Enhancing Oil Recovery from Chalk Reservoirs by a Low-Salinity Water Flooding Mechanism and Fluid/Rock Interactions
Abstract
:1. Introduction
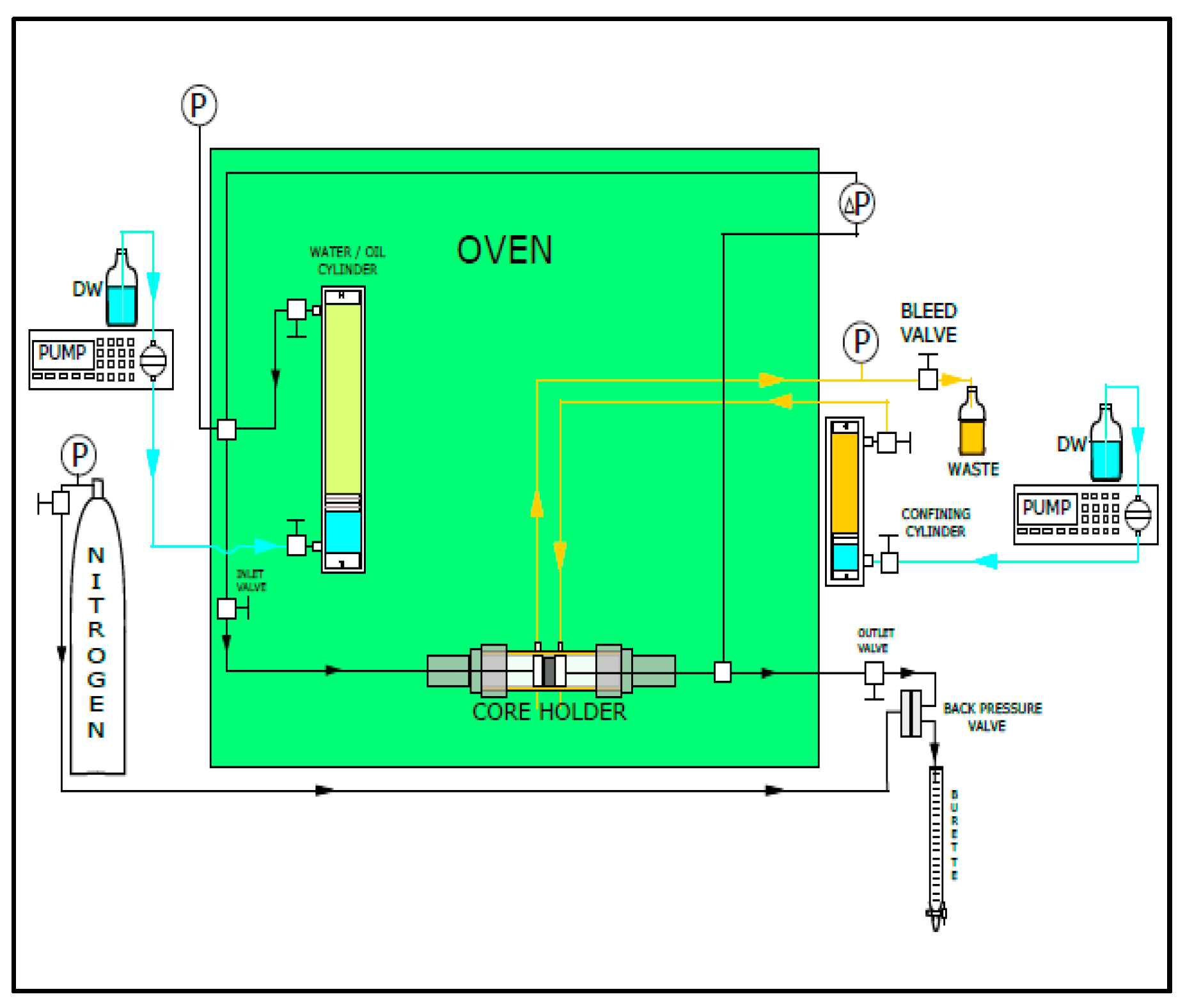
2. Experimental
2.1. Materials
2.1.1. Porous Media
