Development of Heterogeneous Catalysts for Thermo-Chemical Conversion of Lignocellulosic Biomass
Abstract
:1. Introduction
2. The Role of Catalysts in Bio-Oil Production
2.1. Application of Zeolites and Mesoporous Materials
2.2. Modification of Zeolites and Mesoporous Materials by the Addition of Metals
2.3. Application of Metal Oxides and Supported Catalysts
2.4. Bio-Oil Upgrading via Hydrodeoxygenation (HDO)
3. Catalysts for the Production of a Hydrogen-Rich Gas from Lignocellulosic Biomass
4. Summary
5. Perspectives on the High-Temperature Conversion of Lignocellulosic Feedstock
Author Contributions
Conflicts of Interest
References
- Bridgwater, A.V. Review of fast pyrolysis of biomass and product upgrading. Biomass Bioenergy 2012, 38, 68–94. [Google Scholar] [CrossRef]
- Yildiz, G.; Ronsse, F.; van Duren, E.; Prins, W. Challenges in the design and operation of processes for catalytic fast pyrolysis of woody biomass. Renew. Sustain. Energy Rev. 2016, 57, 1596–1610. [Google Scholar] [CrossRef]
- Galadima, A.; Muraza, O. In situ fast pyrolysis of biomass with zeolite catalysts for bioaromatics/gasoline production: A review. Energy Convers. Manag. 2015, 105, 338–354. [Google Scholar] [CrossRef]
- Besson, M.; Gallezot, P.; Pinel, C. Conversion of biomass into chemicals over metal catalysts. Chem. Rev. 2014, 114, 1827–1870. [Google Scholar] [CrossRef] [PubMed]
- Bulushev, D.A.; Ross, J.R.H. Catalysis for conversion of biomass to fuels via pyrolysis and gasification: A review. Catal. Today 2011, 171, 1–13. [Google Scholar] [CrossRef]
- Venderbosch, R.H. A critical view on catalytic pyrolysis of biomass. ChemSusChem 2015, 8, 1306–1316. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Kim, K.H.; Brown, R.C. Catalytic pyrolysis of individual components of lignocellulosic biomass. Green Chem. 2014, 16, 727–735. [Google Scholar] [CrossRef]
- French, R.; Czernik, S. Catalytic pyrolysis of biomass for biofuels production. Fuel Process. Technol. 2010, 91, 25–32. [Google Scholar] [CrossRef]
- Jae, J.; Tompsett, G.A.; Foster, A.J.; Hammond, K.D.; Auerbach, S.M.; Lobo, R.F.; Huber, G.W. Investigation into the shape selectivity of zeolite catalysts for biomass conversion. J. Catal. 2011, 279, 257–268. [Google Scholar] [CrossRef]
- Asadieraghi, M.; Daud, W.M.A.W.; Abbas, H.F. Heterogeneous catalysts for advanced bio-fuel production through catalytic biomass pyrolysis vapor upgrading: A review. RSC Adv. 2015, 5, 22234–22255. [Google Scholar] [CrossRef]
- Resende, F.L.P. Recent advances on fast hydropyrolysis of biomass. Catal. Today 2016, 269, 148–155. [Google Scholar] [CrossRef]
- Zhang, X.; Lei, H.; Chen, S.; Wu, J. Catalytic co-pyrolysis of lignocellulosic biomass with polymers: A critical review. Green Chem. 2016, 18, 4145–4169. [Google Scholar] [CrossRef]
- Hassan, H.; Lim, J.K.; Hameed, B.H. Recent progress on biomass co-pyrolysis conversion into high-quality bio-oil. Bioresource Technol. 2016, 221, 645–655. [Google Scholar] [CrossRef] [PubMed]
- Stefanidis, S.D.; Kalogiannis, K.G.; Iliopoulou, E.F.; Lappas, A.A.; Pilavachi, P.A. In-situ upgrading of biomass pyrolysis vapors: Catalyst screening on a fixed bed reactor. Bioresource Technol. 2011, 102, 8261–8267. [Google Scholar] [CrossRef] [PubMed]
- Zheng, A.; Zhao, Z.; Chang, S.; Huang, Z.; Wu, H.; Wang, X.; He, F.; Li, H. Effect of crystal size of ZSM-5 on the aromatic yield and selectivity from catalytic fast pyrolysis of biomass. J. Mol. Catal. A 2014, 383–384, 23–30. [Google Scholar] [CrossRef]
- Li, J.; Li, X.; Zhou, G.; Wang, W.; Wang, C.; Komarneni, S.; Wang, Y. Catalytic fast pyrolysis of biomass with mesoporous ZSM-5 zeolites prepared by desilication with NaOH solutions. Appl. Catal. A 2014, 470, 115–122. [Google Scholar] [CrossRef]
- Hoff, T.C.; Gardner, D.W.; Thilakaratne, R.; Wang, K.; Hansen, T.W.; Brown, R.C.; Tessonnier, J.P. Tailoring ZSM-5 zeolites for the fast pyrolysis of biomass to aromatic hydrocarbons. ChemSusChem. 2016, 9, 1473–1482. [Google Scholar] [CrossRef] [PubMed]
- Engtrakul, Ch.; Mukarakate, C.; Starace, A.K.; Magrini, K.A.; Rogers, A.K.; Yung, M.M. Effect of ZSM-5 acidity on aromatic product selectivity during upgrading of pine pyrolysis vapors. Catal. Today 2016, 269, 175–181. [Google Scholar] [CrossRef]
- Fischer, A.; Du, S.; Valla, J.A.; Bollas, G.M. The effect of temperature, heating rate, and ZSM-5 catalyst on the product selectivity of the fast pyrolysis of spent coffee grounds. RSC Adv. 2015, 5, 29252–29261. [Google Scholar] [CrossRef]
- Zhang, B.; Zhong, Z.; Min, M.; Ding, K.; Xie, Q.; Ruan, R. Catalytic fast co-pyrolysis of biomass and food waste to produce aromatics: Analytical Py–GC/MS study. Bioresour. Technol. 2015, 189, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Lei, H.; Zhu, X.; Qian, M.; Yadavalli, G.; Wu, J.; Chen, S. Thermal behavior and kinetic study for catalytic co-pyrolysis of biomass with plastics. Bioresour. Technol. 2016, 220, 233–238. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.-S.; Kim, Y.-M.; Jae, J.; Watanabe, Ch.; Kim, S.; Jung, S.-Ch.; Kim, S.-Ch.; Park, Y.-K. Pyrolysis and catalytic upgrading of Citrus unshiu peel. Bioresour. Technol. 2015, 194, 312–319. [Google Scholar] [CrossRef] [PubMed]
