A Simple Method for the Detection of Long-Chain Fatty Acids in an Anaerobic Digestate Using a Quartz Crystal Sensor
Abstract
:1. Introduction
2. Materials and Methods
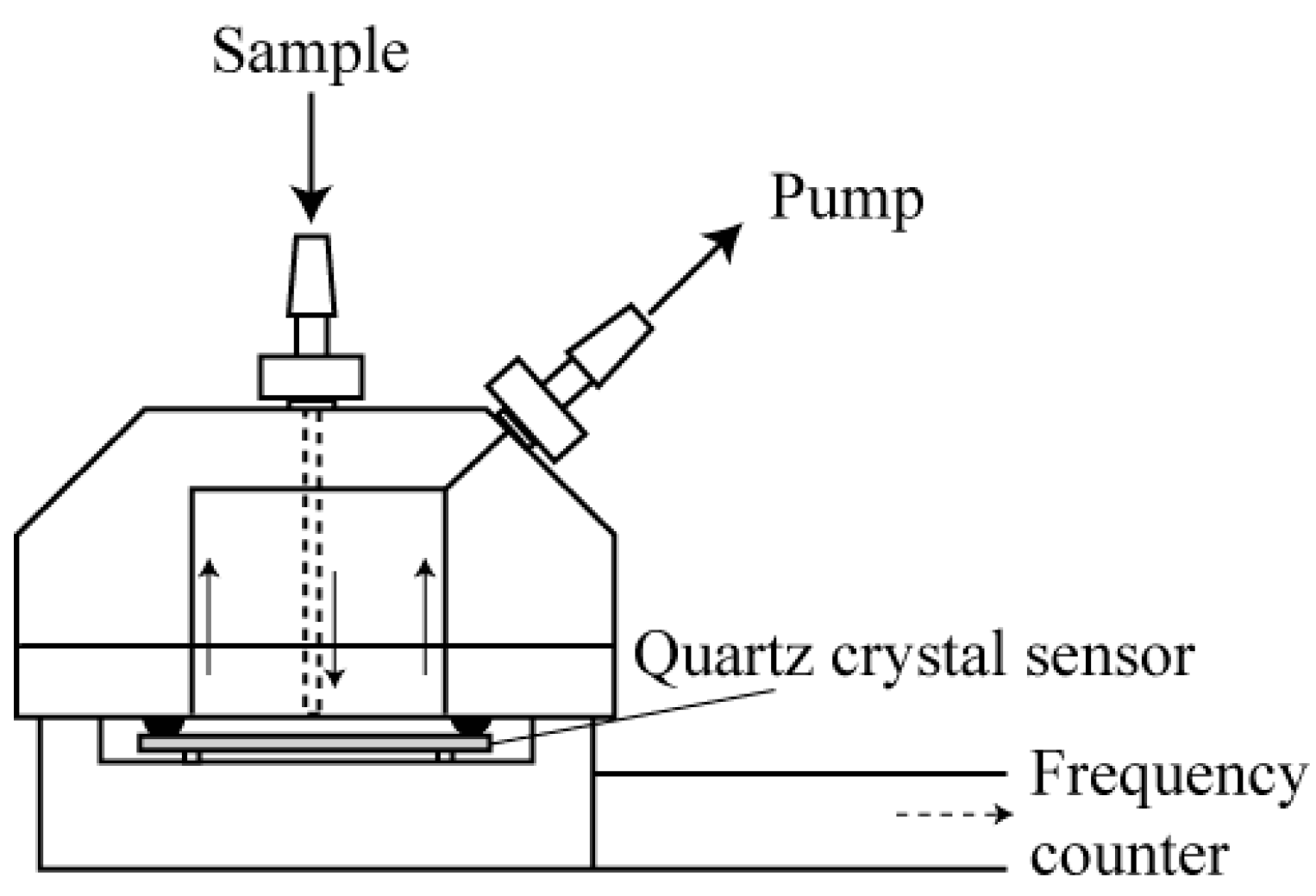
2.1. Materials and Preparation of the Quartz Crystal Microbalance Sensor
2.2. Performance of the Quartz Crystal Microbalance Sensor
2.3. Reactor Set-Up
2.4. Validation of Quartz Crystal Microbalance Measurements
2.5. Analysis
3. Results and Discussion
3.1. Characteristics of the Quartz Crystal Microbalance Sensor
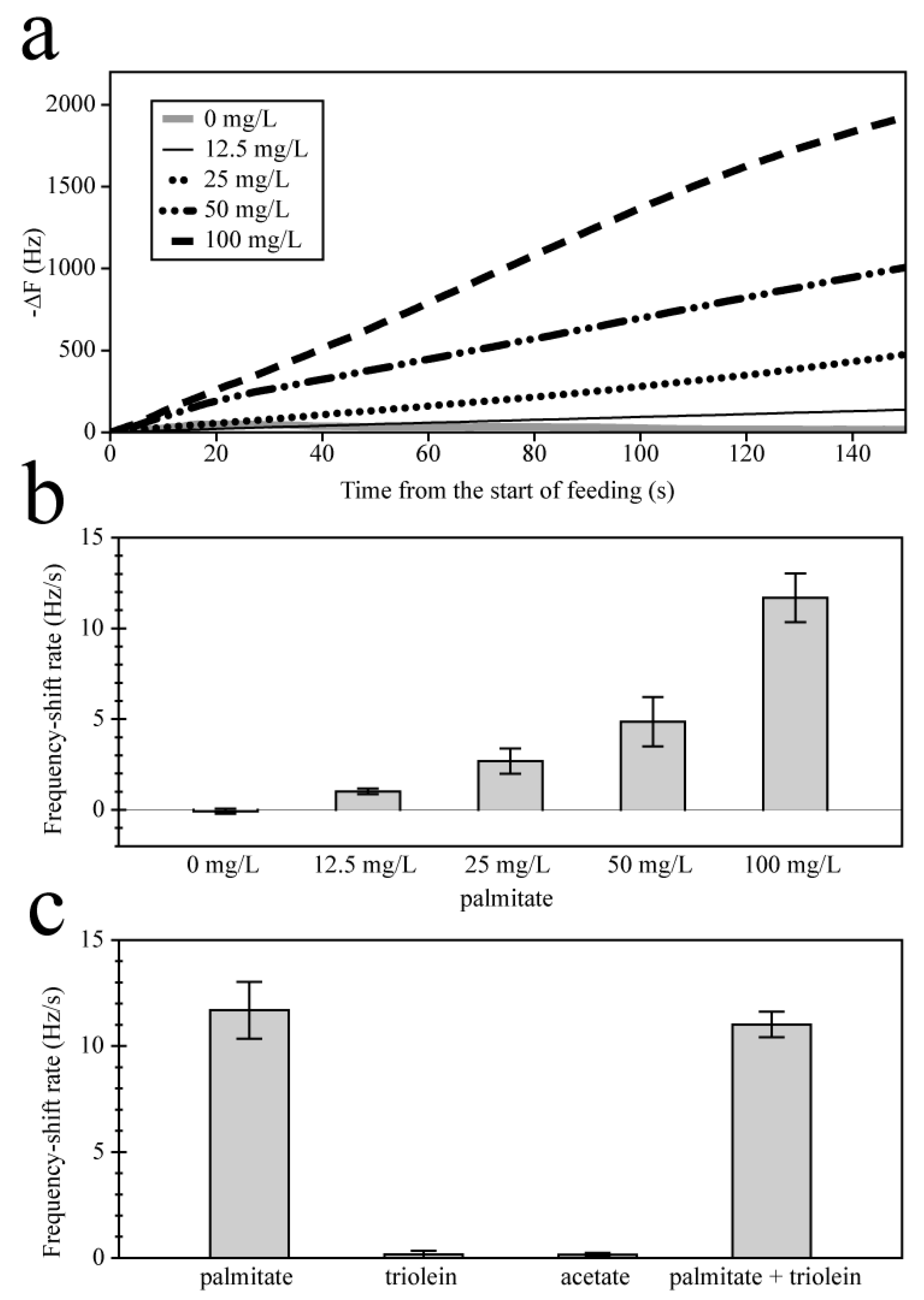
3.1.1. Effect of Dissociation on Quartz Crystal Microbalance Frequency Responses
3.1.2. Linear Range and Selectivity of the Sensor
3.2. Feasibility for Practical Use in Anaerobic Digestion Operation
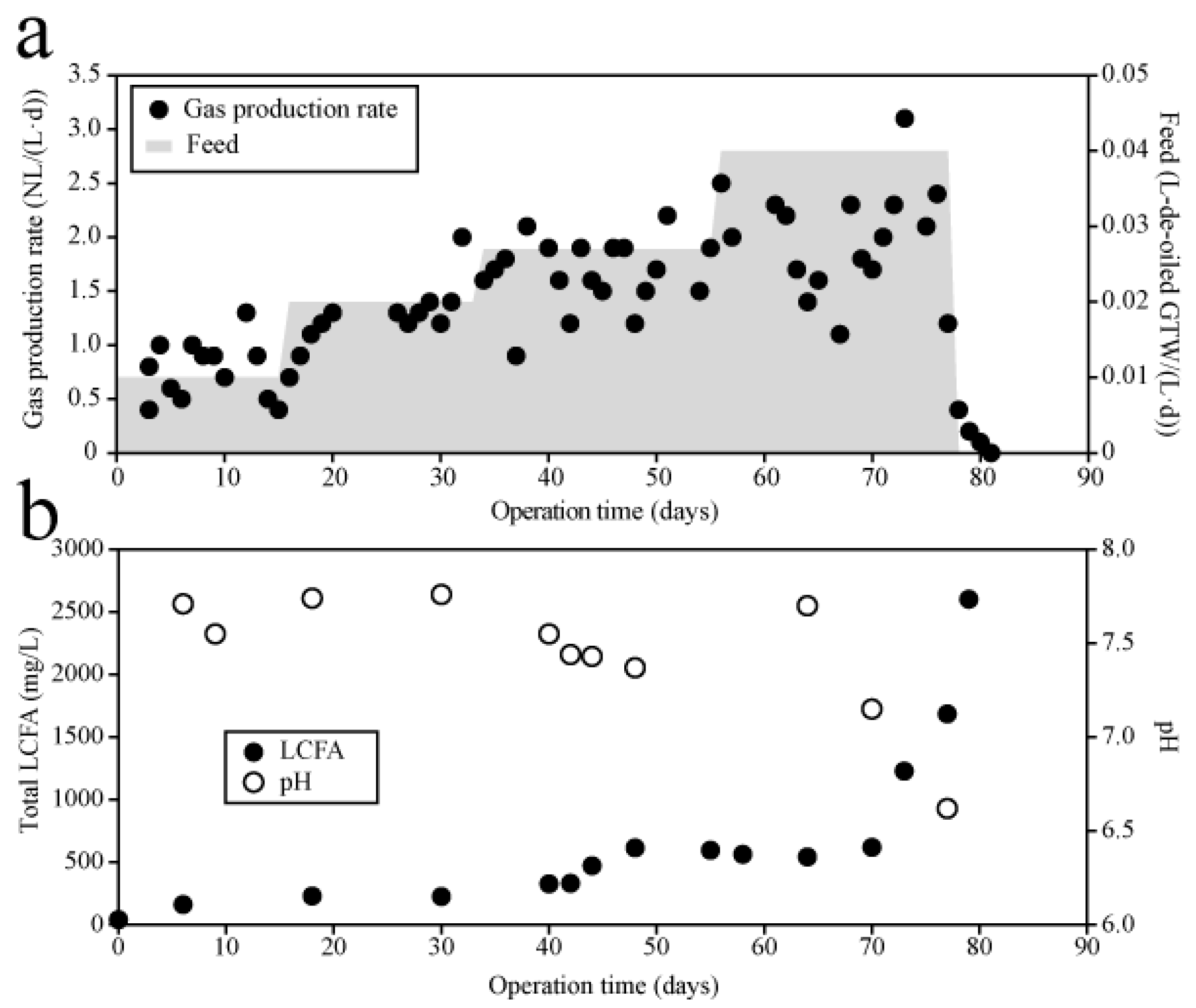
3.2.1. Semi-Continuous Operation of an Anaerobic Digester Treating De-Oiled Grease Trap Waste
3.2.2. Quartz Crystal Microbalance Measurements of Anaerobic Digestate Samples
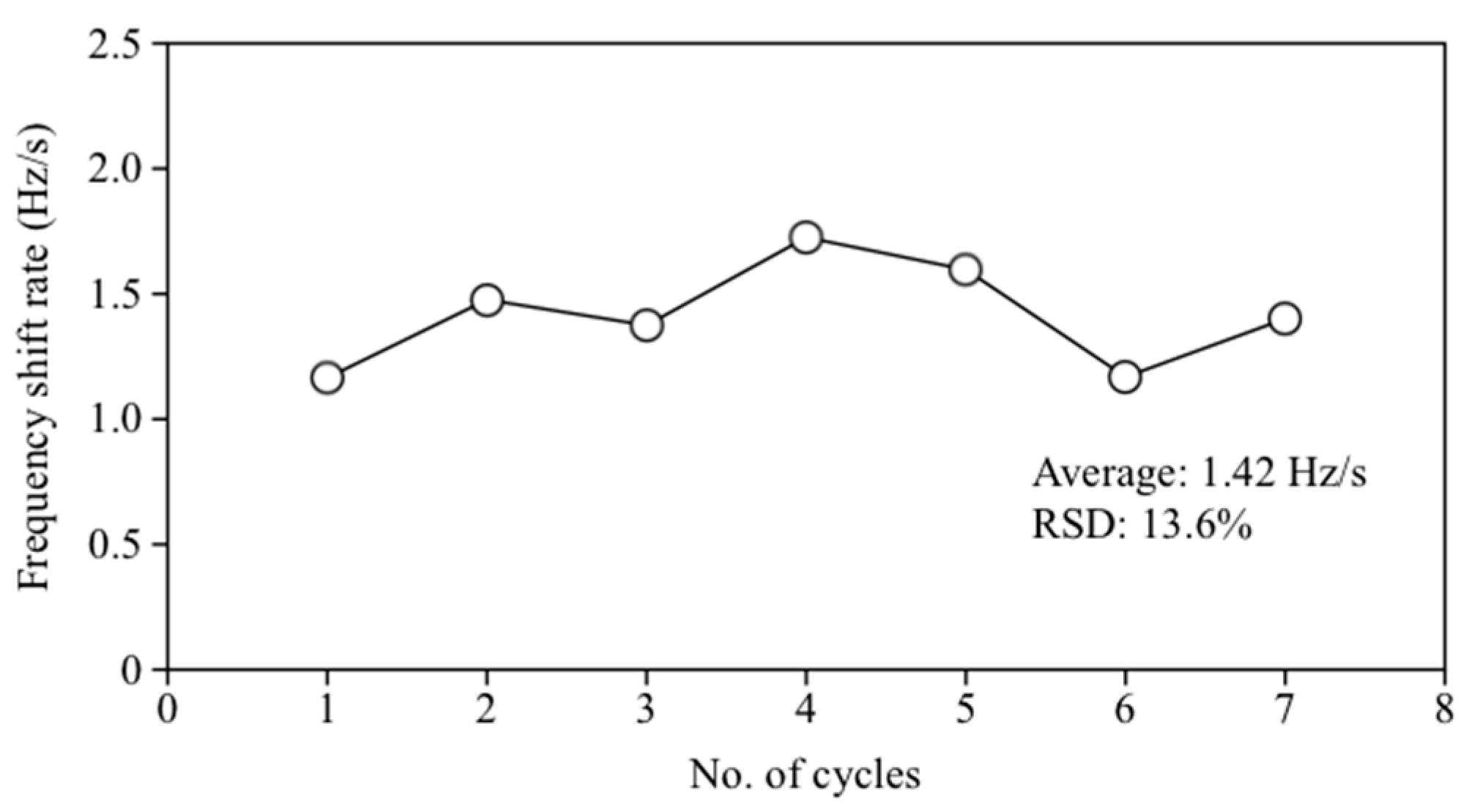
3.3. Validation of the New Method
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kobayashi, T.; Kuramochi, H.; Xu, K.Q. Variable oil properties and biomethane production of grease trap waste derived from different resources. Int. Biodeterior. Biodegrad. 2016. [Google Scholar] [CrossRef]
- Yalcinkaya, S.; Malina, J.F. Model development and evaluation of methane potential from anaerobic co-digestion of municipal wastewater sludge and un-dewatered grease trap waste. Waste Manag. 2015, 40, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Shin, H.; Kim, S.H.; Lee, C.Y.; Nam, S.Y. Inhibitory effects of long-chain fatty acids on VFA degradation and beta-oxidation. Water Sci. Technol. 2003, 47, 139–146. [Google Scholar] [PubMed]
