1. Introduction
The World Health Organisation (WHO) has indicated that approximately 1.8 million deaths and 61.9 million disability-adjusted life years (DALYs) worldwide are attributable to unsafe water, sanitation and poor hygiene. An estimated 99.8% of such deaths occur in developing countries, with children ranking (90%) as the first victims [
1]. Consumption of groundwater and surface water sources contaminated with pathogenic bacteria such as
Escherichia coli O157:H7,
Salmonella typhimurium,
Shigella dysenteriae and
Vibrio cholera continues to be one the major causes of diarrheal diseases and gastrointestinal infections [
2,
3,
4,
5,
6]. This implies that safe drinking water plays a significant role in human health and well-being.
In 2002, the United Nation Millennium Development Goals (MDG) firmly established the issue of water and sanitation on the global agenda. The vision of the MDG is to halve the number of people without access to safe drinking water and sanitation by 2015 [
7]. Although tremendous progress has been made to date, the 2010 updated report by the WHO and the United Nations Children’s Fund (UNICEF) has indicated that 884 million people in the world still lack access to drinking water from improved sources.
The provision of safe drinking water is currently a high priority for the South African government, one of the signatories of the MDG. The percentage of households with access to water infrastructure above or equal to the Reconstruction and Development Programme standard increased from 61.7% in 1994 to 91.8% in March 2009. Based on these data, it is estimated that 93% of the population had access to an improved drinking-water supply in 2010 [
8]. In most of the cases, non-improved drinking water supplies are currently found in rural communities that are widely dispersed and informal peri-urban communities that are continuously expanding. It is therefore difficult to implement centralised systems such as piped systems, which not only require substantial financial support, but also highly skilled personnel to manage and maintain them. The implementation of decentralised systems is therefore needed to provide these rural communities with safe drinking-water sources.
Cost-effective filter materials coated with silver nanoparticles is an alternative technology that could assist the developing countries in meeting the MDG, and South Africa, in particular, in providing a safe drinking-water supply to all scatted rural areas and informal settlements. Silver ion (Ag
+) has long been known as a potential antimicrobial agent and is used in wound dressings to prevent infections in burn patients, to blindness in newborns, for severe chronic osteomyelitis and urinary infection, to control
Legionella bacteria in hospitals and to improve the performance of drinking-water filters [
9,
10,
11,
12]. It can bind to bacterial cells and enzymes (proteins) at multiple sites, damaging them and preventing them from performing their functions and result, to cells death through penetration at specific bacterial DNA and RNA [
9,
13,
14].
Silver in the form of nanoparticles that release silver ions more effectively has a better bactericidal activity due to its high surface-area-to-volume ratio [
15,
16]. Recent studies have shown that distinctively prepared silver nanoparticles display good antibacterial activity [
17,
18]. As a result, researchers have considered silver nanoparticles for drinking-water treatment due to its strong and broad spectrum of antimicrobial activities [
19,
20,
21]. With the advancement of material development, silver nanoparticles can be easily deposited on solid materials for the deactivation of microorganisms in water treatment [
22]. In the case of drinking-water treatment, various forms of silver nanoparticles coated on materials/substrates have been used. These include Ag/sand [
23], Ag/zeolite [
17] and Ag/fibreglass [
24]. Sand filtrations have been used in water purification to control microbiological contamination for over 150 years [
25]. Sand filters are a less expensive, more effective method of water treatment, can be self-constructed and may be constructed by using local skills. Natural zeolites as cation exchangers in water treatment have increased due to their availability, low cost, high surface area and sorptive capacity, negative surface charge, chemical inertness and low or null toxicity for human [
26,
27]. Most kinds of fibreglass are used for thermal and acoustic insulation in building construction, shipbuilding and filtration applications. Fibreglass-reinforced plastics (FRPs) have been used for various types of process equipment in the chemical industry, pulp and paper industry, power and mining industries, municipal sewer treatment and water treatment, as well as many other associated industries handling corrosive equipment [
28]. A number of investigations have been carried out on the use of resins containing silver/silver nanoparticles for oral and dental applications [
29,
30]. Resins are used in ion exchange and constitute a very powerful technology for removing impurities from water and other solutions. There is no health risk with resins, as many industries use resins for multiple purposes (nuclear and thermal power stations, semiconductors, computers), including dental and pharmaceutical applications and drinking-water treatment for the removal of toxic contaminants [
31,
32].