- Karagöz, S.; Kawakami, T.; Kako, A.; Iiguni, Y.; Ohtani, H. Single shot pyrolysis and on-line conversion of lignocellulosic biomass with HZSM-5 catalyst using tandem micro-reactor-GC-MS. RSC Adv. 2016, 6, 46108–46115. [Google Scholar] [CrossRef]
- Stefanidis, S.D.; Kalogiannis, K.G.; Pilavachi, P.A.; Fougret, Ch.M.; Jordan, E.; Lappas, A.A. Catalyst hydrothermal deactivation and metal contamination during the in situ catalytic pyrolysis of biomass. Catal. Sci. Technol. 2016, 6, 2807–2819. [Google Scholar] [CrossRef]
- Zhang, B.; Zhong, Z.; Chen, P.; Ruan, R. Microwave-assisted catalytic fast pyrolysis of biomass for bio-oil production using chemical vapor deposition modified HZSM-5 catalyst. Bioresour. Technol. 2015, 197, 79–84. [Google Scholar] [CrossRef] [PubMed]
- Kelkar, S.; Saffron, Ch.M.; Andreassi, K.; Li, Z.; Murkute, A.; Miller, D.J.; Pinnavaia, T.J.; Kriegel, R.M. A survey of catalysts for aromatics from fast pyrolysis of biomass. Appl. Catal. B 2015, 174–175, 85–95. [Google Scholar] [CrossRef]
- Imran, A.; Bramer, E.A.; Seshan, K.; Brem, G. Catalytic flash pyrolysis of biomass using different types of zeolite and online vapor fractionation. Energies 2016, 9, 187. [Google Scholar] [CrossRef]
- Yoo, M.L.; Park, Y.H.; Park, Y.-K.; Park, S.H. Catalytic pyrolysis of wild reed over a zeolite-based waste catalyst. Energies 2016, 9, 201. [Google Scholar] [CrossRef]
- Custodis, V.B.F.; Karakoulia, S.A.; Triantafyllidis, K.S.; van Bokhoven, J.A. Catalytic fast pyrolysis of lignin over high-surface-area mesoporous aluminosilicates: Effect of porosity and acidity. ChemSusChem 2016, 9, 1134–1145. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Li, D.; Yang, H.; Wang, X.; Chen, H. Effects of Fe-, Zr-, and Co-modified zeolites and pretreatments on catalytic upgrading of biomass fast pyrolysis vapors. Energy Fuels 2016, 30, 3004–3013. [Google Scholar] [CrossRef]
- Mullen, C.A.; Boateng, A.A. Production of aromatic hydrocarbons via catalytic pyrolysis of biomass over Fe-modified HZSM-5 zeolites. ACS Sustain. Chem. Eng. 2015, 3, 1623–1631. [Google Scholar] [CrossRef]
- Cheng, Y.; Wang, Z.; Gilbert, G.J.; Fan, W.; Huber, G.W. Production of p-xylene from biomass by catalytic fast pyrolysis using ZSM-5 catalysts with reduced pore openings. Angew. Chem. Int. Ed. 2012, 51, 11097–11100. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Zhang, X.; Chen, L.; Zhao, B.; Yang, S.; Xie, X. Comparison of catalytic fast pyrolysis of biomass to aromatic hydrocarbons over ZSM-5 and Fe/ZSM-5 catalysts. J. Anal. Appl. Pyrol. 2016, 121, 342–346. [Google Scholar] [CrossRef]
- Veses, A.; Puértolas, B.; Callén, M.S.; García, T. Catalytic upgrading of biomass derived pyrolysis vapors over metal-loaded ZSM-5 zeolites: Effect of different metal cations on the bio-oil final properties. Microporous Mesoporous Mater. 2015, 209, 189–196. [Google Scholar] [CrossRef]
- Yung, M.M.; Starace, A.K.; Mukarakate, C.; Crow, A.M.; Leshnov, M.A.; Magrini, K.A. Biomass catalytic pyrolysis on Ni/ZSM-5: Effects of nickel pretreatment and loading. Energy Fuels 2016, 30, 5259–5268. [Google Scholar] [CrossRef]
- Melligan, F.; Hayes, M.H.B.; Kwapinski, W.; Leahy, J.J. A study of hydrogen pressure during hydropyrolysis of Miscanthus x giganteus and online catalytic vapour upgrading with Ni on ZSM-5. J. Anal. Appl. Pyrol. 2013, 103, 369–377. [Google Scholar] [CrossRef]
- Melligan, F.; Hayes, M.H.B.; Kwapinski, W.; Leahy, J.J. Hydro-pyrolysis of biomass and online catalytic vapor upgrading with Ni-ZSM-5 and Ni-MCM-41. Energy Fuels 2012, 26, 6080–6090. [Google Scholar] [CrossRef]
- Yao, W.; Li, J.; Feng, Y.; Wang, W.; Zhang, X.; Chen, Q.; Komarneni, S.; Wang, Y. Thermally stable phosphorus and nickel modified ZSM-5 zeolites for catalytic co-pyrolysis of biomass and plastics. RSC Adv. 2015, 5, 30485–30494. [Google Scholar] [CrossRef]
- Widayatno, W.B.; Guan, G.; Rizkiana, J.; Yang, J.; Hao, X.; Tsutsumi, A.; Abudula, A. Upgrading of bio-oil from biomass pyrolysis over Cu-modified β-zeolite catalyst with high selectivity and stability. Appl. Catal. B 2016, 186, 166–172. [Google Scholar] [CrossRef]
- Karnjanakom, S.; Guan, G.; Asep, B.; Hao, X.; Kongparakul, S.; Samart, Ch.; Abudula, A. Catalytic upgrading of bio-oil over Cu/MCM-41 and Cu/KIT-6 prepared by β-cyclodextrin-assisted coimpregnation method. J. Phys. Chem. C 2016, 120, 3396–3407. [Google Scholar] [CrossRef]
- Kim, B.-S.; Jeong, C.S.; Kim, J.M.; Park, S.B.; Park, S.H.; Jeon, J.-K.; Jung, S.-C.; Kim, S.C.; Park, Y.-K. Ex situ catalytic upgrading of lignocellulosic biomass components over vanadium contained H-MCM-41 catalysts. Catal. Today 2016, 265, 184–191. [Google Scholar] [CrossRef]
- Budhi, S.; Mukarakate, C.; Iisa, K.; Pylypenko, S.; Ciesielski, P.N.; Yung, M.M.; Donohoe, B.S.; Katahira, R.; Nimlos, M.R.; Trewyn, B.G. Molybdenum incorporated mesoporous silica catalyst for production of biofuels and value-added chemicals via catalytic fast pyrolysis. Green Chem. 2015, 17, 3035–3046. [Google Scholar] [CrossRef]