2.1.2. Oil
2.1.3. Brines
2.1.4. Core Preparations and Flooding
3. Results and Discussion
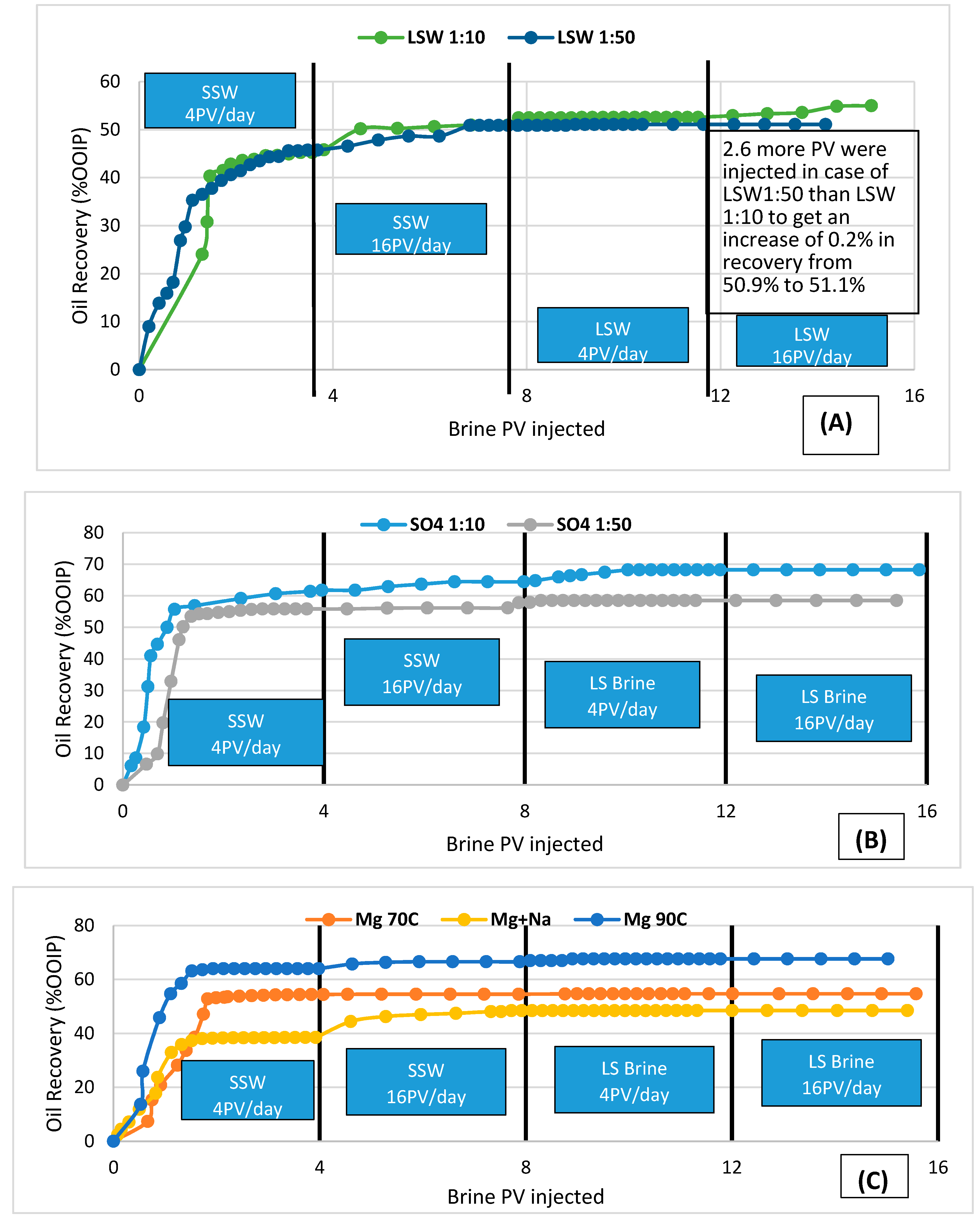
3.1. Oil Recovery from Secondary Flooding with LS
- (1)
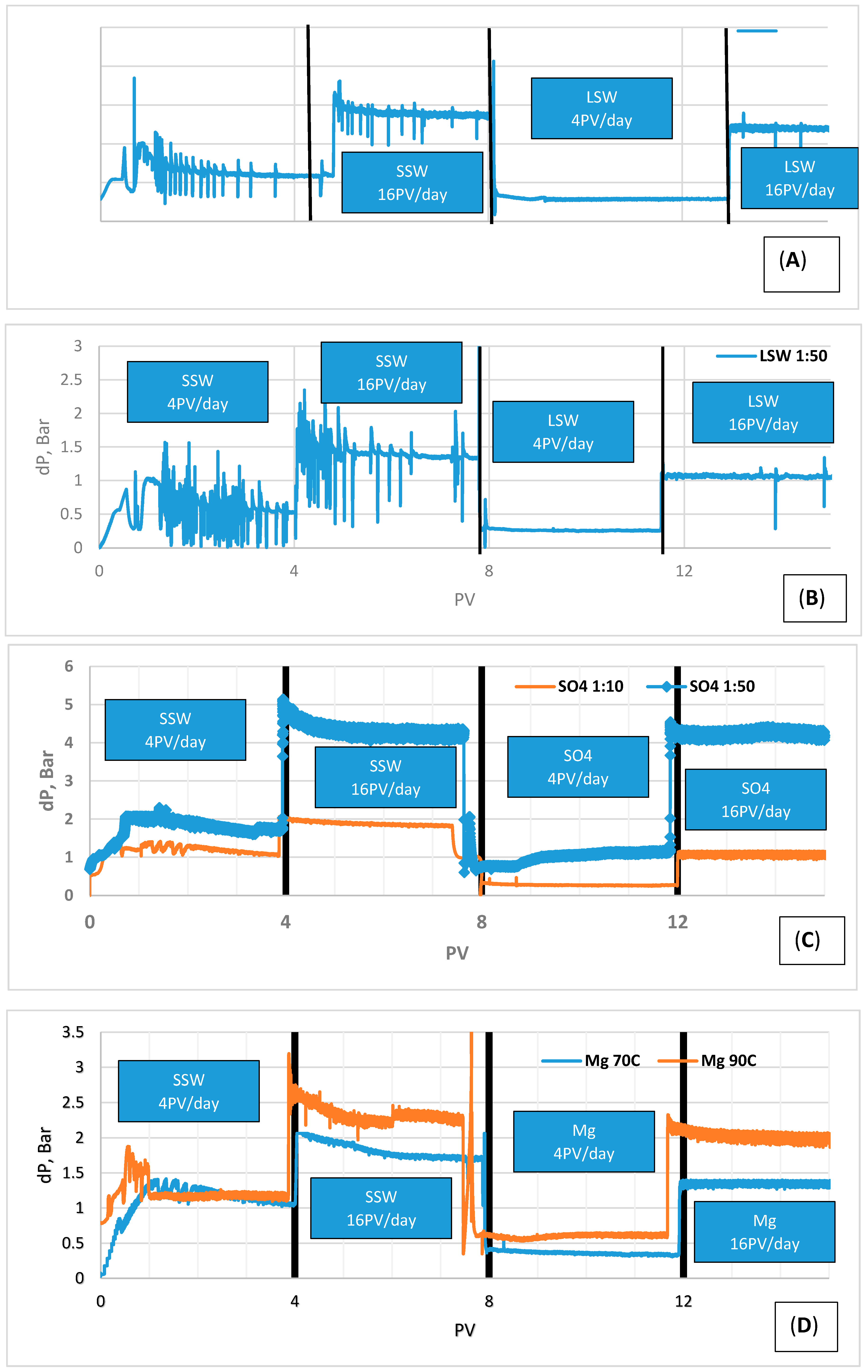
