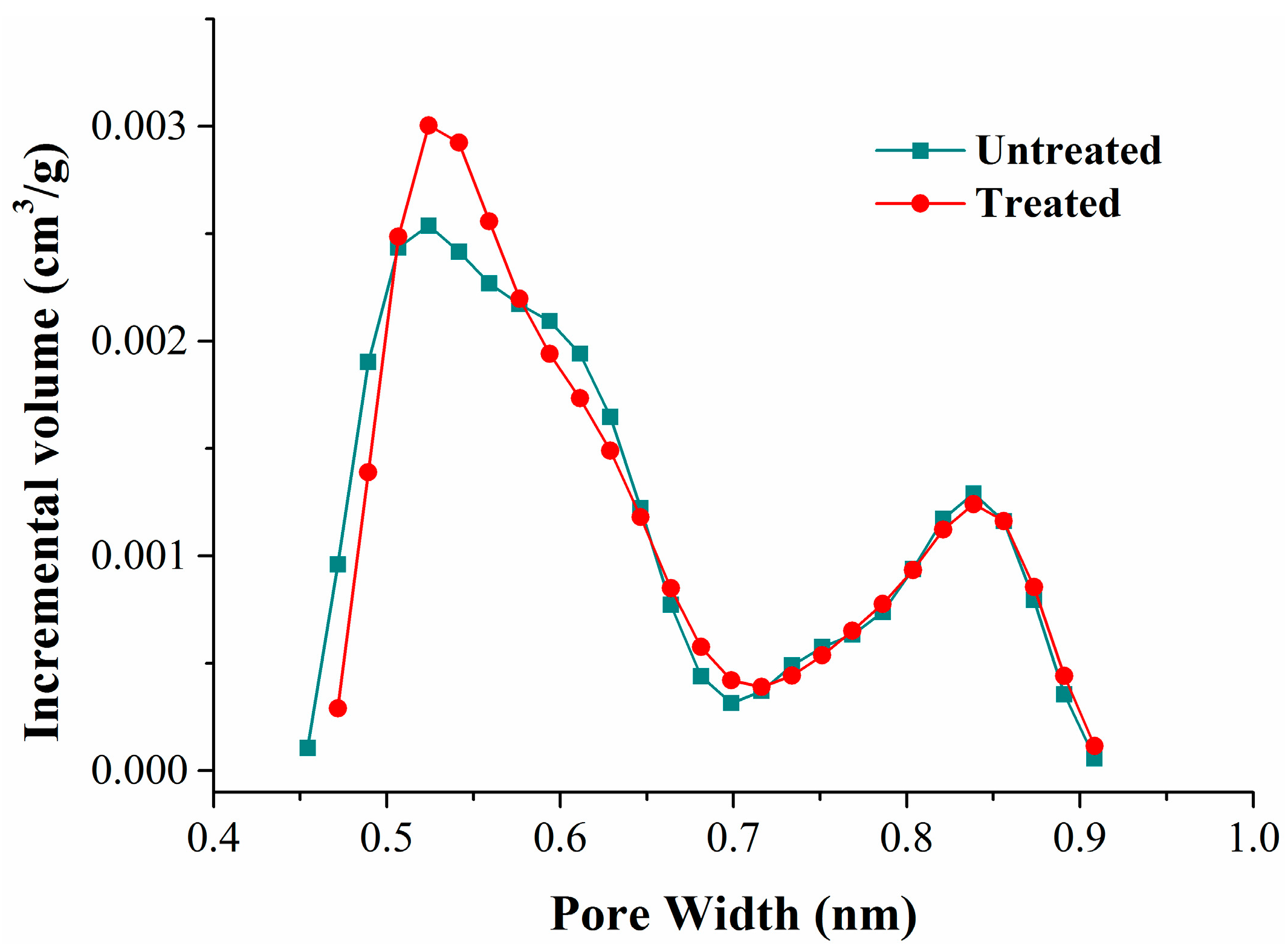
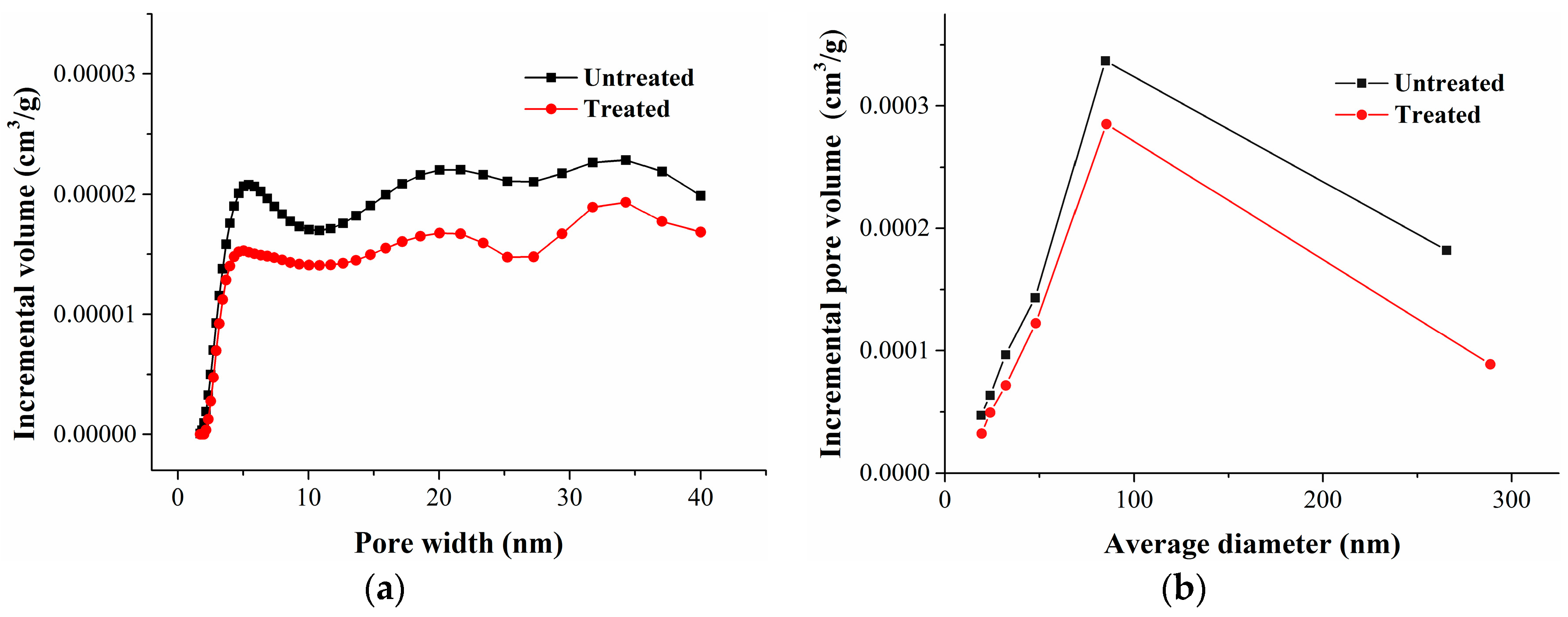
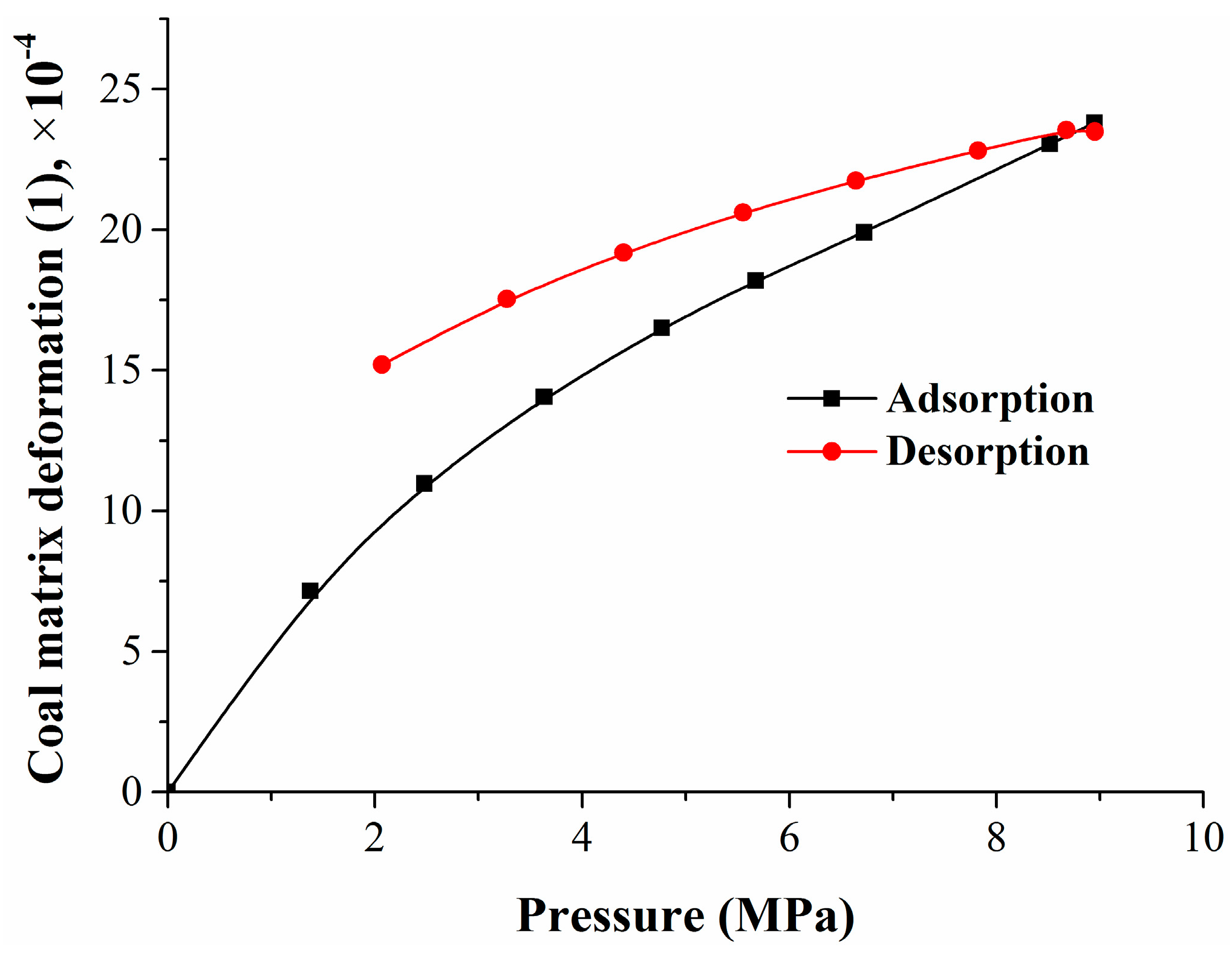
1. Introduction
A coal seam is a kind of typical heterogeneous unconventional natural gas reservoir, developed abundantly in pores and fractures [
1,
2]. Pores in the coal matrix are the main space that adsorbs coalbed methane (CBM), and pores and fractures are also the main channels for CBM diffusion and migration [
3,
4]. Compared to those in other countries, coal reservoirs in China are characterized by low reservoir pressure, low gas saturation, and low permeability. The permeability of most coal seams ranges from 10
−4 to 10
−1 mD [
5,
6,
7,
8]. Nitrogen foam fracturing was introduced into CBM exploitation, as traditional hydraulic fracturing for coal reservoirs cannot sufficiently enhance the permeability significantly in the complex geological conditions of China. Therefore, nitrogen foam fracturing has been introduced into CBM exploitation as a new technique in recent years [
9,
10,
11,
12]. High pressure nitrogen gas is injected into the coal reservoir, which can improve the recovery efficiency of CBM [
5,
13,
14,
15,