- Yang, Z.; Kumar, A.; Apblett, A. Integration of biomass catalytic pyrolysis and methane aromatization over Mo/HZSM-5 catalysts. J. Anal. Appl. Pyrol. 2016, 120, 484–492. [Google Scholar] [CrossRef]
- Mahadevan, R.; Adhikari, S.; Shakya, R.; Wang, K.; Dayton, D.; Lehrich, M.; Taylor, S.E. Effect of alkali and alkaline earth metals on in-situ catalytic fast pyrolysis of lignocellulosic biomass: A microreactor study. Energy Fuels 2016, 30, 3045–3056. [Google Scholar] [CrossRef]
- Zhu, Ch.; Maduskar, S.; Paulsen, A.D.; Dauenhauer, P.J. Alkaline-earth-metal-catalyzed thin-film pyrolysis of cellulose. ChemCatChem 2016, 8, 818–829. [Google Scholar] [CrossRef]
- Nguyen, T.S.; He, S.; Raman, G.; Seshan, K. Catalytic hydro-pyrolysis of lignocellulosic biomass over dual Na2CO3/Al2O3 and Pt/Al2O3 catalysts using n-butane at ambient pressure. Chem. Eng. J. 2016, 299, 415–419. [Google Scholar] [CrossRef]
- Karnjanakom, S.; Guan, G.; Asep, B.; Du, X.; Hao, X.; Yang, J.; Samart, Ch.; Abudula, A. A green method to increase yield and quality of biooil: Ultrasonic pretreatment of biomass and catalytic upgrading of bio-oil over metal (Cu, Fe and/or Zn)/γ-Al2O3. RCS Adv. 2015, 5, 83494–83503. [Google Scholar]
- Karnjanakom, S.; Bayu, A.; Xiaoketi, P.; Hao, X.; Kongparakul, S.; Samart, Ch.; Abudula, A.; Guan, G. Selective production of aromatic hydrocarbons from catalytic pyrolysis of biomass over Cu or Fe loaded mesoporous rod-like alumina. RCS Adv. 2016, 6, 50618–50629. [Google Scholar] [CrossRef]
- Mante, O.F.; Rodriguez, J.A.; Senanayake, S.D.; Babu, S.P. Catalytic conversion of biomass pyrolysis vapors into hydrocarbon fuel precursors. Green Chem. 2015, 17, 2362–2368. [Google Scholar] [CrossRef]
- Grams, J.; Niewiadomski, M.; Ruppert, A.M.; Kwapiński, W. Influence of Ni catalyst support on the product distribution of cellulose fast pyrolysis vapors upgrading. J. Anal. Appl. Pyrol. 2015, 113, 557–563. [Google Scholar] [CrossRef]
- Grams, J.; Niewiadomski, M.; Ruppert, A.M.; Kwapiński, W. Catalytic performance of Ni catalyst supported on CeO2, ZrO2 and CeO2-ZrO2 in the upgrading of cellulose fast pyrolysis vapors. C. R. Chim. 2015, 18, 1223–1228. [Google Scholar] [CrossRef]
- Lazdovica, K.; Liepina, L.; Kampars, V. Catalytic pyrolysis of wheat bran for hydrocarbons production in the presence of zeolites and noble-metals by using TGA-FTIR method. Bioresour. Technol. 2016, 207, 126–133. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Kim, Y.-M.; Lee, I.-G.; Jeon, J.-K.; Jung, S.-Ch.; Chung, J.D.; Choi, W.G.; Park, Y.-K. Recent advances in the catalytic hydrodeoxygenation of bio-oil. Korean J. Chem. Eng. 2016, 33, 3299–3315. [Google Scholar] [CrossRef]
- Sanna, A.; Vispute, T.P.; Huber, G.W. Hydrodeoxygenation of the aqueous fraction of bio-oil with Ru/C and Pt/C catalysts. Appl. Catal. B 2015, 165, 446–456. [Google Scholar] [CrossRef]
- Nolte, M.W.; Shanks, B.H. A perspective on catalytic strategies for deoxygenation in biomass pyrolysis. Energy Technol. 2017, 5, 7–18. [Google Scholar] [CrossRef]
- Robinson, A.M.; Hensley, J.E.; Medlin, J.W. Bifunctional catalysts for upgrading of biomass-derived oxygenates: A review. ACS Catal. 2016, 6, 5026–5043. [Google Scholar] [CrossRef]
- Routray, K.; Barnett, K.J.; Huber, G.W. Hydrodeoxygenation of pyrolysis oils. Energy Technol. 2017, 5, 80–93. [Google Scholar] [CrossRef]
- Pucher, H.; Schwaiger, N.; Feiner, R.; Ellmaier, L.; Pucher, P.; Chernev, B.S.; Siebenhofer, M. Biofuels from liquid phase pyrolysis oil: A two-step hydrodeoxygenation (HDO) process. Green Chem. 2015, 17, 1291–1298. [Google Scholar] [CrossRef]
- Nelson, R.C.; Baek, B.; Ruiz, P.; Goundie, B.; Brooks, A.; Wheeler, M.C.; Frederick, B.G.; Grabow, L.C.; Austin, R.N. Experimental and theoretical insights into the hydrogen-efficient direct hydrodeoxygenation mechanism of phenol over Ru/TiO2. ACS Catal. 2015, 5, 6509–6523. [Google Scholar] [CrossRef]
- Xu, G.-Y.; Guo, J.-H.; Qu, Y.-C.; Zhang, Y.; Fu, Y.; Guo, Q.-X. Selective hydrodeoxygenation of lignin-derived phenols to alkyl cyclohexanols over a Ru-solid base bifunctional catalyst. Green Chem. 2016, 18, 5510–5517. [Google Scholar] [CrossRef]
- De Souza, P.M.; Rabelo-Neto, R.C.; Borges, L.E.P.; Jacobs, G.; Davis, B.H.; Graham, U.M.; Resasco, D.E.; Noronha, F.B. Effect of zirconia morphology on hydrodeoxygenation of phenol over Pd/ZrO2. ACS Catal. 2015, 5, 7385–7398. [Google Scholar] [CrossRef]
- Hellinger, M.; Carvalho, H.W.P.; Baier, S.; Wang, D.; Kleist, W.; Grunwaldt, J.-D. Catalytic hydrodeoxygenation of guaiacol over platinum supported on metal oxides and zeolites. Appl. Catal. A 2015, 490, 181–192. [Google Scholar] [CrossRef]
- Sullivan, M.W.; Bhan, A. Acetone hydrodeoxygenation over bifunctional metallic-acidic molybdenum carbide catalysts. ACS Catal. 2016, 6, 1145–1152. [Google Scholar] [CrossRef]
- Foo, G.S.; Rogers, A.K.; Yung, M.M.; Sievers, C. Steric Effect and evolution of surface species in the hydrodeoxygenation of bio-oil model compounds over Pt/HBEA. ACS Catal. 2016, 6, 1292–1307. [Google Scholar] [CrossRef]
