Biosorptive Removal of Ni(Ii) from Wastewater and Industrial Effluent
Abstract
:Introduction
Materials and Methods
Preparation of Biosorbent Material (FBM)
Preparation of Chemically Treated Leached Biomass (LBM)
Preparation of Nickel Solution
Instrumentation
Batch Mode Adsorption Study
- Ci-initial concentration of metal ion mg/L;
- Ce – Equilibrium concentration of metal ion mg/L;
- m – mass of adsorbent g/L;
- qe – Amount of metal ion adsorbed per gram of adsorbent.
Column Mode Adsorption Studies
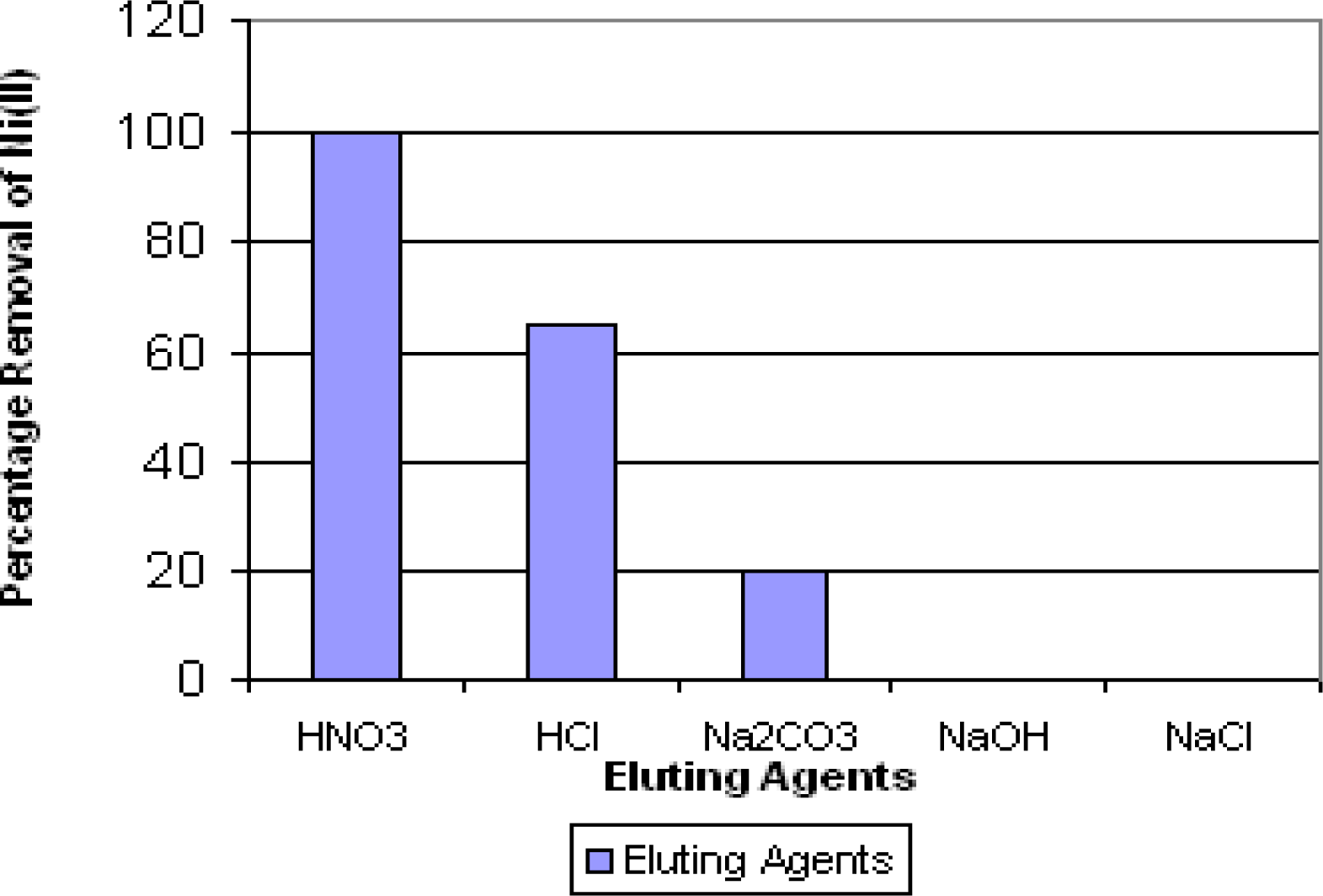
Desorption Study
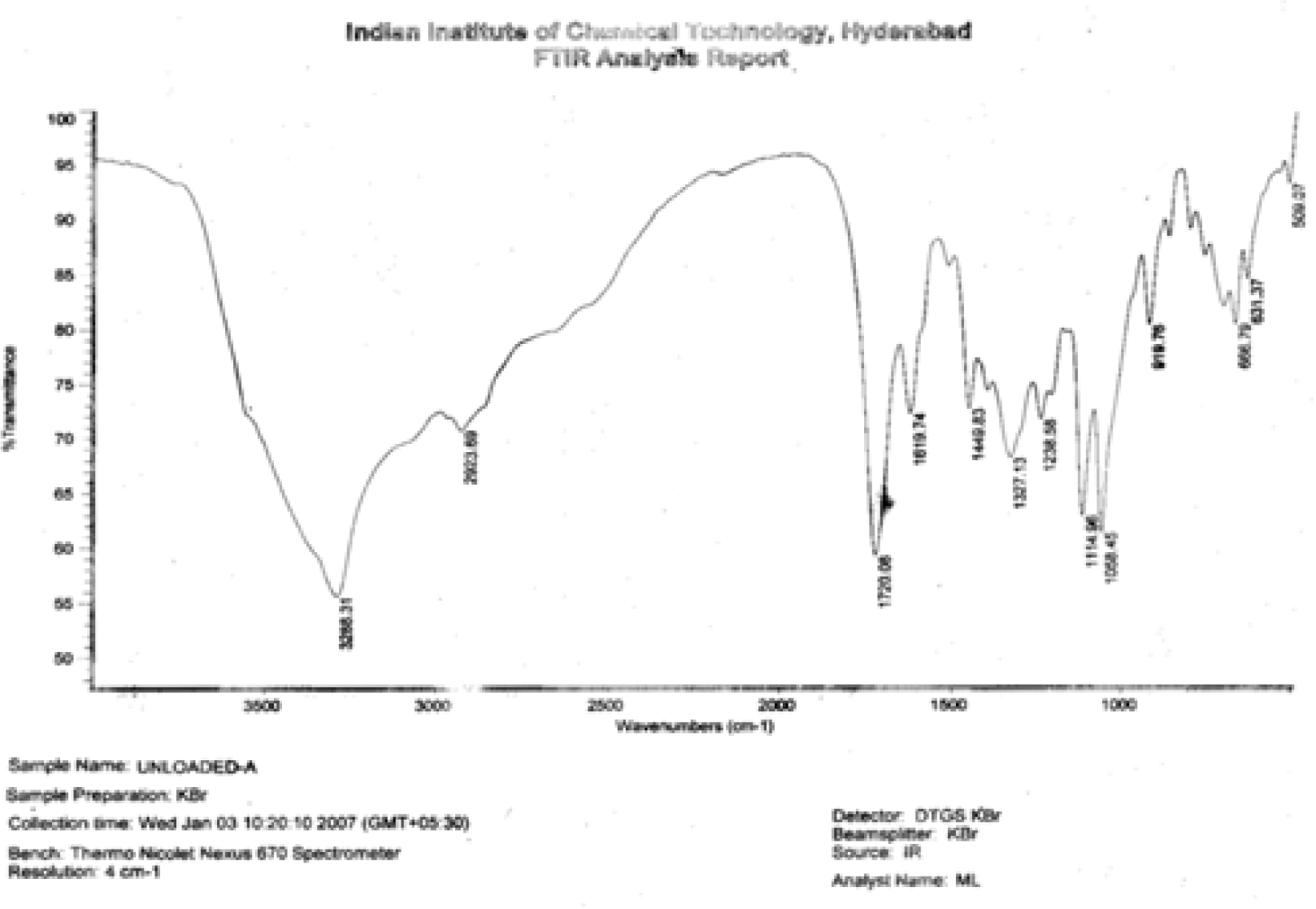
FT-IR Method
Results and Discussion
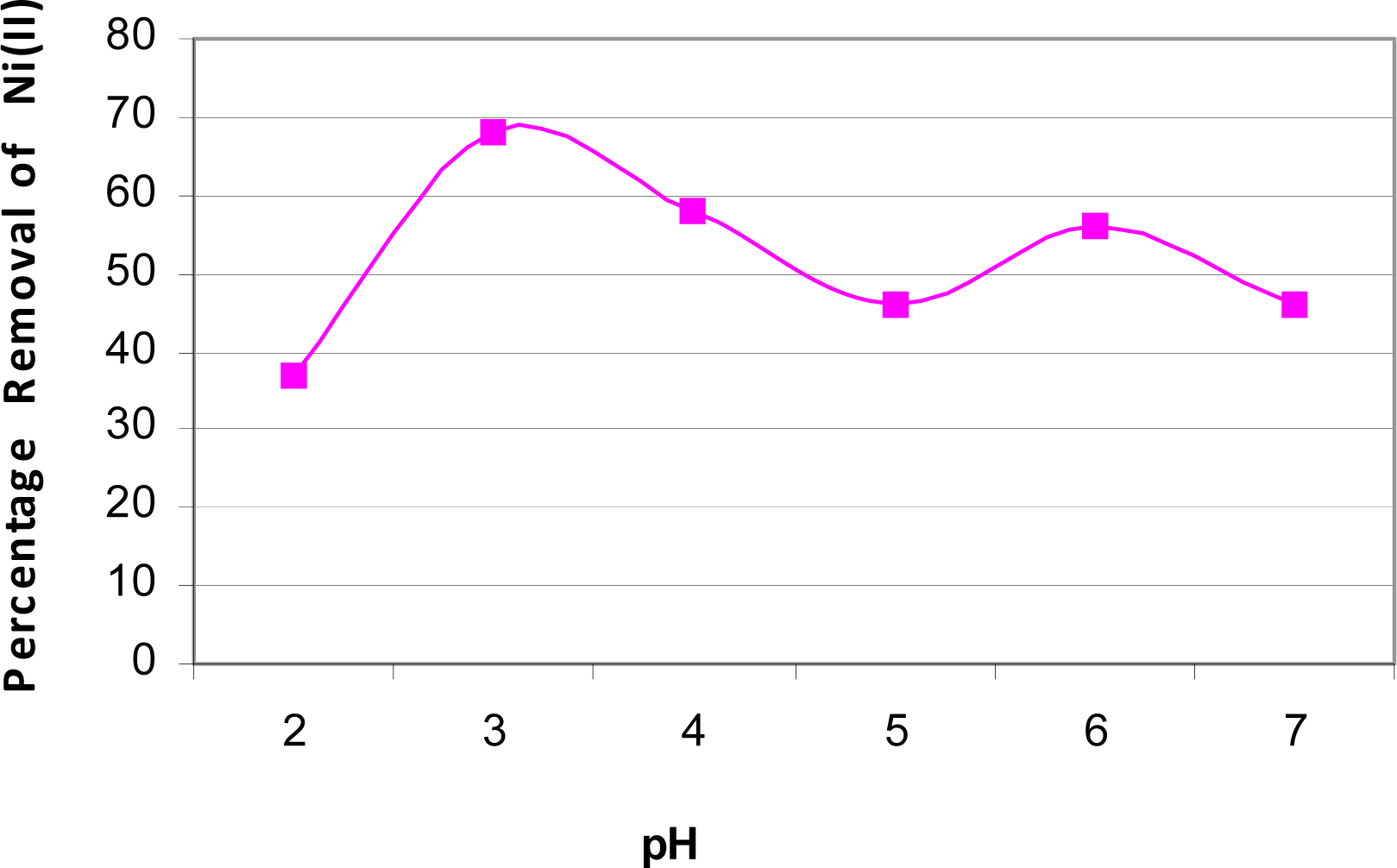
Effect of pH on Biosorption
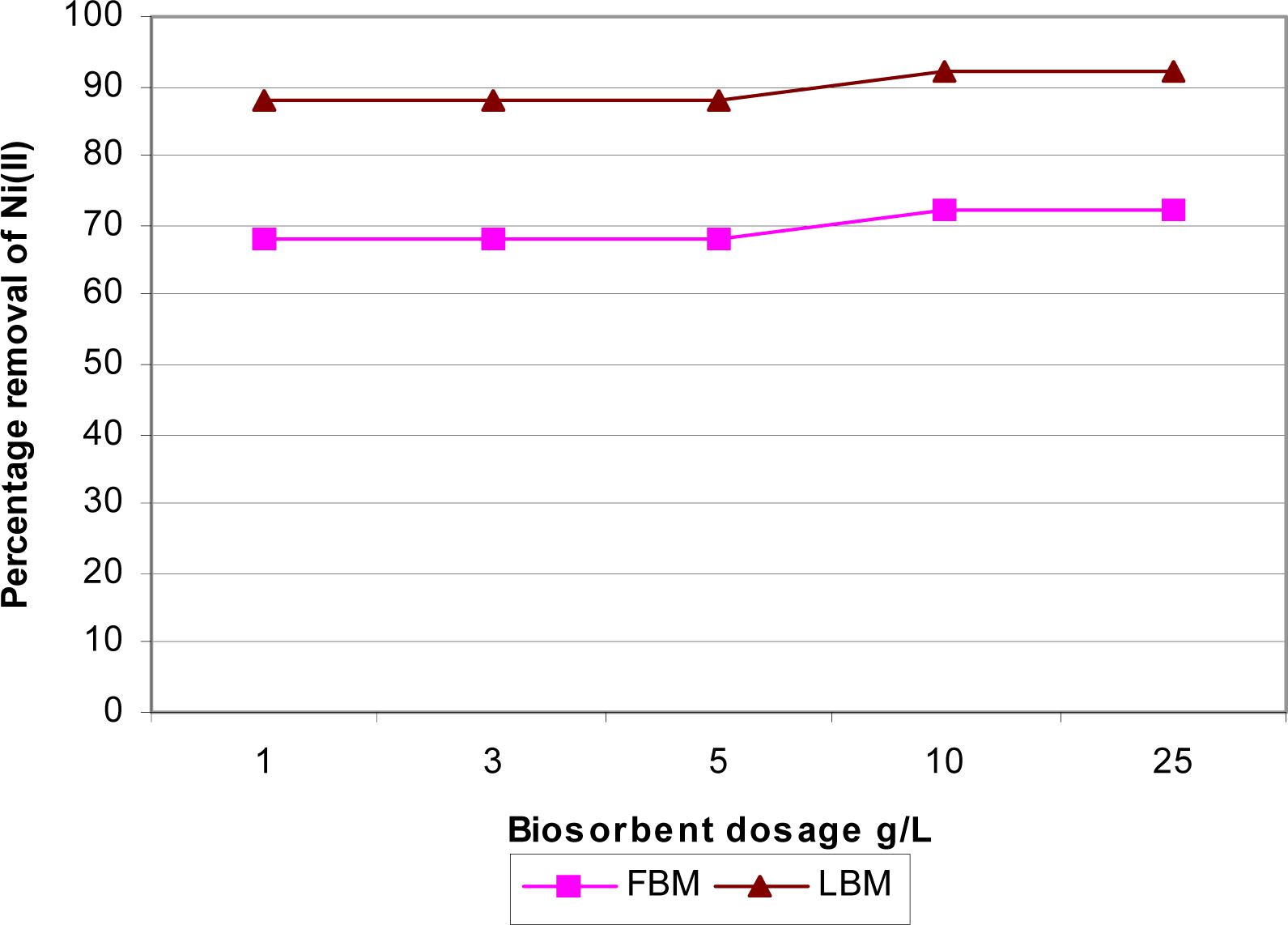
Effect of Biosorbent Dose
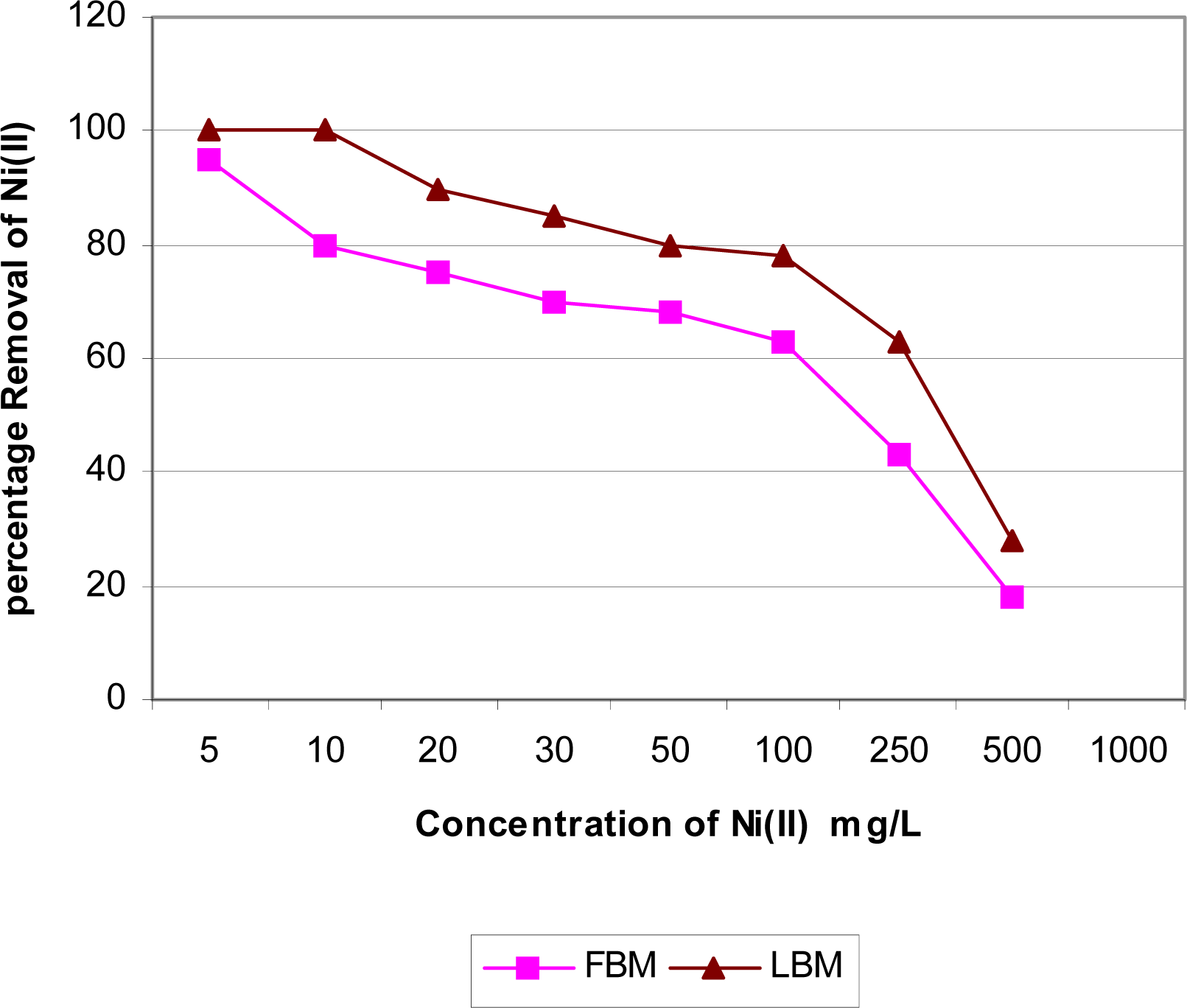
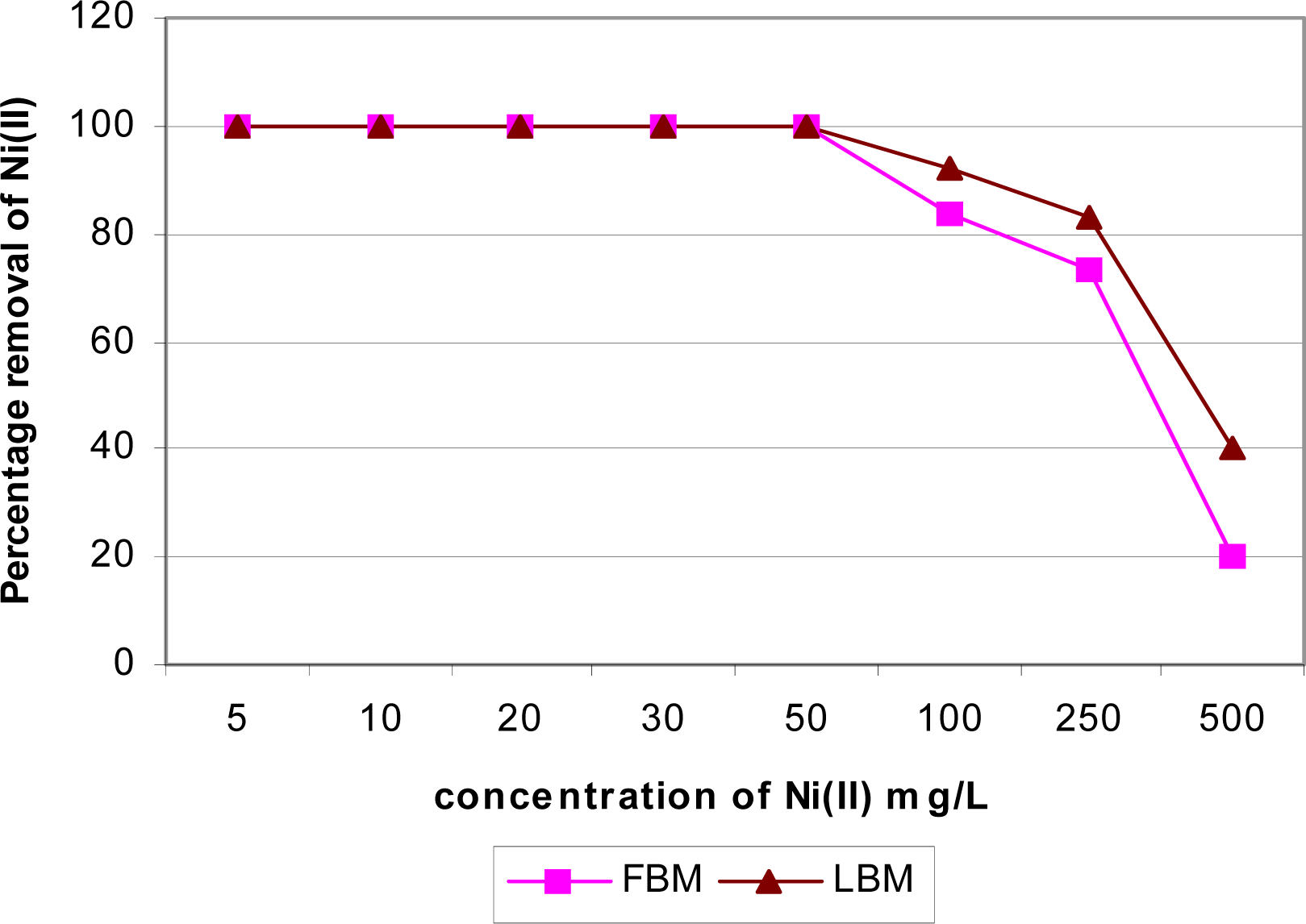
Effect of Initial Metal Concentration
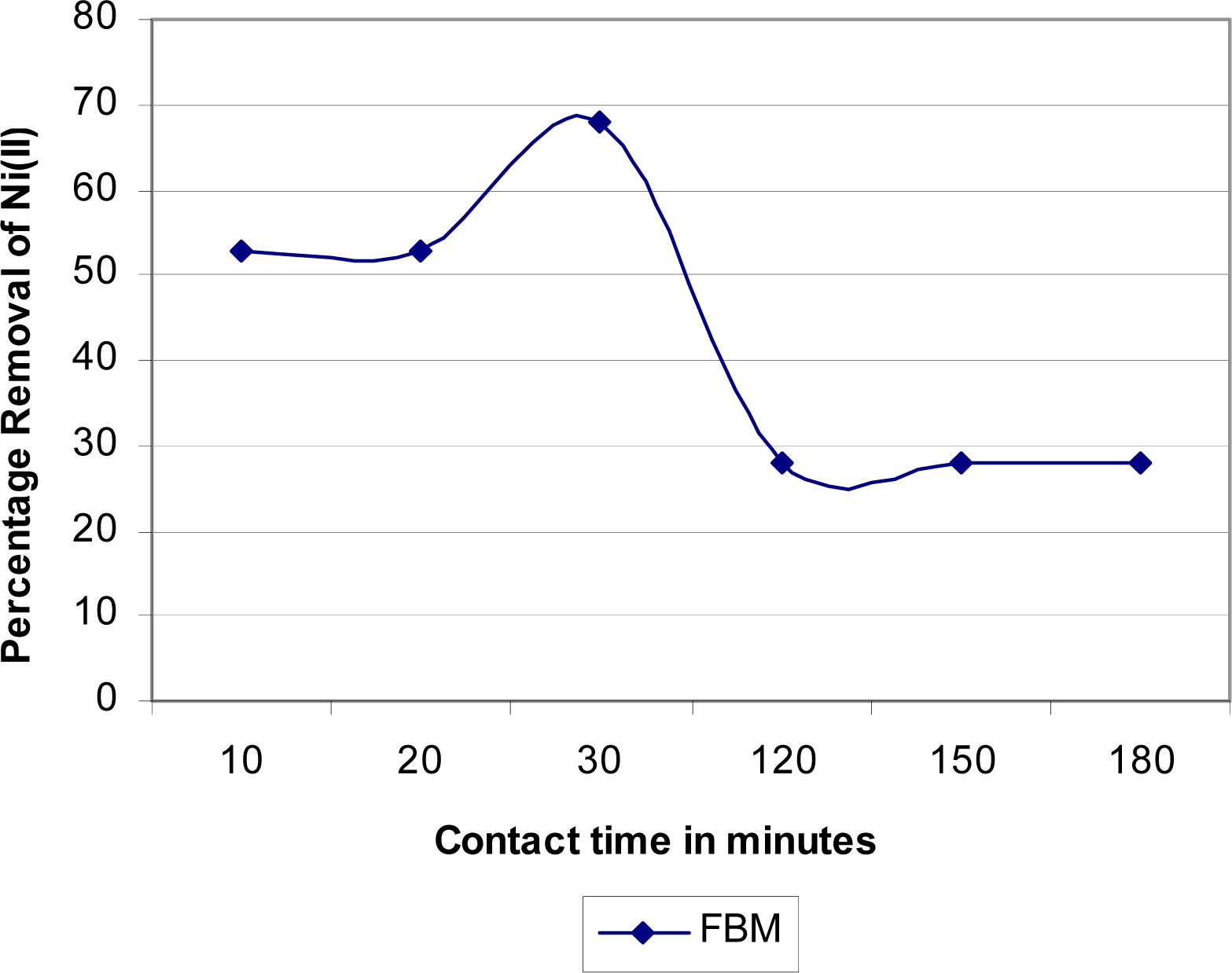