16].
Nitrogen foam fracturing has many advantages for CBM reservoir reconstruction. However, due to nitrogen adsorption, the pressure of the reservoir increases and the coal matrix swells, which leads to a decrease in coal reservoir permeability. Studies on the relationship between the swelling deformation of the coal matrix and the adsorption of methane and nitrogen are helpful for precise control of the nitrogen volume in the fracturing process.
Nitrogen foam fracturing involves multicomponent gas adsorption/desorption, replacement, and interaction. The coal matrix swells and shrinks, and coal reservoir permeability decreases and increases accordingly [
17,
18,
19,
20]. As is widely known, methane adsorption in coal is through physical adsorption. The adsorption quantity of coal is not only associated with its own properties (pore features, coal–rock composition, coal rank, and moisture content), but also associated with temperature, gas types, and coal particle size [
21,
22,
23,
24].
In recent years, research on coal matrix deformation has mainly focused on adsorption gas types, adsorption quantity, gas pressure, and so on. Lin and Zhou calculated the deformation of coal samples by experiments; the deformation amplitude was CO
2 > CH
4 > N
2. As the gas pressure and adsorption quantity increases, the coal sample swelling deformation basically follows the Langmuir equation. In the desorption process, the shrinkage deformation of coal samples decreases exponentially [
25]. A study has shown that coal matrix deformation is associated with the adsorption quantities of different gases: the nitrogen adsorption quantity is the lowest. Its deformation is lower than other gases, and has a linear relationship with adsorption quantity [
26]. Through laboratory experiments or numerical simulations, the relationship between coal matrix deformation and the pressure or adsorption quantity of gas can be estimated. However, due to the differences in the experimental apparatus, measuring methods, selection of samples, and simulation calculation models, there are differences in the relationship between coal matrix deformation and the pressure or adsorption quantity of gas [
27,
28,
29,
30,
31]. The deformation of the coal matrix plays an important role in the change of coal reservoir permeability [
32,
33].
