Can We Modify the Intrauterine Environment to Halt the Intergenerational Cycle of Obesity?
Abstract
:1. Introduction—What is the Problem?
2. Why Are We Concerned About Mom?
2.1. Maternal Obesity
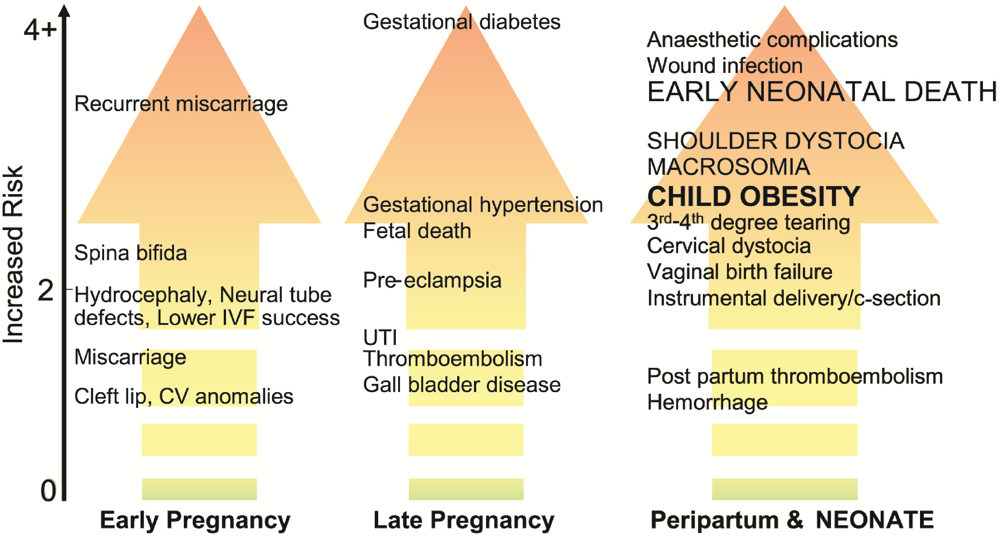
2.2. Gestational Weight Gain

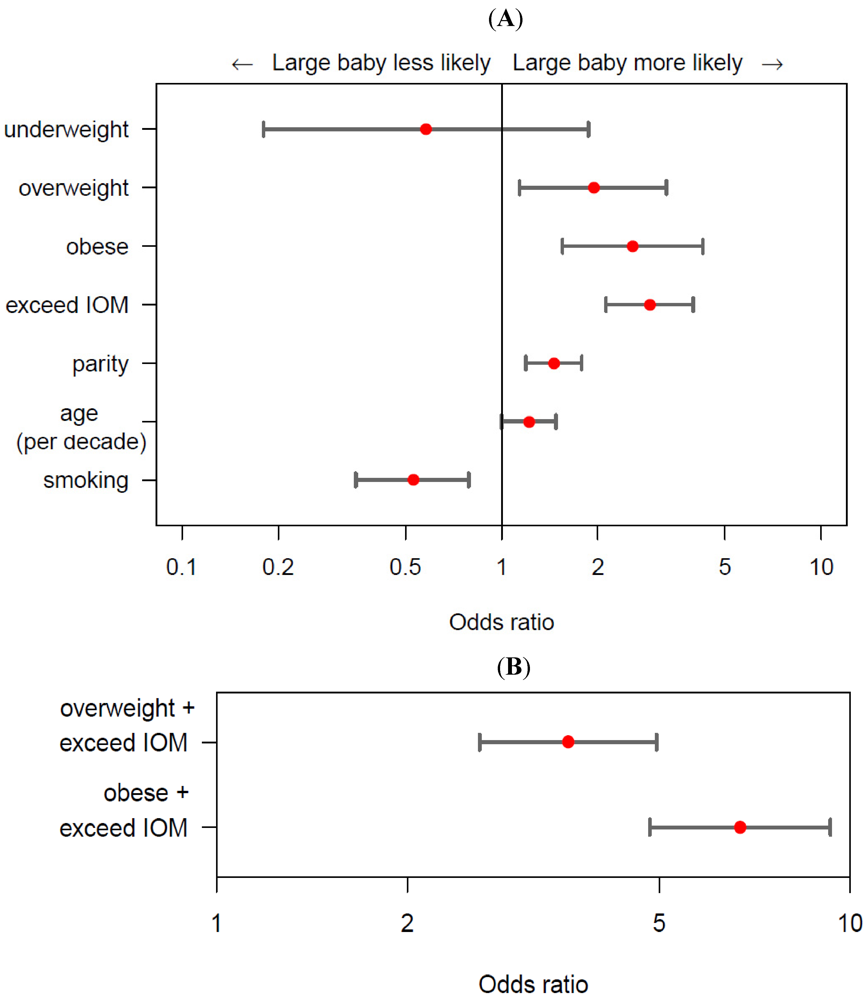

2.3. Gestational Diabetes
2.4. Importance of the Intrauterine Environment
2.5. Epigenetics
3. What Can We Do About It?
3.1. Modifiable Targets—Importance of Physical Activity & Nutrition
3.2. Pregnancy Specific Interventions
| Author | Population | Objective | Intervention | Primary Outcome | Findings: Maternal Outcome | Findings: Neonatal Outcome |
|---|---|---|---|---|---|---|
| 27 RCTs | ||||||
| Rae 2000 [191] f | GDM population Australia 110% ideal BMI n (I) = 66 n (C) = 58 | To identify if treatment of obese women with GDM could reduce insulin therapy and incidence of macrosomia | Nutrition Energy restriction diet (70% of recommended intake) | Need for maternal insulin therapy & infant macrosomia | No difference in requirement for insulin (but trend toward need later in pregnancy and for lower dose in intervention) | No difference in BW |
| Clapp 2000 [176] h | Sedentary, non-overweight n (I) = 22 n (C) = 24 | To identity the effect of beginning moderate-intensity exercise in early pregnancy on fetoplacental growth | Exercise 20 min of monitored, weight-bearing activity 3–5 times/wk @ 55–60% of VO2max (treadmill, step aerobics or stair stepper) | Antenatal placental growth Neonatal and placental morphometry | No difference in GWG | No difference in gestational age. BW & length > in exercise group because of > lean body mass, lower % BF Placenta: exercisers > growth rate & volume, and > functional volume |
| Marquez-Sterling 2000 [192] h | Sedentary non-obese primigravida USA n (I) = 9 n (C) = 6 | To examine the effects of exercise on physical and psychological variables | Exercise 3-1 h supervised sessions/wk ‘aggressive’ aerobic training; combination of rowing, cycling, walking/jogging, rhythmic calisthetics and step classes | Significant improvement in aerobic fitness (p = 0.035) Improvement in several scores on the Body Cathexis Scale (p < 0.05) No difference in GWG or body composition | No difference in BW or APGAR | |
| Polley 2002 [193] a,b,c,d,e,f,i | Low-income USA BMI > 19.8 Age > 18 years n (I) = 57 n (C) = 53 | To determine whether a stepped care, behavioral intervention will decrease the percentage of women who exceed the 1990 IOM GWG recommendation. | Nutrition & Exercise Stepped-care behavioural counseling sessions at prenatal appointments re: recommended GWG, nutrition & exercise. Provision of personalised graph of weight gain trajectory.Bi-weekly education re: healthy eating and exercise delivered via mail | Reduce proportion of women who exceed GWG recommendations | Overall no significant difference. Normal weight subgroup: significant reduction in GWG reduction in those exceeding 1990 IOM recommendations (p < 0.05). Overweight subgroup: Opposite trend overweight women (32 versus 59%, p = 0.09). | No difference in BW or complications during pregnancy/delivery |
| Bechtel-Blackwell 2002 [194] b | African-American teens USA Age 13–18 years n (I) = 22 n (C) = 24 | To conduct computer-assisted self-interview (CASI) nutrition assessment in pregnant, adolescents to compare the effect of a nutrition education intervention with the standard dietitian consult on GWG patterns and postpartum weight retention. | Nutrition Patient education. Group sessions. Repeated nutritional assessment. | Reduction in GWG and PPWR at 6 weeks | 1st trimester; less GWG (p < 0.000) 2nd trimester; no difference (p = 0.056) 3rd trimester; higher GWG (p < 0.006) higher PPWR in control group at 6 weeks (p < 0.0024) | |
| Prevedel 2003 [195] h Prospective, random cohort study | low-risk nulliparous Brazil n (I)=22 n (C ) =19 | Aimed to study maternal (body composition andcardiovascular capacity) and perinatal (weight and prematurity) effects of hydrotherapy during pregnancy | Exercise Hydrotherapy throughout gestation | Maternal body composition and cardiovascular capacity. Perinatal weight and Prematurity. | Intervention group maintained their fat index and VO2 max. Control group increase their fat and saw a reduction in VO2max. | No difference in prematurity or weight loss in newborns |
| Barakat 2008 [196] | Sedentary gravidae Caucasian Spain n (I) = 72 n (C ) = 70 | This study aimed to determine the possible cause–effect relationship between regular exercise during the 2nd and3rd trimesters of pregnancy by previously sedentary, healthy gravidae and gestational age at the moment of delivery | Exercise The supervised training programme focused mainly on very light resistance and toning exercises and included ,80 sessions (three times/week, 35 min/session from weeks 12–13 to weeks 38–39 of pregnancy) | Risk of preterm delivery and neonatal APGAR scores | no difference in gestational age or APGAR scores | |
| Barakat 2009 [167,197] h | Sedentary gravidae Spain n (I) =80 n (C ) = 80 | Examined the effect of light-intensity resistance exercise training performed during the 2nd and3rd trimester of pregnancy by previously sedentary and healthy women on the type of delivery and on the dilation, expulsion, and childbirth time [197] and birth size [167] | Exercise The training programme focused on light resistance and toning exercises (3 times/wk, 35–40 min per session) | Main outcomes were maternal and newborn characteristics, the type of delivery (normal, instrumental, or cesarean), and dilation, expulsion, childbirth time and neonatal size at birth | No difference between groups with regard to delivery type (normal, instrumental, or cesarean) The mean dilation, expulsion, and childbirth time did not differ between groups | No differences between control and intervention in Apgar score, BW, birth length, and head circumference of the newborn |
| Santos 2005 [198] h | OW-BMI 25-30 Brazil n (I) = 37 n (C) = 35 | To evaluate the effect of aerobic training on submaximal cardiorespiratory capacity in overweight pregnant women | Exercise 3- 1 h aerobic exercise session/wk @ 50-60% max predicted HR never exceeding 140 bpm | Cardiorespiratory fitness | Improvement in VO2 at aerobic threshold (p <0.002) Improvement in ventilation at aerobic threshold (p = 0.02) No difference in weight or GWG | No difference in BW, prevalence of low BW, premature birth, APGAR |
| Garshasbi 2005 [199] h | Primigravida Mean BMI ~ 26 Iran n (I) = 107 n (C) = 105 | To investigate the effect of exercise on the intensity of low back pain and kinematics of the spine | Exercise 1 h supervised program 3 ×’s /wk @ < 140 bpm included walking, anaerobic exercise, and other specific strengthening exercises | Prevention or reduction of low back pain | Sign difference in intensity of low back pain favouring exercise Sign reduction in flexibility of spine in both groups but greater reduction in exercising group No difference in GWG or length of pregnancy | No difference in BW |
| Hui 2006 [200] a,b,c,d,i | Socioeconomically deprived women in urban core Canada n (I) = 24 n (C) = 21 | To deter mine the feasibility of implementing a community based exercise/dietary intervention program aiming to reduce risks of obesity and diabetes | Nutrition & Exercise Group exercise sessions and home-based exercise (3–5 ×/week for 30–45 min per session) also recommended. Video exercise instruction was provided to assist.Intervention also included computer-assisted Food Choice Map dietary interviews and counselling | Improve pregnancy outcomes | No significant difference in GWG or adherence to guidelines PA level sign higher (p = 0.005) | No difference in BW |
| Wolff 2008 [201] a,b,d,g,i | Caucasian, non smoking Denmark. BMI > 30 n (I) = 23 n (C) = 27 | To investigate whether restriction of GWG in obese women can be achieved via diet counseling | Nutrition Individual dietary consultations on 10 separate occasions during pregnancy. Healthful diet instruction and restriction of energy intake | Reduction in pregnancy induced increases in insulin, leptin and glucose | Less GWG in the intervention group (p = 0.002) lower energy intake (p = 0.001) less perturbation in insulin & leptin (p = 0.004) less PPWR in intervention (p = 0.003) | |
| Asbee 2009 [202] a,b,c,d,i | USA BMI < 40.5 Age 18–49 years n (I) = 57 n (C) = 43 | To estimate whether an organized, consistent program of dietary and lifestyle counseling prevents excessive GWG | Nutrition & Exercise 1× consultation with dietician in early pregnancy. (40% CHO, 30% PRO, 30% FAT) Information about IOM recommendations and weight grid provided. Moderate exercise recommended 3–5 ×/wk if not following guidelines—Diet & exercise regime reviewed and modified | Reduce proportion of women who exceed GWG recommendations | Intervention sign < GWG (p = 0.01) But no significant difference in adherence to IOM GWG recommendations (p = 0.21). No difference in preeclampsia, GDM | Trend for lower c-section rate in intervention (p = 0.09) Higher c-section rate in control due to ‘failure to progress’ |
| Jeffries 2009 [203] a | Australia n (I) = 125 n (C) = 111 | To asses effect of a personalized GWG recommendation with regular measurement on GWG | Women were given optimal GWG range and asked to self-monitor weight at various time points over course of pregnancy | Reduce excessive GWG | Reduced GWG in OW women (p = 0.01) No difference in adherence to 1990 GWG guidelines | No difference in gestational age, BW, complications or APGAR score |