Analysis of the Kinetic of Ni(II) Removals
- Kad – is the rate constant of adsorbent
- q- is the amount of metal ion adsorbed at time t (mg/g)
- qe is the amount of metal ions adsorbed at equilibrium (mg/g)
- Kad – is the rate constant of adsorbent
- q- is the amount of metal ion adsorbed at time t (mg/g)
- qe is the amount of metal ions adsorbed at equilibrium (mg/g)
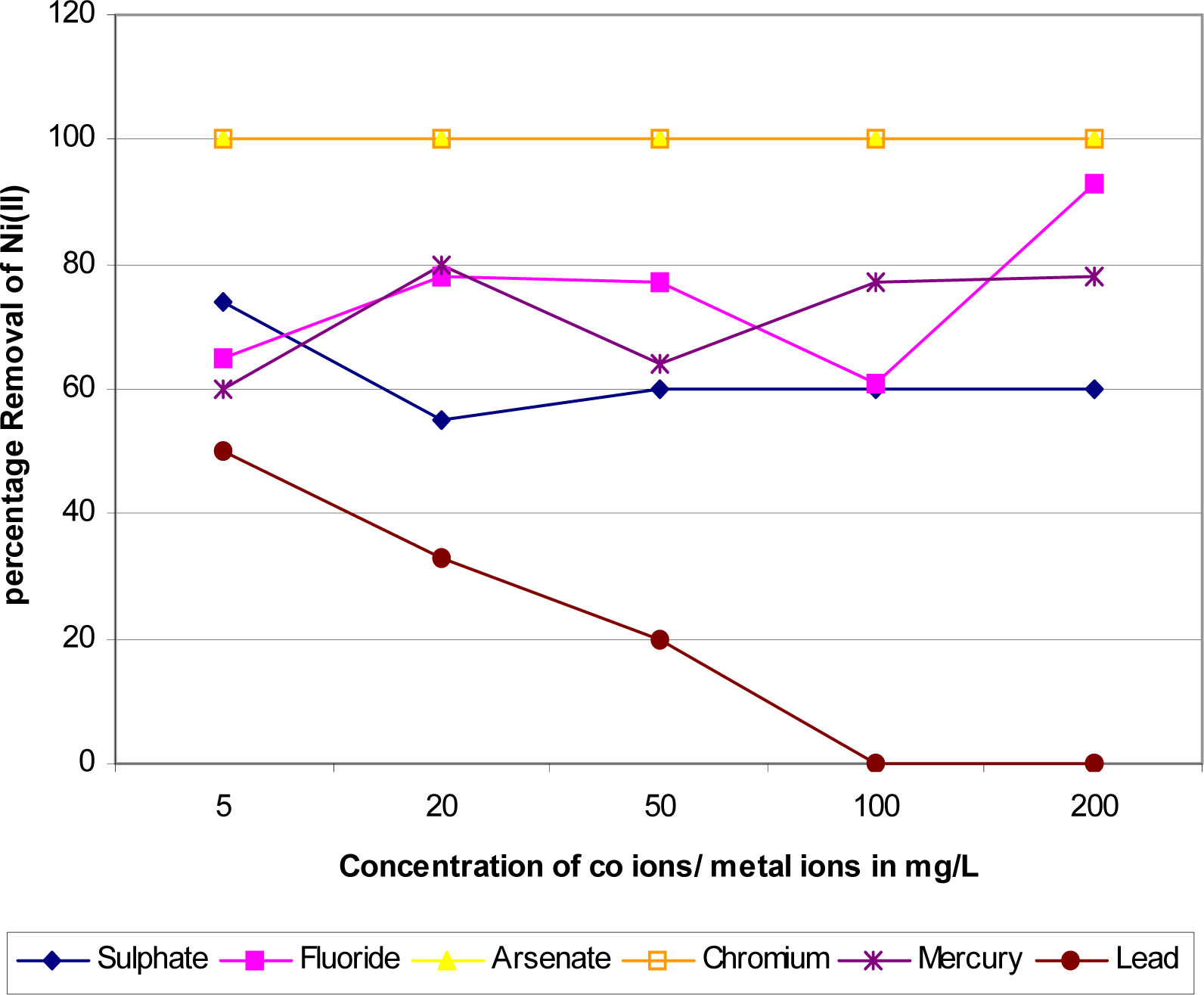
Effect of Background Ions
Column Study
