In Situ analysis of performance degradation of a PEMFC under nonsaturated humidification. Electrochem. Solid State Lett. 2005, 8, A156–A158. [Google Scholar]
- Mehta, V.; Cooper, J.S. Review and analysis of PEM fuel cell design and manufacturing. J. Power Sources 2003, 113, 32–53. [Google Scholar]
- Borup, R.; Meyers, J.; Pivovar, B.; Kim, Y.S.; Mukundan, R.; Garland, N.; Myers, D.; Wilson, M.; Garzon, F.; Wood, D.; Zelenay, P.; More, K.; Stroh, K.; Zawodinski, T.; Boncella, J.; McGrath, J.E.; Inaba, M.; Miyatake, K.; Hori, M.; Ota, K.; Ogumi, Z.; Miyata, S.; Nishikata, A.; Siroma, Z.; Uchimoto, Y.; Yasuda, K.; Kimijima, K.; Iwashita, N. Scientific aspects of polymer electrolyte Fuel Cell durability and degradation. Chem. Rev. 2007, 107, 3904–3951. [Google Scholar]
- Steele, B.C.H.; Heinzel, A. Materials for fuel-cell technologies. Nature 2001, 414, 345–352. [Google Scholar]
- Carrette, L.; Friendrich, K.A.; Stimming, U. Fuel cells: Principles, types, fuels, and applications. Chemphyschem 2000, 1, 162–193. [Google Scholar] [PubMed]
- Wilson, M.S.; Garzon, F.H.; Sickafus, K.E.; Gottesfeld, S. Surface area loss of supported platinum in polymer electrolyte fuel cells. J. Electrochem. Soc. 1993, 140, 2872–2877. [Google Scholar] [CrossRef]
- Barnett, B.M.; Teagan, W.P. The role of fuel cells in our energy future. J. Power Sources 1992, 37, 15–31. [Google Scholar] [CrossRef]
- Melle, F.D. The global and urban environment: the need for clean power systems. J. Power Sources 1998, 71, 7–11. [Google Scholar] [CrossRef]
- Bruijn, F.D. The current status of fuel cell technology for mobile and stationary applications. Green Chem. 2005, 7, 132–150. [Google Scholar] [CrossRef]
- Shao, Y.; Yin, G.; Wang, Z.; Gao, Y. Proton exchange membrane fuel cell from low temperature to high temperature: Material challenges. J. Power Sources 2007, 167, 235–242. [Google Scholar] [CrossRef]
- Aricoa, A.S.; Srinivasanb, S.; Antonuccia, V. DMFCs: From fundamental aspects to technology development. Fuel Cells 2001, 1, 133–161. [Google Scholar] [CrossRef]
- Carrette, L.; Friendrich, K.A.; Stimming, U. Fuel cells–Fundamentals and applications. Fuel Cells 2001, 1, 5–39. [Google Scholar] [CrossRef]
- Gasteiger, H.A.; Kocha, S.S.; Sompalli, B.; Wagner, F.T. Activity benchmarks and requirements for Pt, Pt-alloy, and non-Pt oxygen reduction catalysts for PEMFCs. Appl. Catal. B. 2005, 56, 9–35. [Google Scholar] [CrossRef]
- Litster, S.; McLean, G. PEM fuel cell electrodes. J. Power Sources 2004, 130, 61–76. [Google Scholar] [CrossRef]
- Bernardi, D.M.; Verbrugge, M.K. A mathematical model of the solid-polymer-electrolyte. J. Electrochem. Soc. 1992, 139, 2477–2491. [Google Scholar] [CrossRef]
- Bensebaa, F.; Farah, A.A.; Wang, D.; Bock, C.; Du, X.; Kung, J.; Page, Y.L. Microwave synthesis of polymer-embedded Pt-Ru catalyst for direct methanol fuel cell. J. Phys. Chem. B 2005, 109, 15339–15344. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; He, R.; Jensen, J.O.; Bjerrum, N.J. Approaches and recent development of polymer electrolyte membrane for fuel cells operating above 100 °C. Chem. Mater. 2003, 15, 4896–4915. [Google Scholar] [CrossRef]
- Alberti, G.; Casciola, M. Composite membranes for medium-temperature PEM fuel cells. Annu. Rev. Mater. Res. 2003, 33, 129–154. [Google Scholar] [CrossRef]