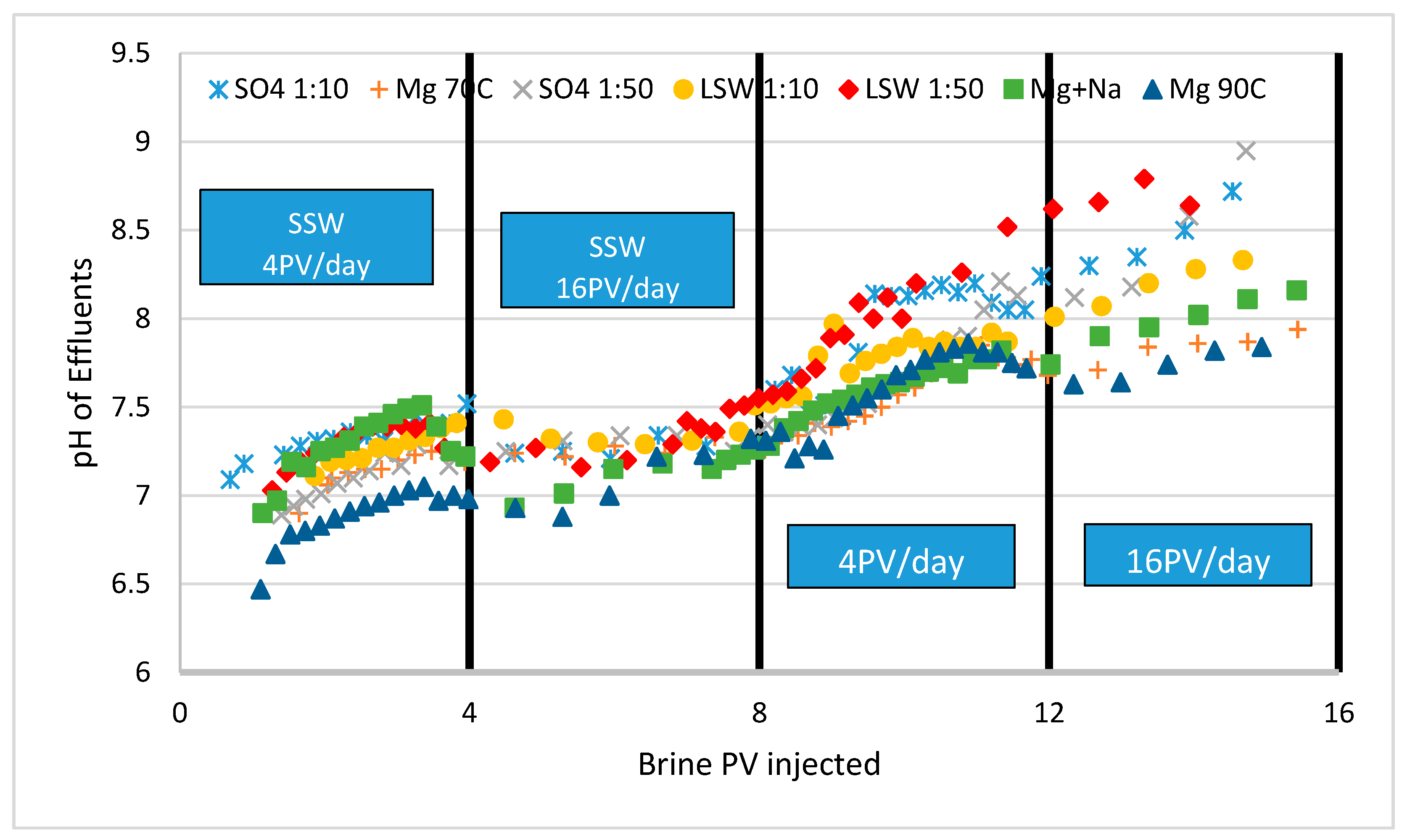
- Higher fluctuations were observed at 16 PV/day than 4 PV/day in the case of LSW 1:10 and 1:50 flooding. This may mean occasional resistance to the flow, hence a possible increase of the sweep efficiency.