- Alves, M.M.; Mota, V.J.A.; Álvares, P.R.M.; Pereira, M.A.; Mota, M. Effects of lipids and oleic acid on biomass development in anaerobic fixed-bed reactors. Part II: Oleic acid toxicity and biodegradability. Water Res. 2001, 35, 264–270. [Google Scholar] [CrossRef]
- Cho, H.S.; Moon, H.S.; Lim, J.Y.; Kim, J.Y. Effect of long chain fatty acids removal as a pretreatment on the anaerobic digestion of food waste. J. Mater. Cycles Waste Manag. 2013, 15, 82–89. [Google Scholar] [CrossRef]
- Pereira, M.A.; Pires, O.C.; Mota, M.; Alves, M.M. Anaerobic biodegradation of oleic and palmitic acids: Evidence of mass transfer limitation caused by long chain fatty acid accumulation onto anaerobic sludge. Biotechnol. Bioeng. 2005, 92, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, T.; Kuramochi, H.; Maeda, K.; Tsuji, T.; Xu, K.Q. Dual-fuel production from restaurant grease trap waste: Bio-fuel oil extraction and anaerobic methane production from the post-extracted residue. Bioresour. Technol. 2014, 169, 134–142. [Google Scholar] [CrossRef] [PubMed]
- Palatsi, J.; Laureni, M.; Andrés, M.V.; Flotats, X.; Nielsen, H.B.; Angelidaki, I. Strategies for recovering inhibition caused by long chain fatty acids on anaerobic thermophilic biogas reactors. Bioresour. Technol. 2009, 100, 4588–4596. [Google Scholar] [CrossRef] [PubMed]