- Herring, A.M. Inorganic–polymer composite membranes for proton exchange membrane fuel cells. J. Macromol. Sci. Part C 2006, 46, 245–296. [Google Scholar] [CrossRef]
- Liu, H.; Songa, C.; Zhang, L.; Zhang, J.; Wang, H.; Wilkinson, D.P. A review of anode catalysis in the direct methanol fuel cell. J. Power Sources 2006, 155, 95–110. [Google Scholar] [CrossRef]
- Arico, A.S.; Bruce, P.; Scrosati, B.; Tarascon, J.; Schalkwijk, W. Nanostructured materials for advanced energy conversion and storage devices. Nat. Mater. 2005, 4, 366–377. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Zhou, X.; Sun, X.; Li, R.; Murphy, M.; Ding, Z.; Sun, X.; Sham, T. Interaction between Pt nanoparticles and carbon nanotubes–An X-ray absorption near edge structures (XANES) study. Chem. Phys. Lett. 2007, 437, 229–232. [Google Scholar] [CrossRef]
- Faubert, G.; CGti, R.; Dodelet, J.P.; Lefevre, M.; Bertrand, P. Oxygen reduction catalysts for polymer electrolyte fuel cells from the pyrolysis of Fe acetate adsorbed on 3,4,9,l0-perylenetetracarboxylic dianhydride. Electrochim. Acta 1999, 44, 2589–2603. [Google Scholar] [CrossRef]
- Jaouen, F.; Marcotte, S.; Dodelet, J.; Lindbergh, G. Oxygen reduction catalysts for polymer electrolyte fuel cells from the pyrolysis of iron acetate adsorbed on various carbon supports. J. Phys. Chem. B 2003, 107, 1376–1386. [Google Scholar] [CrossRef]
- Bashyam, R.; Zelenay, P. A class of non-percious metal composite catalysts for fuel cells. Nature 2006, 443, 63–66. [Google Scholar] [CrossRef] [PubMed]
- Yuasa, M.; Yamaguchi, A.; Itsuki, H.; Tanaka, K.; Yamamoto, M.; Oyaizu, K. Modifying carbon particles with polypyrrole for adsorption of cobalt ions as electrocatatytic site for oxygen reduction. Chem. Mater. 2005, 17, 4278–4281. [Google Scholar] [CrossRef]
- Serov, A.; Kwak, C. Review of non-platinum anode catalysts for DMFC and PEMFC application. Appl. Catal. B. 2009, 90, 313–320. [Google Scholar] [CrossRef]
- Smotkin, E.S.; Diaz-Morales, R.R. New electrocatalysts by combinatorial methods. Annu. Rev. Mater. Res. 2003, 33, 557–579. [Google Scholar] [CrossRef]
- Zhang, L.; Zhang, J.; Wilkinson, D.P.; Wang, H. Progress in preparation of non-noble electrocatalysts for PEM fuel cell reactions. J. Power Sources 2006, 156, 171–182. [Google Scholar] [CrossRef]
- Fournier, J.; Faubert, G.; Tilquin, J.Y.; Cote, R.; Guay, D.; Dodelet, J.R. High-performance, low Pt content catalysts for the electrorecluction of oxygen in polymer-electrolyte fuel cells. J. Electrochem. Soc. 1997, 144, 145–154. [Google Scholar] [CrossRef]
- Vante, N.A.; Jaegermann, W.; Tributsch, H.; Honle, W.; Yvod, K. Electrocatalysis of oxygen reduction by chalcogenides containing mixed transition metal clusters. J. Am. Chem. Soc. 1987, 109, 3251–3257. [Google Scholar] [CrossRef]
- Matter, P.H.; Zhang, L.; Ozkan, U.S. The role of nanostructure in nitrogen-containing carbon catalysts for the oxygen reduction reaction. J. Catal. 2006, 239, 83–96. [Google Scholar] [CrossRef]
- Serov, A.A.; Min, M.; Chai, G.; Han, S.; Kang, S.; Kwak, C. Preparation, characterization, and high performance of RuSe/C for direct methanol fuel cells. J. Power Sources 2008, 175, 175–182. [Google Scholar] [CrossRef]