Study on the Regeneration of Impregnated Biomass
Adsorption Isotherms
- S = moles sorbed at equilibrium per mass of sorbent (mg/g) i.e. (Ce/m)
- KF = Freundlich isotherm constant (L/g)
- N = Freundlich isotherm constant; N ≥ 1
- Cs = Sorbate concentration in solution at equilibrium (mg/L)
- S = moles sorbed at equilibrium /mass of sorbent (mg/g) i.e. Ce/m
- AM = maximum sorption capacity of the sorbent (mg/g)
- KL = Langmuir adsorption constant, related to binding energy of the Sorbate (l/mg)
- Cs = Sorbate concentration in solution at equilibrium (mg/L)
- Am = the maximum sorption capacity of the sorbent
Identification of Ni (II) Binding Functional Group
Application of Biomass for Removal of Nickel from Industrial Effluents
Conclusion

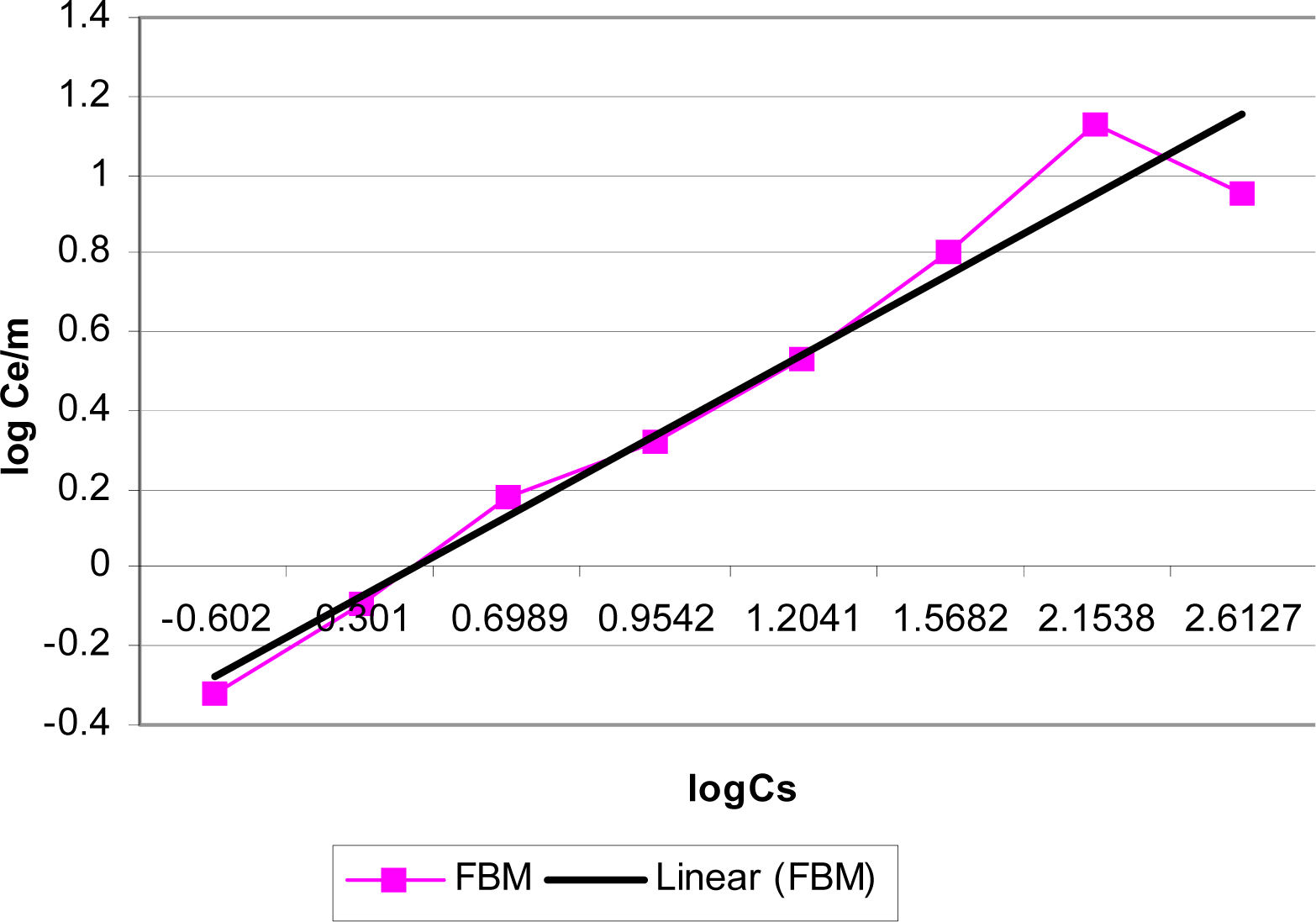


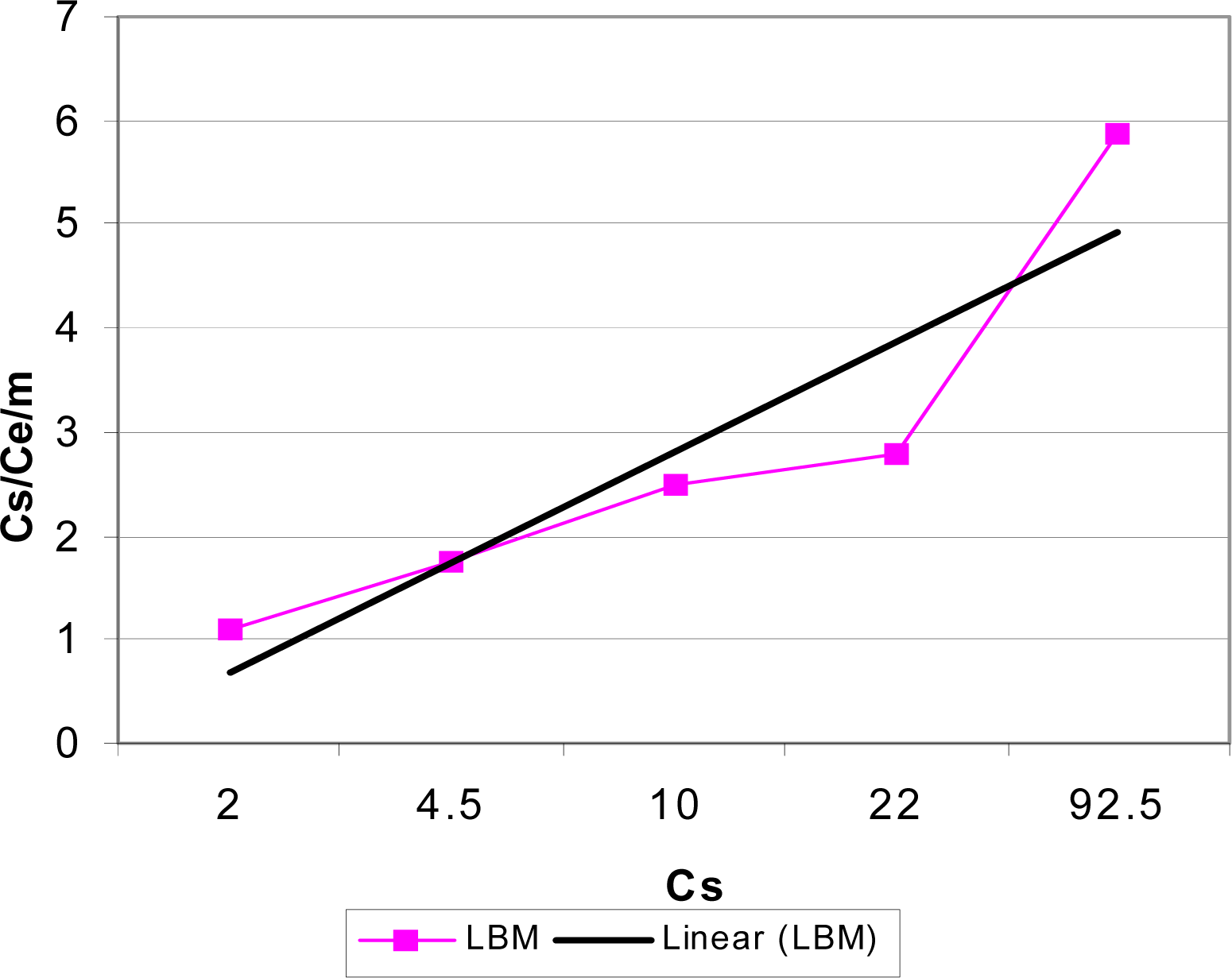

| Biomass | Adsorption Mode | R2 | 1/N | KF |
|---|---|---|---|---|