- Roy, F.; Albagnac, G.; Samain, E. Influence of calcium addition on growth of highly purified syntrophic cultures degrading long-chain fatty acids. Appl. Environ. Microbiol. 1985, 49, 702–705. [Google Scholar] [PubMed]
- Koster, I.W. Abatement of long-chain fatty acid inhibition of methanogenesis by calcium addition. Biolog. Wastes 1987, 22, 295–301. [Google Scholar] [CrossRef]
- Wu, L.; Kobayashi, T.; Kuramochi, H.; Li, Y.Y.; Xu, K.Q. Recovery strategies of inhibition for mesophilic anaerobic sludge treating the de-oiled grease trap waste. Int. Biodeterior. Biodegrad. 2015, 104, 315–323. [Google Scholar] [CrossRef]
- Lalman, J.A.; Bagley, D.M. Anaerobic degradation and methanogenic inhibitory effects of oleic and stearic acids. Water Res. 2001, 35, 2975–2983. [Google Scholar] [CrossRef]
- Neves, L.; Oliveira, R.; Alves, M.M. Fate of LCFA in the co-digestion of cow manure, food waste and discontinuous addition of oil. Water Res. 2009, 43, 5142–5150. [Google Scholar] [CrossRef] [PubMed]
- Pitk, P.; Palatsi, J.; Kaparaju, P.; Fernández, B.; Vilu, R. Mesophilic co-digestion of dairy manure and lipid rich solid slaughterhouse wastes: Process efficiency, limitations and floating granules formation. Bioresour. Technol. 2014, 166, 168–177. [Google Scholar] [CrossRef] [PubMed]
- Si, P.; Mortensen, J.; Komolov, A.; Denborg, J.; Møller, P.J. Polymer coated quartz crystal microbalance sensors for detection of volatile organic compounds in gas mixtures. Anal. Chim. Acta 2007, 597, 223–230. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.H.; Rao, Y.K.; Wu, T.Z.; Tzeng, Y.M. Direct characterization and quantification of volatile organic compounds by piezoelectric module chips sensor. Sens. Actuators B 2009, 137, 741–746. [Google Scholar] [CrossRef]
