Efficiency of Silver Impregnated Porous Pot (SIPP) Filters for Production of Clean Potable Water
Abstract
:1. Introduction
- Evaluate the removal efficiency for calcium (Ca2+), iron (Fe3+), magnesium (Mg2+), arsenic (As3+), total organic carbon (TOC) and fluorides (F−) from contaminated water.
- Determine the flow rate of the filter and compare with results given in the literature.
- Determine whether the filter removes suspended particles (turbidity) from contaminated water.
- Evaluate the effect of turbidity on the flow rate of the filter.
- Determine the relationship between chlorophyll a in the feed water and noted flow rate of the SIPP filter.
2. Materials and Methods
2.1. Filter Description
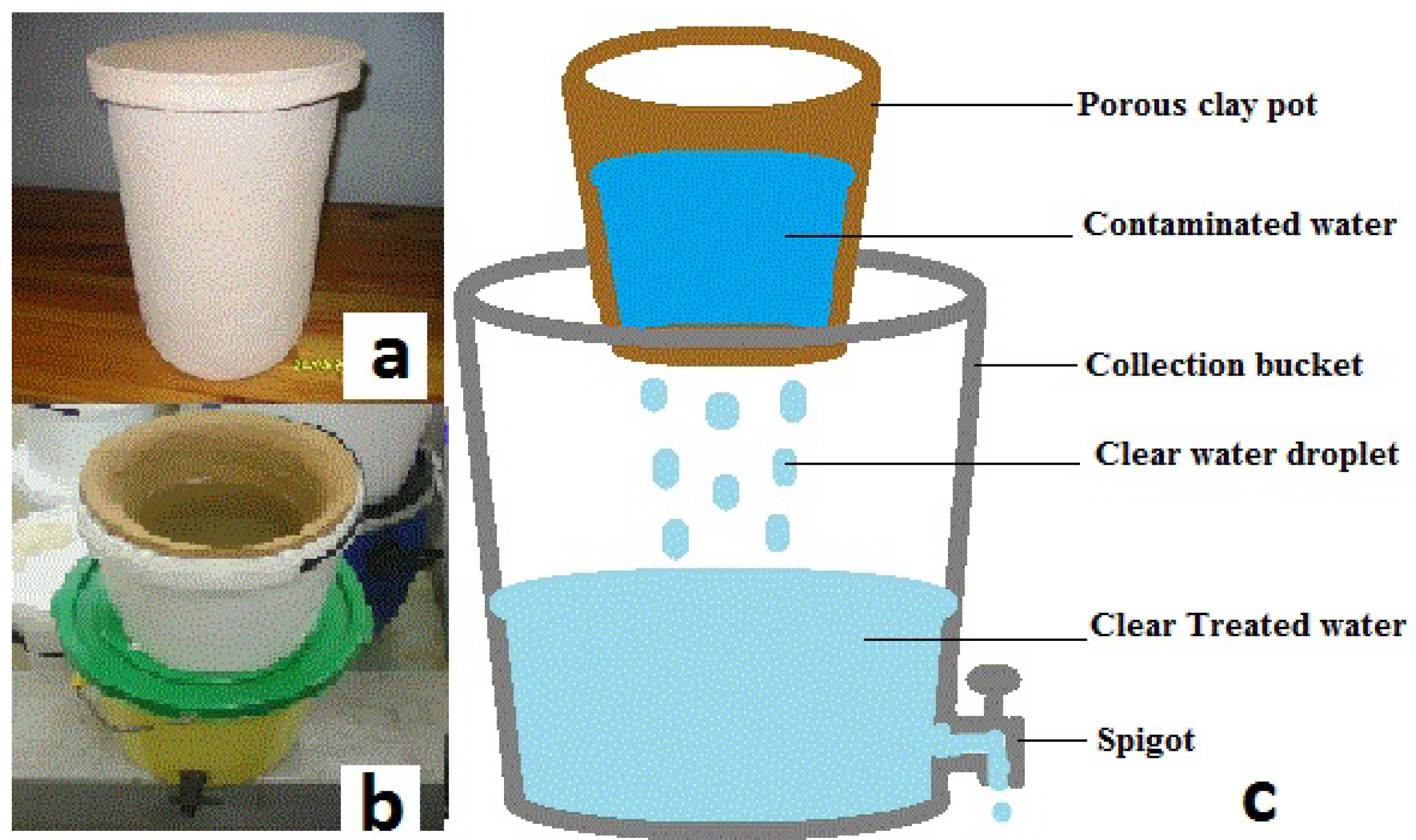
2.2. Baseline Determination of Flow Rate and Filtration of Spiked Samples
2.3. Sample Collection and Filtration
2.4. Flow Rate and Turbidity Determination
2.5. Determination of the Contaminant Removal Efficiency of the SIPP Filters
2.6. Fluoride Analysis
2.7. Analysis of Total Organic Carbon (TOC)
2.8. Chlorophyll a Analysis
2.9. Statistical Analysis
2.10. Investigation of Cost and Maintenance of the Filters
3. Results and Discussion
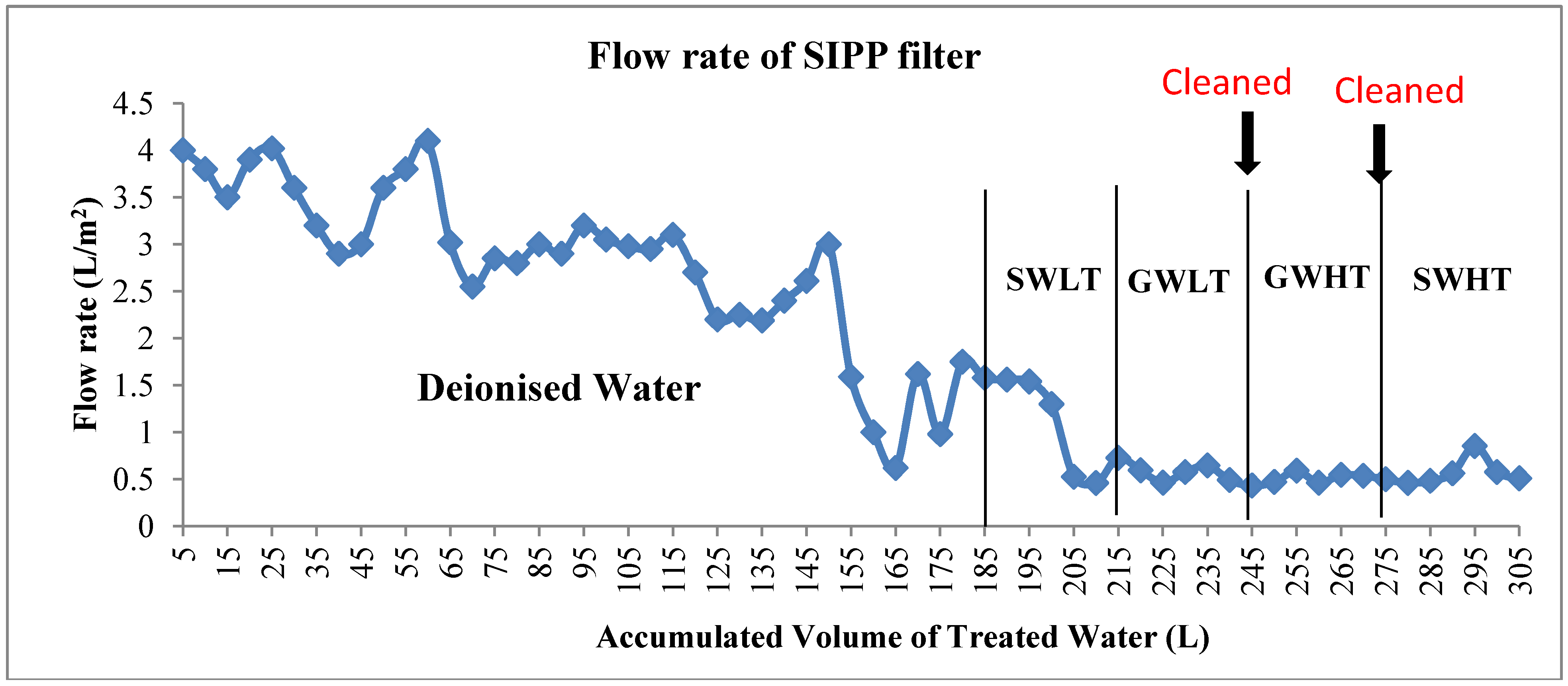
3.1. Flow Rates and Turbidity Reductions
3.2. Reduction of Chemical Contaminants
3.2.1. Removal of Chemical Contaminants from Spiked Water Samples
| Contaminant of interest | Guidelines (mg/L) | Spiked levels (mg/L) | Removal (%) | No. of samples | |
|---|---|---|---|---|---|
| SANS 241 | WHO | ||||
