Introduction
Schistosomiasis is water borne disease and it is estimated to affect 200 million people in 76 countries and approximately 600 million people have been estimated to be at risk of infection [
1].
Schistosoma haematobium is spread through contact by uninfected persons with fresh water, which is contaminated with the urine of infected persons. The disease continues to spread and intensify with the expanding water resource projects, which are needed to meet the increasing food demands in Cameroon. Schistomiasis is a public health problem in Cameroon and contributes to a third of the morbidity among school-age children in Kumba.
The southwest Province of Cameroon has an Equatorial climate, with a 9-months rainy season and a 3 months dry season each year. Rainfall is very heavy 1500–2000mm/year. The snail hosts of the schistosome parasites of man are scare throughout the area [
2] and only a few isolated foci of
schistosoma haematobium are found [
3].
Kumba, an administrative and commercial centre with a population of 80 000, lies in the South-West Province, on the banks of the Kumba River. Since its inhabitants come from a wide variety of cultural backgrounds and socio-economic groups, a study of the effects of these factors on several tropical diseases is ideal. Dombeu and collaborators reported the existence of a focus of
S. haematobium in Kumba [
4]. This is possibly the first reported even though the population has been well studied in earlier years. The aims of the present study, conducted in Kumba, were to quantify the potential risk of schistosome infection from the activities involving water contact and schistosome infection and to identify any behavioural or socio-cultural factors that increased the risk of the infection.
Materials and Methods
A preliminary survey was carried out to identify the areas of Kumba town where the prevalence of
S. haematobium infection in the population was >10%. A complete census of all housing units was then carried out in these areas and urine samples were collected in 50-ml tubes from all members of each household who were aged ≥1 year. Urine was collected between 11.00 and 14.00 hours, the period of peak intensity of infection in the sample [unpublished], preserved with sodium azide and examined using sedimentation. Sedimentation appears to be as sensitive a method as centrifugation or filtration [
5].
The data collected from each household included the occupation, ethnic group and religion of each member and objective data used to calculate a socio-economic index. A questionnaire was administered to sub-sample of 665 individuals, to gather information on their level of domestic, recreational and occupational water contact, and their level of knowledge about schistosomiasis transmission. The main water-contact activities were bathing, laundry, food preparation, dishwashing, playing, fishing, and wading. A coefficient indicating the risk of each activity, based on the average duration of the water contact and the proportion of the body exposed [both measured by direct observation], the use of soap and the location where the activity occurred, was calculated.
Table 1 presents the activity coefficients used to quantify risk behaviours.
A water-contact index was then calculated for each subject by multiplying the water-contact coefficient for each activity by the number of times the individual performed the activity each week and then summing the results for all activities. Indices of [0, 1–9, 10–25, and 26–53] indicated no, low, medium and high levels of contact, respectively.
Knowledge about schistosomiasis was evaluated by asking five simple questions, each subject being given a point for each correct answer, giving scores of 0–5. The questions explored knowledge about the cause, clinical symptoms, and availability of treatment, transmission and prevention of schistosomiasis.
All subjects found to be infected with schistosomes were given a single oral dose of 40 mg praziquantel/kg, which was well tolerated.
Results
Population
The census in area with >10% schistosomiasis prevalence involved 171 households with 1025 residents. Most of the subjects were young 60% <20 years, 31% 20–44 years and only 9% >45 years with a slight preponderance of females 55%. On average each household comprised six individuals range 1–2, usually an extended family. Although the subjects belonged to 27 ethnic groups they could be classified in five main categories: Bakundus from the Douala coast; Bamileke from the western, hill region; Hausa Foulbe, Fulani and Hausa from northern Cameroon; and Nigerians and Tikaris from the north-western, hill region. Hausas were predominantly Muslims, the Bamileke Roman Catholics and the Bakundus, Tikaris and Nigerians Protestants. Two thirds of the population 65% were from the lower socioeconomic group [labourers and unskilled workers], 30% from the middle (semi-skilled or skilled workers) and 5% from the high professionals.