Most researchers have focused on pure or mixed gas adsorption-induced coal matrix deformation. Studies on the continuous deformation characteristics of the coal matrix during the process of methane replacement by nitrogen injection, which involves two kinds of gas adsorption/desorption and their interactions, are relatively few in number.
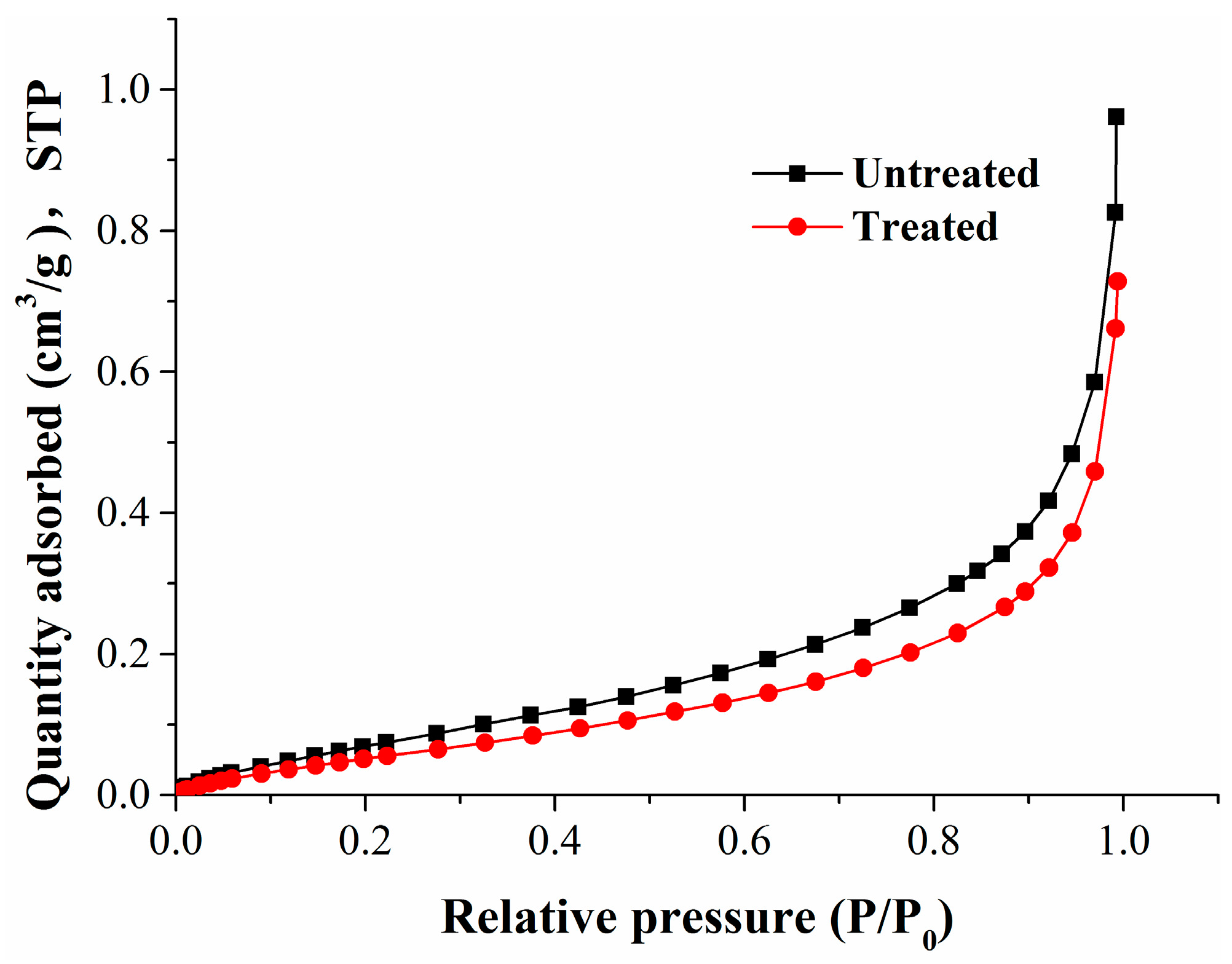
This paper studied the coal matrix deformation models at different stages by performing isothermal adsorption/desorption experiments of methane replacement by nitrogen injection. Combining the experimental results, the deformation law of the coal matrix and the relationship between the coal matrix and its adsorption quantity or pressure was studied, which can provide technical support and a theoretical basis for tuning injection parameters in nitrogen foam fracturing and predicting coal reservoir permeability. By comparing the characteristics of pore development in the samples before and after isothermal adsorption experiments, the mechanism of coal matrix deformation was discussed. Finally, the results revealed whether the output and migration of CBM was of benefit.