- Escuderos, M.E.; Sanchez, S.; Jimenez, A. Quartz crystal microbalance (QCM) sensor arrays selection for olive oil sensory evaluation. Food Chem. 2011, 124, 857–862. [Google Scholar] [CrossRef]
- Escuderos, M.E.; Sánchez, S.; Jiménez, A. Virgin olive oil sensory evaluation by an artificial olfactory system, based on quartz crystal microbalance (QCM) sensors. Sens. Actuators B 2010, 147, 159–164. [Google Scholar] [CrossRef]
- Yang, Z.P.; Zhang, C.J. Designing of MIP-based QCM sensor for the determination of Cu(II) ions in solution. Sens. Actuators B 2009, 142, 210–215. [Google Scholar] [CrossRef]
- Gupta, V.K.; Yola, M.L.; Eren, T.; Atar, N. Selective QCM sensor based on atrazine imprinted polymer: Its application to wastewater sample. Sens. Actuators B 2015, 218, 215–221. [Google Scholar] [CrossRef]
- Maeda, K.; Hayashi, A.; Iimura, K.; Suzuki, M.; Hirota, M.; Asakuma, Y.; Fukui, K. Generation of nanometer-scale crystals of hydrophobic compound from aqueous solution. Chem. Eng. Process. 2005, 44, 941–947. [Google Scholar] [CrossRef]
- Ichihara, K.; Fukubayashi, Y. Preparation of fatty acid methyl esters for gas-liquid chromatography. J. Lipid Res. 2010, 51, 635–640. [Google Scholar] [CrossRef] [PubMed]
- Kanicky, J.R.; Poniatowski, A.F.; Mehta, N.R.; Shah, D.O. Cooperativity among molecules at interfaces in relation to various technological processes: Effect of chain length on the pKa of fatty acid salt solutions. Langmuir 2000, 16, 172–177. [Google Scholar] [CrossRef]