Prevalence and Intensity of Infection
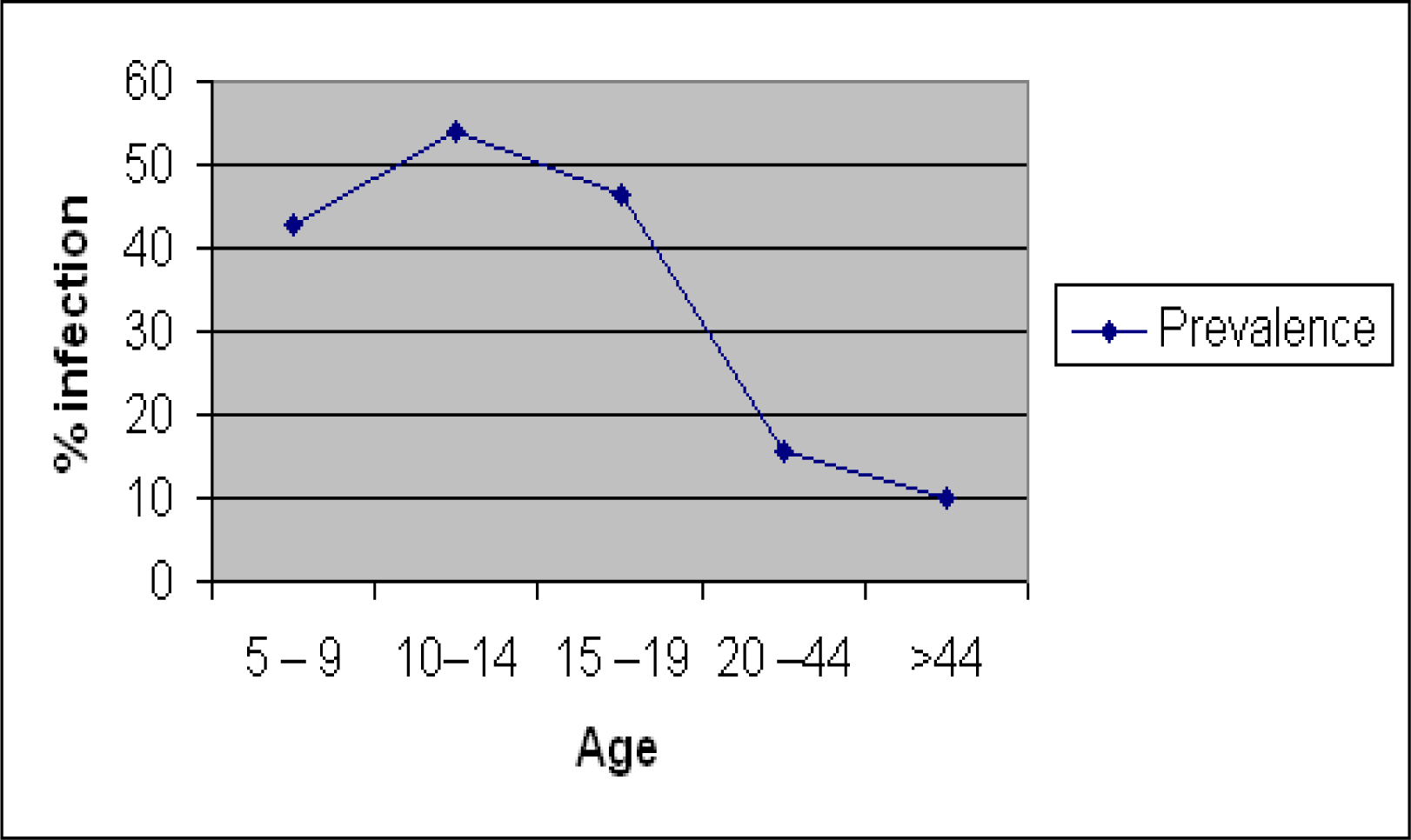
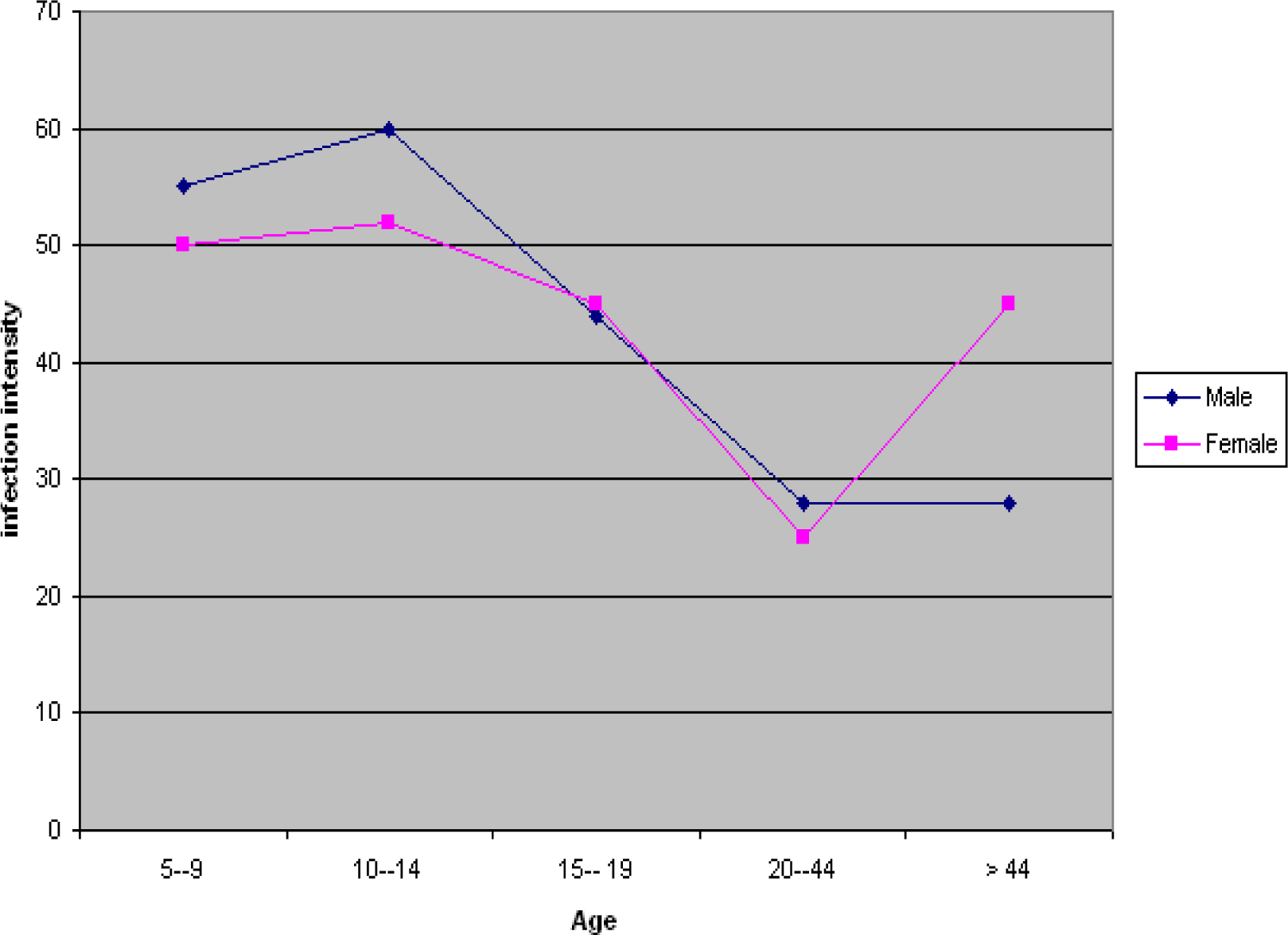
The prevalence and intensity of infection are presented in
Table 2, and graphically illustrated in
Figure 1 and
Figure 2. The prevalence rates were relatively high in the 10–14 and 15–19 age groups compared to the 5–9, 20–44 and >44 age groups. The overall infection rates were 42.5, 54.1, 46.5, 15.8, and 10.0% in the 5–9, 10–14, 15–19, 20–44 and >44 age group, respectively. In the male subjects, the intensities of infection were 55, 60, 44, 42 and 28 eggs per 10 ml urine, respectively in the 5–9, 10–14, 15–19, 20–44 and >44 age group. In the female subjects, infection intensities of 50, 52, 45, 51 and 25 eggs per 10 ml urine, were recorded for the 5–9, 10–14, 15–19, 20–44 and >44 age group respectively.