Even though a number of studies have been conducted on bacterial removal with Ag/zeolite, Ag/sand, Ag/fibreglass and Ag/resin nanoparticle substrates, there is no information on comparative studies related to the use of these technologies for the removal of pathogenic bacteria from drinking-water sources. This study therefore concentrated on the development of these substrates modified with silver nanoparticles and compared their effectiveness in removing pathogenic bacteria (Escherichia coli, Vibrio cholerae, Shigella dysenterae and Salmonella typhimurium) from polluted groundwater sources. Our main intention was to find the alternative cost-effective technology with the best concentration of silver nanoparticles loaded on the substrates, which could completely remove pathogenic bacteria from test water and result in the production of safe drinking water for rural communities.
2. Experimental Methodology
2.1. Preparation of Substrates
Locally available materials for silver deposition were utilised in the present study. Silver was coated on natural zeolite, sand, fibreglass, anion resin and cation resin. Natural clinoptilolite zeolite purchased from Ajax Industries CC (Cape Town, South Africa) was conditioned in a 500 mL solution of 2 M NaCl (Merck, South Africa), followed by stirring at room temperature (between 20 and 25 °C) for 36 h. The solid-liquid mixture was separated by centrifugation at 3,000 rpm for 15 min. Liquids were discarded and the solids were washed with deionised water three times, and then oven dried at 105 °C for 8 h. Silica sand purchased from Eggo Sand (Pty) Ltd (Pretoria, South Africa) was submitted to a cleaning process by stirring 200 g of sand in a litre of 30% nitric acid (Merck, South Africa) solution with a reciprocating shaker at 210 rpm at room temperature for 24 h. The sand was allowed to settle and separated from the solution by decantation, and thereafter rinsed three times with deionised water and oven dried at 105 °C for 24 h. Fibreglass chopped-strand mat was purchased from Collins Fibreglass Plastics (Johannesburg, South Africa) and cleaned by immersion in an ultrasonic bath containing isopropanol (Sigma, South Africa) for 2 h. The substrate was rinsed three times with deionised water and oven dried at 105 °C for 24 h.
2.2. Synthesis of Silver Nanoparticle-Coated Substrates
2.2.1. Coating of Zeolite, Sand and Fibreglass Substrates
Silver nitrate (AgNO3) (Merck, South Africa) stock solution (1 mM) was prepared by adding 169.87 mg of silver nitrate to a litre of deionised water. Thereafter concentrations of 0.01, 0.03, 0.05 and 0.1 mM silver nitrate (250 mL) were prepared by diluting silver stock solution. The substrates (20 g) were separately immersed in aqueous solution containing concentrations of 0.01, 0.03, 0.05 and 0.1 mM silver nitrate for 24 h. They were incubated in a thermostatic shaker at a speed of 250 rpm in the dark at room temperature for 24 h. Substrates containing silver were separated from the mixture by centrifugation at 3,000 rpm for 15 min and washed with deionised water, and then oven dried for 24 h at 105 °C. For the reduction of silver ions to silver nanoparticles, the substrates containing silver were heated in an N2 furnace (Lenntech, South Africa) at a flow rate of 400 mL/min for 1 h at 120 °C and the furnace was ramped up to 350 °C for 3 h.