- (2)
- The magnitude of dP was higher in case of dilution ratio 1:50 than 1:10, this is perhaps due to a higher availability of Ca2+ promoting precipitation of sulfate salt over the limit if diverting flow increasing the trapped oil.
- (3)
- Higher recovery in the case of dilution ratio 1:10 than 1:50, which has also been observed in the case of sulfate salt single brine flooding may support the above point (2). In [14], several dilutions of LSW were investigated and concluded that the dilution of 1:10 gave the best incremental oil recovery.
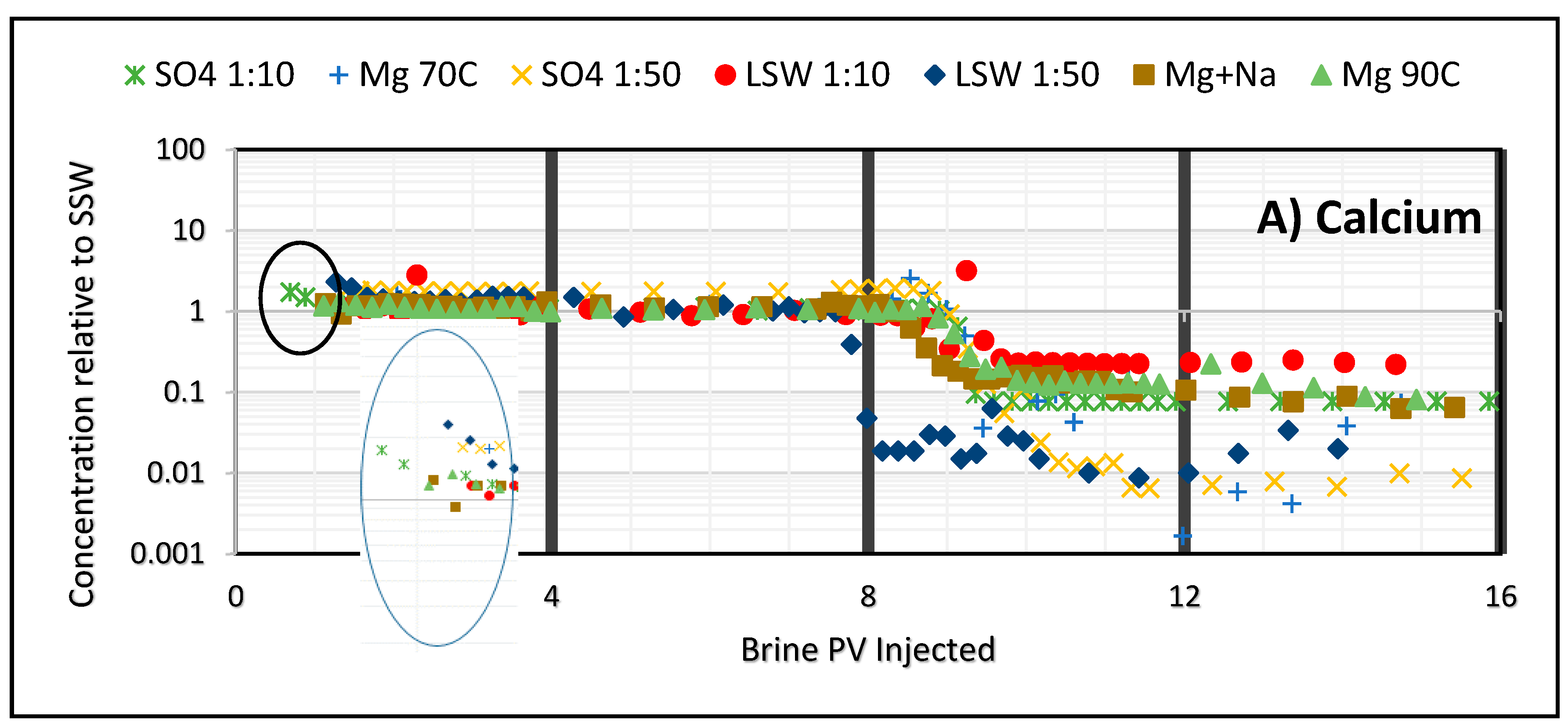
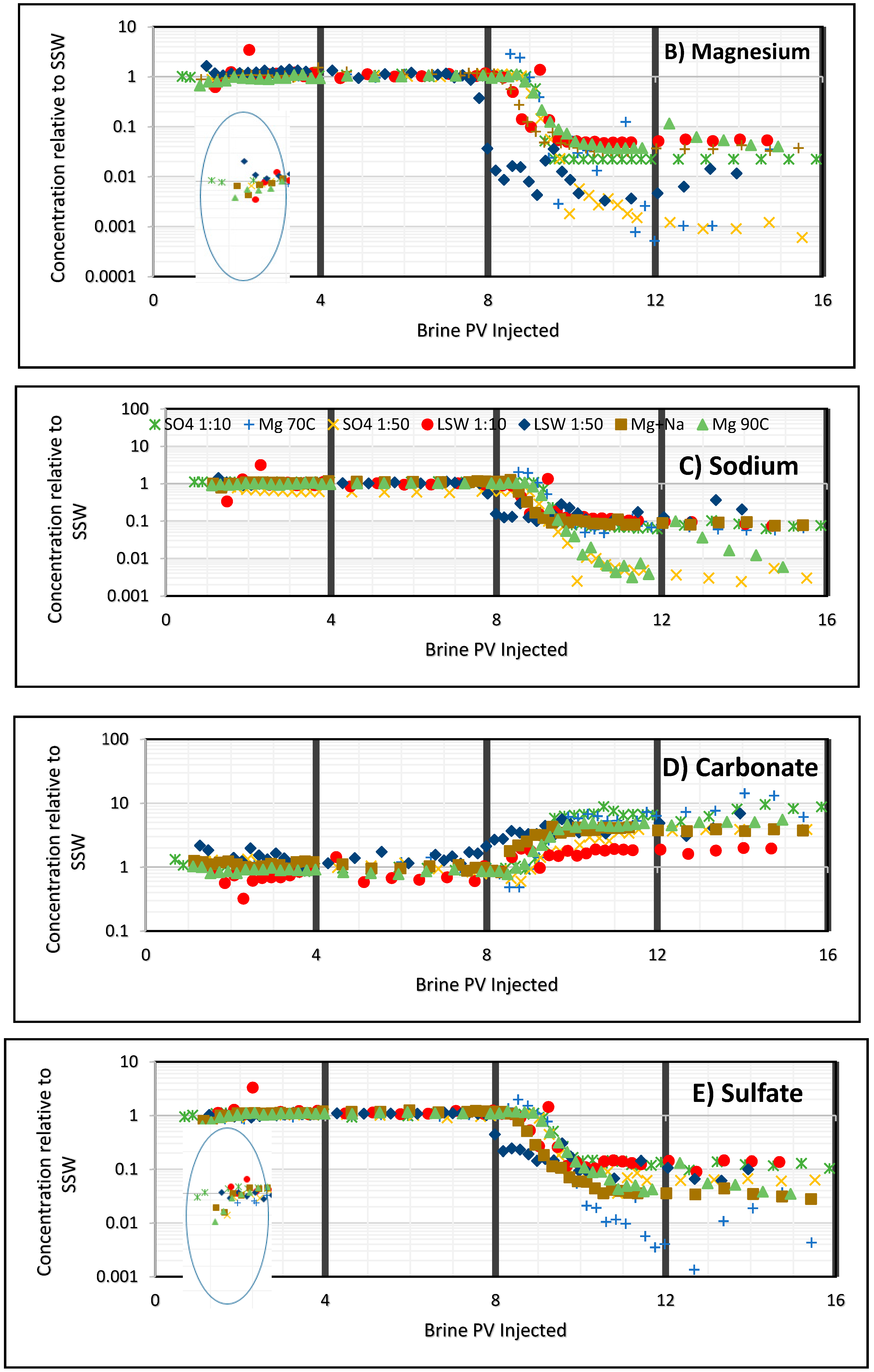
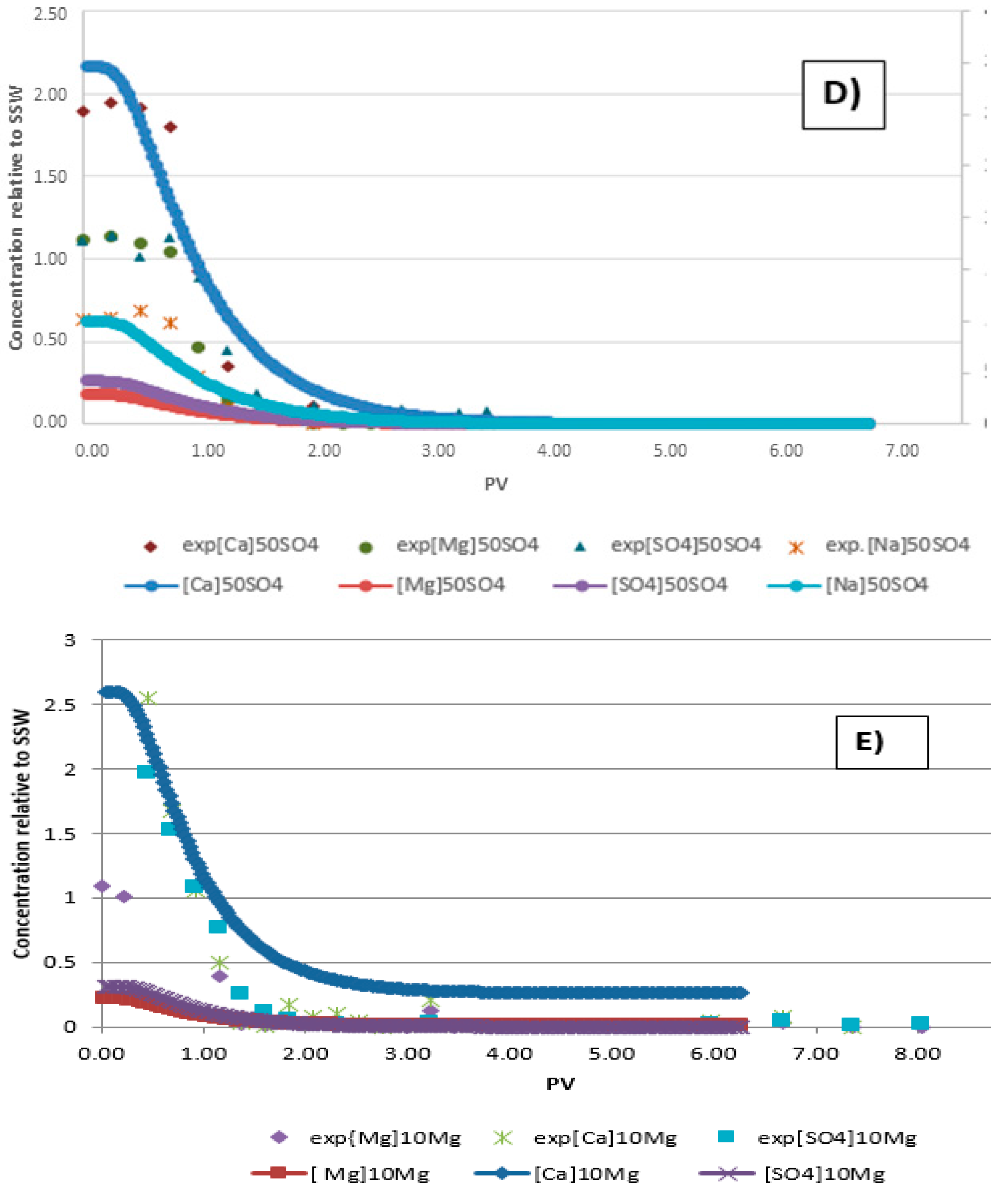
3.2. Ion Tracking from Secondary Flooding by LS Brines
4. Summary and Conclusions
- (1)
- Experimentally it is concluded that oil recovery response time depends on the ion dilution factor of the brine. LSW 1:10 gives earlier response than the LSW (1:50).