- Lu, B.; Li, J.; Lv, G.; Qi, Y.; Wang, Y.; Deng, T.; Hou, X.; Yang, Y. Obtaining a high value branched bio-alkane from biomass-derived levulinic acid using RANEY® as hydrodeoxygenation catalyst. RSC Adv. 2016, 6, 93956–93962. [Google Scholar] [CrossRef]
- Ayodele, O.O.; Dawodu, F.A.; Yan, D.; Lu, X.; Xin, J.; Zhang, S. Hydrodeoxygenation of angelica lactone dimers and trimers over silica-alumina supported nickel catalyst. Renew. Energy 2016, 86, 943–948. [Google Scholar] [CrossRef]
- Ni, M.; Leung, D.Y.C.; Leung, M.K.H.; Sumathy, K. An overview of hydrogen production from biomass. Fuel Proc. Technol. 2006, 87, 461–472. [Google Scholar] [CrossRef]
- Saxena, R.C.; Seal, D.; Kumar, S.; Goyal, H.B. Thermo-chemical routes for hydrogen rich gas from biomass: A review. Renew. Sustain. Energy Rev. 2008, 12, 1909–1927. [Google Scholar] [CrossRef]
- Balat, M.; Balat, M.; Kirtay, E.; Balat, H. Main routes for the thermo-conversion of biomass into fuels and chemicals. Part 1: Pyrolysis systems. Energy Convers. Manag. 2009, 50, 3147–3157. [Google Scholar] [CrossRef]
- Tanksale, A.; Beltramini, J.N.; Lu, G.M. A review of catalytic hydrogen production processes from biomas. Renew. Sustain. Energy Rev. 2010, 14, 166–182. [Google Scholar] [CrossRef]
- Grams, J.; Potrzebowska, N.; Goscianska, J.; Michalkiewicz, B.; Ruppert, A.M. Mesoporous silicas as supports for Ni catalyst used in cellulose conversion to hydrogen rich gas. Int. J. Hydrogen Energy 2016, 41, 8656–8667. [Google Scholar] [CrossRef]
- Cheah, S.; Jablonski, W.S.; Olstad, J.L.; Carpenter, D.L.; Barthelemy, K.D.; Robichaud, D.J.; Andrews, J.C.; Black, S.K.; Oddo, M.D.; Westover, T.L. Effects of thermal pretreatment and catalyst on biomass gasification efficiency and syngas composition. Green Chem. 2016, 18, 6291–6304. [Google Scholar] [CrossRef]
- Arregi, A.; Lopez, G.; Amutio, M.; Barbarias, I.; Bilbao, J.; Olazar, M. Hydrogen production from biomass by continuous fast pyrolysis and in-line steam reforming. RSC Adv. 2016, 6, 25975–25985. [Google Scholar] [CrossRef]
- Esfahani, R.A.M.; Osmieri, L.; Specchia, S.; Yusup, S.; Tavasoli, A.; Zamaniyan, A. H2-rich syngas production through mixed residual biomass and HDPE waste via integrated catalytic gasification and tar cracking plus bio-char upgrading. Chem. Eng. J. 2017, 308, 578–587. [Google Scholar] [CrossRef]
- Kumagai, S.; Alvarez, J.; Blanco, P.H.; Wu, Ch.; Yoshioka, T.; Olazar, M.; Williams, P.T. Novel Ni–Mg–Al–Ca catalyst for enhanced hydrogen production for the pyrolysis–gasification of a biomass/plastic mixture. J. Anal. Appl. Pyrol. 2015, 113, 15–21. [Google Scholar] [CrossRef]
- Chen, F.; Wu, Ch.; Dong, L.; Vassallo, A.; Williams, P.T.; Huang, J. Characteristics and catalytic properties of Ni/CaAlOx catalyst for hydrogen-enriched syngas production from pyrolysis-steam reforming of biomass sawdust. Appl. Catal. B 2016, 183, 168–175. [Google Scholar] [CrossRef]
- Dong, L.; Wu, Ch.; Ling, H.; Shi, J.; Williams, P.T.; Huang, J. Promoting hydrogen production and minimizing catalyst deactivation from the pyrolysis-catalytic steam reforming of biomass on nanosized NiZnAlOx catalysts. Fuel 2017, 188, 610–620. [Google Scholar] [CrossRef]
- Matras, J.; Niewiadomski, M.; Ruppert, A.; Grams, J. Activity of Ni catalysts for hydrogen production via biomass pyrolysis. Kinet. Catal. 2012, 53, 565–569. [Google Scholar] [CrossRef]
- Ruppert, A.; Niewiadomski, M.; Grams, J.; Kwapiński, W. Optimization of Ni/ZrO2 catalytic performance in thermochemical cellulose conversion for enhanced hydrogen production. Appl. Catal. B 2014, 145, 85–90. [Google Scholar] [CrossRef]
- Grams, J.; Niewiadomski, M.; Ryczkowski, R.; Ruppert, A.M.; Kwapiński, W. Activity and characterization of Ni catalyst supported on CeO2-ZrO2 for thermo-chemical conversion of cellulose. Int. J. Hydrogen Energy 2016, 41, 8679–8687. [Google Scholar] [CrossRef]
- Shao, S.; Shi, A.; Liu, C.; Yang, R.; Dong, W. Hydrogen production from steam reforming of glycerol over Ni/CeZrO catalysts. Fuel Process. Technol. 2014, 125, 1–7. [Google Scholar] [CrossRef]
- Ryczkowski, R.; Niewiadomski, M.; Michalkiewicz, B.; Skiba, E.; Ruppert, A.M.; Grams, J. Effect of alkali and alkaline earth metals addition on Ni/ZrO2 catalyst activity in cellulose conversion. J. Therm. Anal. Calorim. 2016, 126, 103–110. [Google Scholar] [CrossRef]
- Ryczkowski, R.; Ruppert, A.M.; Przybysz, P.; Chałupka, K.; Grams, J. Hydrogen production from biomass woodchips using Ni/CaO–ZrO2 catalysts. Reacct. Kinet. Mech. Catal. 2017. [Google Scholar] [CrossRef]
- Kang, K.; Shakouri, M.; Azargohar, R.; Dalai, A.K.; Wang, H. Application of Ni-Co/Mg-Al catalyst system for hydrogen production via supercritical water gasification of lignocellulosic biomass. Catal. Lett. 2016, 146, 2596–2605. [Google Scholar] [CrossRef]
- Kang, K.; Azargohar, R.; Dalai, A.K.; Wang, H. Systematic screening and modification of Ni based catalysts for hydrogen generation from supercritical water gasification of lignin. Chem. Eng. J. 2016, 283, 1019–1032. [Google Scholar] [CrossRef]