2.2.2. Coating of Anion Resin Beads Substrate
Amberlite-IRA-458 anion exchange resin (in chloride form) was purchased from Lenntech (Johannesburg, South Africa). Silver was coated on the anion resin beads using the method previously described by [
33]. Briefly, the silver nanoparticle-resins were prepared following a two-step procedure. A known amount (20 g) of anion exchange resin was used. Firstly, 30 mL of 1 M HCl (Merck, South Africa) solution were added dropwise to 200 mL stirred, freshly prepared aqueous solution of concentrations of 0.01, 0.03, 0.05 and 0.1 mM AgNO
3 to form white precipitates of silver chloride. The precipitates were washed three times with deionised water to remove HNO
3 and dried in a water bath at 65 °C for 2 h. The silver precursor [AgCl
2]
¯ complex was prepared by dissolving 0.3 g of solid AgCl in a concentrated HCl solution and the mixture was placed in an ultrasonic bath for dissolution. Secondly, the silver precursor ions were allowed to exchange with Cl
¯ ions of the neat chloride form of anion-exchange resin beads (R
+Cl
¯) and the mixture was kept overnight. The resin beads, on which silver precursor ions were immobilised, were washed three times with water to drain out the liberated HCl and un-exchanged [AgCl
2]
¯ and then reduced with a freshly prepared ice-cold aqueous solution of 0.01 M sodium borohydride. The prepared shining reddish-black silver-coated beads [R(Ag)
0]
+Cl¯ were washed thoroughly with deionised water and dried at room temperature in a vacuum.
2.2.3. Coating of Cation Resin Beads Substrate
The methods described by [
34] were also used for the coating of cation resin. The silver amine complex, [Ag(NH
3)
2]
+, was prepared by adding 10 mL of 25% ammonia solution dropwise to each of 200 mL aqueous solution of concentrations of 0.01, 0.03, 0.05 and 0.1 mM AgNO
3. A known amount (20 g) of cation exchange resin (R
–H
+) was added to each of these mixtures, followed by a mixing process for 3 h using a magnetic stirrer. The resin silver amine moiety [R–Ag(NH
3)
2] was washed three times with deionised water and heated in an oven at 150 °C for 1 h. The yellow colour of the resin beads was transformed to black due to the formation of resin silver oxide composite [R(Ag
2O)]¯H
+. Then, this complex was reduced with an aqueous solution of freshly prepared 0.01 M sodium borohydride to form silver nanoparticle-coated resin beads [R(Ag
0)]¯H
+, with a white colour. The silver nanoparticle-resin beads were washed three times with deionised water and finally dried on a water bath at 65 °C for 2 h to obtain dry silver-coated resin beads.
2.3. Characterisation of Substrates Coated with Silver Nanoparticles
The surface morphology of the silver nanoparticles-coated substrates was examined with a scanning electron microscope (SEM) (JEOL JSM-5800LV, JEOL Ltd, Tokyo, Japan) coupled with energy-dispersive spectroscopy (EDS) to confirm the chemical content on the substrates. Transmission electron microscope (TEM) analysis was performed with a JEOL 2100F (JEOL Ltd, Tokyo, Japan) that operated at 100 kV to examine the morphology and particle size distribution of the silver nanoparticle substrates. X-ray diffraction (XRD) was used to determine the crystal phase of the substrates. The patterns of the silver nanoparticles were recorded with a Bruker D8 Advance using Cu Kα radiation with 1.5416 Å wavelengths. The structure of the silver on the substrates was studied by scanning the media in 2θ ranges from 30 to 80 °C in a continuous scan mode. The crystallite size of the silver substrates was determined from X-ray line broadening using the Debye-Scherrer equation as follows:
![Ijerph 09 00244 i001]()
where D = Crystallite size, A (Angstroms), K = Crystallite-shape factor = 0.9, λ = X-ray wavelength, 1.5416 Å for CuKα, θ = Observed peak angle, degree, β = X-ray diffraction broadening, radian.