- Mustain, W.E.; Kepler, K.; Prakash, J. Investigations of carbon-supported CoPd3 catalysts as oxygen cathodes in PEM fuel cells. Electrochem. Commun. 2006, 8, 406–410. [Google Scholar] [CrossRef]
- Ye, S.; Vijh, A.K.; Dao, L.H. Oxygen reduction on a new electrocatalyst based on highly porous carbonized polyacrylonitrile microcellular foam with very low platinum loading. J. Electroanal. Chem. 1996, 415, 115–121. [Google Scholar] [CrossRef]
- Hu, F.P.; Shen, P.K. Ethanol oxidation on hexagonal tungsten carbide single nanocrystal-supported Pd electrocatalyst. J. Power Sources 2007, 173, 877–881. [Google Scholar] [CrossRef]
- Helia, H.; Jafariana, M.; Mahjania, M.G.; Gobalb, F. Electro-oxidation of methanol on copper in alkaline solution. Electrochim. Acta 2004, 49, 4999–5006. [Google Scholar] [CrossRef]
- Joo, S.H.; Choi, S.J.; Oh, I.; Kwak, J.; Liu, Z.; Terasaki, O.; Ryoo, R. Ordered nanoporous arrays of carbon supporting high dispersions of platinum nanoparticles. Nature 2001, 412, 169–172. [Google Scholar] [CrossRef] [PubMed]
- Fujimoto, T.; Terauchi, S.; Umehara, H.; Kojima, I.; Henderson, W. Sonochemical preparation of single-dispersion metal nanoparticles from metal salts. Chem. Mater. 2001, 13, 1057–1060. [Google Scholar] [CrossRef]
- Boxall, D.L.; Deluga, G.A.; Kenik, E.A.; King, W.D.; Lukehart, C.M. Rapid synthesis of a Pt1Ru1/Carbon nanocomposite using microwave irradiation: A DMFC anode catalyst of high relative performance. Chem. Mater. 2001, 13, 891–900. [Google Scholar] [CrossRef]
- Bensebaa, F.; Patrito, N.; Page, Y.L.; L’Ecuyer, P.; Wang, D. Tunable platinum-ruthenium nanoparticle properties using microwave synthesis. J. Mater. Chem. 2004, 14, 3378–3384. [Google Scholar] [CrossRef]
- Jalan, V.; Taylor, E.J. Importance of interatomic spacing in catalytic reduction of oxygen in phosphoric acid. J. Electrochem. Soc. 1983, 130, 2299–2302. [Google Scholar] [CrossRef]
- Bessel, C.A.; Laubernds, K.; Rodriguez, N.M.; Baker, R.T.K. Graphite Nanofibers as an electrode for fuel cell applications. J. Phys. Chem. B 2001, 105, 1115–1118. [Google Scholar] [CrossRef]
- Wang, C.; Waje, M.; Wang, X.; Tang, J.M.; Haddon, R.C.; Yan, Y. Proton exchange membrane fuel cells with carbon nanotube based electrodes. Nano Lett. 2004, 4, 345–348. [Google Scholar] [CrossRef]
- Gasteiger, H.A.; Panels, J.E.; Yan, S.G. Dependence of PEM fuel cell performance on catalyst loading. J. Power Sources 2004, 127, 162–171. [Google Scholar] [CrossRef]
- Mukerjee, S.; Srinivasan, S.; Soriaga, M.P. Role of structural and electronic properties of Pt and Pt alloys on electrocatalysis of oxygen reduction. J. Electrochem. Soc. 1995, 142, 1409–1422. [Google Scholar] [CrossRef]
- Shao, Y.; Yin, G.; Gao, Y. Understanding and approaches for the durability issues of Pt-based catalysts for PEM fuel cell. J. Power Sources 2007, 171, 558–566. [Google Scholar] [CrossRef]
- Christian, J.B.; Smith, S.P.E.; Whittingham, M.S.; Abruna, H.D. Tungsten based electrocatalyst for fuel cell applications. Electrochem. Commun. 2007, 9, 2128–2132. [Google Scholar] [CrossRef]
- Levy, R.L.; Boudart, M. Platinum-like behavior of tungsten carbide in surface catalysis. Science 1973, 181, 547–549. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.S.; Hyun, T.H. Metal carbides. In Encyclopedia of Catalysis, 1st ed.; Horvath, I.T., Ed.; John Wiley & Sons: New York, NY, USA, 2003. [Google Scholar]