2. Deformation Model
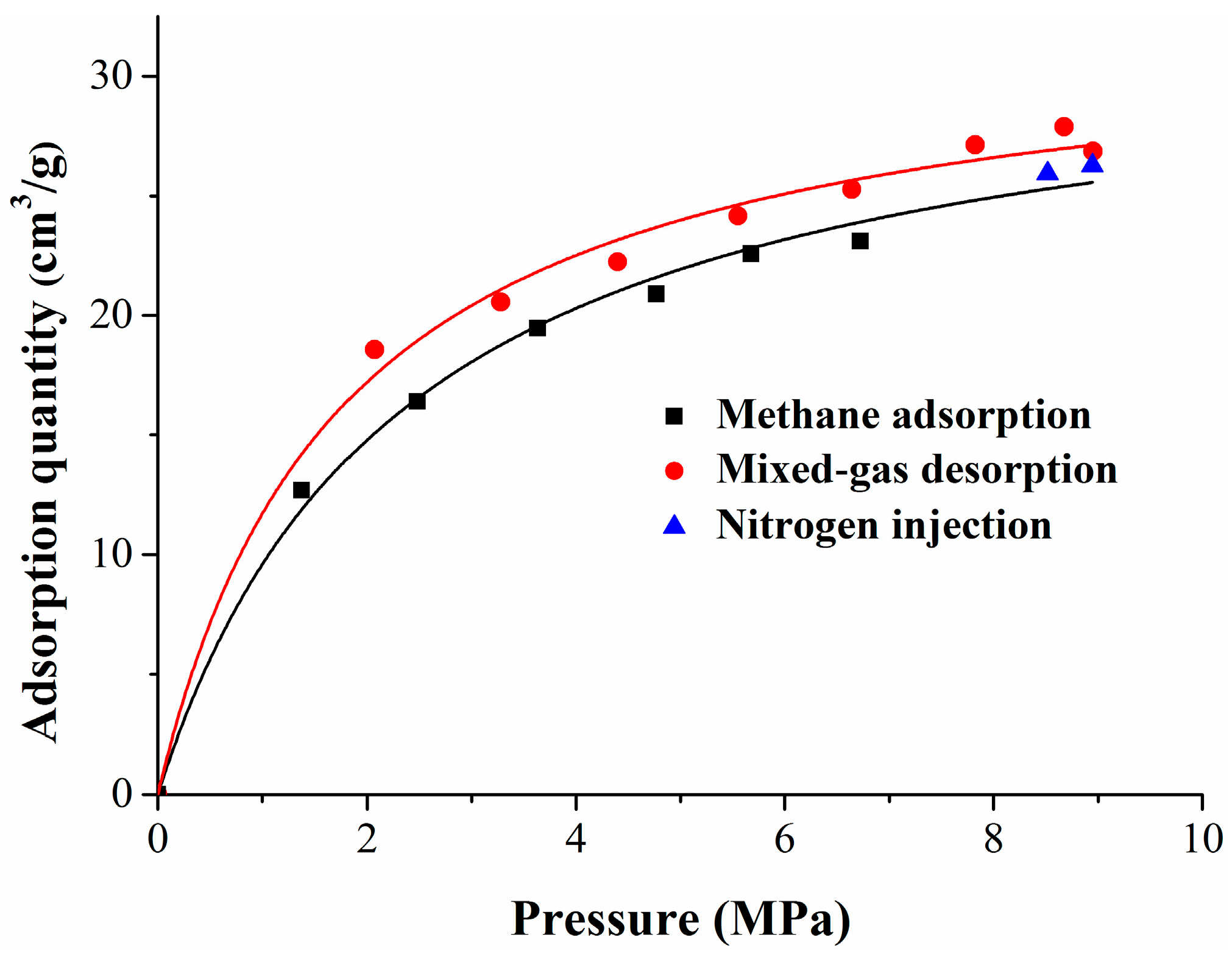
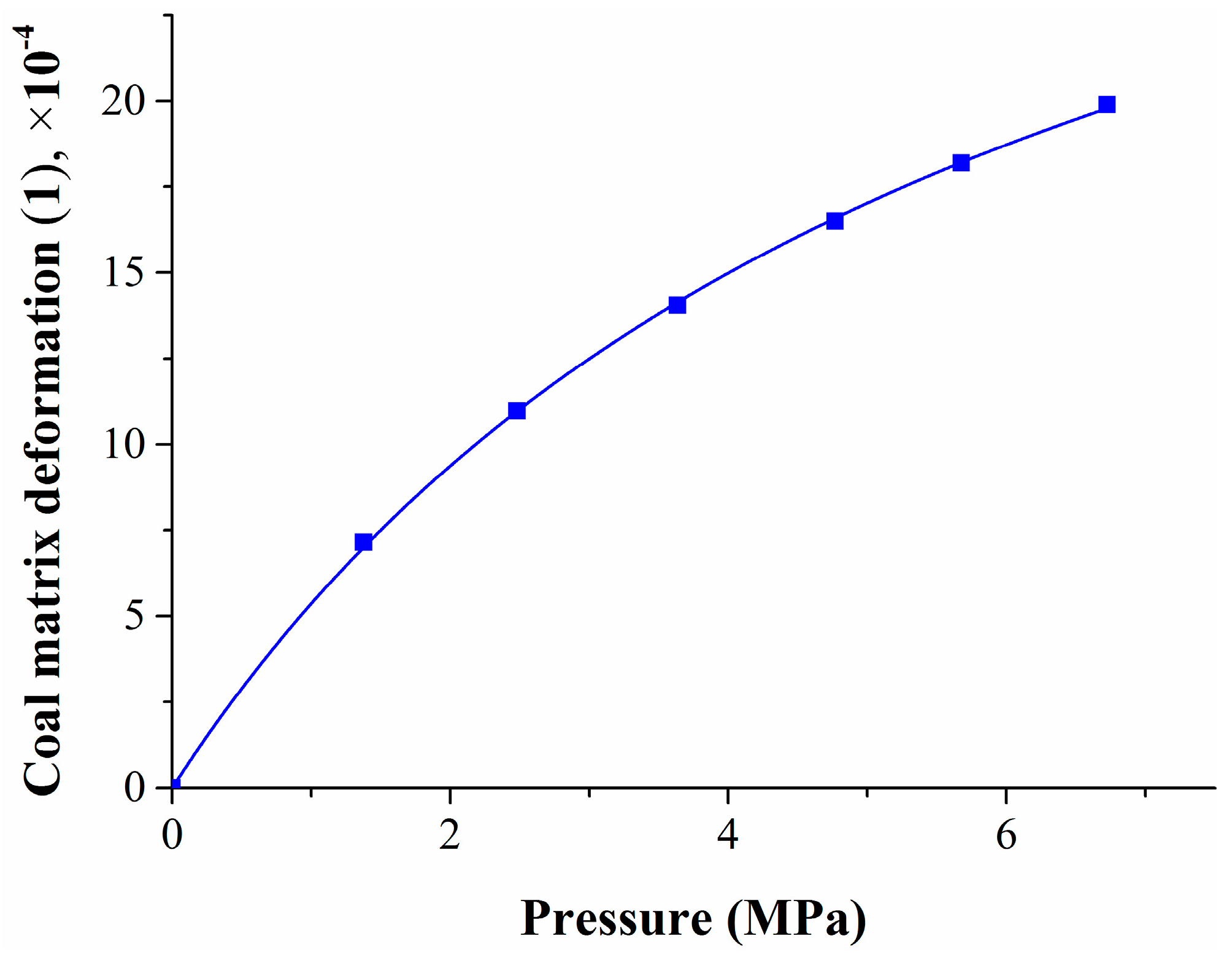
The coal matrix deformation experiment was divided into three stages: the first stage is the methane adsorption process; the second stage is the nitrogen injection process; and the third stage is the desorption process by pressure decrease. Assuming that the temperature of the gas adsorption and desorption process remains unchanged, the gas pressure does not affect the compressibility of the coal matrix under the low-pressure condition. Based on the theories of surface free energy, the Langmuir equation, and elastic mechanics, the deformation models were established at different stages.
2.1. Methane Adsorption Swelling Model
Assuming that the coal matrix is an isotropic elastic body, the change in the elastic energy equals the change in the surface energy of the coal matrix caused by the adsorption. Bangham considered that the swelling deformation of the coal matrix was proportional to its surface free energy decrease, and that the surface free energy could be considered as the surface tension of the unit length, so the swelling deformation of the coal matrix was proportional to the decrease in surface tension [
34,
35]. The relationship between the swelling deformation and the surface tension reduction of the coal matrix is expressed in Equation (1):
where
ε is the coal matrix deformation, 1;
ρc is the coal matrix density, cm
3/g;
S is the specific surface area, m
2/g; Δ
r is the surface tension reduction, N·m
−1; and
E is the elastic modulus, Pa.