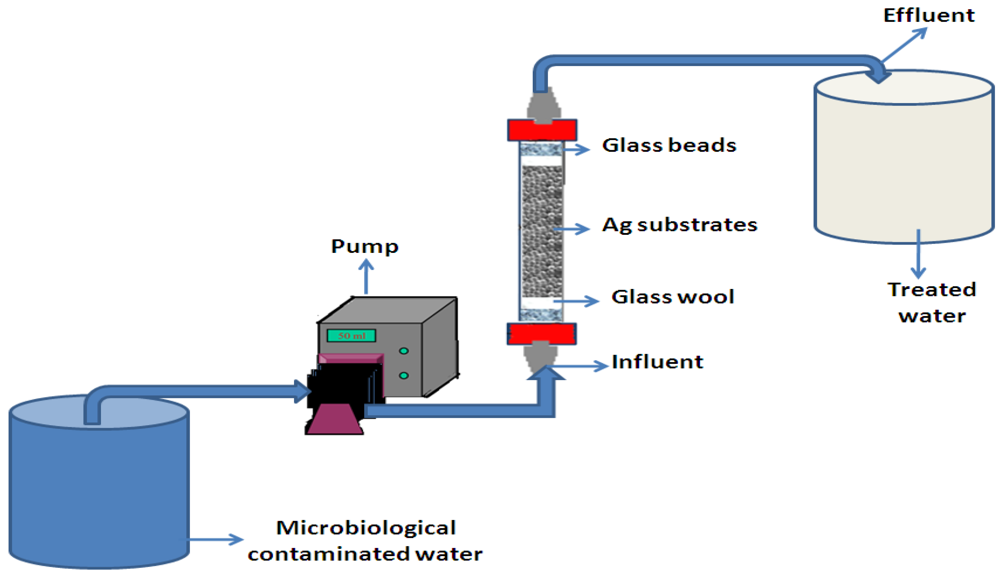
2.4. Production of Combined Substrate-Silver Nanoparticle Filter Systems
The filter systems consisted of a polyvinyl chloride (PVC) column of 2 cm diameter and 20 cm length (
Figure 1). Each column was packed with one type of the substrate coated with silver nanoparticles at a depth of 10 cm. With reference to various substrates (sand, zeolite, fibreglass, anion resin and cation resin), five filters were used during the study period. Glass beads (2 cm) and glass wool (2 cm) were placed in the upper and bottom ends of each column. Glass beads were positioned to prevent the substrates from pilling up at one end. A 10 L bucket served as storage container for contaminated influent water, which was fed into the filter system with a 1 m length of 8 mm diameter latex tubing connected to a Rainin Dynamax peristaltic pump (Rainin Instrument Co., Woburn, MA, USA). The effluent sample (treated water) was collected at the top of the filter with a 1 m length of 8 mm diameter latex tubing into a 10 L bucket container.
Figure 1 illustrates the schematic diagram of a laboratory-scale setup with an example of a combined substrate-Ag nanoparticle filter system.
Figure 1.
Schematic diagram of laboratory-scale setup to evaluate the antibacterial efficiency of Ag nanoparticle-coated substrates.
Figure 1.
Schematic diagram of laboratory-scale setup to evaluate the antibacterial efficiency of Ag nanoparticle-coated substrates.
2.5. Testing the Efficiency of Filter Systems in Removing Pathogenic Bacteria
The performance of the combined substrates-silver nanoparticle filter systems in removing pathogenic bacteria was studied in two phases. In the first phase, the five substrates coated with various concentrations of AgNO
3 were tested against
Escherichia coli (ATCC 43895) spiked in synthetic water samples. The main objective of this part of the study was to determine the best loading concentration of AgNO
3 that could result in the total removal of pathogenic bacteria from a test water source. In the second phase, the performance of the five combined substrates-silver nanoparticle filter systems was tested using groundwater, and only the best concentration of silver nanoparticles loaded on the substrates was investigated against the four different pathogenic bacteria-
E. coli,
S. typhimurium,
S. dysenteriae and
V. cholerae. In cases where these pathogens were not present in the intake groundwater source, the water was spiked with the pathogens. This was mainly done to evaluate the efficiency of each filter in reaching the allowable recommended limits set by the
South African National Standards for domestic use [
35,
36]. The bacterial removal efficiency was obtained by comparing the concentrations (Log
10 cfu/100 mL) of target organisms before and after treatment. In each series of the experimental study, a control filter constituted of the substrate without silver nanoparticles was included. The experimental study for each combined substrates-silver nanoparticle filter system was performed in three different trials.