- Storms, E.K. The Refractory Carbides; Academic Press: New York, NY, USA, 1967. [Google Scholar]
- Toth, L.E. Transition Metal Carbides and Nitrides; Academic Press: New York, NY, USA, 1971. [Google Scholar]
- Levi, R.B. Advanced Materials in Catalysis; Academic Press: New York, NY, USA, 1977. [Google Scholar]
- Oyama, S.T. The Chemistry of Transition Metal Carbides and Nitrides; Blackie Academic and Professional: Glasgow, UK, 1996. [Google Scholar]
- Gogotsi, Y.G.; Andrievski, R.A. Materials Science of Carbides, Nitrides and Borides; Kluwer Academic publisher: Dordrecht, The Netherlands, 1999. [Google Scholar]
- Freer, R. The Physics and Chemistry of Carbide, Nitrides, and Borides; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1990. [Google Scholar]
- Gubanov, V.A.; Ianovsky, A.L.; Zhukov, V.P. Electronic Structure of Refractory Carbides and Nitrides; Cambridge University Press: Cambridge, UK, 1994. [Google Scholar]
- Johansson, L.I. Electronic and structural properties of transition-metal carbide and nitride surfaces. Surf. Sci. Rep. 1995, 21, 177–250. [Google Scholar] [CrossRef]
- Chen, J.G. NEXAFS investigations of transition metal oxides, nitrides, carbides, sulfides and other interstitial compounds. Surf. Sci. Rep. 1997, 30, 1–152. [Google Scholar] [CrossRef]
- Hwu, H.H.; Chen, J.G. Surface chemistry of transition metal carbides. Chem. Rev. 2005, 105, 185–212. [Google Scholar] [CrossRef] [PubMed]
- Oyama, S.T.; Kieffer, R. Kirk-Othmer Encyclopedia of Chemical Technology; John Wiley & Sons: New York, NY, USA, 1992. [Google Scholar]
- Ettmayer, P.; Lengauer, W. Encyclopedia of Inorganic Chemistry; John Wiley & Sons: Chichester, UK, 1994. [Google Scholar]
- Oyama, S.T. Preparation and catalytic properties of transition metal carbides and nitrides. Catal. Today 2007, 15, 179–200. [Google Scholar] [CrossRef]
- Chen, J.G. Carbide and nitride overlayers on early transition metal surfaces: Preparation, characterization, and reactivities. Chem. Rev. 1996, 96, 1477–1498. [Google Scholar] [CrossRef] [PubMed]
- Heine, V. s-d Interaction in transition metals. Phys. Rev. 1967, 153, 673–682. [Google Scholar] [CrossRef]
- Zellner, M.B.; Chen, J.G. Surface science and electrochemical studies of WC and W2C PVD films as potential electrocatalysts. Catal. Today 2005, 99, 299–307. [Google Scholar] [CrossRef]
- Brady, C.D.A.; Rees, E.J.; Burstein, G.T. Electrocatalysis by nanocrystalline tungsten carbides and the effects of codeposited silver. J. Power Sources 2008, 179, 17–26. [Google Scholar] [CrossRef]
- Andersson, K.M.; Bergstrom, L. Oxidation and dissolution of tungsten carbide powder in water. Int. J. Refract. Mer. H. 2000, 18, 121–129. [Google Scholar] [CrossRef]
- Jain, M.; Sadangi, R.K.; Cannon, W.R.; Kear, B.H. Processing of functionally graded WC/Co/diamond nanocomposites. Scr. Mater. 2001, 44, 2099–2103. [Google Scholar] [CrossRef]
- Ghandehari, M.H. Anodic behavior of cemented WC-6% Co alloy in phosphoric acid solutions. J. Eletrochem. Soc. 1980, 127, 2144–2147. [Google Scholar] [CrossRef]
- Binder, H.; Kohling, A.; Kuhn, W.; Lindner, W.; Sanstede, G. Nature 1969, 224, 1299–1300.