| FBM | Batch | 0.9561 | 0.2042 | 0.6174 |
| LBM | Batch | 0.9802 | 0.2354 | 0.9630 |
| Biomass | Adsorption Mode | R2 | AM | KL |
|---|---|---|---|---|
| FBM | Batch | 0.7728 | 0.606 | −0.9712 |
| LBM | Batch | 0.83 | 0.9451 | −0.3477 |
| Sample | pH | Percentage removal of nickel |
|---|---|---|
| Lab | 3.0 | 85 |
| Wastewater | 2.5 | 56 |
| Wastewater(adjusted pH) | 3.0 | 70 |
Reference
- Gangaiya, P. Land based pollution sources in Kiribiti: A case study. SPREP series # 80. South Pacific Regional Environmental Program, Apia Western Samoa 1994. [Google Scholar]
- Davey, EW; Morgan, MJ; Erickson, SJ. A biological measurement of copper complexation capacity in seawater. Limnol Oceanogr 1973, 18, 993–997. [Google Scholar]
- Nies, DH. Resistance to cadmium, cobalt, zinc and nickel in microbes. Volume Plasmid 27, pp. 17–28. Parker, P, Ed.; In Encyclopaedia of environmental science, 2nd edn; 1980; McGraw Hill: New York, 1992.