| Calcium | 150–300 | 100–300 | 200 | 56.3 (±11.1) | 30 |
| Magnesium | 70–100 | 200 | 100 | 66.7 (±6.7) | 30 |
| Iron | 0.2–2.0 | 1–3 | 5 | 88.7 (±12.4) | 30 |
| Arsenic | 0.0005 | 0.0002 | 10 | 53.9 (±20.4) | 30 |
| Fluoride | 1.0–1.5 | 1.5 | 5 | 43.5 (±31.5) | 30 |
3.2.2. Removal of Chemical Contaminants from Environmental Water Samples
| Water Type | Turbidity (NTU) | Analyte Concentration (mg/L) | |||||
|---|---|---|---|---|---|---|---|
| Ca | Mg | Fe | As | F | TOC | ||
| SWLT | 11.9 (±10.2) | 137.1 (±48.7) | 65.9 (±42.9) | 1.03 (±0.5) | 4.90 (±1.2) | 3.39 (±1.7) | 7.71 (±0.5) |
| GWLT | 2.17 (±0.8) | 158.5 (±47.2) | 58.5 (±35.9) | 0.20 (±0.2) | 9.48 (±2.9) | 7.72 (±11.1) | 7.12 (±1.0) |
| GWHT | 8.39 (±5.4) | 23.9 (±9.6) | 59.6 (±38.9) | 0.23 (±0.0) | 8.12 (±1.5) | 0.49 (±0.1) | 5.87 (±0.8) |
| SWHT | 40.4 (±4.1) | 14.9 (±2.67) | 25.6 (±1.1) | 0.29 (±0.0) | 5.03 (±0.9) | 0.85 (±0.1) | 4.81 (±0.7) |
| Water type | Analyte Reduction (%) (average ± standard deviation) | ||||||
|---|---|---|---|---|---|---|---|
| Turbidity | Ca | Mg | Fe | As | F | TOC | |
| SWLT | 69.4 (±24.5) | 43.4 (±19.2) | 49.5 (±19.6) | 70.0 (±8.1) | 57.3 (±18.8) | 39.6 (±13.4) | 15.8 (±3.0) |
| GWLT | 50.4 (±10.3) | 67.5 (±6.8) | 39.8 (±13.7) | 92.5 (±2.6) | 42.6 (±14.6) | 42.9 (±19.5) | 12.3 (±3.1) |
| GWHT | 45.5 (±9.6) | 36.5 (±17.9) | 71.5 (±7.9) | 87.8 (±4.6) | 52.6 (±19.1) | 56.9 (±18.5) | 18.6 (±6.6) |
| SWHT | 82.5 (±4.5) | 68.8 (±11.8) | 98.4 (±0.1) | 79.1 (±3.3) | 82.9 (±7.0) | 95.1 (±3.6) | 35.2 (±5.3) |
3.2.2.1. Filter Performance in Treating SWLT