- Patil, P.R.; Pawar, S.H.; Pail, P.S. The electrochromic properties of tungsten oxide thin films deposited by solution thermolysis. Sol. State Ionics 2000, 136, 505–511. [Google Scholar] [CrossRef]
- Sutthiruangwong, S.; Mori, G. Corrosion properties of Co-based cemented carbides in acidic solutions. Int. J. Refract. Met. H. 2003, 21, 135–145. [Google Scholar] [CrossRef]
- Scholl, H.; Hofman, B.; Rauscher, A. Anodic polarization of cemented carbides of the type [(WC, M); M = Fe, Ni or Co] in sulphuric acid solution. Electrochim. Acta 1992, 37, 447–452. [Google Scholar] [CrossRef]
- Lee, K.; Ishihara, A.; Mitsushima, S.; Kamiya, N.; Ota, K. Stability and electrocatalytic activity for oxygen reduction in WC + Ta catalyst. Electrochim. Acta 2004, 49, 3479–3485. [Google Scholar] [CrossRef]
- Weigert, E.C.; Esposito, D.V.; Chen, J.G. Cyclic voltammetry and X-ray photoelectron spectroscopy studies of electrochemical stability of clean and Pt-modified tungsten and molybdenum carbide (WC and Mo2C) electrocatalysts. J. Power Sources 2009, 193, 501–506. [Google Scholar] [CrossRef]
- Jeon, M.K.; Daimon, H.; Lee, K.R.; Nakahara, A.; Woo, S.I. CO tolerant Pt/WC methanol electro-oxidation catalyst. Electrochem. Commun. 2007, 9, 2692–2695. [Google Scholar] [CrossRef]
- McIntyre, D.R.; Burstein, G.T.; Vossen, A. Effect of carbon monoxide on the electrooxidation of hydrogen by tungsten carbide. J. Power Sources 2002, 107, 67–73. [Google Scholar] [CrossRef]
- Ham, D.J.; Kim, Y.K.; Han, S.H.; Lee, J.S. Pt/WC as an anode catalyst for PEMFC: Activity and CO tolerance. Catal. Today 2008, 132, 117–122. [Google Scholar] [CrossRef]
- Hwu, H.H.; Polizzotti, B.D.; Chen, J.G. Potential application of tungsten carbides as electrocatalysts. 2. Coadsorption of CO and H2O on carbide-modified W(111). J. Phys. Chem. B 2001, 105, 10045–10053. [Google Scholar] [CrossRef]
- Weigert, E.C.; Zellner, M.B.; Stottlemyer, A.L.; Chen, J.G. A combined surface science and electrochemical study of tungsten carbides as anode electrocatalysts. Top. Catal. 2007, 46, 349–357. [Google Scholar] [CrossRef]
- Hara, Y.; Minami, N.; Itagaki, H. Synthesis and characterization of high-surface area tungsten carbides and application to electrocatalytic hydrogen oxidation. Appl. Catal. A. 2007, 323, 86–93. [Google Scholar] [CrossRef]
- Ganesan, R.; Lee, J.S. Tungsten carbide microspheres as a noble-metal-economic electrocatalyst for methanol oxidation. Angew. Chem. Int. Ed. 2005, 44, 6557–6560. [Google Scholar] [CrossRef]
- Ganesan, R.; Ham, D.J.; Lee, J.S. Platinized mesoporous tungsten carbide for electrochemical methanol oxidation. Electrochem. Commun. 2007, 9, 2576–2579. [Google Scholar] [CrossRef]
- Nagai, M.; Yoshida, M.; Tominaga, H. Tungsten and nickel tungsten carbides as anode electrocatalysts. Electrochim. Acta 2007, 52, 5430–5436. [Google Scholar] [CrossRef]
- Yang, X.G.; Wang, C.Y. Nanostructured tungsten carbide catalysts for polymer electrolyte fuel cells. Appl. Phys. Lett. 2005, 86, 224104. [Google Scholar] [CrossRef]