- (2)
- Divalent Ions have an effect in wettability alteration. Ca/Mg contributes largely in enhancing the sweep efficiency. But this effect increases in presence of SO42−. Highest recovery is obtained while flooding with SO4 brine than any other brine which shows that the presence of sulfate ion may contribute to the wettability alteration.
- (3)
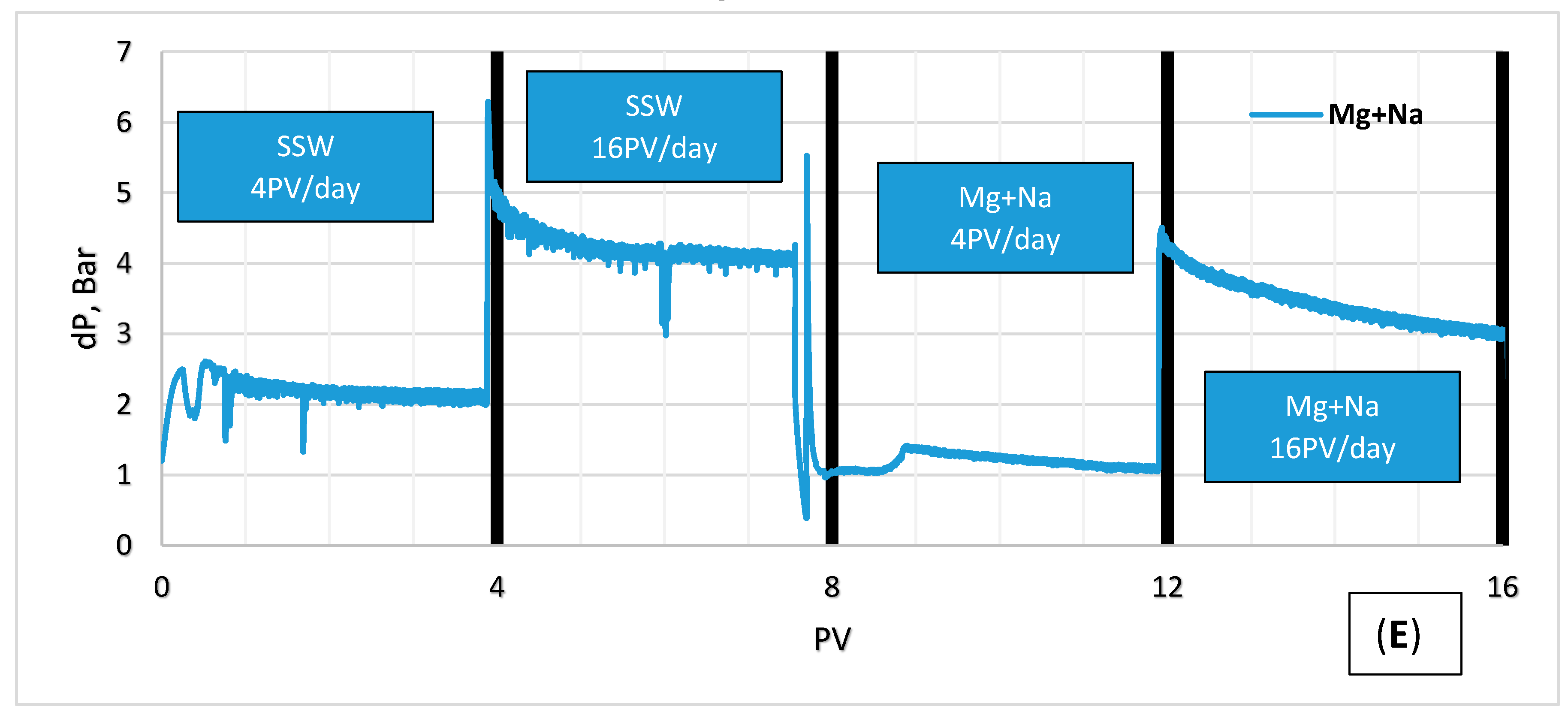
- Increase in ion concentrations of Mg2+ and Ca2+ in the later part of modified brine injection confirms ion exchange between the ions and thus precipitation of magnesium.