| Thornton 2009 [204] g,i | OB-BMI > 30 USA n (I) = 116 n (C)= 116 | To assess effect o nutritional intervention (energy restriction) on perinatal outcomes. | Nutrition Balanced dietary program with energy restriction and food diary monitoring (18 to 24 kcal/kg balanced nutritional regimen, consisting of 40% CHO, 30% PRO, and 30% FAT; not < 2000 kcal/day) | To reduce negative perinatal outcomes | Reduced GWG (p < 0.001) Reduced gestational hypertension, p < 0.046 less 6-week PPWR p < 0.001 no difference in preeclampsia or GDM | No difference in BW, macrosomia, c-section, APGAR score |
| Landon 2009 [205] | Mild GDM USA n (I) = 485 n (C) = 473 | to determine whether treatment of women with mild GDM reduces perinatal and obstetrical complications | Nutrition Formal nutrition counseling and diet therapy, as per the American Diabetes Association’s recommendations and interventions for diabetes. Self-monitoring of blood glucose, and insulin therapy (if necessary) | composite of stillbirth or perinatal death and neonatal complications, including hyperbilirubinemia, hypoglycemia, hyperinsulinemia, and birth trauma | Fewer cesarean deliveries in the treatment group. Lower frequency of pre-eclampsia and gestational hypertension in the treatment group. BMI at delivery and GWG was lower in the treatment group | No significant difference between the groups in the frequency of composite primary perinatal outcome. Mean BW, neonatal fat mass and frequency of LGA and macrosomia was significantly reduced in the treatment group |
| Baciuk 2008 Cavalcante 2009 [206,207] h | Low-risk sedentary Brazil n (I) = 34 n (C) = 37 | To evaluate the effectiveness and safety of a water aerobics program for low risk, sedentary pregnant women on the maternal cardiovascular capacity during pregnancy, labor and neonatal outcomes evolution of pregnancy | Exercise regular, moderate practice of water aerobics for 50 min, 3 ×/wk @ 70% of predicted max HR | Maternal BMI, GWG, blood pressure, cardiovascular capacity, labour type and duration, mode of delivery and neonatal outcomes (BW, viability) | No difference in GWG, maternal BMI, or % body fat, blood pressure, heart rate, maternal cardiovascular capacity, duration of labour, or the type of delivery between the two groups | No differences in incidence of preterm birth, vaginal births, low BW, or adequate weight for gestation |
| Ong 2009 [148] h | Sedentary, OB women Australia n (I) = 6 n (C ) =6 | To investigate the effect of a supervised 10-week, home-based, exercise programme, beginning at week 18 of gestation, on glucose tolerance and aerobic fitness | Exercise Intervention—10 weeks of supervised home-based exercise - 3 sessions/wk of stationary cycling.10 min warm-up followed by one or two 15 min bouts of cycling (with rest periods if necessary) at an intensity of 50–60% HRmax. As the weeks progressed, the exercise intensity was increased to 60–70% HRmax, while the duration was increased to 40–45 min | Glucose and insulin responses to an oral glucose tolerance test (OGTT), as well as their aerobic fitness | Exercise had favourable effects on glucose tolerance and fitness in obese pregnant women compared to control | |
| Guelinckx 2010 [208] a,c,d,g,i 3-arm RCT (passive vs. Active vs. Control) | BMI > 29 Non-diabetic Belgium n (I passive) = 65 n (I active) = 65 n (C) = 65 | To study whether a lifestyle intervention based on a brochure or on active education can improve dietary habits, increase PA, and reduce GWG in obese pregnant women | Nutrition & Exercise Information and counseling re: PA during pregnancy. Group nutritional counseling about healthful eating and nutritionally sound substitutions | Reduction in GWG | No significant difference in GWG or adherence to guidelines | No difference in BW, LGA, c-section rate or infant length |
| Hopkins 2010 [209] h | Nulliparous aged 20–40 yrs New Zealand n (I ) = 47 n (C) = 37 | To determine the effects of aerobic exercise training in the second half of pregnancy on maternal insulin sensitivity and neonatal outcomes | Exercise home-based stationary cycling 5 ×/week, 40 minutes/session from 20 wk gestation to delivery | Maternal insulin sensitivity, neonatal auxology, body composition, and growth-related peptides in cord blood | No difference in maternal insulin sensitivity | lower birth weight (p < 0.03) and body mass index at birth (p < 0.04). Exercise offspring had lower cord serum IGF-I (p < 0.03) and IGF-II (p < 0.04) |
| Korpi-Hyovalti 2011 [210] | At risk of GDM Finland n (I) = 27 n (C) = 27 | To evaluate if a lifestyle intervention during pregnancy is feasible in improving the glucose tolerance of women at a high-risk for GDM | Nutrition & Exercise Diet: 50–55% carbohydrate, 15 g fibre/1000kcal, fat 30%, protein 15–20%. 30 kcal/kg/day for normal weight, and 25 kcal/kg/day for OW. Exercise: moderate intensity PA was encouraged during pregnancy and 6 appointments with a physiotherapist to encourage PA. | Maternal glucose tolerance, the incidence of GDM and perinatal complications. | No differences in change in glucose tolerance from baseline to weeks 26–28 of gestation. Trend towards less GWG in the intervention. | Mean BW was higher in the intervention group, but not difference in macrosomia. No differences in neonatal outcomes. |
| Hui 2011 [211] | Non-diabetic, urban-living Canada <26 wks N (I) = 102 N (C) = 88 | To examine the effect of an exercise and dietary intervention during pregnancy on excessive GWG, dietary habits and PA habits | Nutrition & Exercise Provided with community-based group exercise sessions, instructed home exercise (total of 3–5 ×/wk) and 2 dietary counseling sessions (upon enrolment and 2 months in) | Reduce prevalence of excessive GWG, levels of PA and dietary intake | After 2 months the intervention group reported lower daily intake of calories, fat, sat. fat, chol. (p < 0.01) and higher PA compared with control (p < 0.01) Lifestyle intervention reduced excessive GWG (p < 0.01) | |
| Luoto 2011 [212] Cluster RCT | BMI ≥ 25, or GDM or previous macrosomic newborn Finland n (I) = 219 n (C) = 180 | To examine if GDM or high BW can be prevented by lifestyle counseling in high risk women. | Nutrition & Exercise Individualized counseling on PA (to meet recommendations of 800 MET minutes/wk), healthful diet (high fibre, low fat, low sugar choices) , and GWG at 5 antenatal visits | Incidence of GDM and LGA neonate | No difference in incidence of GDM (ES 1.36, 95% CI: 0.71–2.62, p = 0.36) | Lower BW (p = 0.008) and proportion of LGA neonates (p = 0.042) |
| Phelan 2011 [213] i | Normal weight or OW/OB USA n (I) = 182 n (C) = 176 | To examine if a behavioural intervention could reduce the number of women exceeding 1990 GWG guidelines and increase the number of women returning to pregravid weight by 6 months post-partum | Nutrition & Exercise One face-to-face visit, weekly mailed educations material promoting appropriate GWG, healthy eating and exercise. After each clinic visit individual GWG graphs were provided and 3, 10-15 min telephone calls from dietitian. Additional calls were placed to those not on track with GWG guidelines | Reduce prevalence of excessive GWG and PPWR | Reduced number of normal weight women exceeded GWG guidelines (p = 0.003) Increased number of normal and overweight/obese women who return to the pregravid weight (p = 0.005) | |
| Quinlivan 2011 [214] g | BMI ≥ 25 Australia n (I) = 63 n (C) = 61 | To evaluate whether a 4-step multidisciplinary protocol of antenatal care for OW and OB women would reduce the incidence of GDM | 1. Continuity of care by a single maternity care provider; 2. assessing weight gain at each antenatal visit; 3. brief intervention (5 min) by a food tech before each visit; 4. assess by clinical psych, if difficulties identified, an individualized solution-focused treatment plan was implemented. | Reduce prevalence of combined diagnoses of decreased gestational glucose tolerance and GDM. | Intervention was associated with a sign reduction in incidence of GDM (OR 0.17 95% CI 0.03–0.95, p = 0.04).Intervention also assoc with reduced GWG (p < 0.0001) | No difference in BW (p = 0.16) |
| Nascimento 2011 [215] | OW/OB-BMI ≥ 26 Gest age: 14-24 wks Brazil n (I) = 40 n (C) = 42 | To evaluate the effectiveness and safety of physical exercise in terms of maternal/ perinatal outcomes and the perception of quality of life (QoL) | Exercise Weekly exercise class under supervision and received home exercise counseling to be performed 5 ×/wk | Reduction of GWG and proportion exceeding the GWG guidelines. | No difference in absolute GWG or numbers exceeding guidelines (47 vs. 57%). No difference in QoL The overweight women in the intervention gained sign. less weight (p = 0.001) | |
| Haakstad 2011 [216,217] | Sedentary, nulliparous Norway n (I) = 52 n (C) = 53 | To examine the effect of a supervised exercise-program on birth weight, gestational age at delivery and Apgar-score | Exercise - supervised aerobic dance and strength training : 60 minutes, 2 ×/wk for a minimum of 12 wks, + 30 min of self-imposed PA on the non-supervised days. All aerobic activities were performed at moderate intensity or RPE of 12–14 (somewhat hard) on Borg’s scale | BW, gestational age at delivery and APGAR-score | More women in the intervention met GWG guidelines Intervention participants who attended 24 exercise sessions (n = 14) differed significantly from controls with regard to weight gain during pregnancy (p < 0.01) and postpartum weight retention (p < 0.01) | Intervention was not associated with reduction in BW, preterm birth rate or neonatal well-being |