Knowledge about Schistosomiasis
Most respondents 68% knew that haematuria was the main sign of schistosomiasis, half (50%) knew that a drug was available to treat it, 42% knew that the cause was a worm, 36% knew that urinating in water was the main mode of transmission and only 25% knew that avoiding contact with certain water bodies was the best preventive action. Scores of 0, 1, 2, 3, 4 and 5 were achieved by 8%, 16%, 26%, 19%, 16% and 14% of respondents, respectively. Those aged 15–19 years had the highest mean score and those aged >44 the lowest, with male having significantly higher level of knowledge than females. The Nigerian had the highest water-contact levels and the lowest knowledge scores.
Table 3 presents the intensity of reported surface water contact by age group and sex.
Multi-factorial Analysis
In order to determine the factors that were associated with infection, the data was first analysed in a hierarchical log-linear model after categorization of the variables. From this first step it appeared that occupation, housing and socio-economic status were not important. The more significant variables of age, sex, ethnic group, religion, knowledge and reported water-contact were then analysed by step-wise logistics regression
table 4. All age groups had a relative risk, RR> 1; those aged 10–19 years having the highest RR 3.6. Nigerians had a relatively high RR 2.2; all other ethnic groups having RR of about 1. In terms of water contact, only the high-contact group were at increased risk of infection. Paradoxically, better knowledge about schistosomiasis was associated with a higher relative risk of being infected.
Table 4 presents the stepwise logistic regression of predictors of schistosomiasis.
Discussion
Although clean water is available at numerous fountains throughout Kumba, many inhabitants still use surface water for their domestic activities. Unless sufficient clean water is readily available in a house itself or nearby, its inhabitants will use surface water to carry out their domestic chores.
Age, as expected and as observed in most schistosomiasis surveys, was a major determinant of schistosomiasis infection in Kumba; middle age children being at the highest risk of infection. At similar levels of exposure, children are more susceptible to schistosomiasis, probably because they lack the immunity built up in adults as the result of previous infections [
6].
Only a very intense level of contact with surface water could be related to high relative risk of infection, possibly because the estimation of water contact from interview data is not very accurate. Although a significant association between risk of infection, and the number and duration of water contact with infected waters has often been reported [
7,
8], it appears that the respective roles of age and intensity of water contact need to be considered together [
9,
10].
Most surface-water contact by the children, the group most affected by schistosomiasis, occurs during playing and swimming and it is likely that health education would have a significant impact on these recreational activities. The positive association between knowledge and schistosomiasis was initially surprising. After all, the aim of health-education programmes is to teach people about disease so that they can take preventive action. It may be that individuals with high-risk behaviours have been infected in the past and having gone for treatment, learned more about the disease through their contact with health professionals. If this is so, their experience was not sufficient to trigger the changes in their behaviour that would reduce their risk of infection, as many have been re-infected.
The reasons why ethnic group should be a significant determinant of infection in Kumba are not clearly understood. It may be that the specific behaviours and attitudes of particular ethnic groups lead to levels of surface-water contact that differ from those of other groups. Further interviews are required to elucidate this problem.
There are several ways in which the Kumba focus may be controlled. It is obvious that, although clean water is available, it is not close enough to many households; it should be made easier for all households to obtain clean water than surface water. Given the large population in Kumba, the low cost of unskilled labour and the limited areas where the intermediate host occurs, it should also be possible to reduce infection risk by regularly clearing the snail-infested sites on the stream and river banks. It is probably unrealistic to believe that health education of the children or their parents would stop them playing or swimming in surface water; those most at risk, the middle age children, are little parental control. Health education should not only improve local knowledge about the risk of surface water contact but encourage long-term changes in behaviour and rapid treatment as soon as the symptoms of schistosomiasis develop.