Aflatoxin Contamination in Food and Body Fluids in Relation to Malnutrition and Cancer Status in Cameroon
Abstract
:1. Introduction
2. Material and Methods
2.1. Sample Collection
2.2. Chemical Analysis
2.3. Statistical Analysis
3. Results
3.1. Aflatoxin Content in Egg Samples
3.2. Aflatoxin in Milk
3.3. AFB1 in Urine Samples of Malnourished Children
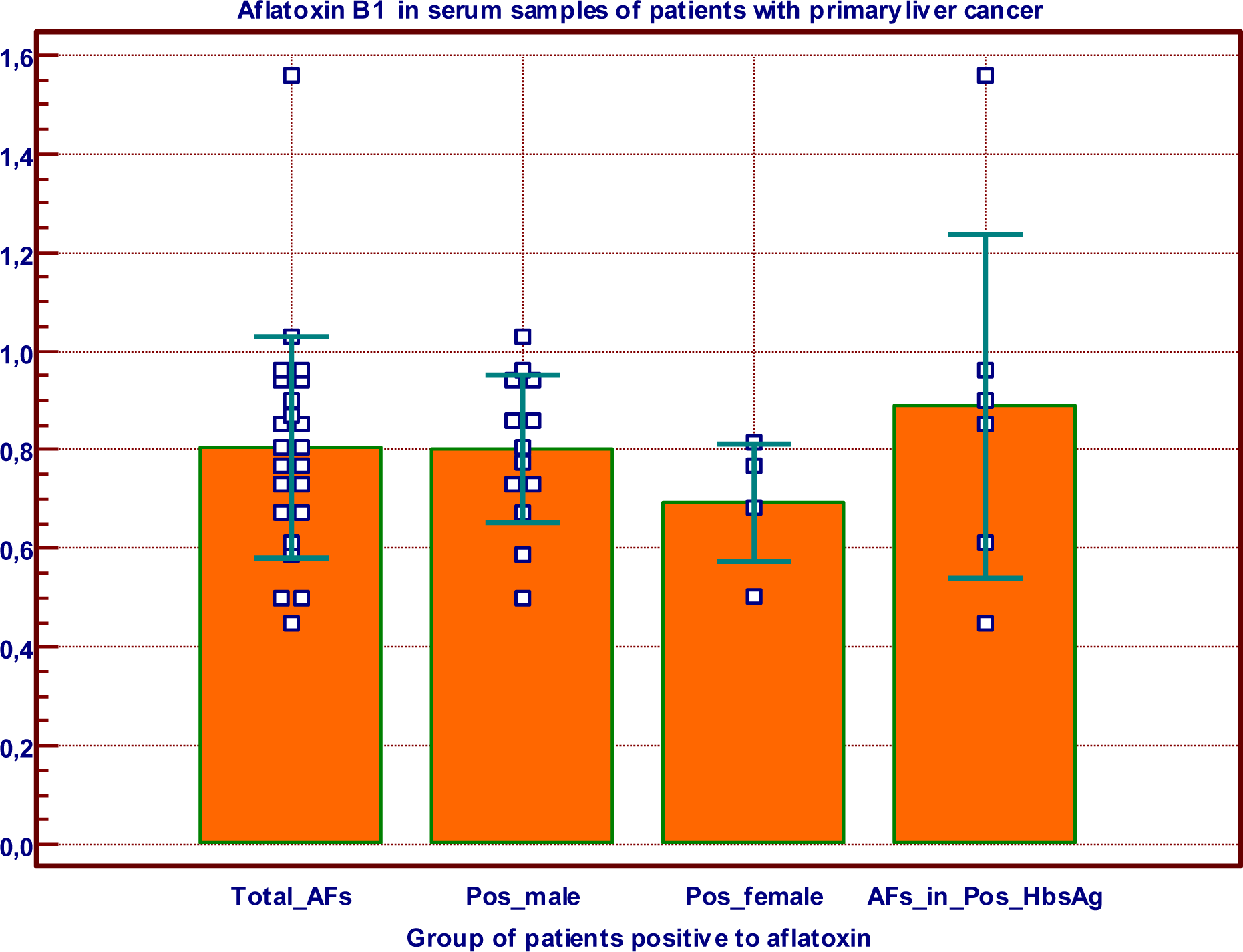
3.4. AFB1 in and HbsAg in Primary Liver Cancer Patients
4. Discussion
Acknowledgments
References
- Gourama, H; Bullerman, LB. Aspergillus flavus: aflatoxigenic fungi of concern in foods and feed. J. Food Protection 1995, 58, 1395–1404. [Google Scholar]
- Williams, JH; Phillips, TD; Jolly, PE; Stiles, JK; Jolly, CM; Aggarwal, D. Human aflatoxicosis in developing countries: a review of toxicology, exposure, potential health consequences, and interventions. Am. J. Clin. Nutr 2004, 80, 1106–1122. [Google Scholar]
- Jian, Y; Jolly, PE; Ellis, WO; Wang, J-S; Phillips, TD; Williams, JH. Aflatoxin B1 albumin adduct levels and cellular immune status in Ghanians. Int. Immunol 2005, 17, 807–814. [Google Scholar]
- Loveland, PM; Wilcox, JS; Hendricks, JD; Bailey, GS. Comparative metabolism and DNA binding of aflatoxin B1, aflatoxin M1, aflatoxicol and aflatoxicol-M1 in hepatocytes from rainbow trout (Salmo gairdneri). Carcinogenesis 1988, 9, 441–446. [Google Scholar]
- Cardwell, KF; Henry, SH. Risk of Exposure to and Mitigation of Effects of Aflatoxin on Human Health: A West African Example. In Aflatoxin and Food Safety; Abbas, HK, Ed.; Taylor and Francis Group, LLC; New York, NY, USA, 2005; pp. 214–235. [Google Scholar]