2.5.1. Preparation of Bacterial Stock Suspensions
The microbial strains used in the experimental study included
Escherichia coli (ATCC 43895) and
Salmonella typhimurium (ATCC 14028) obtained from the American Type Culture Collection (Rockville, MD, USA),
Vibrio cholera and
Shigella dysenteriae obtained from the Council for Scientific and Industrial Research (CSIR, Pretoria, South Africa) bacterial stock cultures. Prior to use, these bacterial strains were confirmed by cultural tests using selective agar medium (Chromocult agar for
E. coli, XLD agar for
S. typhimurium and
S. dysenteriae, TCBS agar for
V. cholera) according to the Standard Method [
37]. One loop full of each bacterial culture was separately inoculated in 100 mL sterile nutrient broth (Merck, South Africa) medium and incubated aerobically at 37 °C in a shaking incubator (Scientific Model 353, Lasec, South Africa) at 120 rpm for 24 h. The bacteria were harvested by centrifugation at 4,000 rpm for 10 min and the pellet was washed twice with 50 mL of sterile 0.01 M phosphate-buffered saline (PBS, pH 7.2).
The stock suspensions of E. coli, S. typhimurium, S. dysenteriae and V. cholera were prepared by re-suspending the final pellets in 10 mL of 0.01 M PBS solution. The initial concentrations of bacterial cells harvested were determined with the spread-plate technique, after serial dilution of each culture in sterile saline solution (0.9% w/v NaCl). The plates were incubated at 37 °C for 24 h. The resulting colonies were counted and expressed as cfu/mL.
2.5.2. Preparation of Synthetic Contaminated Water
For each filter system, an aliquot of the stock suspension of E. coli (ATCC 43895) corresponding to 6 log cfu/100 mL was inoculated into 10 L final volumes of sterile saline water (8.5% NaCl). The spiked water samples were shaken vigorously several times and 1 mL of this water source was used to determine the initial concentration of the target organism before passing the remaining contaminated water through the filter systems.
2.5.3. Collection and Analysis of the Quality of Groundwater Samples
Groundwater samples were collected from a borehole at Delmas (A7) in the Mpumalanga Province of South Africa. The study was conducted between June and July 2010 and the water samples were collected three times during this period. It is important to note that, during the study period, this groundwater supply was used by the community without prior treatment. The water samples were collected in sterile 50 L plastic buckets. Samples were also collected in sterile 1 L glass bottles in order to detect and enumerate the initial concentrations of the target bacteria and selected physicochemical parameters before treatment. The samples were transported to the laboratory and the quality of the water was determined for microbiological contamination and selected physicochemical parameters within 6 h [
37].
Escherichia coli,
S. typhimurium,
S. dysenteriae and
V. cholerae were detected and enumerated from groundwater samples according to Standard Methods [
37]. As mentioned above, in cases where these organisms were not detected in groundwater samples, they were spiked with 10
2 cfu/mL stock suspension in these test water sources using the same method as described for synthetic water samples. All the tests were conducted in aseptic conditions.
The pH and the turbidity were measured on site using a pH meter (Metrohm Co. Model 713) and a microprocessor turbidity meter (Eutech Instrument Turbidimeter TN-100), respectively. Nitrates and fluoride concentrations were determined in the laboratory using the Spectroquant Nova 400 manual water analyser (Merck) and photometric test kits (Merck), while the concentrations of magnesium, calcium in the water sample were determined by atomic absorption spectrophotometry (SpectrAA 220FS), according to Standard Methods [
37].