- Kanicky, J.R.; Shah, D.O. Effect of degree, type, and position of unsaturation on the pKa of long-chain fatty acids. J. Colloid Interface Sci. 2002, 256, 201–207. [Google Scholar] [CrossRef] [PubMed]
- Schwarzenbach, R.P.; Gschwend, P.M.; Imboden, D.M. Environmental Organic Chemistry, 2nd ed.; Wiley-Interscience: Hoboken, NJ, USA, 2003; p. 1313. [Google Scholar]
- Yalkowsky, S.H.; He, Y.; Jain, P. Handbook of Aqueous Solubility Data, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2010; p. 1620. [Google Scholar]
- Ying, Z.; Jiang, Y.; Du, X.; Xie, G.; Yu, J.; Wang, H. PVDF coated quartz crystal microbalance sensor for DMMP vapor detection. Sens. Actuators B 2007, 125, 167–172. [Google Scholar] [CrossRef]
- Ayad, M.M.; Torad, N.L. Alcohol vapours sensor based on thin polyaniline salt film and quartz crystal microbalance. Talanta 2009, 78, 1280–1285. [Google Scholar] [CrossRef] [PubMed]
- Reipa, V.; Purdum, G.; Choi, J. Measurement of nanoparticle concentration using quartz crystal microgravimetry. J. Phys. Chem. B 2010, 114, 16112–16117. [Google Scholar] [CrossRef] [PubMed]
- Buck, R.P.; Lindner, E.; Kutner, W.; Inzelt, G. Piezoelectric chemical sensors. Pure Appl. Chem. 2004, 76, 1139–1160. [Google Scholar] [CrossRef]
- Wu, L.; Kobayashi, T.; Kuramochi, H.; Li, Y.Y.; Xu, K.Q. Improved biogas production from food waste by co-digestion with de-oiled grease trap waste. Bioresour. Technol. 2016, 201, 237–244. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, K.; Maeda, K.; Kuramochi, H.; Nakagawa, K.; Asakuma, Y.; Fukui, K.; Osako, M.; Sakai, S. Solid-Liquid Eduilibria in Fatty Scid/Triglycerol Systems. J. Chem. Eng. Data 2011, 56, 1613–1616. [Google Scholar] [CrossRef]