- Shephard, SG. Aflatoxin and Food Safety: Recent African Perspectives. In Aflatoxin and Food Safety; Abbas, HK, Ed.; Taylor and Francis Group, LLC: New York, NY, USA, 2005; pp. 13–28. [Google Scholar]
- Domngang, F; Kamdem, L; Moundipa, P. Presence d’aflatoxines dans certains aliments comsommés au Cameroun. Ann. Fac. Sc. Biol. Biochem 1984, 2, 93–101. [Google Scholar]
- Domngang, MF; Tchana, A; Moundipa, P. Aflatoxines dans les aliments prêts à la consummation au Cameroun. Cam. J. Biol. Biochem Sc 1988, 1, 65–78. [Google Scholar]
- Tchouanguep, MF; Tchana, A; Moundipa, FP. Aflatoxins in chicken feeds and naturally contaminated eggs. Microb. Hyg. Ali 1994, 6, 7–12. [Google Scholar]
- van Egmond, HP; Jonker, MA. Worldwide regulations on aflatoxins. In Aflatoxin and Food Safety; Abbas, HK, Ed.; Taylor and Francis Group, LLC: New York, NY, USA, 2005; pp. 77–93. [Google Scholar]
- Sun, Z; Lu, P; Gail, MH; Pee, D; Zhang, Q; Ming, L; Wang, J; Wu, Y; Liu, G; Wu, Y; Zhu, Y. Increased risk of hepatocellular carcinoma in male hepatitis B surface antigen carriers with chronic hepatitis who have detectable urinary aflatoxin metabolite M1. Hepatology 1999, 30, 379–383. [Google Scholar]
- Kirk, GD; Bah, E; Montesano, R. Molecular epidemiology of human liver cancer: insights into etiology, pathogenesis and prevention from The Gambia, West Africa. Carcinogenesis 2006, 27, 2070–2082. [Google Scholar]
- Neba, A. Modern Geography of the Republic of Cameroon, 3rd ed; Neba Publishers: Bamenda, Cameroon, 1999. [Google Scholar]