2.5.5. Elution of Silver Ions from Silver Nanoparticles Substrates Filter Systems
The degree of elution of silver from each combined substrates-silver nanoparticle filter system used in the first phase with spiked synthetic water was measured. The silver content in the treated water samples was determined with atomic absorption spectroscopy (AAS) by using a Spectra AA-220FFS (Varian Medical Systems, Inc., Palo Alto, CA, USA).
2.6. Statistical Analysis
All data were analysed statistically using the SPSS computer software version 11.0. Testing of significance was carried out using the one-way analysis of variance (ANOVA) at a 95% confidence interval. Comparisons were made between the treatment means of each filter system to determine if there were significant differences in treatments.
4. Discussion
The increasing demand for access to safe drinking water and the problems associated with centralised systems in developing countries have made decentralised systems vital for the development of new technologies to address these challenges, especially in scattered communities depending totally on groundwater supplies. Therefore, this study explored the use of nanosized silver impregnated onto cost-effective materials locally available in South Africa for possible use in drinking water disinfection. Using hydrothermal and chemical methods, it was revealed that silver nanoparticles were successfully deposited onto sand, zeolite, fibreglass, anion and cation resin substrates. However, in the case of the silver coatings, the antibacterial effect was found to reduce with time and the coatings had minimal antibacterial properties at 30 min. The ion elution studies (
Figure 7) indicated that between 60 and 90% of silver loaded on the Ag/zeolite, Ag/sand, Ag/fibreglass and Ag/anion resin substrates eluted into the treated samples. Consequently the low level of Ag ions remaining in the surface substrates after 30 min elution is responsible for the decrease of bacterial removal at this time point. It was also discovered by [
38] that silver eluted into water when using Ag-zeolite. The elution of silver from the substrates might be due to the weak attachment of silver nanoparticles to the surface substrates. High concentrations of silver ions eluted in water can be toxic to human cells and potentially cause adverse effects in the case of long-term implants. The amount of silver ions eluted from Ag/zeolite, Ag/sand, Ag/fibreglass and Ag/anion resin exceeded by far the recommended limit set by the World Health Organisation and US Environmental Protection Agency (USEPA), which is less than 0.10 mg/l in drinking water used for human consumption. The amount of silver ions eluted from Ag/cation resin complied with the WHO and USEPA limits [
39,
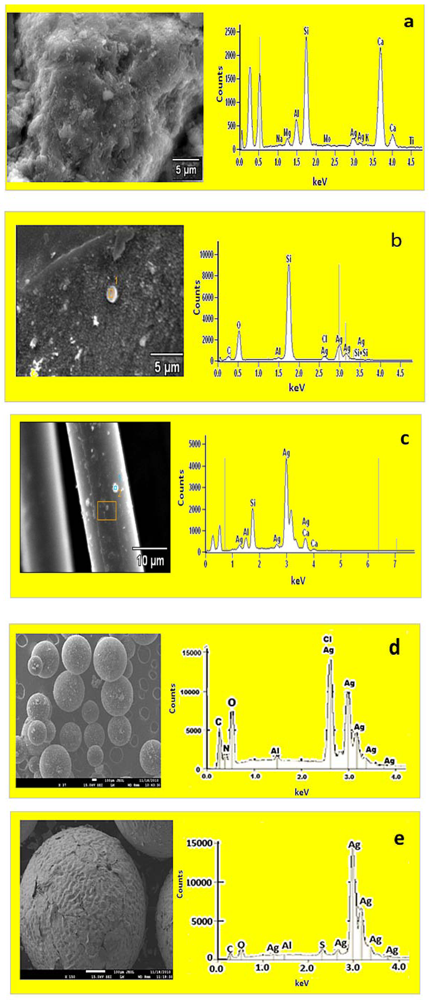
40]. Conversely, the high levels of ions (95%) still present in the Ag/cation resin surface after 30 min would explain the retention of the antimicrobial properties of that surface over the 90 min of the trial. Scanning electron microsope images revealed that the Ag nanoparticles on the substrates were predominantly small and spherical with EDS confirming the presences of silver peaks, as shown in
Figure 2. The TEM images also revealed spherical-shaped particles that aggregated to each other on the silver nanoparticle substrates, which indicated a particle size distribution ranging between 5 and 90 nm. This result was in accordance with the results obtained from the XRD pattern lattice measurement corresponding to the (111) silver plane (
Figure 4). The crystalline nature of Ag nanoparticles was confirmed by the XRD experimental study. The diffraction pattern observed from the XRD matches the face-centered cubic (fcc) structure of silver as described by previous investigators [
41,
42]. It was reported by [
33] that the two broad reflection peaks corresponding to (111) and (200) planes indicate that the silver particles are nanocrystals with cubic symmetry. A similar observation was reported by [
43]. According to these authors, both the nanoscale size and the presence of a (111) plane of Ag nanoparticles promote the biocidal property of
E. coli [
33,
43].