After the coal matrix absorbs methane, the surface tension will decrease. The surface tension reduction can be calculated by the Gibbs formula [
36]:
In Equation (2), R is the universal gas constant, 8.314 J/(mol·K); T is the absolute temperature, K; Γ is the surface excess, mol/m2; and P is the adsorption gas pressure, Pa.
According to the principle of minimum energy, the carbon atoms in the surface of the coal matrix can reduce its specific surface energy by adsorption of methane molecules, so that the methane adsorption concentration in the pores or fractures is lower than that on the surface. The methane concentration difference is known as surface excess, which can be expressed as
where
V is the standard state volume of the adsorbed gas, cm
3; and
V0 is the molar volume of gas, cm
3/mol [
37,
38].
When the methane pressure increases from
P1 to
P2, by substituting Equations (2) and (3) into Equation (1), the coal matrix deformation can be expressed as follows:
The adsorption volume of coal matrix is usually expressed by the Langmuir equation [
39]:
where
VCH4 is the volume of methane adsorption, cm
3/g;
a is the maximum adsorption quantity, cm
3/g; and
b is the adsorption equilibrium constant, MPa
−1.
When the methane pressure increases from P1 to P2, the coal matrix swelling deformation can be expressed as
Equation (6) shows that the adsorption quantity of the coal matrix increases as the pressure increases, and the coal matrix swells. The coal matrix deformation is associated with the gas pressure, temperature, and the characteristics of the adsorbent. Compared with the adsorption model of coal–rock by [
40], the adsorption swelling model in this paper only considered changes of the adsorption quantity and the pressure, from which the coal matrix deformation under different gas pressure can easily be calculated.
2.2. Nitrogen Injection Swelling Model
Previous studies have shown that the adsorption quantity of the coal matrix with regards to different gases is different, and that the adsorption quantity of methane is higher than that of nitrogen [
21,
41].
After nitrogen injection, the nitrogen and the methane will compete for adsorption. The calculation of multicomponent gas adsorption quantity is based on the extended Langmuir equation [
42,
43]:
where
VN2 and
VCH4 are the adsorption quantities of nitrogen and methane, respectively, cm
3/g;
aN2 and
aCH4 are the maximum adsorption quantities of nitrogen and methane, respectively, cm
3/g;
bN2 and
bCH4 are the adsorption equilibrium constants of nitrogen and methane, respectively, MPa
−1; and
PN2 and
PCH4 are the partial pressures of nitrogen and methane, respectively, in the adsorption equilibrium state, MPa.
In the adsorption equilibrium state, the equilibrium pressure is the sum of the two partial pressures:
where
P is the equilibrium pressure, MPa.
In Equation (10), XN2 and XCH4 are the molar fractions of nitrogen and methane, respectively, in the free phase, %. Their values can be tested by gas chromatograph, and XN2 + XCH4 = 1.
From the initial to the equilibrium state, set the nitrogen pressure from 0 to
P3, and the methane pressure from
P2 to
P4. According to Equation (4), during the process of nitrogen replacing methane, the coal matrix deformation can be deduced from Equations (11) and (12):
where Δ
εN2 and Δ
εCH4 are the coal matrix deformations induced by nitrogen and methane adsorption, respectively, 1.
As the nitrogen injection rate is slow and constant, the partial pressure of nitrogen can be considered to increase linearly. The relationship between the partial pressure of nitrogen and the equilibrium pressure is given by Equation (13):
where
u and
w are the fitting constants given by the experimental results.
From Equations (9), (10), and (13), the relationship between the partial pressure of nitrogen and methane can be obtained.