- (4)
- 10 times SSW dilution ratio gives the best outcome. This is also in agreement with the case of single salt brine injection. For SO4 1:10 dilution, higher recovery was obtained compared to that with SO4 1:50.
- (5)
- Pressure drop in the secondary flooding may indicate fine migration during injection of single salt brine and LSW, though fines were not observed in the effluent samples during our experiments. This may be due to size of the particles being too small to be observed or the migration took place in the core, fines were trapped and the pressure was not high enough to overcome the trapping resistance of the particles.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Al Shalabi, E.W.; Sepehrnoori, K.; Delshad, M. Mechanisms Behind Low Salinity Water Flooding in Carbonate Reservoirs. In Proceedings of the SPE Western Regional & AAPG Pacific Section Meeting 2013 Joint Technical Conference, Monterey, CA, USA, 19–25 April 2013. [Google Scholar]
- Smith, K.W. Brines as Flooding Liquids. In Proceedings of the 7th annual technical meetings of the Bradford District Research Group, Penn. State College, PA, USA, 6–7 November 1942. [Google Scholar]
- Lager, A.; Webb, K.J.; Black, C.J. Impact of brine chemistry on oil recovery. In Proceedings of the 14th European Symposium on IOR, Cairo, Egypt, 22–24 April 2007. [Google Scholar]
- Lager, A.K.; Webb, K.J.; Collins, I.R.; Richmond, D.M. LoSalTM enhanced oil recovery: Evidence of enhanced oil recovery at the reservoir scale. In Proceedings of the SPE/DOE Symposium on Improved Oil Recovery, Tulsa, Oklahoma, 20–23 April 2008. [Google Scholar]
- Webb, K.J.; Black, C.J.J.; Edmonds, I.J. Low salinity oil recovery—The role of reservoir condition core floods. In Proceedings of the 13h European Symposium on Improved Oil Recovery, Budapest, Hungary, 25–27 April 2005. [Google Scholar]
- Webb, K.; Lager, A.; Black, C. Comparison of high/low salinity water/oil relative permeability. In Proceedings of the International Symposium of the Society of Core Analysts, Abu Dhabi, UAE, 29 October–2 November 2008. [Google Scholar]
- Tang, G.-Q.; Morrow, N.R. Influence of brine composition and fines migration on crude oil/brine/rock interactions and oil recovery. J. Pet. Sci. Eng. 1999, 24, 99–111. [Google Scholar] [CrossRef]
- Zhang, P.; Tweheyo, M.T.; Austad, T. Wettability alteration and improved oil recovery in chalk: The effect of calcium in the presence of sulfate. Energy & fuels 2006, 20, 2056–2062. [Google Scholar]
- Yousef, A.A.; Al-Saleh, S.; Al-Jawfi, M.S. The impact of the injection water chemistry on oil recovery from carbonate reservoirs. In Proceedings of the SPE EOR Conference at Oil and Gas West Asia, Muscat, Oman, 16–18 April 2012. [Google Scholar]
- Al-Attar, H.H.; Mahmoud, M.Y.; Zekri, A.Y.; Almehaideb, R.; Ghannam, M. Low-salinity flooding in a selected carbonate reservoir: Experimental approach. J. Pet. Explor. Prod. Technol. 2013, 3, 139–149. [Google Scholar] [CrossRef]
- Petrovich, R.; Hamouda, A. Dolomitization of Ekofisk Oil Field Reservoir Chalk by Injected Seawater. In Proceedings of the Ninth International Symposium on Water–Rock Interactions, Taupo, New Zealand, 30 March–3 April 1998. [Google Scholar]
- Hamouda, A.; Pranoto, A. Synergy between Low Salinity Water Flooding and CO2 for Eor in Chalk Reservoirs. In Proceedings of the SPE EOR Conference at Oil and Gas West Asia, Muscat, Oman, 21–23 March 2016; Society of Petroleum Engineers: Richardson, TX, USA. [Google Scholar]
- Hamouda, A.A.; Rezaei Gomari, K.A. Influence of Temperature on Wettability Alteration of Carbonate Reservoirs. In Proceedings of the SPE/DOE Symposium on Improved Oil Recovery, Tulsa, OK, USA, 22–26 April 2006; Society of Petroleum Engineers: Richardson, TX, USA. [Google Scholar]
- Yousef, A.A.; Al-Saleh, S.H.; Al-Kaabi, A.; Al-Jawfi, M.S. Laboratory investigation of the impact of injection-water salinity and ionic content on oil recovery from carbonate reservoirs. SPE Reservoir Eval. Eng. 2011, 14, 578–593. [Google Scholar] [CrossRef]