The physicochemical analyses of the tested groundwater source were within the recommended limits of no risk for drinking except for turbidity which was above the SANS 241 limit [
35]. High turbidity levels are associated with poor water quality and promote the survival of microorganisms [
36]. Turbidity can also protect microorganisms from the effects of disinfection, stimulate the growth of bacteria and give rise to a significant disinfection demand [
42].
Escherichia coli was found to be present at the highest concentration of 3 log cfu/100 mL in groundwater samples collected from Delmas, while the limit recommended by SANS 241 is 0 cfu/100 mL for drinking water that is meant for human consumption. Although
S. typhimurium,
S. dysenteriae and
V. cholerae were not detected in the groundwater samples, these pathogenic bacteria were seeded into the groundwater samples at a concentration of approximately 3 log cfu/100 mL in order to determine the removal efficiency of the filters. In the first part of this study, the antibacterial efficiency of the combined substrates-silver nanoparticle filter systems were determined, using various concentrations of silver nitrate against
E. coli as the test organism. The aim of this part of the study was to determine the concentration of silver that would have the most effective antibacterial property against the
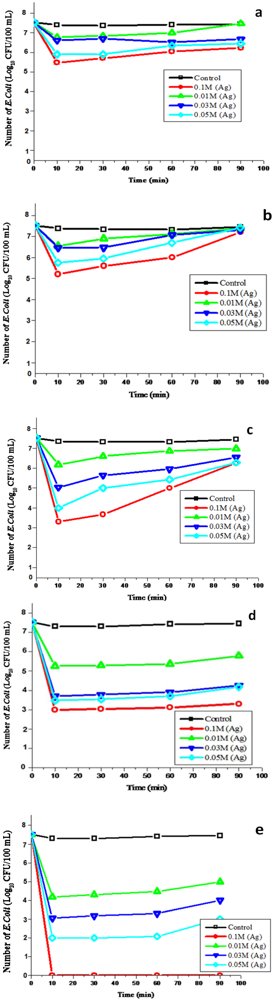
E. coli. The results indicated that all the filter systems containing uncoated substrates (
Figure 5(a–e)) were unable to deactivate
E. coli from synthetic water when compared to the combined substrates-silver nanoparticles filter systems. The bactericidal effect of silver nanoparticle substrates depended on the concentrations of the silver nitrate as well as on the type of substrates. The higher the concentration of Ag added to modify the substrates, the greater was the removal of
E. coli. In analyses regarding the effect of silver nanoparticles in a size range of 5–90 nm, significant reductions in the
E. coli population were noted when using filter materials coated with 0.01 mM, 0.03 mM, 0.05 mM and 0.1 mM silver concentrations as compared to the control (
P < 0.05). The overall results indicated a significantly higher bactericidal efficiency with 0.1 mM AgNO
3 (
P < 0.05) compared to other silver concentrations, despite the phenomenon of bacterial regrowth that resulted in a progressive increase of bacterial counts in the treated water during the subsequent operation of all the filters, except for the Ag/cation resin filter (
Figure 5(a–e)). This filter showed the best performance, which resulted in the complete removal of
E. coli from synthetic water without the occurrence of bacterial regrowth when the cation resin was loaded with 0.1 mM silver (
Figure 5(e)). The particle sizes of silver ranging between 1 and 100 nm have been reported to have an effect on the antibacterial properties of nanoparticles [