| Vinter 2011 [218] | Obese, BMI 30–45 Denmark n (I) = 150 n (C) = 154 | To study the effects of lifestyle intervention on gestational weight gain (GWG) and obstetric outcomes. | Nutrition & Exercise Individualized dietary counseling at 4 time points to assist in limiting GWG to 5 kg. Encouraged to engage in moderate PA 30–60 min daily. Were provided with a pedometer and a fitness membership for 6 months, which included private classes with an exercise specialist. Women also had 4–6 group meetings with specialist who assisted them with integrating of PA in pregnancy and daily life. | Obstetric and neonatal outcomes: GWG, preeclampsia, pregnancy-induced hypertension (PIH), GDM, cesarean section, macrosomia/large for gestational age (LGA), and admission to neonatal intensive unit. | Significantly lower GWG, p = 0.01 Trend for fewer intervention women to exceed IOM recommendations (35% vs. 47%, p = 0.058) No difference in c-section, pre-eclampsia/PIH, GDM | Higher BW in intervention group (3,742 vs. 3,593 g, p = 0.039) |
| 8 Non-RCTs | ||||||
| Gray-Donald 2000 [219] a,b,c Historical control | Cree First Nations population. Canada. n (I) = 112 n (C) = 107 | To evaluate an intervention aimed at improving dietary intake during pregnancy, optimizing GWG, glycemic levels and BW, and avoiding unnecessary PPWR | Nutrition & Exercise Exercise/walking groups. Nutrition information re: improving healthful food intake via radio broadcasts, booklets, supermarket tours and cooking demos | Improve dietary I/T, optimize GWG, glycemia, birthweight & PPWR | No sign difference in GWG, glycemic levels, or PPWR | No difference in BW |
| Olson 2004 [220] a,b,c Prospective cohort & Historical control | BMI 19.8–29.0 USA. Age > 18 years n (I) = 179 n (C) = 381 | To evaluate the efficacy of an intervention directed at preventing excessive GWG. | Nutrition & Exercise Education of healthcare providers. Personalized GWG grid. Participant education about PA by-mail. Dietary ‘health checkbook’ and self-monitoring tips and newsletters | Prevention of excessive GWG | No overall significant difference in GWG (p = 0.3). Significant difference in GWG and adherence to guidelines in ‘low-income’ subgroup (p = 0.01). Less PPWR in low income OW subgroup | No difference in infant BW |
| Kinnunen 2007 [221] a,b,c,i Controlled trial | Primipara Finland Age > 18 years n (I) = 49 n (C) = 56 | To investigate whether individual counselling on diet and physical activity during pregnancy can have positive effects on diet and leisure time physical activity and prevent excessive GWG | Nutrition & Exercise Information provided about GWG guidelines. Individual counseling concerning diet (4 sessions) and physical activity (5 sessions). Option to attend group classes | Improve diet and PA and prevention of GWG | No significant difference in total GWG (p = 0.77). No significant difference in proportion exceeding IOM recommendations (p = 0.053) | Significant difference in BW: 15% LGA in control vs. none in intervention (p = 0.006) |
| Claesson 2008 [222] a,b,c Prospective case-Historical control | OB-BMI > 30 Sweden n (I) = 155 n (C) = 193 | To minimize obese women’s GWG to less than 7 kg and to investigate the delivery and neonatal outcome | Nutrition & Exercise CBT Patient education and motivational interview. Frequent individual sessions. Weekly aqua aerobic exercise and information about nutrition during pregnancy | Reduce GWG to <7 kg | Significantly less weight gain in the intervention group (p < 0.001) Better adherence to GWG guidelines (p = 0.003). No difference in pregnancy outcomes | No difference in mode of delivery |
| Shirazian 2010 [223] c cohort-matched historical control | OB-BMI > 30 USA n (I) = 21 n (C) = 20 | To investigate if a comprehensive lifestyle modification program would limit GWG and reduce obesity-related complications | Nutrition & Exercise Written material, seminars, and counseling sessions for both encouraging walking (self monitor via pedometer), and healthful eating (food diary, calorie counting) | Reduce GWG | Significantly less GWG in intervention group (p = 0.003) | No difference in BW, gestational age at delivery, preeclampsia, gestational HTN, GDM, c-section, fetal complications and labour complications |
| Mottola 2010 [224] Single arm-historical matched control | OW/OB- BMI ≥ 25 Canada n (I) = 65 n (C) = 260 | To determine the effect of a nutrition and exercise program on GWG, birthweight, and PPWR. | Nutrition & Exercise Individualized nutrition plan with E/I~2000 kcal/d and walking program 3–4 ×/wk | Prevent excessive GWG, BW and PPWR | 80% of intervention women meet GWG recommendations 53% of NELIP women were within 2 kg of pre-pregnancy weight at 2 months post partum | No difference in BW |
| Lindholm 2010 [225] Prospective intervention No control group | OB-BMI > 30 n = 27 | To control GWG among obese women by a dietary and physical activity program | Nutrition & Exercise - meeting with midwife bi-weekly - 2 support group sessions - 1 dietary consultation - food diaries & PA diaries - aqua fitness class 1×/wk and encouraged to exercise for 30 min on the other days | To limit GWG to ≤6 kg | - 56% met the goal of ≤6 kg | All AGA babies |
| Artal 2007 [145] Prospective intervention (self-enrolled) | OB with GDM USA n (Ex+Diet) = 39 n (Diet) n= 57 | To examine whether weight gain restriction, with or without exercise, would impact glycemic control, pregnancy outcome and total GWG | Nutrition & Exercise All patients were provided a eucaloric or hypocaloric consistent carbohydrate meal plan and instructed in self-monitoring blood glucose. Exercise and diet group prescribed an exercise routine equal to 60% symptom-limited VO2max (1 time/wk supervised in the lab and 6 days/wk independently) | Improved glycemic control, pregnancy outcome and total GWG | Weight gain was significantly lower in subjects in the exercise and diet group No difference in complications or c-section delivery | No difference in gestation age. Fewer macrosomic neonates in moms who restricted intake and exercised |
3.3. Systematic Reviews
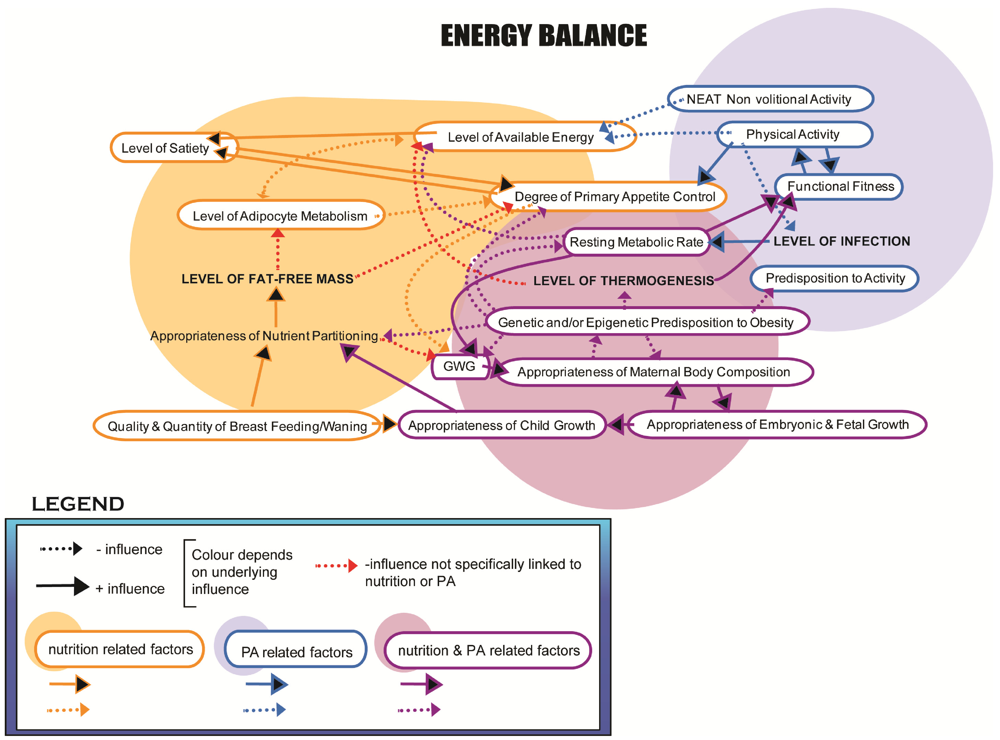
4. Novel Mechanisms
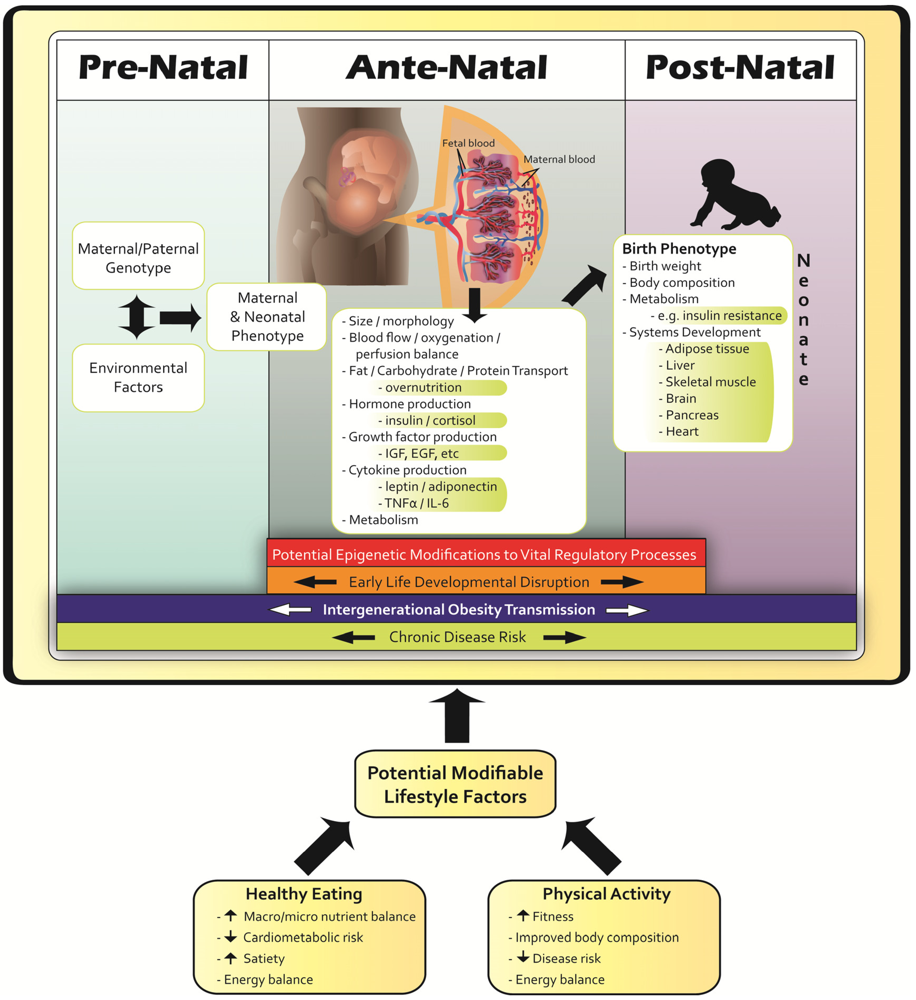
5. Conclusions
Acknowledgments
Conflict of Interest
References
- Thow, A.M.; Jan, S.; Leeder, S.; Swinburn, B. The effect of fiscal policy on diet, obesity and chronic disease: A systematic review. Bull. World Health Organ. 2010, 88, 609–614. [Google Scholar]
- Shields, M. Overweight and obesity among children and youth. Health Rep. 2006, 17, 27–42. [Google Scholar]
- Freedman, D.S.; Khan, L.K.; Dietz, W.H.; Srinivasan, S.R.; Berenson, G.S. Relationship of childhood obesity to coronary heart disease risk factors in adulthood: The Bogalusa Heart Study. Pediatrics 2001, 108, 712–718. [Google Scholar]
- Magarey, A.M.; Daniels, L.A.; Boulton, T.J.; Cockington, R.A. Predicting obesity in early adulthood from childhood and parental obesity. Int. J. Obes. Relat. Metab. Disord. 2003, 27, 505–513. [Google Scholar] [Green Version]
- Must, A. Does overweight in childhood have an impact on adult health? Nutr. Rev. 2003, 61, 139–142. [Google Scholar] [CrossRef]
- Freedman, D.S.; Dietz, W.H.; Srinivasan, S.R.; Berenson, G.S. The relation of overweight to cardiovascular risk factors among children and adolescents: The Bogalusa Heart Study. Pediatrics 1999, 103, 1175–1182. [Google Scholar]
- Fagot-Campagna, A. Emergence of type 2 diabetes mellitus in children: Epidemiological evidence. J. Pediatr. Endocrinol. Metab. 2000, 13, 1395–1402. [Google Scholar]
- Sinha, R.; Fisch, G.; Teague, B.; Tamborlane, W.V.; Banyas, B.; Allen, K.; Savoye, M.; Rieger, V.; Taksali, S.; Barbetta, G.; et al. Prevalence of impaired glucose tolerance among children and adolescents with marked obesity. N. Engl. J. Med. 2002, 346, 802–810. [Google Scholar]
- Eminoglu, T.F.; Camurdan, O.M.; Oktar, S.O.; Bideci, A.; Dalgic, B. Factors related to non-alcoholic fatty liver disease in obese children. Turk. J. Gastroenterol. 2008, 19, 85–91. [Google Scholar]
- Tominaga, K.; Fujimoto, E.; Suzuki, K.; Hayashi, M.; Ichikawa, M.; Inaba, Y. Prevalence of non-alcoholic fatty liver disease in children and relationship to metabolic syndrome, insulin resistance, and waist circumference. Environ. Health Prev. Med. 2009, 14, 142–149. [Google Scholar]
- Daniels, S.R. The consequences of childhood overweight and obesity. Future Child. 2006, 16, 47–67. [Google Scholar]
- Kim, S.Y.; Dietz, P.M.; England, L.; Morrow, B.; Callaghan, W.M. Trends in pre-pregnancy obesity in nine states, 1993–2003. Obesity (Silver Spring) 2007, 15, 986–993. [Google Scholar] [CrossRef]
- Robinson, H.E.; O'Connell, C.M.; Joseph, K.S.; McLeod, N.L. Maternal outcomes in pregnancies complicated by obesity. Obstet. Gynecol. 2005, 106, 1357–1364. [Google Scholar]
- Yogev, Y.; Catalano, P.M. Pregnancy and obesity. Obstet. Gynecol. Clin. North Am. 2009, 36, 285–300, viii.. [Google Scholar]
- Hedley, A.A.; Ogden, C.L.; Johnson, C.L.; Carroll, M.D.; Curtin, L.R.; Flegal, K.M. Prevalence of overweight and obesity among U.S. children, adolescents, and adults, 1999–2002. JAMA 2004, 291, 2847–2850. [Google Scholar]
- Department of Health, Health Survey for England 2002- Latest Trends; DoH: London, UK, 2003.
