Systems Biology: New Approaches to Old Environmental Health Problems
Abstract
:Introduction
The Envirome
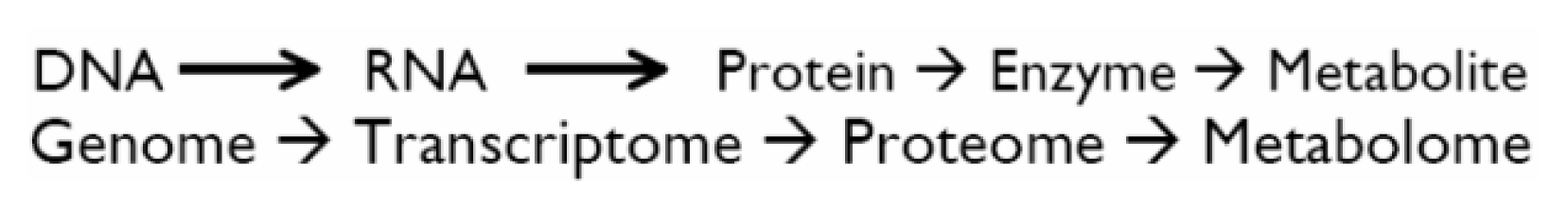
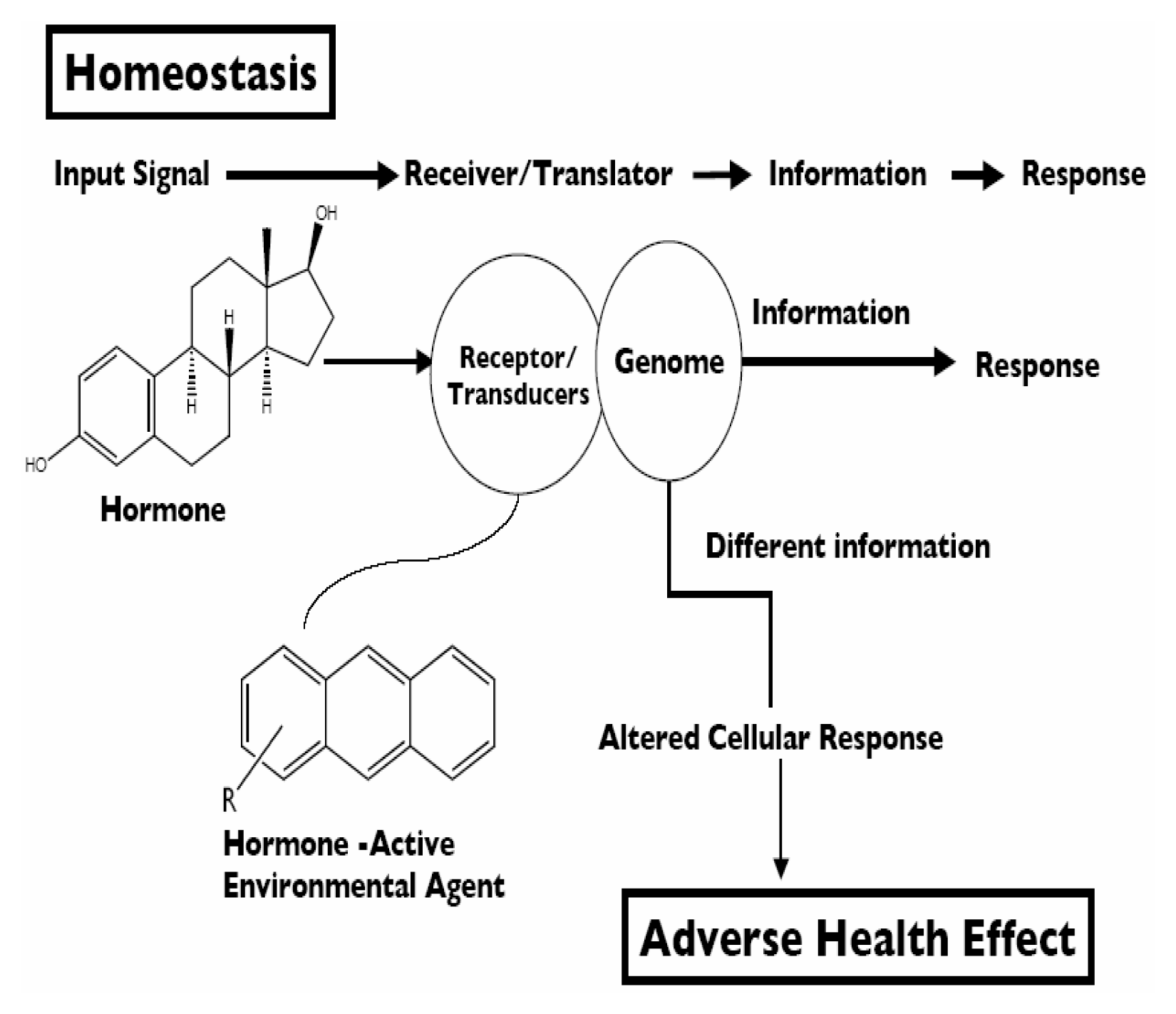
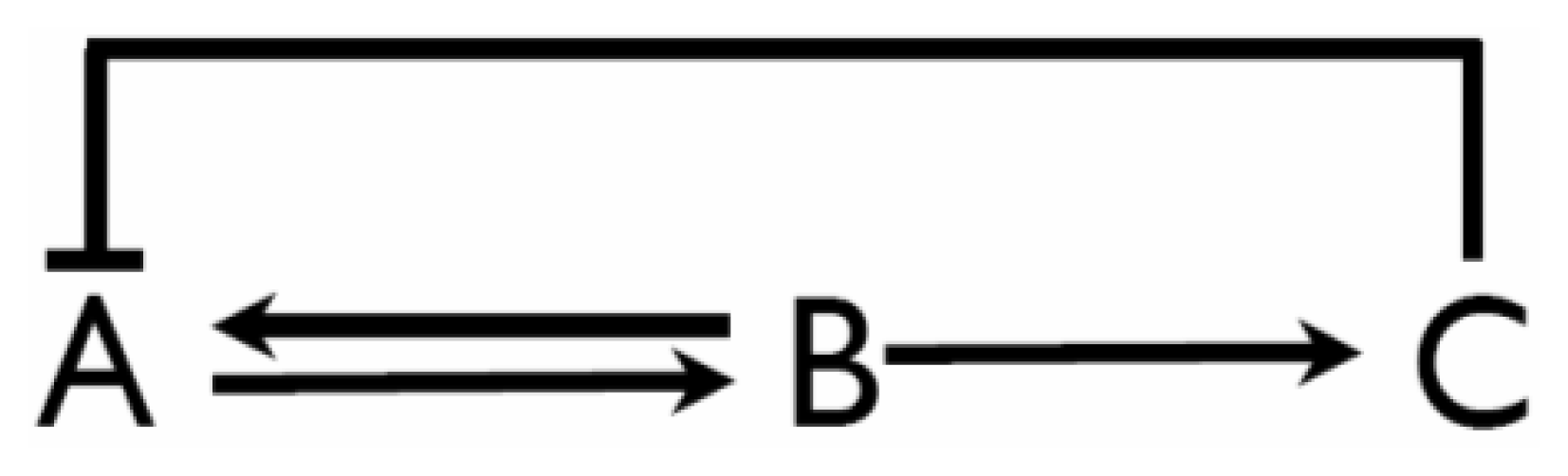
Environmental Systems Biology
Where Do We Go From Here?