- Kang, K.; Azargohar, R.; Dalai, A.K.; Wang, H. Hydrogen production from lignin, cellulose and waste biomass via supercritical water gasification: Catalyst activity and process optimization study. Energy Convers. Manag. 2016, 117, 528–537. [Google Scholar] [CrossRef]
- Nanda, S.; Reddy, S.N.; Dalai, A.K.; Kozinski, J.A. Subcritical and supercritical water gasification of lignocellulosic biomass impregnated with Nikel nanocatalyst for hydrogen production. Int. J. Hydrogen Energy 2016, 41, 4907–4921. [Google Scholar] [CrossRef]
- Elif, D.; Neziche, A. Hydrogen production by supercritical water gasification of fruit pulp in the presence of Ru/C. Int. J. Hydrogen Energy 2016, 41, 8073–8083. [Google Scholar] [CrossRef]
- Chen, F.; Wu, Ch.; Dong, L.; Jin, F.; Williams, P.T.; Huang, J. Catalytic steam reforming of volatiles released via pyrolysis of wood sawdust for hydrogen-rich gas production on Fe–Zn/Al2O3 nanocatalysts. Fuel 2015, 158, 999–1005. [Google Scholar] [CrossRef]
- Yang, J.; Kaewpanha, M.; Karnjanakom, S.; Guan, G.; Hao, X.; Abudula, A. Steam reforming of biomass tar over calcined egg shell supported catalysts for hydrogen production. Int. J. Hydrogen Energy 2016, 41, 6699–6705. [Google Scholar] [CrossRef]
- Lang, Ch.; Sécordel, X.; Kinnemann, A.; Courson, C. Water gas shift catalysts for hydrogen production from biomass steam gasification. Fuel Proc. Technol. 2017, 156, 246–252. [Google Scholar] [CrossRef]
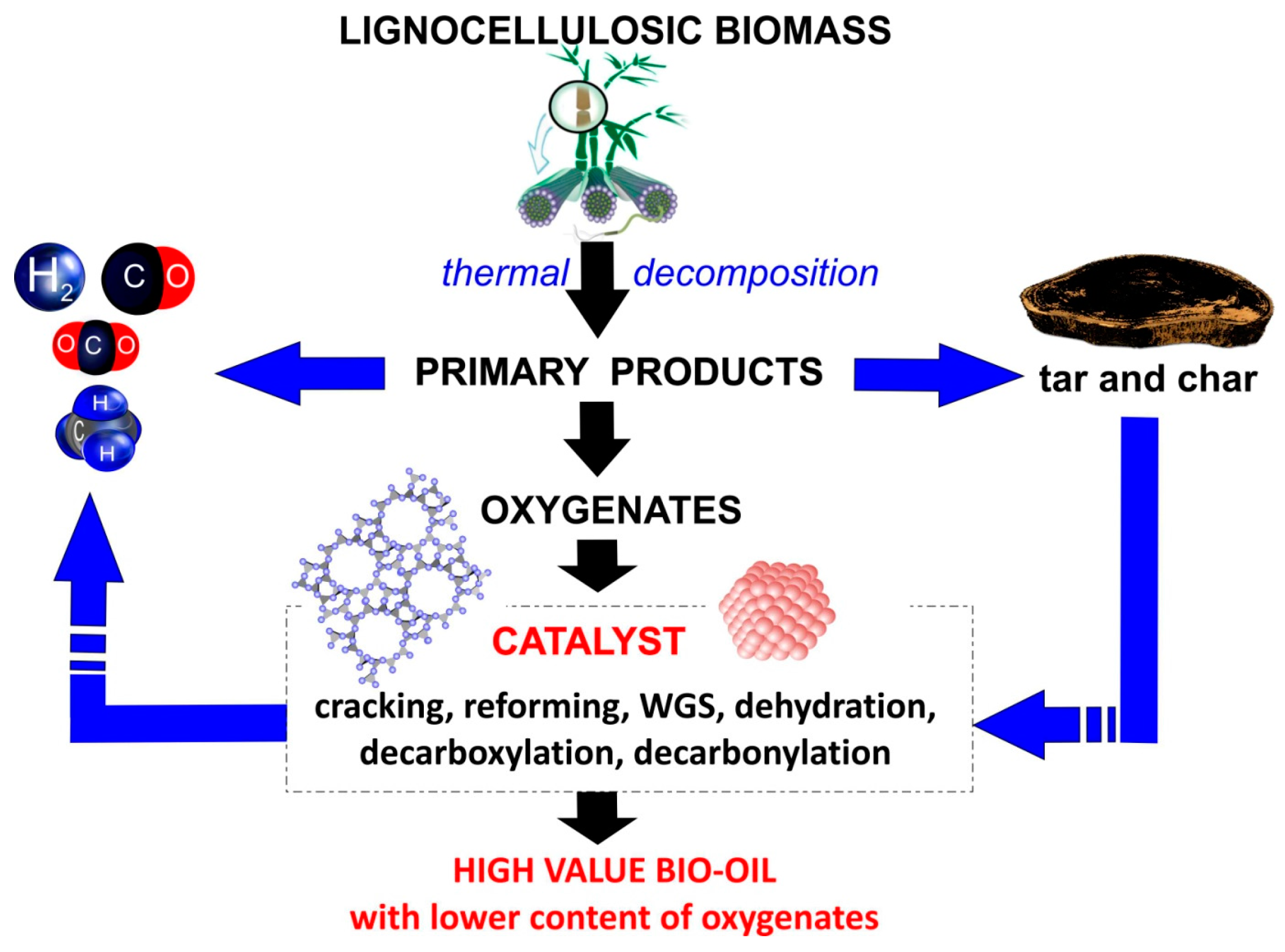
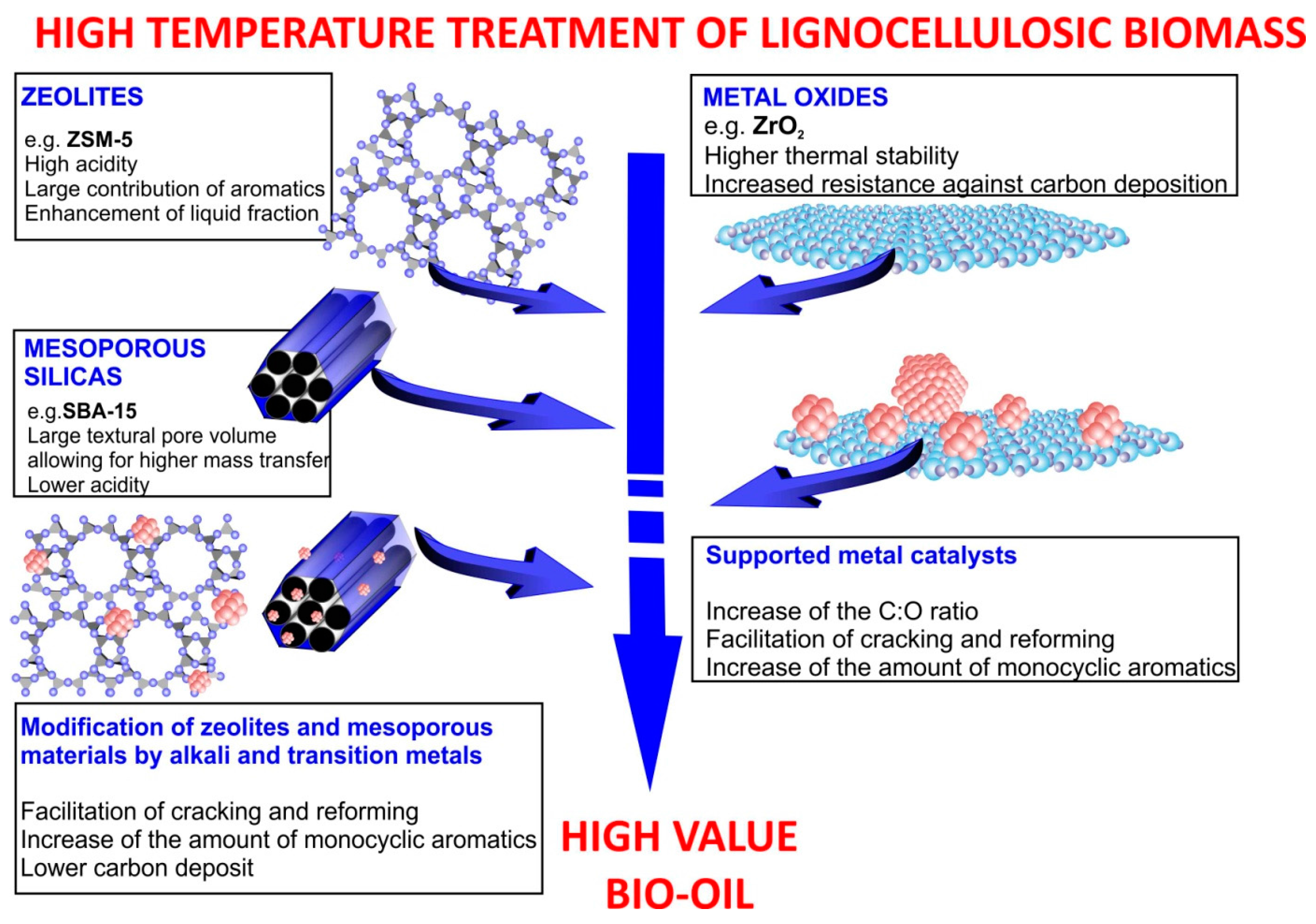
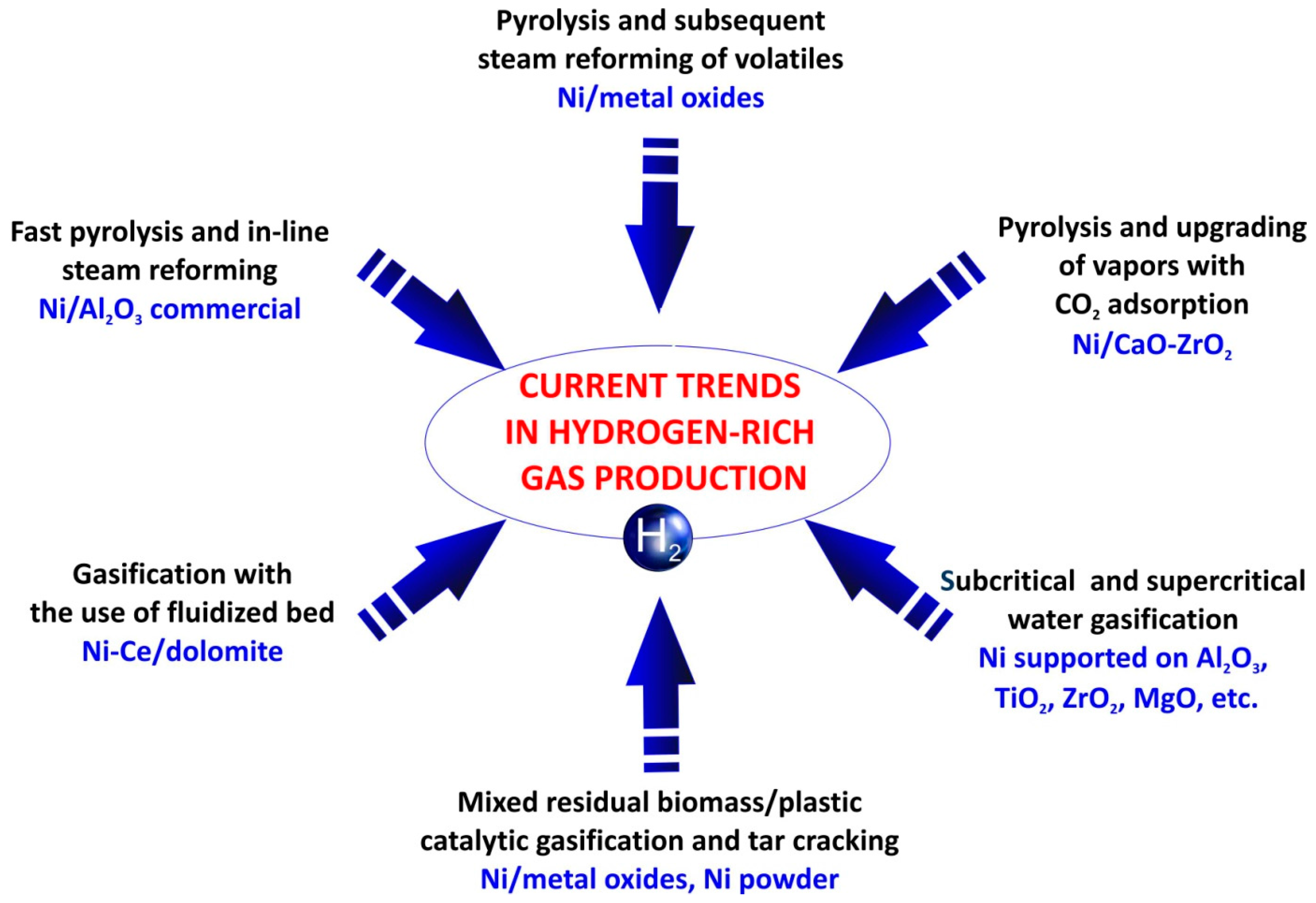
| No. | Catalyst | Feedstock | Products and Remarks Regarding Influence of Catalyst | Conditions of Pyrolysis Process | Reference |
|---|---|---|---|---|---|
| 1 | ZSM-5 | Cellulose, hemicelluloses, lignin | Carbon yields of aromatics from cellulose, hemicelluloses and lignin were 28.8%, 19.4 and 7.4%, respectively. | Micro-furnace pyrolyzer equipped with an autoshot sampler at 400 °C–800 °C, analysis of products by GCMS (Gas Chromatograph coupled with Mass Spectrometer) | [7] |
| 2 | ZSM-5 | Pine, corncob and straw cellulose, hemicellulose and lignin | Fast pyrolysis, the highest yield of aromatics (38.4% for cellulose and 25.4% for pine) was obtained in the presence of catalyst. | Py-GCMS, residence time and heating rate were fixed to 600 °C, 50 s and 20,000 °C/s, catalyst to feedstock weight ratio = 9 | [15] |
| 3 | desilicated ZSM-5 | Beech wood | Fast pyrolysis, desilicated ZSM-5 more active towards production of aromatics than unmodified material (26.2% and 30.2%, respectively) and slightly limits coke formation (from 41.2% to 39.9%). | Py-GCMS, maximal temperature, residence time and heating rate were fixed to 650 °C, 60 s and 20,000 °C/s, catalyst to feedstock weight ratio = 10 | [16] |
| 4 | ZSM-5 | Cellulose | 32% of aromatics in the presence of ZSM-5 with optimized structure | Micro-pyrolyzer (equipped with an autoshot sampler) at 700 °C, catalyst to biomass weight ratio = 20, analysis of products by GCMS | [17] |
| 5 | ZSM-5 | Spent coffee grounds | Considerable increase in the amount of aromatic hydrocarbons in comparison to non-catalyzed process | (a) Py-GCMS and (b) fixed bed reactor (a)—maximal temperature, residence time and heating rate were fixed to 490 °C–590 °C, 10 s and 2000 °C/s, catalyst to feedstock weight ratio = 1 (b)—temperature—490 °C–590 °C, residence time in thermal bed 1.5 s and 2.5 s in catalytic bed, catalyst to feedstock weight ratio = 1 | [19] |
| 6 | ZSM-5 | Corn stalk and food waste | Co-pyrolysis, relative content of aromatics about 30% | Py-GCMS, maximal temperature, residence time and heating rate were fixed to 500 °C–700 °C, 20 s and 20,000 °C/s, catalyst to feedstock weight ratio = 1 | [20] |
| 7 | HZSM-5 | Citrus unshiu peel | Ex situ catalytic pyrolysis, yield of aromatics about 7% | Tandem micro-reactor consisting of two sequential furnaces coupled to GCMS, pyrolysis—500 °C, upgrading—400 °C–600 °C, catalyst to feedstock weight ratio = 1 | [22] |
| 8 | HZSM-5 | Beech wood, cellulose, microalgae | The relative yield of total BTX (benzene, toluene, xylene) was the highest for beech wood among studied feedstocks | Tandem micro-reactor consisting of two sequential furnaces coupled to GCMS, temperature 400 °C–600 °C, catalyst to feedstock weight ratio = 5–20 | [23] |
| 9 | ZSM-5 | Beech wood | Investigations of the influence of various metals contamination on the efficiency of in situ catalytic pyrolysis of biomass | Bubbling fluidized bed pyrolysis reactor, temperature 500 °C, catalyst 150 g, biomass 100 g (fed to the reactor at a rate of 5 g/min) | [24] |
| 10 | HZSM-5 | Corn stover | Microwave-assisted catalytic fast pyrolysis conducted with the presence of HZSM-5 (H form of Zeolite Socony Mobil-5) modified by chemical vapor deposition with TEOS (tetraethyl orthosilicate), relative content of aromatics 38%–39% | Microwave oven containing semi-continuous biomass feeder, temperature 500 °C, HZSM-5 was mixed with SiC particles (used as the microwave absorbent) | [25] |