- Hamouda, A.A.; Valderhaug, O.M. Investigating enhanced oil recovery from sandstone by low-salinity water and fluid/rock interaction. Energy Fuels 2014, 28, 898–908. [Google Scholar] [CrossRef]
- Linke, W.F.; Seidell, A. Solubilities: Inorganic and Metal-Organic Compounds: A Compilation of Solubility Data from the Periodical Literature; American Chemical Society: Washington, DC, USA, 1958. [Google Scholar]
- Nasralla, R.A.; Nasr-El-Din, H.A. Coreflood Study of Low Salinity Water Injection in Sandstone Reservoirs. In Proceedings of the SPE/DGS Saudi Arabia Section Technical Symposium and Exhibition, Al-Khobar, Saudi Arabia, 15–18 May 2011; Society of Petroleum Engineers: Richardson, TX, USA. [Google Scholar]
- Gandomkar, A.; Rahimpour, M.R. Investigation of low-salinity waterflooding in secondary and tertiary enhanced oil recovery in limestone reservoirs. Energy Fuels 2015, 29, 7781–7792. [Google Scholar] [CrossRef]
- Hamouda, A.A.; Maevskiy, E. Oil recovery mechanism (s) by low salinity brines and their interaction with chalk. Energy Fuels 2014, 28, 6860–6868. [Google Scholar] [CrossRef]
- Lehmann, O.; Birnhack, L.; Lahav, O. Design aspects of calcite-dissolution reactors applied for post treatment of desalinated water. Desalination 2013, 314, 1–9. [Google Scholar] [CrossRef]



| Oil Used in Saturation of Cores | Core | Length (cm) | Diameter (cm) | swi (%) | sor (%) | Pore Volume (mL) | Porosity (%) | Flooding Sequence of Brines in the Core |
|---|---|---|---|---|---|---|---|---|
| Model Oil (n-decane + stearic acid, SA) | 1 | 5.92 | 3.78 | 23 | 24.4 | 34.23 | 51.8 | SSW/SO4 (1:10) |
| 2 | 6.01 | 3.78 | 21 | 35.8 | 34.23 | 51.8 | SSW/Mg (1:10) | |
| 4 | 5.95 | 3.78 | 22.3 | 32.2 | 31.94 | 50.12 | SSW/SO4 (1:50) | |
| 7 | 6.00 | 3.78 | 21.8 | 40.2 | 34.8 | 52.22 | SSW/Mg+Na (1:10) | |
| 8 | 6.00 | 3.78 | 28.5 | 23.1 | 33 | 50.99 | SSW/Mg (1:10) at 90 °C | |
| Crude Oil X | 5 | 6.008 | 3.78 | 19.01 | 32.5 | 32.50 | 50.55 | SSW/LSW (1:10) |
| 6 | 6.00 | 3.78 | 21.1 | 38.4 | 34.50 | 52.04 | SSW/LSW (1:50) |
| Property | Temperature | Model Oil (n-decane + 0.005M SA) | Crude Oil X |
|---|---|---|---|
| Density (g/cc) | 20 °C | 0.73 | 0.7827 |
| 50 °C | 0.705 | 0.7009 | |
| 70 °C | 0.67 | 0.7537 | |
| Dynamic Viscosity (cP) | 70 °C | 0.41 | 0.4976 |
| Components | Mole Fraction |
|---|---|
| i-C5 | 1.79 × 10−5 |
| n-C5 | 0.000117 |
| C6 | 0.002371 |
| C7 | 0.013287 |
| C8 | 0.039608 |
| C9 | 0.062886 |
| C10 | 0.881712 |
| Ions/Brine | SSW | LSW 1:10 | LSW 1:50 | Mg+Na | SO42− Brine | SO42− Brine | Mg2+ Brine |
|---|---|---|---|---|---|---|---|
| (mole/L) | (mole/L) | (mole/L) | 1 to 10 | 1 to 10 | 1 to 50 | 1 to 10 | |
| (mole/L) | (mole/L) | (mole/L) | (mole/L) | ||||
| HCO3− | 0.002 | 0.0002 | 0.00004 | ||||
| Cl− | 0.525 | 0.0525 | 0.0105 | 0.0493 | 0.009 | ||
| SO42− | 0.024 | 0.0024 | 0.00048 | 0.0024 | 0.00048 | ||
| Mg+2 | 0.045 | 0.0045 | 0.0009 | 0.0045 | 0.0045 | ||
| Ca+2 | 0.013 | 0.0013 | 0.00026 | ||||
| Na+ | 0.45 | 0.045 | 0.009 | 0.0403 | 0.0046 | 0.00092 | |
| K+ | 0.01 | 0.001 | 0.0002 | ||||
| TDS (ppm) | 33,388 | 3338.8 | 667.76 | 2785 | 336.2 | 67.24 | 423 |
| TDS (g/L) | 33.33 | 3.33 | 0.667 | 2.78 | 0.336 | 0.067 | 0.423 |
| Ionic Strength | 0.657 | 0.0657 | 0.01314 | 0.0538 | 0.007 | 0.0014 | 0.0135 |
| pH | 7.83 | 7.32 | 6.74 | 5.85 | 7.12 | 6.74 | 6.11 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hamouda, A.A.; Gupta, S. Enhancing Oil Recovery from Chalk Reservoirs by a Low-Salinity Water Flooding Mechanism and Fluid/Rock Interactions. Energies 2017, 10, 576. https://doi.org/10.3390/en10040576
Hamouda AA, Gupta S. Enhancing Oil Recovery from Chalk Reservoirs by a Low-Salinity Water Flooding Mechanism and Fluid/Rock Interactions. Energies. 2017; 10(4):576. https://doi.org/10.3390/en10040576
Chicago/Turabian StyleHamouda, Aly A., and Sachin Gupta. 2017. "Enhancing Oil Recovery from Chalk Reservoirs by a Low-Salinity Water Flooding Mechanism and Fluid/Rock Interactions" Energies 10, no. 4: 576. https://doi.org/10.3390/en10040576