14]. Silver nanoparticles cause irreversible damage to the cellular membrane [
18,
43,
44], which enables the accumulation of nanoparticles in the cytoplasm. The action of silver nanoparticles is due to this damage and not to its toxicity [
45]. Previous investigators have pointed out that Ag nanoparticles bind to the outer membrane of
E. coli, causing the inhibition of active transport, dehydrogenase and periplasmic enzyme activity and eventually the inhibition of RNA, DNA and a decrease in the cell permeability, which finally results in cell lysis [
18,
45,
46,
47]. While microorganisms carry a negative charge, the Ag ions carry a positive charge, which is crucial for its antimicrobial activity through the electrostatic attraction between the negatively charged cell membrane of microorganisms and positively charged nanoparticles [
48,
49,
50]. [
51,
52] indicated that the higher the concentration, the better the antibacterial activity will be. The percentage removal of
E. coli from synthetic water was also in accordance with findings by previous investigations using a silver-coated ceramic water filter and silver-coated cylindrical polypropylene filters [
53,
54].
In the second part of the study, the antibacterial activities of combined substrates-silver nanoparticle filter systems prepared from 0.1 mM AgNO
3 were investigated against
E. coli,
S. typhimurium,
S. dysenteriae and
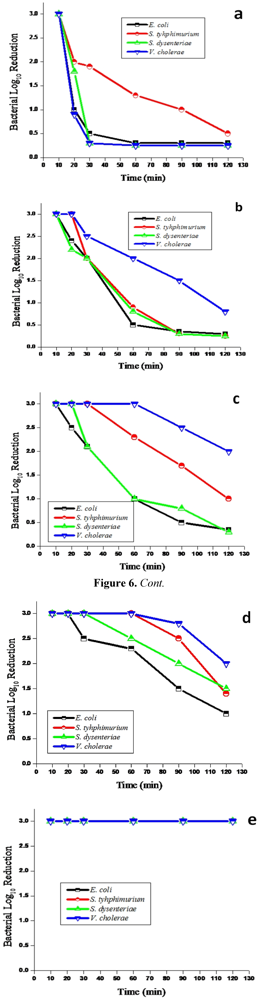
V. cholerae found or spiked in groundwater samples. The results of this part of the study also showed a decrease in bacterial concentrations from groundwater samples by all filters (
Figure 6). While the regrowth of targeted pathogenic bacteria occurred in water treated by Ag/zeolite, Ag/sand, Ag/fibreglass and Ag/anion resin, this phenomenon did not occur in drinking water treated with the Ag/cation resin nanoparticle filter system. This filter produced drinking water that complied with the limit of
E. coli 0 cfu /100 mL as set by the South African guidelines [
35,
36]. The silver cation resin nanoparticle filter system achieved a 100% removal of all the targeted pathogenic bacteria during the entire 120 min of the filter operation. This performance of the Ag/cation resin nanoparticle filter system, namely removing 100%
E. coli, was also reported by other researchers who used silver nanoparticle filters [
55,
56]. The results achieved in the first 10 min of the filter operation with a Ag/fibreglass nanoparticle filter system in removing
E. coli confirmed those reported by [
23] when these authors used a similar filter system for the purification of drinking water. Taking into account the performance of resin-silver nanoparticle filters in removing pathogenic bacteria and their level of silver ion elution in the drinking water, this suggests the use of this type of filter system as an alternative decentralisation technology for the production of safe drinking water for communities depending on groundwater supplies.