- Ferraro, Z.M.; Barrowman, N.; Prud'homme, D.; Walker, M.; Wen, S.W.; Rodger, M.; Adamo, K.B. Excessive gestational weight gain predicts large for gestational age neonates independent of maternal body mass index. J Matern.-Fetal Neonatal. Med. 2011, 1–5, Early Online PMID: 22081936. [Google Scholar]
- Gillman, M.W.; Rifas-Shiman, S.; Berkey, C.S.; Field, A.E.; Colditz, G.A. Maternal gestational diabetes, birth weight, and adolescent obesity. Pediatrics 2003, 111, 221–226. [Google Scholar]
- Parsons, T.J.; Power, C.; Manor, O. Fetal and early life growth and body mass index from birth to early adulthood in 1958 British cohort: Longitudinal study. Br. Med. J. 2001, 323, 1331–1335. [Google Scholar]
- Whitaker, R.C. Predicting preschooler obesity at birth: the role of maternal obesity in early pregnancy. Pediatrics 2004, 114, 29–36. [Google Scholar]
- Oken, E.; Taveras, E.M.; Kleinman, K.P.; Rich-Edwards, J.W.; Gillman, M.W. Gestational weight gain and child adiposity at age 3 years. Am. J. Obstet. Gynecol. 2007, 196, 322–328. [Google Scholar]
- Catalano, P.M.; Farrell, K.; Thomas, A.; Huston-Presley, L.; Mencin, P.; de Mouzon, S.H.; Amini, S.B. Perinatal risk factors for childhood obesity and metabolic dysregulation. Am. J. Clin. Nutr. 2009, 90, 1303–1313. [Google Scholar]
- Whitaker, R.C.; Dietz, W.H. Role of the prenatal environment in the development of obesity. J. Pediatr. 1998, 132, 768–776. [Google Scholar]
- Yu, Z.B.; Han, S.P.; Zhu, G.Z.; Zhu, C.; Wang, X.J.; Cao, X.G.; Guo, X.R. Birth weight and subsequent risk of obesity: A systematic review and meta-analysis. Obes. Rev. 2011, 12, 525–542. [Google Scholar]
- Salihu, H.M.; Weldeselasse, H.E.; Rao, K.; Marty, P.J.; Whiteman, V.E. The impact of obesity on maternal morbidity and feto-infant outcomes among macrosomic infants. J. Matern.-Fetal Neonatal. Med. 2011, 24, 1088–1094. [Google Scholar]
- Arendas, K.; Qiu, Q.; Gruslin, A. Obesity in pregnancy: Pre-conceptional to postpartum consequences. J. Obstet. Gynaecol. Can. 2008, 30, 477–488. [Google Scholar]
- Guelinckx, I.; Devlieger, R.; Beckers, K.; Vansant, G. Maternal obesity: Pregnancy complications, gestational weight gain and nutrition. Obes. Rev. 2008, 9, 140–150. [Google Scholar]
- Huda, S.S.; Brodie, L.E.; Sattar, N. Obesity in pregnancy: Prevalence and metabolic consequences. Semin. Fetal. Neonatal. Med. 2010, 15, 70–76. [Google Scholar]
- Mehta, S.H. Nutrition and pregnancy. Clin. Obstet. Gynecol. 2008, 51, 409–418. [Google Scholar]
- Satpathy, H.K.; Fleming, A.; Frey, D.; Barsoom, M.; Satpathy, C.; Khandalavala, J. Maternal obesity and pregnancy. Postgrad. Med. 2008, 120, E01–E09. [Google Scholar]
- Ong, K.K.; Ahmed, M.L.; Emmett, P.M.; Preece, M.A.; Dunger, D.B. Association between postnatal catch-up growth and obesity in childhood: Prospective cohort study. Br. Med. J. 2000, 320, 967–971. [Google Scholar]
- Ong, K.K. Size at birth, postnatal growth and risk of obesity. Horm. Res. 2006, 65, S65–S69. [Google Scholar]
- Toschke, A.M.; Beyerlein, A.; von Kries, R. Children at high risk for overweight: A classification and regression trees analysis approach. Obes. Res. 2005, 13, 1270–1274. [Google Scholar]
- Reilly, J.J.; Armstrong, J.; Dorosty, A.R.; Emmett, P.M.; Ness, A.; Rogers, I.; Steer, C.; Sherriff, A. Early life risk factors for obesity in childhood: Cohort study. Br. Med. J. 2005, 330, 1357. [Google Scholar]
- Karaolis-Danckert, N.; Buyken, A.E.; Kulig, M.; Kroke, A.; Forster, J.; Kamin, W.; Schuster, A.; Hornberg, C.; Keil, T.; Bergmann, R.L.; Wahn, U.; Lau, S. How pre- and postnatal risk factors modify the effect of rapid weight gain in infancy and early childhood on subsequent fat mass development: Results from the Multicenter Allergy Study 90. Am. J. Clin. Nutr. 2008, 87, 1356–1364. [Google Scholar]
- Nader, P.R.; O'Brien, M.; Houts, R.; Bradley, R.; Belsky, J.; Crosnoe, R.; Friedman, S.; Mei, Z.; Susman, E.J. Identifying risk for obesity in early childhood. Pediatrics 2006, 118, e594–e601. [Google Scholar]
- Guihard-Costa, A.M.; Papiernik, E.; Kolb, S. Maternal predictors of subcutaneous fat in the term newborn. Acta Paediatr. 2004, 93, 346–349. [Google Scholar]
- Hull, H.R.; Thornton, J.C.; Ji, Y.; Paley, C.; Rosenn, B.; Mathews, P.; Navder, K.; Yu, A.; Dorsey, K.; Gallagher, D. Higher infant body fat with excessive gestational weight gain in overweight women. Am. J. Obstet. Gynecol. 2011, 205, 211–217. [Google Scholar]
- Ludwig, D.S.; Currie, J. The association between pregnancy weight gain and birthweight: A within-family comparison. Lancet 2010, 376, 984–990. [Google Scholar]
- Schack-Nielsen, L.; Michaelsen, K.F.; Gamborg, M.; Mortensen, E.L.; Sorensen, T.I. Gestational weight gain in relation to offspring body mass index and obesity from infancy through adulthood. Int. J. Obes. (Lond.) 2010, 34, 67–74. [Google Scholar] [CrossRef]
- Wrotniak, B.H.; Shults, J.; Butts, S.; Stettler, N. Gestational weight gain and risk of overweight in the offspring at age 7 y in a multicenter, multiethnic cohort study. Am. J. Clin. Nutr. 2008, 87, 1818–1824. [Google Scholar]
- Institute of Medicine, Weight Gain During Pregnancy: Reexamining the Guidelines; The National Academies Press: Washington, DC, USA, 2009.
- Kinnunen, T.I.; Luoto, R.; Gissler, M.; Hemminki, E. Pregnancy weight gain from 1960s to 2000 in Finland. Int. J. Obes. Relat. Metab. Disord. 2003, 27, 1572–1577. [Google Scholar]
- Weisman, C.S.; Hillemeier, M.M.; Downs, D.S.; Chuang, C.H.; Dyer, A.M. Preconception predictors of weight gain during pregnancy: Prospective findings from the Central Pennsylvania Women's Health Study. Womens Health Issues 2010, 20, 126–132. [Google Scholar]
- Olson, C.M.; Strawderman, M.S. Modifiable behavioral factors in a biopsychosocial model predict inadequate and excessive gestational weight gain. J. Am. Diet. Assoc. 2003, 103, 48–54. [Google Scholar]
- Stotland, N.E.; Haas, J.S.; Brawarsky, P.; Jackson, R.A.; Fuentes-Afflick, E.; Escobar, G.J. Body mass index, provider advice, and target gestational weight gain. Obstet. Gynecol. 2005, 105, 633–638. [Google Scholar]
- Wells, C.S.; Schwalberg, R.; Noonan, G.; Gabor, V. Factors influencing inadequate and excessive weight gain in pregnancy: Colorado, 2000–2002. Matern. Child Health J. 2006, 10, 55–62. [Google Scholar]
- Gore, S.A.; Brown, D.M.; West, D.S. The role of postpartum weight retention in obesity among women: A review of the evidence. Ann. Behav. Med. 2003, 26, 149–159. [Google Scholar]
- Cedergren, M. Effects of gestational weight gain and body mass index on obstetric outcome in Sweden. Int. J. Gynaecol. Obstet. 2006, 93, 269–274. [Google Scholar]
- Cedergren, M.I. Optimal gestational weight gain for body mass index categories. Obstet. Gynecol. 2007, 110, 759–764. [Google Scholar]
- Oken, E.; Kleinman, K.P.; Belfort, M.B.; Hammitt, J.K.; Gillman, M.W. Associations of gestational weight gain with short- and longer-term maternal and child health outcomes. Am. J. Epidemiol. 2009, 170, 173–180. [Google Scholar]
- Park, S.; Sappenfield, W.M.; Bish, C.; Salihu, H.; Goodman, D.; Bensyl, D.M. Assessment of the Institute of Medicine recommendations for weight gain during pregnancy: Florida, 2004–2007. Matern. Child Health J. 2011, 15, 289–301. [Google Scholar]
- Hinkle, S.N.; Sharma, A.J.; Dietz, P.M. Gestational weight gain in obese mothers and associations with fetal growth. Am. J. Clin. Nutr. 2010, 92, 644–651. [Google Scholar]
- Catalano, P.M.; Ehrenberg, H.M. The short- and long-term implications of maternal obesity on the mother and her offspring. BJOG 2006, 113, 1126–1133. [Google Scholar]
- Ekblad, U.; Grenman, S. Maternal weight, weight gain during pregnancy and pregnancy outcome. Int. J. Gynaecol. Obstet. 1992, 39, 277–283. [Google Scholar]
- Stevens-Simon, C.; McAnarney, E.R. Determinants of weight gain in pregnant adolescents. J. Am. Diet. Assoc. 1992, 92, 1348–1351. [Google Scholar]
- Ananth, C.V.; Wen, S.W. Trends in fetal growth among singleton gestations in the United States and Canada, 1985 through 1998. Semin. Perinatol. 2002, 26, 260–267. [Google Scholar]
- Choi, S.K.; Park, I.Y.; Shin, J.C. The effects of pre-pregnancy body mass index and gestational weight gain on perinatal outcomes in Korean women: A retrospective cohort study. Reprod. Biol. Endocrinol. 2011, 9, 6. [Google Scholar]
- Saxena, P.; Tyagi, S.; Prakash, A.; Nigam, A.; Trivedi, S.S. Pregnancy outcome of women with gestational diabetes in a tertiary level hospital of north India. Indian J. Community Med. 2011, 36, 120–123. [Google Scholar]
- Abrams, B.; Parker, J.D. Maternal weight gain in women with good pregnancy outcome. Obstet. Gynecol. 1990, 76, 1–7. [Google Scholar]
- Gunderson, E.P.; Abrams, B. Epidemiology of gestational weight gain and body weight changes after pregnancy. Epidemiol. Rev. 1999, 21, 261–275. [Google Scholar]
- Gunderson, E.P.; Abrams, B.; Selvin, S. The relative importance of gestational gain and maternal characteristics associated with the risk of becoming overweight after pregnancy. Int. J. Obes. Relat. Metab. Disord. 2000, 24, 1660–1668. [Google Scholar]
- Keppel, K.G.; Taffel, S.M. Pregnancy-related weight gain and retention: implications of the 1990 Institute of Medicine guidelines. Am. J. Public Health. 1993, 83, 1100–1103. [Google Scholar]
- McKeown, T.; Record, R.G. The influence of weight and height on weight changes associated with pregnancy in women. J. Endocrinol. 1957, 15, 423–429. [Google Scholar]
- Luke, B.; Hediger, M.L.; Scholl, T.O. Point of diminishing returns: When does gestational weight gain cease benefiting birthweight and begin adding to maternal obesity? J. Matern. Fetal Med. 1996, 5, 168–173. [Google Scholar]
- Catalano, P.M. Obesity and pregnancy—The propagation of a viscous cycle? J. Clin. Endocrinol. Metab. 2003, 88, 3505–3506. [Google Scholar] [CrossRef]
- Swinburn, B.A.; Sacks, G.; Hall, K.D.; et al. The global obesity pandemic: Shaped by global drivers and local environments. Lancet 2011, 378, 804–814. [Google Scholar]
- Shankar, K.; Harrell, A.; Liu, X.; Gilchrist, J.M.; Ronis, M.J.; Badger, T.M. Maternal obesity at conception programs obesity in the offspring. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2008, 294, R528–R538. [Google Scholar]
- Villamor, E.; Cnattingius, S. Interpregnancy weight change and risk of adverse pregnancy outcomes: A population-based study. Lancet 2006, 368, 1164–1170. [Google Scholar]