Acknowledgements
References
- Vegetti, M. Law and nature in the Hippocratic treatise “De aere”. Med. Secoli. (Italy) 1995, 7, 611–619. [Google Scholar]
- Alvarez, M. C. Graeco-Roman case histories and their influence on Medieval Islamic clinical accounts. Soc. Hist. Med 1999, 12, 19–43. [Google Scholar]
- Geisen, G. L. The Private Science of Louis Pasteur; Princeton University Press: Princeton, NJ, 1995. [Google Scholar]
- Baxter, A. G. Louis Pasteur’s beer of revenge. Nat Rev Immunol 2001, 1, 229–232. [Google Scholar]
- Prost, A. From disease to health: The individual, society, environment and culture. Sante (France) 1995, 5, 331–333. [Google Scholar]
- Waller, J. The discovery of the germ: Twenty years that transformed the way we think about disease; Columbia University Press: NY, 2002. [Google Scholar]
- Roberts, L. Polio Endgame: The exit strategy. Science 2004, 303, 1960–1071. [Google Scholar]
- John, T. J. The golden jubilee of vaccination against poliomyelitis. Indian J. Med. Res 2004, 119, 1–17. [Google Scholar]
- Franklin, R. E.; Gosling, R. G. Molecular Configuration in Sodium Thymonucleate. Nature (London) 1953, 171, 740–741. [Google Scholar]
- Watson, J. D.; Crick, F. H. Molecular structure of nucleic acids: A structure for deoxyribose nucleic acid. Nature (London) 1953, 171, 737–738. [Google Scholar]
- Seegmiller, J. E. Genetic and Molecular Basis of Human Hereditary Diseases. Clin. Chem 1967, 13, 554–564. [Google Scholar]
- Day, I. N. M.; Gu, D.; Ganderton, R. H.; Spanakis, E.; Ye, S. Epidemiology and the genetic basis of disease. Int. J. Epidemiol 2001, 30, 661–667. [Google Scholar]
- Fitzgerald, P. J. From Demons Evil Spirite to Cancer Genes; American Registry of Pathology: Washington, DC, 2000. [Google Scholar]
- Carson, R. Silent Spring; Houghton Mifflin Co.: Boston, MA, 1962; Reprinted 2002. [Google Scholar]
- Mazur, A. Hazardous Inquiry: The Rashomon Effect at Love Canal; Harvard University Press: Cambridge, MA, 1997. [Google Scholar]
- Brown, P.; Clapp, R. Looking back on Love Canal. Public Health Rep 2002, 117, 9598. [Google Scholar]
- Renn, O. Perception of Risks. Toxicol. Lett 2004, 149, 403–413. [Google Scholar]
- Grandjean, P. Implications of the precautionary principle for primary prevention and research. Annu. Rev. Public Health 2004, 25, 199–223. [Google Scholar]
- Rushton, L.; Elliott, P. Evaluating evidence on environmental health risks. Br. Med. Bull 2003, 68, 113–128. [Google Scholar]
- Kaufman, J. S.; Poole, C. Looking back on “causal thinking in the health sciences”. Annu. Rev. Public Health 2000, 21, 101–119. [Google Scholar]
- Maldonado, G.; Greenland, S. Estimating causal effects. Int. J. Epidemiol 2002, 31, 422–429. [Google Scholar]
- Collins, F. S.; Morgan, M.; Patrinos, A. The Human Genome Project: lessons from large-scale biology. Science 2003, 300, 286–90. [Google Scholar]
- Austin, M. A. The impact of the completed human genome sequence on the development of novel therapeutics for human disease. Annu. Rev. Med 2004, 55, 1–13. [Google Scholar]
- Thomas, P. D.; Kejariwal, A. Coding single-nucleotide polymorphisms associated with complex vs. Mendelian disease: Evolutionary evidence for differences in molecular effects. Proc. Natl. Acad. Sci. (USA) 2004. [Google Scholar]
- Primrose, S. B.; Twyman, R. M. Genomics: Applications in Human Biology; Blackwell Scientific: Malden, MA, 2004. [Google Scholar]
- Bentley, D. R. The human genome project -An overview. Med. Res. Rev 2000, 20, 189186. [Google Scholar]
- Crick, F. H. Central dogma of molecular biology. Nature (London) 1970, 227, 561–563. [Google Scholar]
- Bentley, D. R. Genomes for medicine. Nature (London) 2004, 429, 440–445. [Google Scholar]
- Orcutt, B. C.; George, D. G.; Dayhoff, M. O. Protein and nucleic acid sequence database systems. Annu. Rev. Biophys. Bioeng 1983, 12, 419–441. [Google Scholar]
- Ouzounis, C. A.; Valencia, A. Early bioinformatica: The birth of a discipline--A Personal View. Bioinformatics 2003, 19, 2176–2179. [Google Scholar]
- Mori, C. M. K.; Adachi, T.; Sakurai, K.; Nishimura, D.; Takashima, K.; Todaka, E. Application of toxicogenomic analysis to risk assessment of delayed long-term effects of multiple chemicals, including endocrine disruptors in human fetuses. Environ Health Perspect 2002, 111, 7–13. [Google Scholar]