3.2.2.2. Filter Performance in Treating GWLT
3.2.2.3. Filter Performance in Treating GWHT
3.2.2.4. Filter Performance in Treating SWHT
3.3. Statistical Analysis of Correlation between Removals and Initial Concentrations per Water Source
| Water Type | Relation between contaminant removal and initial concentration in source water ( r value) | ||||||
|---|---|---|---|---|---|---|---|
| Turbidity | Ca | Mg | Fe | As | F | TOC | |
| SWLT | 0.90 | 0.91 | 0.09 | 0.50 | −0.23 | 0.78 | −0.92 |
| GWLT | 0.68 | −0.87 | 0.79 | 0.05 | −0.78 | 0.52 | −0.99 |
| GWHT | 0.24 | −0.55 | −0.63 | 0.81 | −0.77 | 0.58 | −0.99 |
| SWHT | 0.56 | 0.93 | −0.96 | 0.99 | −0.99 | 0.62 | −0.68 |
3.4. Statistical Analysis of Variance in Contaminant Removal between the Water Sources
3.5. Association of Chlorophyll a with the Flow Rate of SIPP
4. Cost and Maintenance Guidelines of the SIPP Filter
5. Conclusions
6. Study Limitations
Acknowledgments
Conflict of interest
References
- Peng, F.F.; Di, P. Removal of arsenic from aqueous solution by adsorbing colloid flotation. Ind. Eng. Chem. Res 1994, 33, 922–928. [Google Scholar] [CrossRef]
- Kau, P.M.H.; Smith, D.W.; Binning, P. Fluoride retention by kaolin clay. J. Contam. Hydrol. 1997, 28, 267–288. [Google Scholar] [CrossRef]
- Central American Institute of Industrial Research Technology (ICAITI), Identification and Evaluation of Design Alternatives for a Low Cost Domestic Filter for Drinking Water; Central American Institute of Industrial Technology: Guatemala City, Guatemala, 1984.