- Ehrlich, S.F.; Hedderson, M.M.; Feng, J.; Davenport, E.R.; Gunderson, E.P.; Ferrara, A. Change in body mass index between pregnancies and the risk of gestational diabetes in a second pregnancy. Obstet. Gynecol. 2011, 117, 1323–1330. [Google Scholar]
- Ehrenberg, H.M.; Huston-Presley, L.; Catalano, P.M. The influence of obesity and gestational diabetes mellitus on accretion and the distribution of adipose tissue in pregnancy. Am. J. Obstet. Gynecol. 2003, 189, 944–948. [Google Scholar]
- Sohlstrom, A.; Wahlund, L.O.; Forsum, E. Total body fat and its distribution during human reproduction as assessed by magnetic resonance imaging. Basic Life Sci. 1993, 60, 181–184. [Google Scholar]
- Kinoshita, T.; Itoh, M. Longitudinal variance of fat mass deposition during pregnancy evaluated by ultrasonography: The ratio of visceral fat to subcutaneous fat in the abdomen. Gynecol. Obstet. Invest. 2006, 61, 115–118. [Google Scholar]
- Denison, F.C.; Roberts, K.A.; Barr, S.M.; Norman, J.E. Obesity, pregnancy, inflammation, and vascular function. Reproduction 2010, 140, 373–385. [Google Scholar]
- Giguere, I.; Prud'homme, D.; Strychar, I.; Doucet, E.; Adamo, K.B. Gestational Weight gain, Post-partum Weight Loss or Retention, Body Composition and Metabolic profile of Pre-menopausal Women: A MONET Group Study. Am. J. Obstet. Gynecol. 2012. submitted for publication.. [Google Scholar]
- Lawlor, D.A.; Lichtenstein, P.; Fraser, A.; Langstrom, N. Does maternal weight gain in pregnancy have long-term effects on offspring adiposity? A sibling study in a prospective cohort of 146,894 men from 136,050 families. Am. J. Clin. Nutr. 2011, 94, 142–148. [Google Scholar]
- O'Brien, T.E.; Ray, J.G.; Chan, W.S. Maternal body mass index and the risk of preeclampsia: A systematic overview. Epidemiology 2003, 14, 368–374. [Google Scholar]
- Frederick, I.O.; Rudra, C.B.; Miller, R.S.; Foster, J.C.; Williams, M.A. Adult weight change, weight cycling, and prepregnancy obesity in relation to risk of preeclampsia. Epidemiology 2006, 17, 428–434. [Google Scholar]
- Rudra, C.B.; Williams, M.A.; Lee, I.M.; Miller, R.S.; Sorensen, T.K. Perceived exertion during prepregnancy physical activity and preeclampsia risk. Med. Sci. Sports Exerc. 2005, 37, 1836–1841. [Google Scholar]
- Sebire, N.J.; Jolly, M.; Harris, J.P.; Wadsworth, J.; Joffe, M.; Beard, R.W.; Regan, L.; Robinson, S. Maternal obesity and pregnancy outcome: A study of 287,213 pregnancies in London. Int. J. Obes. Relat. Metab. Disord. 2001, 25, 1175–1182. [Google Scholar]
- Nucci, L.B.; Schmidt, M.I.; Duncan, B.B.; Fuchs, S.C.; Fleck, E.T.; Santos Britto, M.M. Nutritional status of pregnant women: prevalence and associated pregnancy outcomes. Rev. Saude Publica 2001, 35, 502–507. [Google Scholar]
- Callaway, L.K.; Prins, J.B.; Chang, A.M.; McIntyre, H.D. The prevalence and impact of overweight and obesity in an Australian obstetric population. Med. J. Aust. 2006, 184, 56–59. [Google Scholar]
- Langer, O.; Yogev, Y.; Xenakis, E.M.; Brustman, L. Overweight and obese in gestational diabetes: the impact on pregnancy outcome. Am. J. Obstet. Gynecol. 2005, 192, 1768–1776. [Google Scholar]
- Langer, O.; Yogev, Y.; Most, O.; Xenakis, E.M. Gestational diabetes: the consequences of not treating. Am. J. Obstet. Gynecol. 2005, 192, 989–997. [Google Scholar]
- Catalano, P.M.; Hauguel-De, M.S. Is it time to revisit the Pedersen hypothesis in the face of the obesity epidemic? Am. J. Obstet. Gynecol. 2011, 204, 479–487. [Google Scholar] [CrossRef]
- Silverman, B.L.; Rizzo, T.A.; Cho, N.H.; Metzger, B.E. Long-term effects of the intrauterine environment. The Northwestern University Diabetes in Pregnancy Center. Diabetes Care 1998, 21 (Suppl. 2:B), 142–149. [Google Scholar]
- Kitajima, M.; Oka, S.; Yasuhi, I.; Fukuda, M.; Rii, Y.; Ishimaru, T. Maternal serum triglyceride at 24–32 weeks' gestation and newborn weight in nondiabetic women with positive diabetic screens. Obstet. Gynecol. 2001, 97, 776–780. [Google Scholar]
- Knopp, R.H.; Bergelin, R.O.; Wahl, P.W.; Walden, C.E. Relationships of infant birth size to maternal lipoproteins, apoproteins, fuels, hormones, clinical chemistries, and body weight at 36 weeks gestation. Diabetes 1985, 34, S71–S77. [Google Scholar]
- Nolan, C.J.; Riley, S.F.; Sheedy, M.T.; Walstab, J.E.; Beischer, N.A. Maternal serum triglyceride, glucose tolerance, and neonatal birth weight ratio in pregnancy. Diabetes Care 1995, 18, 1550–1556. [Google Scholar]
- Barker, D.J. The fetal and infant origins of adult disease. Br. Med. J. 1990, 301, 1111. [Google Scholar]
- Barker, D.J.; Bull, A.R.; Osmond, C.; Simmonds, S.J. Fetal and placental size and risk of hypertension in adult life. BMJ 1990, 301, 259–262. [Google Scholar]
- Barker, D.J. In utero programming of cardiovascular disease. Theriogenology 2000, 53, 555–574. [Google Scholar]
- Dabelea, D.; Hanson, R.L.; Lindsay, R.S.; et al. Intrauterine exposure to diabetes conveys risks for type 2 diabetes and obesity: A study of discordant sibships. Diabetes 2000, 49, 2208–2211. [Google Scholar] [CrossRef]
- Gluckman, P.D.; Hanson, M.A.; Cooper, C.; Thornburg, K.L. Effect of in utero and early-life conditions on adult health and disease. N. Engl. J. Med. 2008, 359, 61–73. [Google Scholar]
- Murtaugh, M.A.; Jacobs, D.R., Jr.; Moran, A.; Steinberger, J.; Sinaiko, A.R. Relation of birth weight to fasting insulin, insulin resistance, and body size in adolescence. Diabetes Care 2003, 26, 187–192. [Google Scholar]
- Wilkin, T.J.; Metcalf, B.S.; Murphy, M.J.; Kirkby, J.; Jeffery, A.N.; Voss, L.D. The relative contributions of birth weight, weight change, and current weight to insulin resistance in contemporary 5-year-olds: The EarlyBird Study. Diabetes 2002, 51, 3468–3472. [Google Scholar]
- Huang, R.C.; Burke, V.; Newnham, J.P.; et al. Perinatal and childhood origins of cardiovascular disease. Int. J. Obes. (Lond.) 2007, 31, 236–244. [Google Scholar] [CrossRef]
- Druet, C.; Ong, K.K. Early childhood predictors of adult body composition. Best Pract. Res. Clin. Endocrinol. Metab. 2008, 22, 489–502. [Google Scholar]
- Ozanne, S.E.; Fernandez-Twinn, D.; Hales, C.N. Fetal growth and adult diseases. Semin. Perinatol. 2004, 28, 81–87. [Google Scholar]
- Pettitt, D.J.; Jovanovic, L. Birth weight as a predictor of type 2 diabetes mellitus: The U-shaped curve. Curr. Diab. Rep. 2001, 1, 78–81. [Google Scholar]
- Wei, J.N.; Sung, F.C.; Li, C.Y.; et al. Low birth weight and high birth weight infants are both at an increased risk to have type 2 diabetes among schoolchildren in taiwan. Diabetes Care 2003, 26, 343–348. [Google Scholar] [CrossRef]
- Bayol, S.A.; Farrington, S.J.; Stickland, N.C. A maternal 'junk food' diet in pregnancy and lactation promotes an exacerbated taste for 'junk food' and a greater propensity for obesity in rat offspring. Br. J. Nutr. 2007, 98, 843–851. [Google Scholar]
- Bayol, S.A.; Simbi, B.H.; Bertrand, J.A.; Stickland, N.C. Offspring from mothers fed a 'junk food' diet in pregnancy and lactation exhibit exacerbated adiposity that is more pronounced in females. J. Physiol. 2008, 586, 3219–3230. [Google Scholar]
- Kral, J.G.; Biron, S.; Simard, S.; et al. Large maternal weight loss from obesity surgery prevents transmission of obesity to children who were followed for 2 to 18 years. Pediatrics 2006, 118, e1644–e1649. [Google Scholar] [CrossRef]
- Smith, J.; Cianflone, K.; Biron, S.; et al. Effects of maternal surgical weight loss in mothers on intergenerational transmission of obesity. J. Clin. Endocrinol. Metab. 2009, 94, 4275–4283. [Google Scholar]
- Gluckman, P.D.; Pinal, C.S. Regulation of fetal growth by the somatotrophic axis. J. Nutr. 2003, 133, 1741S–1746S. [Google Scholar]
- Gluckman, P.D.; Hanson, M.A. Developmental and epigenetic pathways to obesity: an evolutionary-developmental perspective. Int. J. Obes. (Lond.) 2008, 32, S62–S71. [Google Scholar] [CrossRef]
- Gluckman, P.D.; Lillycrop, K.A.; Vickers, M.H.; et al. Metabolic plasticity during mammalian development is directionally dependent on early nutritional status. Proc. Natl. Acad. Sci. USA 2007, 104, 12796–12800. [Google Scholar]
- McMillen, I.C.; Rattanatray, L.; Duffield, J.A.; et al. The early origins of later obesity: Pathways and mechanisms. Adv. Exp. Med. Biol. 2009, 646, 71–81. [Google Scholar] [CrossRef]
- Levin, B.E. Epigenetic influences on food intake and physical activity level: review of animal studies. Obesity (Silver Spring) 2008, 16, S51–S54. [Google Scholar] [CrossRef]
- Godfrey, K.M.; Sheppard, A.; Gluckman, P.D.; et al. Epigenetic gene promoter methylation at birth is associated with child's later adiposity. Diabetes 2011, 60, 1528–1534. [Google Scholar] [CrossRef]
- Cawley, J. The economics of childhood obesity. Health Aff. (Millwood) 2010, 29, 364–371. [Google Scholar] [CrossRef]
- Olson, C.M. Tracking of food choices across the transition to motherhood. J. Nutr. Educ. Behav. 2005, 37, 129–136. [Google Scholar]
- Phelan, S. Pregnancy: A "teachable moment" for weight control and obesity prevention. Am. J. Obstet. Gynecol. 2010, 202, 135–138. [Google Scholar]
- Boney, C.M.; Verma, A.; Tucker, R.; Vohr, B.R. Metabolic syndrome in childhood: Association with birth weight, maternal obesity, and gestational diabetes mellitus. Pediatrics 2005, 115, e290–e296. [Google Scholar]
- Finer, L.B.; Zolna, M.R. Unintended pregnancy in the United States: Incidence and disparities, 2006. Contraception 2011, 84, 478–485. [Google Scholar]
- Nohr, E.A.; Vaeth, M.; Baker, J.L.; Sorensen, T.I.; Olsen, J.; Rasmussen, K.M. Combined associations of prepregnancy body mass index and gestational weight gain with the outcome of pregnancy. Am. J. Clin. Nutr. 2008, 87, 1750–1759. [Google Scholar]
- World Health Organization, WHO European Ministerial Conference on Counteracting Obesity: Conference Report; World Health Organization Europe: Copenhagen, Denmark, 2007.
- Lau, D.C.; Douketis, J.D.; Morrison, K.M.; Hramiak, I.M.; Sharma, A.M.; Ur, E. 2006 Canadian clinical practice guidelines on the management and prevention of obesity in adults and children [summary]. Can. Med. Assoc. J. 2007, 176, S1–S13. [Google Scholar]
- Committee on the Impact of Pregnancy Weight on Maternal and Child Health, Influence of Pregnancy Weight on Maternal and Child Health: Workshop Report; The National Academies Press: Washington DC, USA, 2007.