- Yui, K.; Itsukaichi, Y.; Kobayashi, T.; Tsuji, T.; Fukui, K.; Maeda, K.; Kuramochi, H. Solid-liquid equilibria in the binary systems of saturated fatty acids or triglycerides (C12 to C18) + hexadecane. J. Chem. Eng. Data 2016. [Google Scholar] [CrossRef]
- Neves, L.; Pereira, M.A.; Mota, M.; Alves, M.M. Detection and quantification of long chain fatty acids in liquid and solid samples and its relevance to understand anaerobic digestion of lipids. Bioresour. Technol. 2009, 100, 91–96. [Google Scholar] [CrossRef] [PubMed]
- Pereira, M.A.; Sousa, D.Z.; Mota, M.; Alves, M.M. Mineralization of LCFA associated with anaerobic Sludge: Kinetics, enhancement of methanogenic activity, and effect of VFA. Biotechnol. Bioeng. 2004, 88, 502–511. [Google Scholar] [CrossRef] [PubMed]
- Pan, M.; Fang, G.; Lu, Y.; Kong, L.; Yang, Y.; Wang, S. Molecularly imprinted biomimetic QCM sensor involving a poly(amidoamine) dendrimer as a functional monomer for the highly selective and sensitive determination of methimazole. Sens. Actuators B 2015, 207, 588–595. [Google Scholar] [CrossRef]
- He, F.; Cui, X.; Ren, J. A Novel QCM-based biosensor for detection of microorganisms producing hydrogen sulfide. Anal. Lett. 2008, 41, 2697–2709. [Google Scholar] [CrossRef]


| Day | Concentration (mg/L) | |||||
|---|---|---|---|---|---|---|
| C16:0 | C16:1 | C18:0 | C18:1 | C18:2 | Total | |
| 30 | 115 | 0 | 36 | 76 | 0 | 227 |
| 40 | 147 | 0 | 48 | 116 | 18 | 328 |
| 42 | 136 | 3 | 51 | 72 | 26 | 333 |