- Van Halem, D.; van de Laan, H.; Heijman, S.G.J.; van Dijk, J.C.; Amy, G.L. Assessing the sustainability of the silver-impregnated ceramic pot filter for low-income drinking water treatment. Phys. Chem. Earth 2009, 34, 36–42. [Google Scholar] [CrossRef]
- Lantagne, D.S.; Quick, R.; Mintz, E.D. Household Water Treatment and Safe Storage Options in Developing Countries: A Review of Current Implementation Practices; Water Supply & Sanitation Collaborative Council: Geneva, Switzerland, 2006. [Google Scholar]
- South African National Standards (SANS). In Drinking Water Part 1 (SANS 241-1) and Part 2 (SANS 241-2); SABS Standards Division: Pretoria, South Africa, 2006.
- Shafiquzzaman, M.; Azam, M.S.; Mishima, I.; Nakajima, J. Technological and social evaluation of arsenic mitigation in rural Bangladesh. J. Health Popul. Nutr. 2009, 27, 674–683. [Google Scholar]
- Shafiquzzaman, M.; Azam, M.S.; Nakajima, J.; Bari, Q.H. Investigation of arsenic removal by a simple iron removal ceramic filter in rural Bangladesh. Desalination 2011, 265, 60–66. [Google Scholar]
- American Public Health Association (APHA)American Water Works Association (AWWA)Water Environment Federation (WEF)Revisions to Standard Methods for the Examination of Water and Wastewater (Supplement); American Public Health Association: Washington, DC, USA, 2001.
- Mwabi, J.K.; Adeyemo, F.E.; Mahlangu, T.O.; Mamba, B.B.; Brouckaert, B.M.; Swartz, C.D.; Offringa, G.; Mpenyana-Monyatsi, L.; Momba, M.N.B. Household water treatment systems: A solution to the production of safe drinking water by the low-income communities of Southern Africa. Phys. Chem. Earth 2011, 6, 1120–1128. [Google Scholar]
- Hamdi, N.; Srasra, E. Removal of fluoride from acidic wastewater by clay mineral: Effect of solid-liquid ratios. Desalination 2007, 206, 238–244. [Google Scholar] [CrossRef]
- Mahlangu, T.O.; Mpenyana-Monyatsi, L.; Mamba, B.B.; Momba, M.N.B. A simplified cost-effective biosand filter (BSFZ) for the removal of chemical contaminants from water. J. Chem. Eng. Mater. Sci. 2010, 2, 156–167. [Google Scholar]
- Mahlangu, T.O.; Mamba, B.B.; Momba, M.N.B. A comparative assessment of chemical contaminant removal by three household water treatment filters. Water SA 2012, 38, 39–48. [Google Scholar]
- World Health Organization (WHO), WHO Guidelines for Drinking-Water Quality, 3rd edWHO: Geneva, Switzerland, 2008; 1.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Mahlangu, O.; Mamba, B.; Momba, M. Efficiency of Silver Impregnated Porous Pot (SIPP) Filters for Production of Clean Potable Water. Int. J. Environ. Res. Public Health 2012, 9, 3014-3029. https://doi.org/10.3390/ijerph9093014
Mahlangu O, Mamba B, Momba M. Efficiency of Silver Impregnated Porous Pot (SIPP) Filters for Production of Clean Potable Water. International Journal of Environmental Research and Public Health. 2012; 9(9):3014-3029. https://doi.org/10.3390/ijerph9093014
Chicago/Turabian StyleMahlangu, Oranso, Bhekie Mamba, and Maggie Momba. 2012. "Efficiency of Silver Impregnated Porous Pot (SIPP) Filters for Production of Clean Potable Water" International Journal of Environmental Research and Public Health 9, no. 9: 3014-3029. https://doi.org/10.3390/ijerph9093014