- Butland, B.; Jebb, S.A.; Kopelman, P.; et al. Foresight. Tackling Obesities: Future Choices; Government Office for Science: London,UK, 2008.
- Krishnamoorthy, U.; Schram, C.M.; Hill, S.R. Maternal obesity in pregnancy: Is it time for meaningful research to inform preventive and management strategies? BJOG 2006, 113, 1134–1140. [Google Scholar] [CrossRef]
- Olafsdottir, A.S.; Skuladottir, G.V.; Thorsdottir, I.; Hauksson, A.; Steingrimsdottir, L. Combined effects of maternal smoking status and dietary intake related to weight gain and birth size parameters. BJOG 2006, 113, 1296–1302. [Google Scholar]
- King, J.C. Physiology of pregnancy and nutrient metabolism. Am. J. Clin. Nutr. 2000, 71, 1218S–1225S. [Google Scholar]
- Anderson, A.S. Symposium on 'nutritional adaptation to pregnancy and lactation'. Pregnancy as a time for dietary change? Proc. Nutr. Soc. 2001, 60, 497–504. [Google Scholar] [CrossRef]
- Weissgerber, T.L.; Wolfe, L.A. Physiological adaptation in early human pregnancy: Adaptation to balance maternal-fetal demands. Appl. Physiol. Nutr. Metab. 2006, 31, 1–11. [Google Scholar]
- Butte, N.F.; Wong, W.W.; Treuth, M.S.; Ellis, K.J.; O'Brian, S.E. Energy requirements during pregnancy based on total energy expenditure and energy deposition. Am. J. Clin. Nutr. 2004, 79, 1078–1087. [Google Scholar]
- Chen, C.Y.; Crott, J.; Liu, Z.; Smith, D.E. Fructose and saturated fats predispose hyperinsulinemia in lean male rat offspring. Eur. J. Nutr. 2010, 49, 337–343. [Google Scholar]
- Ghezzi, A.C.; Cambri, L.T.; Ribeiro, C.; Botezelli, J.D.; Mello, M.A. Impact of early fructose intake on metabolic profile and aerobic capacity of rats. Lipids Health Dis. 2011, 10, 3. [Google Scholar]
- Zhang, Z.Y.; Zeng, J.J.; Kjaergaard, M.; et al. Effects of a maternal diet supplemented with chocolate and fructose beverage during gestation and lactation on rat dams and their offspring. Clin. Exp. Pharmacol. Physiol. 2011, 38, 613–622. [Google Scholar]
- Laraia, B.A.; Bodnar, L.M.; Siega-Riz, A.M. Pregravid body mass index is negatively associated with diet quality during pregnancy. Public Health Nutr. 2007, 10, 920–926. [Google Scholar]
- Brock, K.; Huang, W.Y.; Fraser, D.R.; et al. Low vitamin D status is associated with physical inactivity, obesity and low vitamin D intake in a large U.S. sample of healthy middle-aged men and women. J. Steroid Biochem. Mol. Biol. 2010, 121, 462–466. [Google Scholar] [CrossRef]
- Mojtabai, R. Body mass index and serum folate in childbearing age women. Eur. J. Epidemiol. 2004, 19, 1029–1036. [Google Scholar]
- Maghbooli, Z.; Hossein-Nezhad, A.; Karimi, F.; Shafaei, A.R.; Larijani, B. Correlation between vitamin D3 deficiency and insulin resistance in pregnancy. Diabetes Metab. Res. Rev. 2008, 24, 27–32. [Google Scholar]
- Deierlein, A.L.; Siega-Riz, A.M.; Herring, A. Dietary energy density but not glycemic load is associated with gestational weight gain. Am. J. Clin. Nutr. 2008, 88, 693–699. [Google Scholar]
- Crowther, C.A.; Hiller, J.E.; Moss, J.R.; McPhee, A.J.; Jeffries, W.S.; Robinson, J.S. Effect of treatment of gestational diabetes mellitus on pregnancy outcomes. N. Engl. J. Med. 2005, 352, 2477–2486. [Google Scholar]
- Clapp, J.F.; Capeless, E. The changing glycemic response to exercise during pregnancy. Am. J. Obstet. Gynecol. 1991, 165, 1678–1683. [Google Scholar]
- Clapp, J.F., III. The effects of maternal exercise on early pregnancy outcome. Am. J. Obstet. Gynecol. 1989, 161, 1453–1457. [Google Scholar]
- Clapp, J.F., III. Exercise and fetal health. J. Dev. Physiol. 1991, 15, 9–14. [Google Scholar]
- Davies, G.A.; Wolfe, L.A.; Mottola, M.F.; MacKinnon, C. Joint SOGC/CSEP clinical practice guideline: Exercise in pregnancy and the postpartum period. Can. J. Appl. Physiol. 2003, 28, 330–341. [Google Scholar]
- Kramer, M.S.; McDonald, S.W. Aerobic exercise for women during pregnancy. Cochrane Database Syst. Rev. 2006, 3, CD000180. [Google Scholar]
- Ferraro, Z.M.; Gaudet, L.; Adamo, K.B. The potential impact of physical activity during pregnancy on maternal and neonatal outcomes. Obstet. Gynecol. Surv. 2012, 67, 99–110. [Google Scholar]
- Artal, R. Outcome of fetuses in women with pregestational diabetes mellitus. J. Perinat. Med. 2006, 34, 332. [Google Scholar]
- Dyck, R.; Klomp, H.; Tan, L.K.; Turnell, R.W.; Boctor, M.A. A comparison of rates, risk factors, and outcomes of gestational diabetes between aboriginal and non-aboriginal women in the Saskatoon health district. Diabetes Care 2002, 25, 487–493. [Google Scholar]
- Artal, R.; Catanzaro, R.B.; Gavard, J.A.; Mostello, D.J.; Friganza, J.C. A lifestyle intervention of weight-gain restriction: Diet and exercise in obese women with gestational diabetes mellitus. Appl. Physiol. Nutr. Metab. 2007, 32, 596–601. [Google Scholar]
- Dempsey, J.C.; Butler, C.L.; Sorensen, T.K.; et al. A case-control study of maternal recreational physical activity and risk of gestational diabetes mellitus. Diabetes Res. Clin. Pract. 2004, 66, 203–215. [Google Scholar] [CrossRef]
- Dempsey, J.C.; Sorensen, T.K.; Williams, M.A.; et al. Prospective study of gestational diabetes mellitus risk in relation to maternal recreational physical activity before and during pregnancy. Am. J. Epidemiol. 2004, 159, 663–670. [Google Scholar] [CrossRef]
- Ong, M.J.; Guelfi, K.J.; Hunter, T.; Wallman, K.E.; Fournier, P.A.; Newnham, J.P. Supervised home-based exercise may attenuate the decline of glucose tolerance in obese pregnant women. Diabetes Metab. 2009, 35, 418–421. [Google Scholar]
- Saftlas, A.F.; Logsden-Sackett, N.; Wang, W.; Woolson, R.; Bracken, M.B. Work, leisure-time physical activity, and risk of preeclampsia and gestational hypertension. Am. J. Epidemiol. 2004, 160, 758–765. [Google Scholar]
- Sorensen, T.K.; Williams, M.A.; Lee, I.M.; Dashow, E.E.; Thompson, M.L.; Luthy, D.A. Recreational physical activity during pregnancy and risk of preeclampsia. Hypertension 2003, 41, 1273–1280. [Google Scholar]
- Weissgerber, T.L.; Wolfe, L.A.; Davies, G.A. The role of regular physical activity in preeclampsia prevention. Med. Sci. Sports Exerc. 2004, 36, 2024–2031. [Google Scholar]
- Dempsey, J.C.; Butler, C.L.; Williams, M.A. No need for a pregnant pause: Physical activity may reduce the occurrence of gestational diabetes mellitus and preeclampsia. Exerc. Sport Sci. Rev. 2005, 33, 141–149. [Google Scholar]
- Osterdal, M.L.; Strom, M.; Klemmensen, A.K.; et al. Does leisure time physical activity in early pregnancy protect against pre-eclampsia? Prospective cohort in Danish women. BJOG 2009, 116, 98–107. [Google Scholar] [CrossRef]
- Juhl, M.; Andersen, P.K.; Olsen, J.; et al. Physical exercise during pregnancy and the risk of preterm birth: A study within the Danish National Birth Cohort. Am. J. Epidemiol. 2008, 167, 859–866. [Google Scholar] [CrossRef]
- Penney, D.S. The effect of vigorous exercise during pregnancy. J. Midwifery Womens Health 2008, 53, 155–159. [Google Scholar]
- Artal, R. Exercise and pregnancy. Clin. Sports Med. 1992, 11, 363–377. [Google Scholar]
- Hopkins, S.A.; Cutfield, W.S. Exercise in pregnancy: Weighing up the long-term impact on the next generation. Exerc. Sport Sci. Rev. 2011, 39, 120–127. [Google Scholar]
- Florack, E.I.; Pellegrino, A.E.; Zielhuis, G.A.; Rolland, R. Influence of occupational physical activity on pregnancy duration and birthweight. Scand. J. Work Environ. Health 1995, 21, 199–207. [Google Scholar]
- Horns, P.N.; Ratcliffe, L.P.; Leggett, J.C.; Swanson, M.S. Pregnancy outcomes among active and sedentary primiparous women. J. Obstet. Gynecol. Neonatal Nurs. 1996, 25, 49–54. [Google Scholar]
- Jarrett, J.C.; Spellacy, W.N. Jogging during pregnancy: An improved outcome? Obstet. Gynecol. 1983, 61, 705–709. [Google Scholar]
- Klebanoff, M.A.; Shiono, P.H.; Carey, J.C. The effect of physical activity during pregnancy on preterm delivery and birth weight. Am. J. Obstet. Gynecol. 1990, 163, 1450–1456. [Google Scholar]
- Rabkin, C.S.; Anderson, H.R.; Bland, J.M.; Brooke, O.G.; Chamberlain, G.; Peacock, J.L. Maternal activity and birth weight: a prospective, population-based study. Am. J. Epidemiol. 1990, 131, 522–531. [Google Scholar]
- Rose, N.; Haddow, J.; Palomaki, G.; Knight, G. Self-rated physical activity level during the second trimester and pregnancy outcome. Obstet. Gynecol. 1991, 78, 1078–1080. [Google Scholar]
- Schramm, W.F.; Stockbauer, J.W.; Hoffman, H.J. Exercise, employment, other daily activities, and adverse pregnancy outcomes. Am. J. Epidemiol. 1996, 143, 211–218. [Google Scholar]
- Snyder, S.; Pendergraph, B. Exercise during pregnancy: What do we really know? Am. Fam. Physician 2004, 69, 1053–1056. [Google Scholar]
- Sternfeld, B.; Quesenberry, C.P., Jr.; Eskenazi, B.; Newman, L.A. Exercise during pregnancy and pregnancy outcome. Med. Sci. Sports Exerc. 1995, 27, 634–640. [Google Scholar]
- Barakat, R.; Lucia, A.; Ruiz, J.R. Resistance exercise training during pregnancy and newborn's birth size: A randomised controlled trial. Int. J. Obes. (Lond.) 2009, 33, 1048–1057. [Google Scholar] [CrossRef]
- Clapp, J.F., III. The effects of maternal exercise on fetal oxygenation and feto-placental growth. Eur. J. Obstet. Gynecol. Reprod. Biol. 2003, 110, S80–S85. [Google Scholar]
- Jackson, M.R.; Gott, P.; Lye, S.J.; Ritchie, J.W.; Clapp, J.F., III. The effects of maternal aerobic exercise on human placental development: Placental volumetric composition and surface areas. Placenta 1995, 16, 179–191. [Google Scholar]
- Juhl, M.; Olsen, J.; Anderson, P.; Nohr, E.; Anderson, A. Physical exercise during pregnancy and fetal growth measures: A study within the Danish National Birth Cohort. Am. J. Obstet. Gynecol. 2010, 202, e1–e8. [Google Scholar]
- Clapp, J.F., III.; Capeless, E. Neonatal morphometrics after endurance exercise during pregnancy. Am. J. Obstet. Gynecol. 1990, 163, 1805–1811. [Google Scholar]
- Bell, R.J.P. The effect of vigorous exercise during pregnancy on birth-weight. Aust. N. Z. J. Obstet. Gynaecol. 1995, 35, 46–51. [Google Scholar]
- Campbell, M.; Mottola, M.F. Recreational exercise and occupational activity during pregnancy and birth weight: A case-control study. Am. J. Obstet. Gynecol. 2001, 184, 403–408. [Google Scholar]
- Magann, E.; Evans, S.; Weitz, B.; Newnham, J. Antepartum, intrapartum, and neonatal significance of exercise on healthy low-risk pregnant working women. Obstet. Gynecol. 2002, 99, 466–472. [Google Scholar]
- Rao, S.; Kanade, A.; Margetts, B.M.; et al. Maternal activity in relation to birth size in rural India. The Pune Maternal Nutrition Study. Eur. J. Clin. Nutr. 2003, 57, 531–542. [Google Scholar] [CrossRef]