- Grody, W. W. Molecular genetic risk screening. Annu. Rev. Med 2003, 54, 473–490. [Google Scholar]
- Shastry, B. S. SNP Alleles in human disease and evolution. J. Hum. Genet 2002, 47, 561566. [Google Scholar]
- Crawford, D. C.; Carlson, C. S.; Reider, M. J.; Carrington, D. P.; Yi, Q.; Smith, J. D.; Eberle, M. A.; Kruglyak, L.; Nickerson, D. A. Haplotype diversity across 100 candidate genes for inflammation, lipid metabolism, and blood pressure regulation in two populations. Amer. J. Hum. Genet 2004, 74, 610–622. [Google Scholar]
- Erichsen, H. C.; Chanock, S. J. SNPs in cancer research and treatment. Br J Cancer 2004, 90, 747–51. [Google Scholar]
- Judson, R.; Salisbury, B.; Schneider, J.; Windemuth, A.; Stephens, J. C. How many SNPs does a genome-wide haplotype map require? Pharmacogenomics 2002, 3, 379–391. [Google Scholar]
- Miller, M. S.; Cronin, M. T. Genetic Polymorphisms and Susceptibility to Disease; Taylor & Francis: London, UK, 2000. [Google Scholar]
- Olden, K. Genomics in environmental health research-opportunities and challenges. Toxicology 2004, 198, 19–24. [Google Scholar]
- Bruning, T. R.; Bruning, T.; Roohs, P. H.; Rihs, H. P.; Golka, K.; Ko, Y.; Bolt, H. M. Markers of genetic susceptibility in human environmental hygiene and toxicology: the role of selected CYP, NAT and GST genes. Int. J. Hyg. Environ. Health 2003, 206, 149171. [Google Scholar]
- Rabinowitz, P. M.; Poljak, A. Host-environment medicine: A primary care model for the age of genomics. J. Gen. Intern. Med 2003, 18, 222–227. [Google Scholar]
- Dean, M. Approaches to identify genes for complex human diseases: lessons from Mendelian disorders. Hum. Mutat 2003, 22, 261–274. [Google Scholar]
- Simmons, P. T.; Portier, C. J. Toxicogenomics: the new frontier in risk analysis. Carcinogenesis 2002, 23, 903–905. [Google Scholar]
- Anthony, J. C. The promise of psychiatric enviromics. Br J Psychiatry Suppl 2001, 40, s8. [Google Scholar]
- Khoury, M. J.; Burke, W.; Thompson, E. J. Genetics and public health: A framework for the integration of human genetics into public health practice. Khoury, M. J., Burke, W., Thompson, E. J., Eds.; In Genetics and Public Health in the 21st Century: Using Genetic Information to Improve Health and Prevent Disease; Oxford University Press: London, UK, 2000; pp. 3–23. [Google Scholar]
- Merikangas, K. R.; Risch, N. Genomic priorities and public health. Science 2003, 302, 599–601. [Google Scholar]
- Ishibe, N.; Kelsey, K. T. Genetic susceptibility to environmental and occupational cancers. Cancer Causes Control 1997, 8, 504–513. [Google Scholar]
- Fujii, T. Transgenerational effects of maternal exposure to chemicals on the functional development of the brain in the offspring. Cancer Causes Control 1997, 8, 524–528. [Google Scholar]
- Bennett, W. P.; Hussain, S. P. B.; Vahakangas, K. H.; Kahn, M. A.; Shields, P. C.; Harris, C. C. Molecular epidemiology of human cancer risk: gene-environment interactions and p53 mutation spectrum in human lung cancer. J. Pathol 1999, 187, 8–18. [Google Scholar]
- Wilson, T. M.; Moore, J. T. Genomics versus orphan nuclear receptors--a halftime report. Mol. Endocrinol 2002, 16, 1135–1144. [Google Scholar]
- Colborn, T. Neurodevelopment and endocrine disruption. Environ Health Perspect 2004, 112, 944–949. [Google Scholar]
- Fisher, J. S. Environmental anti-androgens and male reproductive health: Focus on phthalates and testicular dysgenesis syndrome. Reproduction 2004, 127, 305–315. [Google Scholar]
- Nebert, D. W.; Dalton, T. P.; Okey, A. B.; Gonzalez, F. J. Role of aryl hydrocarbon receptor-mediated induction of the CYP1 enzymes in environmental toxicity and cancer. J. Biol. Chem 2004, 279, 23847–23850. [Google Scholar]
- Egger, G.; Liang, G.; Aparicio, A.; Jones, P. A. Epigenetics in human disease and prospects for epigenetic therapy. Nature (London) 2004, 429, 457–463. [Google Scholar]
- Jablonka, E. Epigenetic Epidemiology. Int. J. Epidemiol 2004, 33, 929–935. [Google Scholar]
- National Research Council, N. Risk assessment in the Federal Government: Managing the Process; National Academy Press: Washington, DC, 1983.