- IARC Technical report. Monotoring of aflatoxins in human body fluids and application to field studies. Cancer Res 1985, 45, 922–928. [Google Scholar]
- Micco, C; Brera, C; Miraglia, M; Onori, R. HPLC determination of the total content of aflatoxins in naturally contaminated eggs and free conjugate forms. Food Add. Contamin 1987, 4, 407–414. [Google Scholar]
- Tongtavuch, A; Chutima, C. High performance liquid chromatography of aflatoxins in human urine. J. Chromatography 1983, 275, 387–395. [Google Scholar]
- Wray, BB; Hayes, AW. Aflatoxin B1 in the serum of patient with primary hepatic carcinoma. Environ. Res 1980, 22, 400–403. [Google Scholar]
- Truckness, MW; Stoloff, L. Determination of aflatoxicol, afaltoxins B1 and M1 in egg. JAOAC 1984, 67, 317–320. [Google Scholar]
- Bennet, JW; Klich, M. Mycotoxins. Clin. Microbiol. Rev 2003, 16, 497–516. [Google Scholar]
- Tiwari, RP; Virdi, JS; Gupta, LK; Sainiet, SS; Vadehra, DV. Development of chicks exposed to aflatoxin B1 during embryogenesis. Indian J. Animal. Sci 1989, 59, 1473–1474. [Google Scholar]
- Truckness, MW; Stoloff, L; Young, K. Aflatoxicol, aflatoxin B1 and M1 in eggs and tissues of laying hens consuming aflatoxin contaminated feed. Poultry Sci 1983, 62, 2176–2182. [Google Scholar]
- Smith, JE; Moss, MO. Mycotoxins: Formation, Analysis and significance; John Wiley & Sons: New York, NY, USA, 1985. [Google Scholar]
- van Pee, W; van Brabant, W; Joostens, J. Detection and determination of Aflatoxin M1 in milk and powdered milk. J. Res. Agric. Brux 1977, 30, 403–414. [Google Scholar]