| 11 | HZSM-5, sulfated zirconia, Al–MSU–S alumnosilicate and bauxite waste | Poplar | High selectivity of HZSM-5 towards aromatics, in the case of mesoporous catalysts their mesoporosity, high pore volume and acidity facilitated cracking of pyrolysis intermediates which led to the production of higher amount of permanent gases and coke | Py-GCMS, maximal temperature, residence time and heating rate were fixed to 650 °C, 20 s and 1000 °C/s, catalyst to feedstock weight ratio = 5 | [26] |
| 12 | HZSM-5, H-Y | Woody biomass (LIGNOCEL) | Flash pyrolysis, HZSM-5 allowed for the more efficient decrease in the contribution of acid fraction and production of considerably higher amount of phenolics in comparison to H-Y | Continuous bench scale unit, temperature 450 °C–550 °C, residence time 2–4 s, catalyst to feedstock weight ratio = 0.1–0.5 | [27] |
| 13 | waste FCC catalyst and H-Y | Wild reed | H-Y zeolite allows for the formation of higher amount of aromatics and a decrease in the content of oxygenates in comparison to regenerated FCC catalyst | Batch-type reactor, temperature 500 °C, residence time 14–20 s, catalyst to feedstock weight ratio = 1–10 | [28] |
| 14 | Al-MCM-41, Al-SBA-15 Al-MSU-J and HZSM-5 | Lignin | Al-MCM-41 (Mobil Composition of Matter No. 41) possessing large pore volume and allowing for enhanced mass transfer enabled formation of similar amount of aromatics to that obtained in the case of H-ZSM-5 having noticeably higher acidity | 650 °C at heating rate 20,000 °C/s, reaction time 20 s, catalyst to feedstock weight ratio = 4 | [29] |
| No. | Catalyst | Feedstock | Products and Remarks Regarding Influence of Catalyst | Conditions of Pyrolysis Process | Reference |
|---|---|---|---|---|---|
| 1 | HZSM-5 modified by Fe, Zr and Co | Pine sawdust | Zr/HZSM-5 and Fe/HZSM-5 catalysts promoted formation of aromatic hydrocarbons, the content of aromatics observed for these two materials was noticeably higher (43%–45%) in comparison to that obtained in the presence of Co/HZSM-5 and unmodified HZSM-5 (23% and 33%, respectively). | Two-stage fixed-bed pyrolysis system with continuous feeding, temperature 550 °C, catalyst to feedstock weight ratio = 0.5 | [30] |
| 2 | HZSM-5 modified by Fe | Cellulose, cellobiose, lignin, and switchgrass | Fe/HZSM-5 catalyst favored formation of benzene and naphthalene and inhibited production of p-xylene, ethylbenzene and trimethylbenzene in comparison to unmodified zeolite. | Py-GCMS, maximal temperature and residence time were fixed to 500 °C, 30 s, catalyst to feedstock weight ratio = 5–10 | [31] |
| 3 | Fe/ZSM-5 | Wood sawdust | Fe/ZSM-5 increased efficiency of the production of monocyclic aromatic hydrocarbons and decreased the amount of the formed polycyclic aromatic hydrocarbons in comparison to ZSM-5. | Py-GCMS, maximal temperature, residence time and heating rate were fixed to 600 °C, 25 s and 20,000 °C/s, catalyst to feedstock weight ratio = 10 | [33] |
| 4 | ZSM-5 modified by Ni, Mg, Cu < Ga and Sn | Woody biomass | Ni-ZSM-5 and Ga-ZSM-5 materials led to the formation of the highest amount of haromatics. | Fixed bed reactor, temperature 450 °C | [34] |
| 5 | Ni/ZSM-5 | Pine | Modification of ZSM-5 by Ni resulted in the increase in the amount of aromatics and enhanced conversion of oxygenates in comparison to unmodified material. | Bench-scale, semi-batch reactor system, temperature 500 °C, 35.5 mg of biomass per 2.5 min was inserted into the pyrolysis zone containing 500 mg of catalyst | [35] |
| 6 | Ni/ZSM-5 | Miscanthus | Hydropyrolysis, both hydrogen pressure and the presence of catalyst leaded to the increase in the amount of saturated hydrocarbons. | Py-GCMS, maximal temperature, residence time and heating rate were fixed to 600 °C, 20 s and 20,000 °C/s, catalyst to feedstock weight ratio = 3.3 | [36] |
| 7 | ZSM-5, Ni/ZSM-5, MCM-41, Ni/MCM-41 | Miscanthus, scots pine, mahogany | Fast pyrolysis of biomass pyrolysis vapors, the presence of catalysts increased content of aromatics and lighter phenols. | Py-GCMS, maximal temperature, residence time and heating rate were fixed to 600 °C, 20 s and 20,000 °C/s, catalyst to feedstock weight ratio = 3.3 | [37] |
| 8 | ZSM-5 modified by Ni/P | Pine and low-density polyethylene | Co-pyrolysis, modification of zeolite results in the production of increased amount of olefins and valuable aromatics. Moreover, increased hydrothermal stability of the catalyst and resistance against carbon deposit formation is observed. | Py-GCMS, maximal temperature, residence time and heating rate were fixed to 650 °C, 60 s, catalyst to feedstock weight ratio = 15 | [38] |
| 9 | B-zeolite modified by Cu | Japanese knotweed | Introduction of copper promoted selectivity to hydrocarbons. | Down-draft fixed bed reactor, temperature 600 °C, heating rate 80 °C/min, duration 30 min, catalyst to feedstock weight ratio = 3.3 | [39] |