- Hwu, H.H.; Chen, J.G.; Kourtakis, K.; Lavin, J.G. Potential application of tungsten carbides as electrocatalysts. 1. Decomposition of methanol over carbide-modified W(111). J. Phys. Chem. B 2001, 105, 10037–10044. [Google Scholar] [CrossRef]
- Liu, N.; Kourtakis, K.; Figueroa, J.C.; Chen, J.G. Potential application of tungsten carbides as electrocatalysts III. Reactions of methanol, water, and hydrogen on Pt-modified C/W(111) surfaces. J. Catal. 2003, 215, 254–263. [Google Scholar] [CrossRef]
- Weigert, E.C.; Stottlemyer, A.L.; Zellner, M.B.; Chen, J.G. Tungsten monocarbide as potential replacement of platinum for methanol electrooxidation. J. Phys. Chem. C 2007, 111, 14617–14620. [Google Scholar] [CrossRef]
- Joo, J.B.; Kim, J.S.; Kim, P.; Yi, J. Simple preparation of tungsten carbide supported on carbon for use as a catalyst support in a methanol electro-oxidation. Mater. Lett. 2008, 62, 3497–3499. [Google Scholar] [CrossRef]
- Wang, Z.; Zuo, P.; Liu, B.; Yin, G.; Shi, P. Stable PtNiPb/WC catalyst for direct methanol fuel cells. Electrochem. Solid State Lett. 2009, 12, A13–A15. [Google Scholar] [CrossRef]
- Zhao, Z.; Fang, X.; Li, Y.; Wang, Y.; Shen, P.K.; Xie, F.; Zhang, X. The origin of the high performance of tungsten carbides/carbon nanotubes supported Pt catalysts for methanol electrooxidation. Electrochem. Commun. 2009, 11, 290–293. [Google Scholar] [CrossRef]
- Wang, R.; Tian, C.; Wang, L.; Wang, B.; Zhang, H.; Fu, H. In situ simultaneous synthesis of WC/graphitic carbon nanocomposite as a highly efficient catalyst support for DMFC. Chem. Comm. 2009, 21, 3104–3106. [Google Scholar] [CrossRef] [PubMed]
- Hwu, H.H.; Chen, J.G. Reactions of methanol and water over carbide-modified Mo(110). Surf. Sci. 2003, 536, 75–87. [Google Scholar] [CrossRef]
- Zellner, M.B.; Hwu, H.H.; Chen, J.G. Comparative studies of methanol decomposition on carbide-modified V(110) and Ti(0001). Surf. Sci. 2005, 598, 185–199. [Google Scholar] [CrossRef]
- Hu, F.; Cui, G.; Wei, Z.; Shen, P.K. Improved kinetics of ethanol oxidation on Pd catalysts supported on tungsten carbides/carbon nanotubes. Electrochem. Commun. 2008, 10, 1303–1306. [Google Scholar] [CrossRef]
- Nie, M.; Tang, H.; Wei, Z.; Jiang, S.P.; Shen, P.K. Highly efficient AuPd–WC/C electrocatalyst for ethanol oxidation. Electrochem. Commun. 2007, 9, 2375–2379. [Google Scholar] [CrossRef]
- Chhina, H.; Campbell, S.; Kesler, O. Thermal and electrochemical stability of tungsten carbide catalyst supports. J. Power Sources 2007, 164, 431–440. [Google Scholar] [CrossRef]
- Meng, H.; Shen, P.K. Novel Pt-free catalyst for oxygen electroreduction. Electrochem. Commun. 2006, 8, 588–594. [Google Scholar] [CrossRef]
- Meng, H.; Shen, P.K. Tungsten carbide nanocrystal promoted Pt/C electrocatalysts for oxygen reduction. J. Phys. Chem. B 2005, 109, 22705–22709. [Google Scholar] [CrossRef] [PubMed]
- Nie, M.; Shen, P.K.; Wei, Z. Nanocrystaline tungsten carbide supported Au–Pd electrocatalyst for oxygen reduction. J. Power Sources 2007, 167, 69–73. [Google Scholar] [CrossRef]
- Lee, J.S.; Hyun, T.H. Metal nitrides. In Encyclopedia of Catalysis, 1st ed.; Horvath, I.T., Ed.; John Wiley & Sons: New York, NY, USA, 2003. [Google Scholar]