- Schuller, WE. Aflatoxin M1 in liquid and powdered milk. Zesz. Probl. Postep. Nank Roln 1977, 189, 255–258. [Google Scholar]
- Karaioannoglou, P; Mantis, A; Koufidis, D. Occurrence of aflatoxin M1 in raw and pasteurized milk and in Feta and Teleme cheese samples. Milchwissenschaft 1989, 44, 746–748. [Google Scholar]
- Lamplugh, SM; Hendriskse, RG; Apeagyei, F; Mwanmut, DD. Aflatoxin in brest milk, neonatal cord blood and serum of pregnant women. Br. Med. J 1988, 296, 968. [Google Scholar]
- Moulé, Y. Transformations métaboliques de l’aflatoxin B1 et effet sur la synthèse des acides nucléiques et des protéines. Ann. Nutr. Aliment 1974, 28, 375–384. [Google Scholar]
- Gong, YY; Cardwell, K; Hounsa, A; Egal, S; Turner, PC; Hall, AJ; Wild, CP. Dietary aflatoxin exposure and impaired growth in young children from Benin and Togo: cross sectional study. BMJ 2002, 325, 20–21. [Google Scholar]
- Househam, KCH; Hundt, KL. Aflatoxin Exposure and its Relationship to Kwashiorkor in African Children. J. Trop. Pediat 1991, 37, 300–302. [Google Scholar]
- Boyle, BJ; Stearman, WC, III; Norman, JO; Perterson, HDV. Effect of Aflatoxin B1 on distribution of Fe, Cu, Zn, and Mn in rat Tissues. Vet. Toxicol. Entomol. Res. Lab 1977, 17, 33–39. [Google Scholar]
- Dvořáčková, I. Aflatoxins and Human Health; CRC Press: Boca Raton, FL, USA, 1990. [Google Scholar]
- Perera, FP. Molecular epidemiology: on the path to prevention. J. Natl. Cancer Inst 2000, 92, 602–612. [Google Scholar]
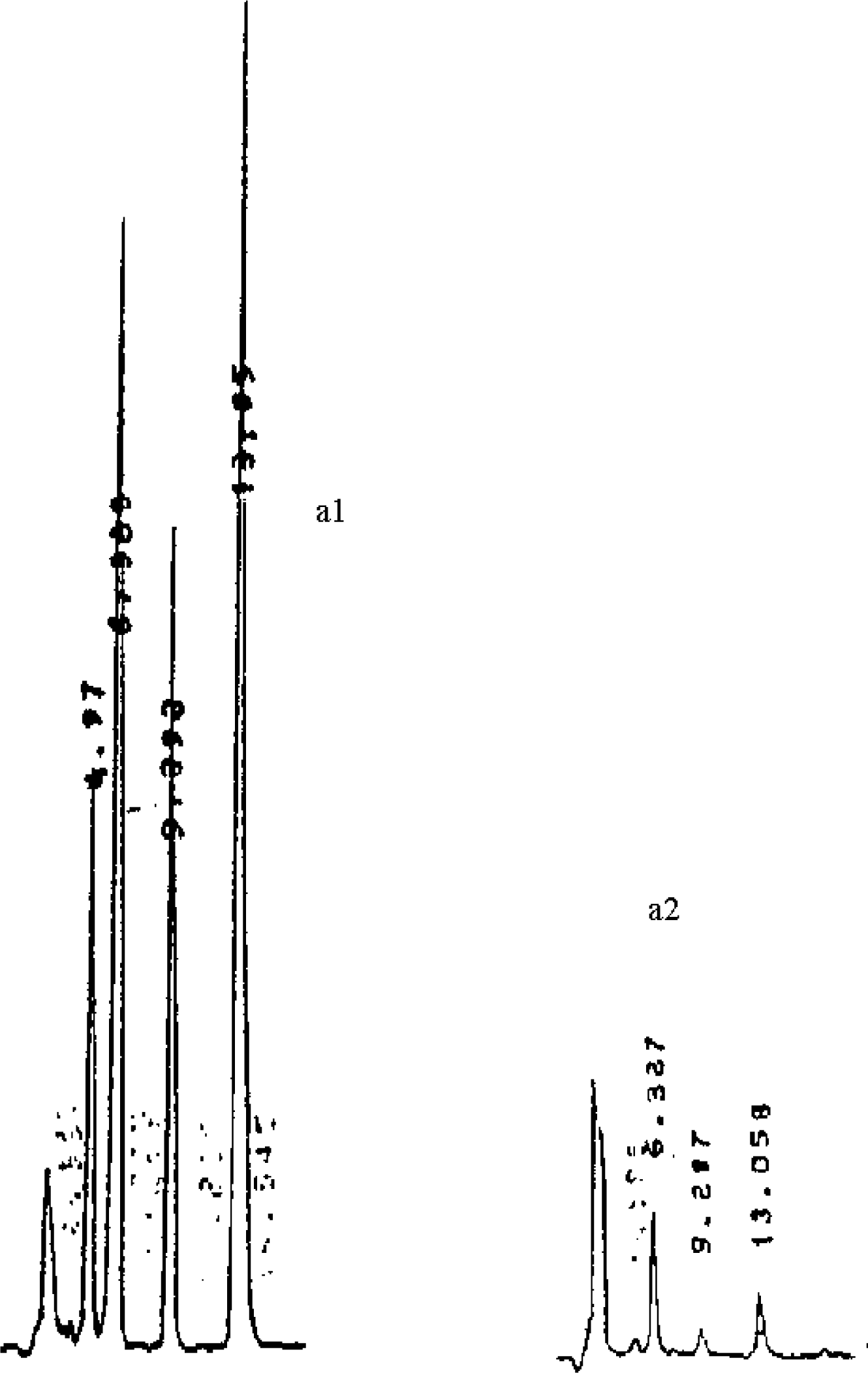