| 10 | Cu/MCM-41 and Cu/KIT-6 | Cedar wood | Catalysts prepared by cyclodextrin-assisted co-impregnation method were more active than the materials synthesized via impregnation. Cu/KIT-6 catalyst was more active in the formation of monocyclic aromatic compounds than Cu/MCM-41 sample. | Fixed bed reactor, reaction temperature, reaction time, and heating rate were fixed at 565 °C, 4 min, and 1000 °C/min, catalyst to feedstock weight ratio = 6 | [40] |
| 11 | V-MCM-41 | Commercial cellulose, xylan kraft lignin and levoglucosan | Ex-situ catalytic pyrolysis, H-V-MCM-41 showed highest catalytic activity for the production of valuable furanic compounds. | Py-GCMS for 3 min at 500 °C, catalyst to feedstock weight ratio = 1 | [41] |
| 12 | Mo/KIT-5 | Pine, lignin and cellulose | Fast pyrolysis, catalyst highly selective for the production of furans. | Horizontal quartz annular flow reactor, temperature 500 °C | [42] |
| No. | Catalyst | Feedstock | Products and Remarks Regarding Influence of Catalyst | Conditions of Pyrolysis Process | Reference |
|---|---|---|---|---|---|
| 1 | Na2CO3/Al2O3 and Pt/Na2CO3/Al2O3 | Pine | Hydropyrolysis—dual-bed system consisting of two separate units containing Na2CO3/Al2O3 and Pt/Al2O3 allowed for a noticeable improvement in the quality of bio-oil | Horizontal quartz annular flow reactor consists of two beds, the catalysts in this study were applied using single-bed and dual-bed modes, temperature 500 °C, vapor residence time 2 s, catalyst to feedstock weight ratio = 0.5 | [46] |
| 2 | Cu, Fe and Zn supported on Al2O3 and SiO2 | Ultrasonically pretreated cedar | The highest yield of hydrocarbons was achieved in the presence of Zn/Al2O3, the largest fraction of aromatic hydrocarbon was produced with the use of Fe/Al2O3 | Fixed bed reactor, reaction temperature, reaction time and heating rate were fixed at 500 °C, 30 min and 20 °C/min, catalyst to feedstock weight ratio = 3.3 | [47] |
| 3 | Cu and Fe supported on mesoporous rod-like Al2O3 | Sunflower stalks | Application of mesoporous Al2O3 with a larger pore size resulted in the formation of more polycyclic aromatic hydrocarbons | Fixed bed reactor, reaction temperature, reaction time and heating rate were fixed at 565 °C, 4 min and 1000 °C/min, catalyst to feedstock weight ratio = 8 | [48] |
| 4 | TiO2, CeO2, CeOx-TiO2, ZrO2 and MgO | Sugar maple, cellulose | CeO2-based catalysts were the most effective in the production of a wide group ketones from oxygenated reaction intermediates | Py-GCMS, temperature 550 °C, residence time 20 s, catalyst to feedstock weight ratio = 8 | [49] |
| 5 | Ni supported on Al2O3, SiO2, MgO, CeO2, ZrO2 and CaO-ZrO2 | Cellulose | Fast pyrolysis, Ni/Al2O3 and Ni/ZrO2 favored formation of hydrocarbons, while Ni/CeO2 and Ni/SiO2 were less effective, the last two catalysts were not capable to the efficient decrease in the amount of carboxylic acids | Py-GCMS, maximal temperature, residence time and heating rate were fixed to 600 °C, 20 s and 2000 °C/s, catalyst to feedstock weight ratio = 5 | [50] |
| 6 | Ni supported on CeO2, ZrO2 and CeO2-ZrO2 | Cellulose | Fast pyrolysis, introduction of ceria to the support structure resulted in the formation of larger amount of olefins and paraffins in comparison to the catalyst supported on monoxide zirconia. Simultaneously, the production of a higher yield of carboxylic acids was also observed. | Py-GCMS, temperature, residence time and heating rate were fixed to 400 °C–600 °C, 20 s and 2000 °C/s, catalyst to feedstock weight ratio = 5 | [51] |
| 7 | Pt/C, Pd/C, ZSM-5, MCM-41 | Wheat bran | Pt/C and Pd/C catalysts efficient in the removal of oxygen from the produced bio-oil and formation of aromatic hydrocarbons | Thermogravimetric analyzer coupled to FTIR spectrometer, dry biomass samples were heated to 700 °C with heating rate of 100 °C/min, catalyst to feedstock weight ratio = 1 | [52] |
| No. | Catalyst | Feedstock | Process, Products and Remarks Regarding Influence of Catalyst | Reaction Conditions | Reference |
|---|---|---|---|---|---|
| 1 | Ni, Ni-Ce supported on olivine | White oak submitted to thermal pretreatment | Gasification with the use of fluidized bed, Ni facilitated the production H2 and reduced the formation of CH4 and tars. | Inconel 800 reactor operated at 800 °C, gas residence time at a fluidizing steam flow rate of 0.8 kg/h was calculated to be 8.6 s | [72] |
| 2 | Commercial Ni/Al2O3 | Pine sawdust | Fast pyrolysis followed by steam reforming process, highest H2 yield observed at 600 °C. | Conical spouted bed reactor followed by in-line steam reforming of formed vapors in a fluidized bed reactor (temperature 500 °C–700 °C), steam biomass ratio 2–5, space time 2–25 gcat/min·g | [73] |
| 3 | Commercial Ni powder | Coconut shells mixed with HDPE | Integrated catalytic gasification and tar cracking, enhanced syngas and hydrogen production. | Bench scale setup consists of two cylindrical reactors with both the fluidized and fixed bed gasifiers, temperature of fluidized bed 650 °C–870 °C, temperature of fixed bed 600 °C | [74] |