- Olden, K.; Guthrie, J. Genomics: implications for toxicology. Mutat Res 2001, 475, 3. [Google Scholar]
- Lash, L. H.; Hines, R. N.; Gonzalez, F. J.; Zacharewski, T. R.; Rothstein, M. A. Genetics and susceptibility to toxic chemicals: do you (or should you) know your genetic profile? J. Pharmacol. Exp. Ther 2003, 305, 403–409. [Google Scholar]
- Pearl, J. Causality; Cambridge University Press: Cambridge, UK, 2000. [Google Scholar]
- Frazier, M. E.; Johnson, G. M.; Thomassen, D. G.; Oliver, C. E.; Patrinos, A. Realizing the potential of the genome revolution: The genomes to life program. Science 2003, 300, 290–293. [Google Scholar]
- Ideker, T.; Galitsky, T.; Hood, L. A New Approach to Decoding Life: Systems Biology. Annu. Rev. Genomics Hum. Gene 2001, 2, 343–372. [Google Scholar]
- Hanahan, D.; Weinberg, R. A. The hallmarks of cancer. Cell 2000, 700, 57–70. [Google Scholar]
- Toyoshiba, H.; Yamanaka, T.; Sone, H.; Parham, F. M.; Walker, N. J.; Martinez, J.; Portier, C. J. Gene interaction network suggests dioxin induces a significant linkage between aryl hydrocarbon receptor and retinoic acid receptor beta. Environ Health Perspect 2004, 112, 1217–1224. [Google Scholar]
- Sanchez, J. C.; Corthals, G. L.; Hochstrasser, D. F. (Eds.) Biomedical Applications of Proteomics; Wiley-VCH: NY, NY, 2004.
- Collins, F. S. The Case for a US Prospective Cohort Study of Genes and Environment. Nature (London) 2004, 429, 475–411. [Google Scholar]
- Brooks, J. The sad and tragic life of Typhoid Mary. Can. Med. J 1996, 154, 915–916. [Google Scholar]
- Gerard, S.; Hayes, M.; Rothstein, M. A. On the edge of tomorrow: fitting genomics into public health policy. J Law Med Ethics 2002, 30, 173–176. [Google Scholar]
- Levy, F.; Lawler, J. F. The potential impact of genetic sequencing on the American health insurance system. J. Health Care Law Policy 2003, 6, 203–221. [Google Scholar]
- Clayton, E. W. Ethical, legal, and social implications of genomic medicine. N. Engl. J. Med 2003, 349, 562–569. [Google Scholar]
- Kelada, S. M.; Eaton, D. L.; Wang, S. S.; Rothman, N. R.; Khoury, M. J. Applications of human genome epidemiology to environmental health. Khoury, M. J., Little, J., Burke, W., Eds.; In Human Genome Epidemiology; Oxford University Press: London, UK, 2004; pp. 145–167. [Google Scholar]
- Khoury, M. J.; Little, J.; Burke, W. Human genome epidemiology: Scope and strategies. Khoury, M. J., Little, J., Burke, W., Eds.; In Human Genome Epidemiology; Oxford University Press: London, UK, 2004; pp. 3–16. [Google Scholar]
© 2005 MDPI. All rights reserved.
Share and Cite
Toscano, W.A.; Oehlke, K.P. Systems Biology: New Approaches to Old Environmental Health Problems. Int. J. Environ. Res. Public Health 2005, 2, 4-9. https://doi.org/10.3390/ijerph2005010004
Toscano WA, Oehlke KP. Systems Biology: New Approaches to Old Environmental Health Problems. International Journal of Environmental Research and Public Health. 2005; 2(1):4-9. https://doi.org/10.3390/ijerph2005010004
Chicago/Turabian StyleToscano, William A., and Kristen P. Oehlke. 2005. "Systems Biology: New Approaches to Old Environmental Health Problems" International Journal of Environmental Research and Public Health 2, no. 1: 4-9. https://doi.org/10.3390/ijerph2005010004
APA StyleToscano, W. A., & Oehlke, K. P. (2005). Systems Biology: New Approaches to Old Environmental Health Problems. International Journal of Environmental Research and Public Health, 2(1), 4-9. https://doi.org/10.3390/ijerph2005010004