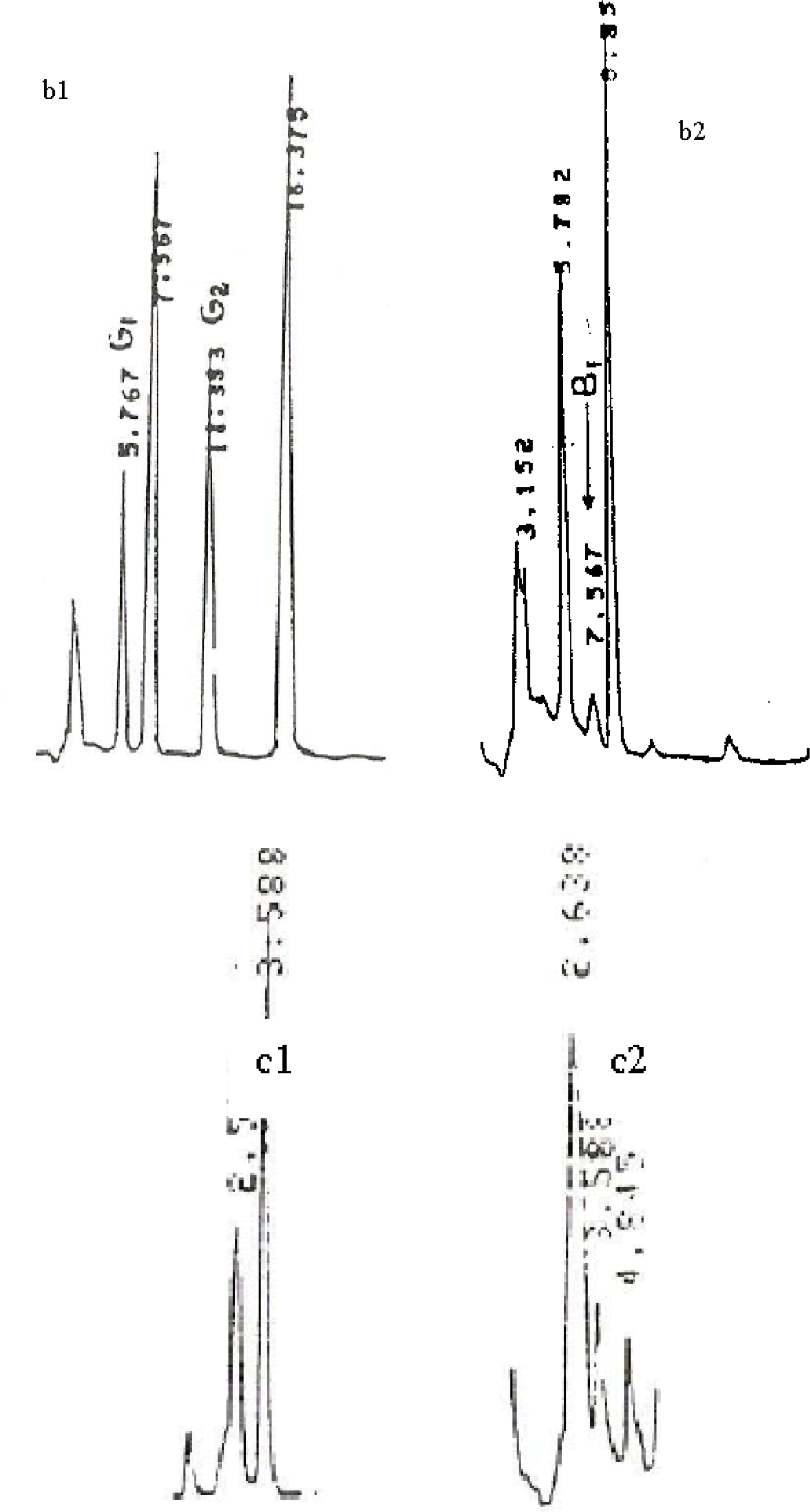
| Climatic areas | Samples | Type of AFs in sample | ||||||
|---|---|---|---|---|---|---|---|---|
| Analysed | Positive(..) | B1 | B2 | B2a | G1 | M1 | Range of AFs content in ppb | |
| Forest | 23 | 12 (52.5) | 1 | 1 | 10 | 4 | 2 | 0.021–7.604 |
| Savannah and steppe | 8 | 2 (25) | 0 | - | 0 | - | 2 | 0.003–0.008 |
| Littoral | 12 | 6 (50) | 0 | - | 5 | 1 | 1 | 0.126–0.768 |
| Mountain | 19 | 8 (42.1) | 3 | 2 | 1 | 5 | 0.002–7.200 | |
| Total | 62 | 28 (45.2) | 4 | 1 | 17 | 6 | 10 | 0,820±1,707 * |
| Type of milk | Sample analysed | Positive Samples | AFM1 content (μg/L) | Sample with level < 0.05* μg·L−1 | Sample with level > 0.05 μg·L−1 |
|---|---|---|---|---|---|
| Cow milk | 63 | 10 (15.9%) | 0.006 to 0.527 | 4 | 6 |
| Breastfeeding milk | 62 | 3 (4.8%) | 0.005 to 0.625 | 2 | 1 |
| Ages (Years) Groups | Gender | 1–3 | 4–5 | 6–9 | 10–12 | Total |
|---|---|---|---|---|---|---|
| Kwashiorkor | Male | 5 | 3 | 3 | 0 | 11 |
| Female | 3 | 1 | 5 | 2 | 11 | |
| Marasmic Kwashiorkor | Male | 4 | 3 | 1 | 1 | 9 |
| Female | 3 | 2 | 4 | 2 | 11 | |
| Control | Male | 2 | 4 | 7 | 7 | 20 |
| Female | 2 | 2 | 6 | 6 | 16 |
| Children Groups | Male | female | Total | AFB1 (μg·L−1) | |||
|---|---|---|---|---|---|---|---|
| Total | Positive | Total | Positive | Total | Positive | ||
| Kwashiorkor | 16 | 7 (43.8%) | 15 | 4 (26.6%) | 31 | 11 (35.5) | 0.109–2.840* |
| Marasmic kwashiorkor | 5 | 3 (60%) | 6 | 2 (33.3%) | 11 | 5 (45.5%) | 0.109–0.864* |
| Control | 20 | 3 (15%) | 16 | 1 (6.3%) | 36 | 4 (11.1%) | 0.07–0.155 |