| 4 | Ni-Mg-Al-Ca | Wood sawdust mixed with polypropylene | Ca addition responsible for enhancement of in situ adsorption of carbon dioxide and limitation of growth of filamentous carbon. | Two-stage stainless tube reactor, pyrolysis was carried out in the first stage and evolved volatiles were passed directly to the second stage reactor where steam gasification of pyrolysis gases took place, gasification and pyrolysis temperature 800 °C and 600 °C, respectively | [75] |
| 5 | Ni/CaAlOx | Wood sawdust | Pyrolysis-steam reforming, presence of calcium resulted in growth of selectivity to carbon oxide and reduction of carbon dioxide content, change of Ca content allowed for control of ratio between hydrogen and carbon oxide in the formed gaseous mixture. | Fixed bed two-stage reaction system, the first stage involved pyrolysis of feedstock and pyrolysis gases were passed directly to the second stage where catalytic reforming took place, temperature of the first unit 500 °C and the second 800 °C | [76] |
| 6 | NiZnAlOx | Wood sawdust | Pyrolysis and subsequent steam reforming reactions, H2 yield increased more than twice while amount of introduced Ni on the catalyst surface changed from 5% to 35%, presence of ZnO protect against the agglomeration of nickel particles and formation of coke deposit. | Two-stage fixed bed reactor, in the first reactor biomass was decomposed into pyrolysis vapors, which were passed to the second reactor for catalytic steam reforming, temperature of the first unit 535 °C and the second 800 °C | [77] |
| 7 | Ni supported on ZrO2, Al2O3, SiO2, CeO2, TiO2 and MgO | Cellulose | Pyrolysis, zirconia the most promising material among the investigated oxides. | Stirred batch reactor at 700 °С for 4 h, 5 g of biomass and 0.2 g of catalyst were used | [78] |
| 8 | Ni supported on ZrO2 prepared by different methods | Cellulose | Pyrolysis, catalyst containing ZrO2 prepared from ZrOCl2 by precipitation with NaOH was the most active in the conversion of cellulose to gaseous products, especially hydrogen. | Stirred batch reactor at 700 °С for 4 h, 5 g of biomass and 0.2 g of catalyst were used | [79] |
| 9 | Ni/CeO2-ZrO2 | Cellulose | Pyrolysis, introduction of Ni on surface of CeO2-ZrO2 synthesized by sol-gel or impregnation allowed for the production of the highest hydrogen yield. | Stirred batch reactor at 700 °С for 4 h, 5 g of biomass and 0.2 g of catalyst were used | [80] |
| 10 | Ni/MexO-ZrO2 (M e = Ca, Mg, Na and K) | Cellulose | Pyrolysis, alkali metals allows for the formation of oxygen vacancies responsible for the removal of the carbon deposit, enhanced adsorption of CO2 shifts an equilibrium of the reactions of thermal treatment of cellulose. | Stirred batch reactor at 700 °С for 4 h, 5 g of biomass and 0.2 g of catalyst were used | [82] |
| 11 | Ni/CaO-ZrO2 | Cellulose and pretreated beech, birch, poplar and pine | Upgrading of pyrolysis vapors, higher resistance against coke formation, decrease in the production of permanent gases in the case of real biomass samples. | Quartz fixed bed two step reactor, biomass decomposition (500 °C) was separated from the catalyst bed (700 °C), 0.4 g of biomass and 0.1 g of the catalyst were used | [83] |
| 12 | Ni supported on SBA-15, SBA-16, KIT-6 and MCM-41 | Cellulose | Pyrolysis, the use of Ni/SBA-15 and Ni/KIT-6 results in the increase in the efficiency in H2 production in comparison to Ni/SiO2 sample | Stirred batch reactor at 700 °С for 4 h, 5 g of biomass and 0.2 g of catalyst were used | [71] |
| 13 | Ni-Co/Mg-Al Ni supported onAl2O3, activated carbon, TiO2, ZrO2, MgO Ni/biomass Ru/C | Lignin, cellulose, wheat straw, timothy grass, canola meal pine wood, fruit pulp | Subcritical and supercritical water gasification of various kinds of lignocellulosic biomass | [ 70,71,72]—batch supercritical water reactor, temperature and pressure—650 °C, 260 atm, water to biomass mass ratio = 5 [73]—batch reactor, temperature 300 °C–500 °C, pressure 230-250 atm, residence time 15–45 min, water to biomass mass ratio = 5–10 [74]—batch reactor, temperature 400 °C–600 °C, reaction time up to 60 min | [84,85,86,87,88] |
| 14 | Fe-Zn/Al2O3 | Wood sawdust | Steam reforming of volatiles from pyrolysis of wood sawdust, hydrogen production increased with the increase of Zn content, catalysts showed high stability in the reaction conditions | Fixed bed two-stage reaction system, the first stage involved pyrolysis of feedstock and pyrolysis gases were passed directly to the second stage where catalytic steam reforming took place, temperature of the first unit 500 °C and the second 800 °C | [89] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grams, J.; Ruppert, A.M. Development of Heterogeneous Catalysts for Thermo-Chemical Conversion of Lignocellulosic Biomass. Energies 2017, 10, 545. https://doi.org/10.3390/en10040545
Grams J, Ruppert AM. Development of Heterogeneous Catalysts for Thermo-Chemical Conversion of Lignocellulosic Biomass. Energies. 2017; 10(4):545. https://doi.org/10.3390/en10040545
Chicago/Turabian StyleGrams, Jacek, and Agnieszka M. Ruppert. 2017. "Development of Heterogeneous Catalysts for Thermo-Chemical Conversion of Lignocellulosic Biomass" Energies 10, no. 4: 545. https://doi.org/10.3390/en10040545