- Clapp, J.F.; Kim, H.; Burciu, B.; Lopez, B. Beginning regular exercise in early pregnancy: Effect on fetoplacental growth. Am. J. Obstet. Gynecol. 2000, 183, 1484–1488. [Google Scholar]
- Clapp, J.F.; Kim, H.; Burciu, B.; Schmidt, S.; Petry, K.; Lopez, B. Continuing regular exercise during pregnancy: Effect of exercise volume on fetoplacental growth. Am. J. Obstet. Gynecol. 2002, 186, 142–147. [Google Scholar]
- Artal, R.; O'Toole, M. Guidelines of the american college of obstetricians and gynecologists for exercise during pregnancy and the postpartum period. Br. J. Sports Med. 2003, 37, 6–12. [Google Scholar] [CrossRef]
- Mottola, M.F. Exercise prescription for overweight and obese women: Pregnancy and postpartum. Obstet. Gynecol. Clin. N. Am. 2009, 36, 301–316, viii.. [Google Scholar]
- Melzer, K.; Schutz, Y.; Boulvain, M.; Kayser, B. Physical activity and pregnancy: Cardiovascular adaptations, recommendations and pregnancy outcomes. Sports Med. 2010, 40, 493–507. [Google Scholar]
- Campbell, F.; Johnson, M.; Messina, J.; Guillaume, L.; Goyder, E. Behavioural interventions for weight management in pregnancy: A systematic review of quantitative and qualitative data. BMC Public Health 2011, 11, 491. [Google Scholar]
- Dodd, J.M.; Crowther, C.A.; Robinson, J.S. Dietary and lifestyle interventions to limit weight gain during pregnancy for obese or overweight women: A systematic review. Acta Obstet. Gynecol. Scand. 2008, 87, 702–706. [Google Scholar]
- Kuhlmann, A.K.; Dietz, P.M.; Galavotti, C.; England, L.J. Weight-management interventions for pregnant or postpartum women. Am. J. Prev. Med. 2008, 34, 523–528. [Google Scholar]
- Quinlivan, J.A.; Julania, S.; Lam, L. Antenatal dietary interventions in obese pregnant women to restrict gestational weight gain to institute of medicine recommendations: A meta-analysis. Obstet. Gynecol. 2011, 118, 1395–1401. [Google Scholar]
- Ronnberg, A.K.; Nilsson, K. Interventions during pregnancy to reduce excessive gestational weight gain: A systematic review assessing current clinical evidence using the Grading of Recommendations, Assessment, Development and Evaluation (GRADE) system. BJOG 2010, 117, 1327–1334. [Google Scholar]
- Skouteris, H.; Hartley-Clark, L.; McCabe, M.; et al. Preventing excessive gestational weight gain: A systematic review of interventions. Obes. Rev. 2010, 11, 757–768. [Google Scholar] [CrossRef]
- Streuling, I.; Beyerlein, A.; von Kries, R. Can gestational weight gain be modified by increasing physical activity and diet counseling? A meta-analysis of interventional trials. Am. J. Clin. Nutr. 2010, 92, 678–687. [Google Scholar]
- Streuling, I.; Beyerlein, A.; Rosenfeld, E.; Hofmann, H.; Schulz, T.; von Kries, R. Physical activity and gestational weight gain: A meta-analysis of intervention trials. BJOG 2011, 118, 278–284. [Google Scholar]
- Tanentsapf, I.; Heitmann, B.L.; Adegboye, A.R. Systematic review of clinical trials on dietary interventions to prevent excessive weight gain during pregnancy among normal weight, overweight and obese women. BMC Pregnancy Childbirth 2011, 11, 81. [Google Scholar]
- Birdsall, K.M.; Vyas, S.; Khazaezadeh, N.; Oteng-Ntim, E. Maternal obesity: A review of interventions. Int. J. Clin. Pract. 2009, 63, 494–507. [Google Scholar]
- Rae, A.; Bond, D.; Evans, S.F.; North, F.; Roberman, B.; Walters, B. A randomized controlled trial of dietary energy restriction in the management of obese women with gestational diabetes. ANZJOG 2008, 40, 416–422. [Google Scholar]
- Marquez-Sterling, S.; Perry, A.C.; Kaplan, T.A.; Halberstein, R.A.; Signorile, J.F. Physical and psychological changes with vigorous exercise in sedentary primigravidae. Med. Sci. Sports Exerc. 2000, 32, 58–62. [Google Scholar]
- Polley, B.A.; Wing, R.R.; Sims, C.J. Randomized controlled trial to prevent excessive weight gain in pregnant women. Int. J. Obes. Relat. Metab. Disord. 2002, 26, 1494–1502. [Google Scholar]
- Bechtel-Blackwell, D.A. Computer-assisted self-interview and nutrition education in pregnant teens. Clin. Nurs. Res. 2002, 11, 450–462. [Google Scholar]
- Prevedel, T.; Calderon, I.; DeConti, M.; Consonni, E.; Rudge, M. Maternal and perinatal effects of hydrotherapy in pregnancy. RGBO 2003, 25, 53–59. [Google Scholar]
- Barakat, R.; Stirling, J.R.; Lucia, A. Does exercise training during pregnancy affect gestational age? A randomised controlled trial. Br. J. Sports Med. 2008, 42, 674–678. [Google Scholar] [Green Version]
- Barakat, R.; Ruiz, J.R.; Stirling, J.R.; Zakynthinaki, M.; Lucia, A. Type of delivery is not affected by light resistance and toning exercise training during pregnancy: A randomized controlled trial. Am. J. Obstet. Gynecol. 2009, 201, 590–596. [Google Scholar]
- Santos, I.A.; Stein, R.; Fuchs, S.C.; et al. Aerobic exercise and submaximal functional capacity in overweight pregnant women: a randomized trial. Obstet. Gynecol. 2005, 106, 243–249. [Google Scholar] [CrossRef]
- Garshasbi, A.; Faghih, Z.S. The effect of exercise on the intensity of low back pain in pregnant women. Int. J. Gynaecol. Obstet. 2005, 88, 271–275. [Google Scholar]
- Hui, A.L.; Ludwig, S.; Gardiner, P.; et al. Community-based exercise and dietary intervention during pregnancy: A pilot study. Can. J. Diabetes 2006, 30, 169–175. [Google Scholar]
- Wolff, S.; Legarth, J.; Vangsgaard, K.; Toubro, S.; Astrup, A. A randomized trial of the effects of dietary counseling on gestational weight gain and glucose metabolism in obese pregnant women. Int. J. Obes. (Lond ) 2008, 32, 495–501. [Google Scholar] [CrossRef]
- Asbee, S.M.; Jenkins, T.R.; Butler, J.R.; White, J.; Elliot, M.; Rutledge, A. Preventing excessive weight gain during pregnancy through dietary and lifestyle counseling: A randomized controlled trial. Obstet. Gynecol. 2009, 113, 305–312. [Google Scholar]
- Jeffries, K.; Shub, A.; Walker, S.P.; Hiscock, R.; Permezel, M. Reducing excessive weight gain in pregnancy: A randomised controlled trial. Med. J. Aust. 2009, 191, 429–433. [Google Scholar]
- Thornton, Y.S. Preventing excessive weight gain during pregnancy through dietary and lifestyle counseling: A randomized controlled trial. Obstet. Gynecol. 2009, 114, 173–174. [Google Scholar]
- Landon, M.B.; Spong, C.Y.; Thom, E.; et al. A multicenter, randomized trial of treatment for mild gestational diabetes. N. Engl. J. Med. 2009, 361, 1339–1348. [Google Scholar] [CrossRef]
- Baciuk, E.P.; Pereira, R.I.; Cecatti, J.G.; Braga, A.F.; Cavalcante, S.R. Water aerobics in pregnancy: Cardiovascular response, labor and neonatal outcomes. Reprod. Health 2008, 5, 10. [Google Scholar]
- Cavalcante, S.R.; Cecatti, J.G.; Pereira, R.I.; Baciuk, E.P.; Bernardo, A.L.; Silveira, C. Water aerobics II: maternal body composition and perinatal outcomes after a program for low risk pregnant women. Reprod. Health 2009, 6, 1. [Google Scholar]
- Guelinckx, I.; Devlieger, R.; Mullie, P.; Vansant, G. Effect of lifestyle intervention on dietary habits, physical activity, and gestational weight gain in obese pregnant women: A randomized controlled trial. Am. J. Clin. Nutr. 2010, 91, 373–380. [Google Scholar]
- Hopkins, S.A.; Baldi, J.C.; Cutfield, W.S.; McCowan, L.; Hofman, P.L. Exercise training in pregnancy reduces offspring size without changes in maternal insulin sensitivity. J. Clin. Endocrinol. Metab. 2010, 95, 2080–2088. [Google Scholar]
- Korpi-Hyovalti, E.A.; Laaksonen, D.E.; Schwab, U.S.; et al. Feasibility of a lifestyle intervention in early pregnancy to prevent deterioration of glucose tolerance. BMC Public Health 2011, 11, 179. [Google Scholar]
- Hui, A.; Back, L.; Ludwig, S.; et al. Lifestyle intervention on diet and exercise reduced excessive gestational weight gain in pregnant women under a randomised controlled trial. BJOG 2012, 119, 70–77. [Google Scholar] [CrossRef]
- Luoto, R.; Kinnunen, T.I.; Aittasalo, M.; et al. Primary prevention of gestational diabetes mellitus and large-for-gestational-age newborns by lifestyle counseling: A cluster-randomized controlled trial. PLoS Med. 2011, 8, e1001036. [Google Scholar] [CrossRef]
- Phelan, S.; Phipps, M.G.; Abrams, B.; Darroch, F.; Schaffner, A.; Wing, R.R. Randomized trial of a behavioral intervention to prevent excessive gestational weight gain: The Fit for Delivery Study. Am. J. Clin. Nutr. 2011, 93, 772–779. [Google Scholar]
- Quinlivan, J.A.; Lam, L.T.; Fisher, J. A randomised trial of a four-step multidisciplinary approach to the antenatal care of obese pregnant women. Aust. N. Z. J. Obstet. Gynaecol. 2011, 51, 141–146. [Google Scholar]
- Nascimento, S.L.; Surita, F.G.; Parpinelli, M.A.; Siani, S.; Pinto e Silva, J.L. The effect of an antenatal physical exercise programme on maternal/perinatal outcomes and quality of life in overweight and obese pregnant women: A randomised clinical trial. BJOG 2011, 118, 1455–1463. [Google Scholar]
- Haakstad, L.A.; Bo, K. Effect of regular exercise on prevention of excessive weight gain in pregnancy: A randomised controlled trial. Eur. J. Contracept. Reprod. Health Care 2011, 16, 116–125. [Google Scholar]
- Haakstad, L.A.; Bo, K. Exercise in pregnant women and birth weight: A randomized controlled trial. BMC Pregnancy Childbirth 2011, 11, 66. [Google Scholar]
- Vinter, C.A.; Jensen, D.M.; Ovesen, P.; Beck-Nielsen, H.; Jorgensen, J.S. The LiP (Lifestyle in Pregnancy) study: A randomized controlled trial of lifestyle intervention in 360 obese pregnant women. Diabetes Care 2011, 34, 2502–2507. [Google Scholar]
- Gray-Donald, K.; Robinson, E.; Collier, A.; David, K.; Renaud, L.; Rodrigues, S. Intervening to reduce weight gain in pregnancy and gestational diabetes mellitus in Cree communities: An evaluation. CMAJ 2000, 163, 1247–1251. [Google Scholar]
- Olson, C.M.; Strawderman, M.S.; Reed, R.G. Efficacy of an intervention to prevent excessive gestational weight gain. Am. J. Obstet. Gynecol. 2004, 191, 530–536. [Google Scholar]
- Kinnunen, T.I.; Pasanen, M.; Aittasalo, M.; et al. Preventing excessive weight gain during pregnancy - a controlled trial in primary health care. Eur. J. Clin. Nutr. 2007, 61, 884–891. [Google Scholar] [CrossRef]
- Claesson, I.M.; Sydsjo, G.; Brynhildsen, J.; et al. Weight gain restriction for obese pregnant women: A case-control intervention study. BJOG 2008, 115, 44–50. [Google Scholar]
- Shirazian, T.; Monteith, S.; Friedman, F.; Rebarber, A. Lifestyle modification program decreases pregnancy weight gain in obese women. Am. J. Perinatol. 2010, 27, 411–414. [Google Scholar]