- Ponce, F.A.; Moustakas, T.D.; Akasaki, I.; Monemar, B.A. III-V Nitrides; Materials Research Society: Warrendale, PA, USA, 1997. [Google Scholar]
- Markel, E.J.; Leaphart, M.E.; Howe-Grant, M.E. Encyclopedia of Chemical Technology; John Wiley & Sons: New York, NY, USA, 1992. [Google Scholar]
- Chen, J.G. Carbide and nitride overlayers on early transition metal surfaces: preparation, characterization, and reactivities. Chem. Rev. 1996, 96, 1477–1498. [Google Scholar] [CrossRef] [PubMed]
- Choi, D.; Kumta, P.N. Synthesis, structure, and electrochemical characterization of nanocrystalline tantalum and tungsten nitrides. J. Am. Ceram. Soc. 2007, 90, 3113–3120. [Google Scholar] [CrossRef]
- Roberson, S.L.; Finello, D.; Davis, R.F. Electrochemical evaluation of molybdenum nitride electrodes in H2SO4 electrolyte. J. Appl. Electrochem. 1999, 29, 75–80. [Google Scholar] [CrossRef]
- Francois, J.C.; Massiani, Y.; Gravier, P.; Grimblot, J.; Gengembre, L. Characterization and optical properties of thin films formed on TiN coatings during electrochemical treatments. Thin Solid Films 1993, 223, 223–229. [Google Scholar] [CrossRef]
- Walther, B.; Schilm, J.; Michaelis, A.; Lohrengel, M.M. Electrochemical dissolution of hard metal alloys. Electrochim. Acta 2007, 52, 7732–7737. [Google Scholar] [CrossRef]
- Subramanian, B.; Jayachandran, M. Electrochemical corrosion behavior of magnetron sputtered TiN coated steel in simulated bodily fluid and its hemocompatibility. Mater. Lett. 2008, 62, 1727–1730. [Google Scholar] [CrossRef]
- Musthafa, O.T.M.; Sampath, S. High performance platinized titanium nitride catalyst for methanol oxidation. Chem. Comm. 2008, 7, 67–69. [Google Scholar] [CrossRef]
- Zhong, H.; Zhang, H.; Liang, Y.; Zhang, J.; Wang, M.; Wang, X. A novel non-noble electrocatalyst for oxygen reduction in proton exchange membrane fuel cells. J. Power Sources 2007, 164, 572–577. [Google Scholar] [CrossRef]
- Xia, D.; Liu, S.; Wang, Z.; Chen, G.; Zhang, L.; Zhang, L.; Hui, S.; Zhang, J. Methanol-tolerant MoN electrocatalyst synthesized through heat treatment of molybdenum tetraphenylporphyrin for four-electron oxygen reduction reaction. J. Power Sources 2008, 177, 296–302. [Google Scholar] [CrossRef]
- Zhong, H.; Zhang, H.; Liu, G.; Liang, Y.; Hu, J.; Yi, B. A novel non-noble electrocatalyst for PEM fuel cell based on molybdenum nitride. Electrochem. Commun. 2006, 8, 707–712. [Google Scholar] [CrossRef]
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ham, D.J.; Lee, J.S. Transition Metal Carbides and Nitrides as Electrode Materials for Low Temperature Fuel Cells. Energies 2009, 2, 873-899. https://doi.org/10.3390/en20400873
Ham DJ, Lee JS. Transition Metal Carbides and Nitrides as Electrode Materials for Low Temperature Fuel Cells. Energies. 2009; 2(4):873-899. https://doi.org/10.3390/en20400873
Chicago/Turabian StyleHam, Dong Jin, and Jae Sung Lee. 2009. "Transition Metal Carbides and Nitrides as Electrode Materials for Low Temperature Fuel Cells" Energies 2, no. 4: 873-899. https://doi.org/10.3390/en20400873