- Chen, CY; Lin, TH. Nickel toxicity to human term placenta: in vitro study on lipid peroxidation. J. Toxicol. Environ. Health Part A. 1998, 54, 37–47, [CrossRef]). [Google Scholar]
- Parker, P. Encyclopaedia of environmental science, 2nd edn; McGraw Hill: New York, 1980. [Google Scholar]
- Revathi, M. Removal of nickel ions from Industrial Plating Effluents Using Activated Alumina as Adsorbent. J. of Environmental & Engg. 2005, 47, 1. [Google Scholar]
- Volesky, B. Biosorption and biosorbents. In Biosorption of heavy metals; Boston, USA; CRC press, 1990; pp. 3–5. [Google Scholar]
- Kapoor, A; Viraraghavan, T. Fungal biosorption- an alternative treatment option for heavy metal bearing wastewater: a review. Bioresource Technolology 1995, 53(3), 195–206. [Google Scholar]
- Viraraghvan, TM; Drohamraju, M. Removal of copper, nickel and Zinc from wastewater by adsorption using peat. Env Sci Health 1993, 28A, 1261–1276. [Google Scholar]
- Dimitrova, SV. Metal sorption on blast-furnace slag. Water Research 1996, 30, 228–232. [Google Scholar]
- Maranon, E; Sastre, H. Heavy Metal Removal in Packed Beds Using Apple Wastes. Biores. Technol. 1991, 38(1), 39–43. [Google Scholar]
- Marshall, WE; Champagne, ET; Evans, WJ. The use of rice milling by products to remove metal ions from solution. J. Env Sci Health 1995, 9A, 1977–1992. [Google Scholar]
- Periasamy, K; Namasivayam, C. Removal of nickel(II) from aqueoussolution and nickel electroplating industry wastewater using an agricultural waste. Peanut hulls Waste Manage 1995, 15, 63–68. [Google Scholar]
- Larson, VJ; Schiernp, HP. Phosphorous in soil advances in agronomy. J. Env Qual. 1981, 10, 188–193. [Google Scholar]
- Meena, AK; et al. Adsorption of Ni(II) and Zn(II) from aqueous solution by chemically treated activated carbon. National conference on carbon (Indo- carbon) Kanpoor 2003, 131–141. [Google Scholar]
- Sharma, YC; Prasad, G; Rupainwar, DC. Removal of Ni(II) from aqueous solutions by sorption. Interl J. of Env. Stud. 1992, 40, 535. [Google Scholar]
- Shumate, ES; Strandberg, WG. Accumulation of metals by microbial cells. Comprehensive Biotechnology 1985, 13, 235–247. [Google Scholar]
- Andres, MYJH; Hubert, CJ. Bacterial biosorption and retention of thorium and uranyl cations by mycobacterium smegmatis. J. of Radio analyses Nuclear Letter 1992, 166, 431–440. [Google Scholar]
- Fourest, E; Roux, CJ. Heavy metal biosorption by fungal mycilial by- products: mechanisms and influence of pH. Applied Microbiology and Biotechnology 1992, 37(3), 399–403. [Google Scholar]
- Hussein, H; Krull, R; Abouel-Ela, SI; Hempel, DC. Interaction of the different heavy metal ions with immobilized bacterial culture degrading xenobiotic wastewater compounds. Conference Proceedings: International Water Association World Water Conference; ss(2º, 15th– 19th October, 2001, Berlin, Germany),. 2001. [Google Scholar]
- Hussein, H; Farag, S; Moawad, H. Isolation and characterisation of Pseudomonas. Resistant to heavy metals contaminants. Arab Journal of Biotechnology 2003, 7, 3–22. [Google Scholar]
- Ashkenazy, R; Gottlieb, L; Yannai, S. Characterization of acetone washed yeast biomass functional groups involved in lead biosorption. Biotechnol, Bioeng 1997, 55(1), 1–10. [Google Scholar]
- Padmarathy, V; Vasudevan, P; Dhingra, SC. Thermal end spectroscopic studies on sorption of Ni(II) ion on protonated baker’s yeast. Chemosphere 2003, 52(10), 1807–1817. [Google Scholar]
© 2007 MDPI All rights reserved.
Share and Cite
Pandey, P.K.; Choubey, S.; Verma, Y.; Pandey, M.; Kamal, S.S.K.; Chandrashekhar, K. Biosorptive Removal of Ni(Ii) from Wastewater and Industrial Effluent. Int. J. Environ. Res. Public Health 2007, 4, 332-339. https://doi.org/10.3390/ijerph200704040009
Pandey PK, Choubey S, Verma Y, Pandey M, Kamal SSK, Chandrashekhar K. Biosorptive Removal of Ni(Ii) from Wastewater and Industrial Effluent. International Journal of Environmental Research and Public Health. 2007; 4(4):332-339. https://doi.org/10.3390/ijerph200704040009
Chicago/Turabian StylePandey, Piyush Kant, Shweta Choubey, Yashu Verma, Madhurima Pandey, S. S. Kalyan Kamal, and K. Chandrashekhar. 2007. "Biosorptive Removal of Ni(Ii) from Wastewater and Industrial Effluent" International Journal of Environmental Research and Public Health 4, no. 4: 332-339. https://doi.org/10.3390/ijerph200704040009