| Age Range in years | Total number of patients | Positive patients to HbsAg | Positive patients to AFB1 | Positive patients for both AFB1 and HbsAg | |||
|---|---|---|---|---|---|---|---|
| Number | Number | Number | |||||
| Male | Female | Male | Female | Male | Female | ||
| 10–20 | 1 | 1 | - | 1 | - | 1 | - |
| 20–30 | 4 | 2 | 1 | 2 | 1 | 2 | - |
| 30–40 | 11 | 6 | 1 | 5 | 3 | 4 | - |
| 40–50 | 6 | 4 | 1 | 3 | - | 2 | - |
| 50–60 | 7 | 6 | - | 4 | - | 3 | - |
| 60–70 | 4 | 2 | - | 2 | - | 1 | - |
| 70–80 | 3 | 1 | - | 2 | - | - | - |
| Total | 36 | 22(61.1%) | 3(8.3%) | 19(52.7%) | 4(11.1%) | 13(36.1%) | - |
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Tchana, A.N.; Moundipa, P.F.; Tchouanguep, F.M. Aflatoxin Contamination in Food and Body Fluids in Relation to Malnutrition and Cancer Status in Cameroon. Int. J. Environ. Res. Public Health 2010, 7, 178-188. https://doi.org/10.3390/ijerph7010178
Tchana AN, Moundipa PF, Tchouanguep FM. Aflatoxin Contamination in Food and Body Fluids in Relation to Malnutrition and Cancer Status in Cameroon. International Journal of Environmental Research and Public Health. 2010; 7(1):178-188. https://doi.org/10.3390/ijerph7010178
Chicago/Turabian StyleTchana, Angele N., Paul F. Moundipa, and Félicité M. Tchouanguep. 2010. "Aflatoxin Contamination in Food and Body Fluids in Relation to Malnutrition and Cancer Status in Cameroon" International Journal of Environmental Research and Public Health 7, no. 1: 178-188. https://doi.org/10.3390/ijerph7010178