- Mottola, M.F.; Giroux, I.; Gratton, R.; et al. Nutrition and exercise prevent excess weight gain in overweight pregnant women. Med. Sci. Sports Exerc. 2010, 42, 265–272. [Google Scholar]
- Lindholm, E.S.; Norman, M.; Kilander, C.P.; Altman, D. Weight control program for obese pregnant women. Acta Obstet. Gynecol. Scand. 2010, 89, 840–843. [Google Scholar]
- Olson, C.M. A call for intervention in pregnancy to prevent maternal and child obesity. Am. J. Prev. Med. 2007, 33, 435–436. [Google Scholar]
- Gavard, J.A.; Artal, R. Effect of exercise on pregnancy outcome. Clin. Obstet. Gynecol. 2008, 51, 467–480. [Google Scholar]
- Stuebe, A.M.; Forman, M.R.; Michels, K.B. Maternal-recalled gestational weight gain, pre-pregnancy body mass index, and obesity in the daughter. Int. J. Obes. (Lond.) 2009, 33, 743–752. [Google Scholar] [CrossRef]
- Dodd, J.M.; Turnbull, D.A.; McPhee, A.J.; Wittert, G.; Crowther, C.A.; Robinson, J.S. Limiting weight gain in overweight and obese women during pregnancy to improve health outcomes: The LIMIT randomised controlled trial. BMC Pregnancy Childbirth 2011, 11, 79. [Google Scholar]
- Althuizen, E.; van Poppel, M.N.; Seidell, J.C.; van der Wijden, C.; van Mechelen, W. Design of the New Life(style) study: A randomised controlled trial to optimise maternal weight development during pregnancy. [ISRCTN85313483]. BMC Public Health. 2006, 6, 168. [Google Scholar] [CrossRef]
- Oostdam, N.; van Poppel, M.N.; Eekhoff, E.M.; Wouters, M.G.; van Mechelen, W. Design of FitFor2 study: The effects of an exercise program on insulin sensitivity and plasma glucose levels in pregnant women at high risk for gestational diabetes. BMC Pregnancy Childbirth 2009, 9, 1. [Google Scholar]
- Adamo, K.B.; Ferraro, Z.; Rutherford, J.; et al. The maternal obesity management (MOM) trial: A lifestyle intervention during pregnancy to minimize downstream obesity. Appl. Physiol. Nutr. Metab. 2010, 35, S2. [Google Scholar]
- Moholdt, T.T.; Salvesen, K.; Ingul, C.B.; Vik, T.; Oken, E.; Morkved, S. Exercise Training in Pregnancy for obese women (ETIP): Study protocol for a randomised controlled trial. Trials 2011, 12, 154. [Google Scholar]
- Lee, J.H.; Reed, D.R.; Price, R.A. Familial risk ratios for extreme obesity: Implications for mapping human obesity genes. Int. J. Obes. Relat. Metab. Disord. 1997, 21, 935–940. [Google Scholar]
- Rooney, K.; Ozanne, S.E. Maternal over-nutrition and offspring obesity predisposition: Targets for preventative interventions. Int. J. Obes. (Lond.) 2011, 35, 883–890. [Google Scholar] [CrossRef]
- Samuelsson, A.M.; Matthews, P.A.; Argenton, M.; et al. Diet-induced obesity in female mice leads to offspring hyperphagia, adiposity, hypertension, and insulin resistance: A novel murine model of developmental programming. Hypertension. 2008, 51, 383–392. [Google Scholar] [CrossRef]
- Barker, D.J.; Osmond, C.; Golding, J.; Kuh, D.; Wadsworth, M.E. Growth in utero, blood pressure in childhood and adult life, and mortality from cardiovascular disease. Br. Med. J. 1989, 298, 564–567. [Google Scholar]
- Barker, D.J.; Gluckman, P.D.; Godfrey, K.M.; Harding, J.E.; Owens, J.A.; Robinson, J.S. Fetal nutrition and cardiovascular disease in adult life. Lancet 1993, 341, 938–941. [Google Scholar]
- Barker, D.J. Fetal origins of coronary heart disease. Br. Med. J. 1995, 311, 171–174. [Google Scholar]
- Godfrey, K.M.; Inskip, H.M.; Hanson, M.A. The long-term effects of prenatal development on growth and metabolism. Semin. Reprod. Med. 2011, 29, 257–265. [Google Scholar]
- Hanson, M.; Godfrey, K.M.; Lillycrop, K.A.; Burdge, G.C.; Gluckman, P.D. Developmental plasticity and developmental origins of non-communicable disease: Theoretical considerations and epigenetic mechanisms. Prog. Biophys. Mol. Biol. 2011, 106, 272–280. [Google Scholar]
- Ainge, H.; Thompson, C.; Ozanne, S.E.; Rooney, K.B. A systematic review on animal models of maternal high fat feeding and offspring glycaemic control. Int. J. Obes. (Lond.) 2011, 35, 325–335. [Google Scholar] [CrossRef]
- George, L.A.; Uthlaut, A.B.; Long, N.M.; et al. Different levels of overnutrition and weight gain during pregnancy have differential effects on fetal growth and organ development. Reprod. Biol. Endocrinol. 2010, 8, 75. [Google Scholar] [CrossRef]
- Muhlhausler, B.S.; Adam, C.L.; Findlay, P.A.; Duffield, J.A.; McMillen, I.C. Increased maternal nutrition alters development of the appetite-regulating network in the brain. FASEB J. 2006, 20, 1257–1259. [Google Scholar]
- Wang, J.; Ma, H.; Tong, C.; et al. Overnutrition and maternal obesity in sheep pregnancy alter the JNK-IRS-1 signaling cascades and cardiac function in the fetal heart. FASEB J. 2010, 24, 2066–2076. [Google Scholar] [CrossRef]
- Alfaradhi, M.Z.; Ozanne, S.E. Developmental programming in response to maternal overnutrition. Front. Genet. 2011, 2, 27. [Google Scholar]
- Matsuzawa-Nagata, N.; Takamura, T.; Ando, H.; et al. Increased oxidative stress precedes the onset of high-fat diet-induced insulin resistance and obesity. Metabolism 2008, 57, 1071–1077. [Google Scholar] [CrossRef]
- Dandona, P.; Mohanty, P.; Ghanim, H.; et al. The suppressive effect of dietary restriction and weight loss in the obese on the generation of reactive oxygen species by leukocytes, lipid peroxidation, and protein carbonylation. J. Clin. Endocrinol. Metab. 2001, 86, 355–362. [Google Scholar] [CrossRef]
- Radaelli, T.; Varastehpour, A.; Catalano, P.; Hauguel-De, M.S. Gestational diabetes induces placental genes for chronic stress and inflammatory pathways. Diabetes 2003, 52, 2951–2958. [Google Scholar]
- Tong, J.F.; Yan, X.; Zhu, M.J.; Ford, S.P.; Nathanielsz, P.W.; Du, M. Maternal obesity downregulates myogenesis and beta-catenin signaling in fetal skeletal muscle. Am. J. Physiol. Endocrinol. Metab. 2009, 296, E917–E924. [Google Scholar]
- Barker, D.J. The developmental origins of chronic adult disease. Acta Paediatr. Suppl. 2004, 93, 26–33. [Google Scholar]
- Fraser, A.; Tilling, K.; Donald-Wallis, C.; et al. Association of maternal weight gain in pregnancy with offspring obesity and metabolic and vascular traits in childhood. Circulation 2010, 121, 2557–2564. [Google Scholar] [CrossRef]
- Gale, C.R.; Javaid, M.K.; Robinson, S.M.; Law, C.M.; Godfrey, K.M.; Cooper, C. Maternal size in pregnancy and body composition in children. J. Clin. Endocrinol. Metab. 2007, 92, 3904–3911. [Google Scholar]
- Lawlor, D.A.; Smith, G.D.; O'Callaghan, M.; et al. Epidemiologic evidence for the fetal overnutrition hypothesis: Findings from the mater-university study of pregnancy and its outcomes. Am. J. Epidemiol. 2007, 165, 418–424. [Google Scholar]
- Lawlor, D.A.; Fraser, A.; Lindsay, R.S.; et al. Association of existing diabetes, gestational diabetes and glycosuria in pregnancy with macrosomia and offspring body mass index, waist and fat mass in later childhood: Findings from a prospective pregnancy cohort. Diabetologia 2010, 53, 89–97. [Google Scholar] [CrossRef]
- McMillen, I.C.; MacLaughlin, S.M.; Muhlhausler, B.S.; Gentili, S.; Duffield, J.L.; Morrison, J.L. Developmental origins of adult health and disease: The role of periconceptional and foetal nutrition. Basic Clin. Pharmacol. Toxicol. 2008, 102, 82–89. [Google Scholar]
- Metzger, B.E.; Lowe, L.P.; Dyer, A.R.; et al. Hyperglycemia and adverse pregnancy outcomes. N. Engl. J. Med. 2008, 358, 1991–2002. [Google Scholar] [CrossRef]
- Whitaker, R.C.; Wright, J.A.; Pepe, M.S.; Seidel, K.D.; Dietz, W.H. Predicting obesity in young adulthood from childhood and parental obesity. N. Engl. J. Med. 1997, 337, 869–873. [Google Scholar]
- Bauer, M.K.; Harding, J.E.; Bassett, N.S.; et al. Fetal growth and placental function. Mol. Cell. Endocrinol. 1998, 140, 115–120. [Google Scholar] [CrossRef]
- Rahnama, F.; Shafiei, F.; Gluckman, P.D.; Mitchell, M.D.; Lobie, P.E. Epigenetic regulation of human trophoblastic cell migration and invasion. Endocrinology 2006, 147, 5275–5283. [Google Scholar]
- Clapp, J.F. Influence of endurance exercise and diet on human placental development and fetal growth. Placenta 2006, 27, 527–534. [Google Scholar]
- Clapp, J.F., III.; Schmidt, S.; Paranjape, A.; Lopez, B. Maternal insulin-like growth factor-I levels (IGF-I) reflect placental mass and neonatal fat mass. Am. J. Obstet. Gynecol. 2004, 190, 730–736. [Google Scholar]
- Jansson, T.; Wennergren, M.; Illsley, N.P. Glucose transporter protein expression in human placenta throughout gestation and in intrauterine growth retardation. J. Clin. Endocrinol. Metab. 1993, 77, 1554–1562. [Google Scholar]
- Jansson, T.; Wennergren, M.; Powell, T.L. Placental glucose transport and GLUT 1 expression in insulin-dependent diabetes. Am. J. Obstet. Gynecol. 1999, 180, 163–168. [Google Scholar]
- Jansson, T.; Ekstrand, Y.; Wennergren, M.; Powell, T.L. Placental glucose transport in gestational diabetes mellitus. Am. J. Obstet. Gynecol. 2001, 184, 111–116. [Google Scholar]
- Jansson, T.; Ylven, K.; Wennergren, M.; Powell, T.L. Glucose transport and system A activity in syncytiotrophoblast microvillous and basal plasma membranes in intrauterine growth restriction. Placenta 2002, 23, 392–399. [Google Scholar]
- Jansson, T.; Ekstrand, Y.; Bjorn, C.; Wennergren, M.; Powell, T.L. Alterations in the activity of placental amino acid transporters in pregnancies complicated by diabetes. Diabetes 2002, 51, 2214–2219. [Google Scholar]
- Jansson, T.; Powell, T.L. IFPA 2005 Award in Placentology Lecture. Human placental transport in altered fetal growth: does the placenta function as a nutrient sensor?—A review. Placenta 2006, 27, S91–S97. [Google Scholar] [CrossRef]
- Roos, S.; Powell, T.L.; Jansson, T. Placental mTOR links maternal nutrient availability to fetal growth. Biochem. Soc. Trans. 2009, 37, 295–298. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Adamo, K.B.; Ferraro, Z.M.; Brett, K.E. Can We Modify the Intrauterine Environment to Halt the Intergenerational Cycle of Obesity? Int. J. Environ. Res. Public Health 2012, 9, 1263-1307. https://doi.org/10.3390/ijerph9041263
Adamo KB, Ferraro ZM, Brett KE. Can We Modify the Intrauterine Environment to Halt the Intergenerational Cycle of Obesity? International Journal of Environmental Research and Public Health. 2012; 9(4):1263-1307. https://doi.org/10.3390/ijerph9041263
Chicago/Turabian StyleAdamo, Kristi B., Zachary M. Ferraro, and Kendra E. Brett. 2012. "Can We Modify the Intrauterine Environment to Halt the Intergenerational Cycle of Obesity?" International Journal of Environmental Research and Public Health 9, no. 4: 1263-1307. https://doi.org/10.3390/ijerph9041263