| 44 | 154 | 11 | 49 | 174 | 42 | 473 |
| 48 | 280 | 4 | 103 | 130 | 33 | 616 |
| 64 | 229 | 12 | 87 | 173 | 21 | 543 |
| 70 | 204 | 14 | 87 | 267 | 31 | 620 |
| 73 | 924 | 14 | 122 | 79 | 0 | 1229 |
| 77 | 1267 | 15 | 140 | 119 | 13 | 1686 |
| 79 | 918 | 85 | 235 | 788 | 352 | 2602 |
| Sample | TSS | Total LCFA in Supernatant | Total LCFA in Sediment | Recovery 1 |
|---|---|---|---|---|
| g/L | mg/L-sludgefed | mg/L-sludgefed | % | |
| Sludge A | 10.4 | 205 ± 74 | 244 ± 22 | 46% |
| Sludge B | 14.6 | 227 ± 29 | 396 ± 65 | 36% |
| Sludge C | 19.1 | 1116 ± 562 | 629 ± 27 | 64% |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kobayashi, T.; Kuramochi, H.; Maeda, K.; Xu, K. A Simple Method for the Detection of Long-Chain Fatty Acids in an Anaerobic Digestate Using a Quartz Crystal Sensor. Energies 2017, 10, 19. https://doi.org/10.3390/en10010019
Kobayashi T, Kuramochi H, Maeda K, Xu K. A Simple Method for the Detection of Long-Chain Fatty Acids in an Anaerobic Digestate Using a Quartz Crystal Sensor. Energies. 2017; 10(1):19. https://doi.org/10.3390/en10010019
Chicago/Turabian StyleKobayashi, Takuro, Hidetoshi Kuramochi, Kouji Maeda, and Kaiqin Xu. 2017. "A Simple Method for the Detection of Long-Chain Fatty Acids in an Anaerobic Digestate Using a Quartz Crystal Sensor" Energies 10, no. 1: 19. https://doi.org/10.3390/en10010019
APA StyleKobayashi, T., Kuramochi, H., Maeda, K., & Xu, K. (2017). A Simple Method for the Detection of Long-Chain Fatty Acids in an Anaerobic Digestate Using a Quartz Crystal Sensor. Energies, 10(1), 19. https://doi.org/10.3390/en10010019