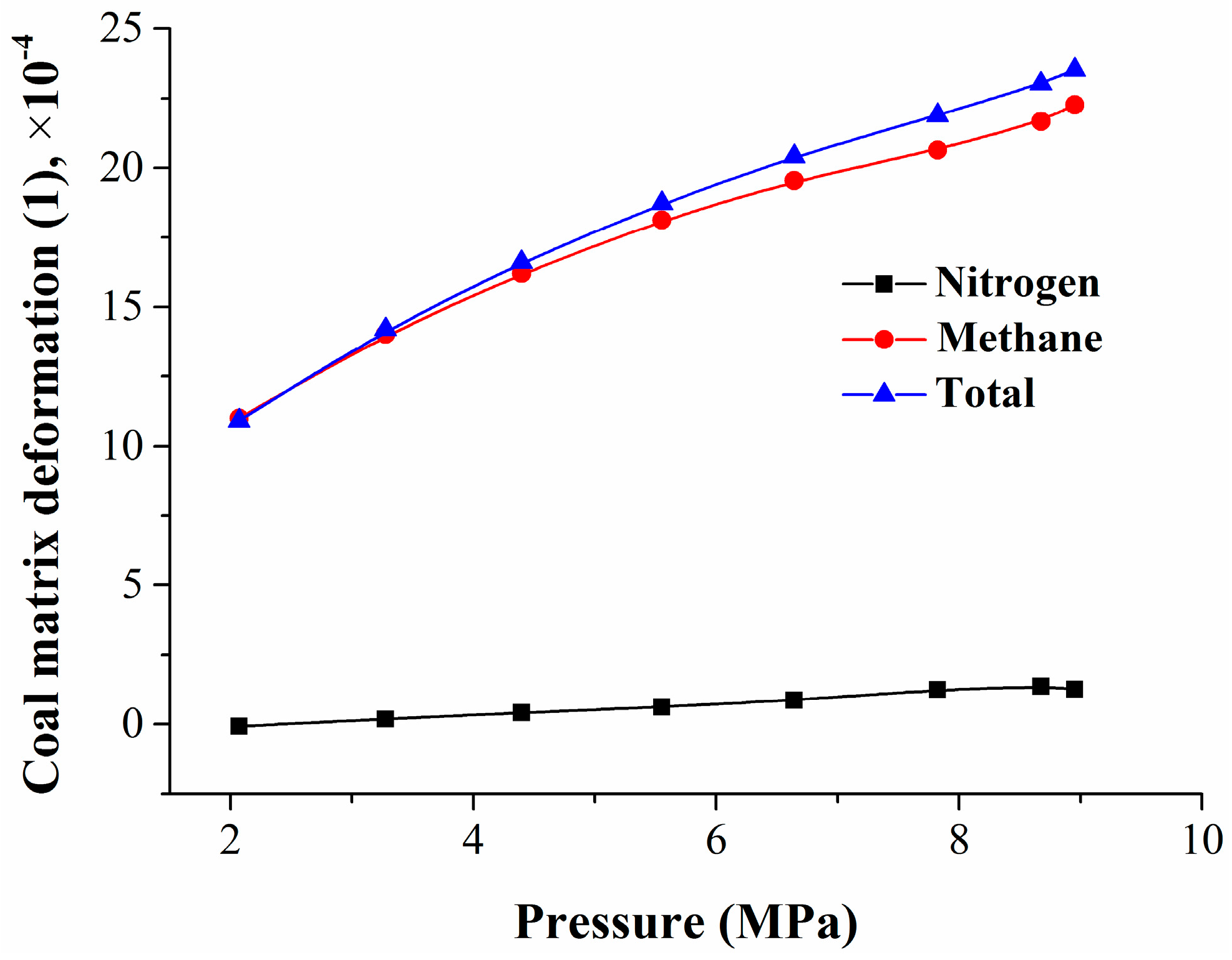
Then, from Equations (7), (11), and (15), the coal matrix deformation model caused by nitrogen adsorption or replacement can be obtained.
Analogously, from Equations (8), (12), and (14), the coal matrix deformation model caused by methane adsorption can be obtained.
In the nitrogen injection process, the total deformation of the coal matrix caused by nitrogen and methane is given by Equation (18):
where Δ
εt is the total deformation of the coal matrix induced by nitrogen and methane, 1.
2.3. Shrinking Model in Methane and Nitrogen Desorption
In the desorption process, as the pressure decreases, the molar fractions of nitrogen and methane in the free phase change dynamically. When the pressure is low, the relationship between the ratio of nitrogen and methane molar fractions and the equilibrium pressure is linear:
where
m and
n are the fitting constants.
According to Equation (10), the relationship between the partial pressures of the two gases and the equilibrium pressure can be expressed as:
Combined with Equation (9), the relationships between the partial pressures of the two gases can be obtained:
During the process of pressure reduction, the partial pressure of nitrogen changes from
PN21 to
PN22, and the partial pressure of methane changes from
PCH41 to
PCH42. According to Equation (4), the coal matrix deformation induced by nitrogen or methane is obtained.
From Equations (7), (22), and (23), the model of nitrogen-induced shrinking deformation of the coal matrix is obtained.
Similarly, from Equations (8), (21), and (24), a model of methane-induced shrinking deformation of the coal matrix is obtained.
In the desorption process, as there are two kinds of gas desorption, the shrinking deformation of the coal matrix was also caused by two kinds of gas desorption. The total deformation is:
where Δ
εj is the total deformation of the coal matrix induced by the two kinds of gas desorption, 1.