Effects of Ascorbic Acid on Osteopontin Expression and Axonal Myelination in the Developing Cerebellum of Lead-Exposed Rat Pups
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Design and Animals
2.2. Chemical Treatment
2.3. Locomotor Coordination Assay (Bar Holding Test and Wire Mesh Ascending Test) in Offspring
2.4. Analysis of Pb Level in Blood Using Atomic Absorption Spectrometry and Measurement of Cerebellar Weight
2.5. Tissue Processing and Histological Analysis
2.6. Statistical Analysis
3. Results
3.1. Body Weight, Cerebellar Weight, and Blood Pb Levels
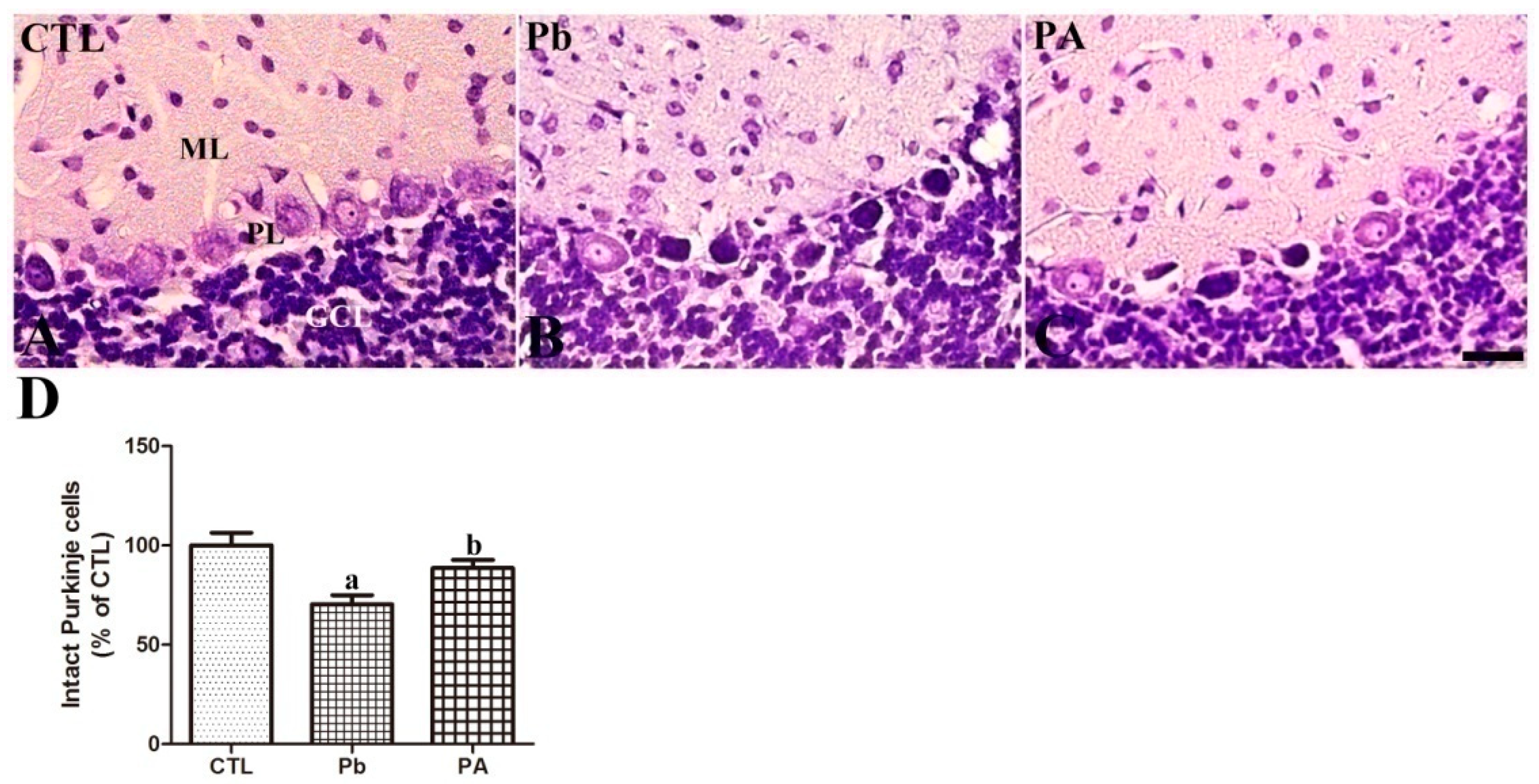
3.2. Effects of Pb Exposure and Ascorbic Acid Treatment on Cerebellar Development
3.3. Effects of Pb Exposure and Ascorbic Acid Treatment on OPN and Brain-Derived Neurotrophic Factor (BDNF)Expression in the Developing Cerebellum
3.4. Effects of Pb Exposure and Ascorbic Acid Treatment on Olig2 Expression in the Developing Cerebellum
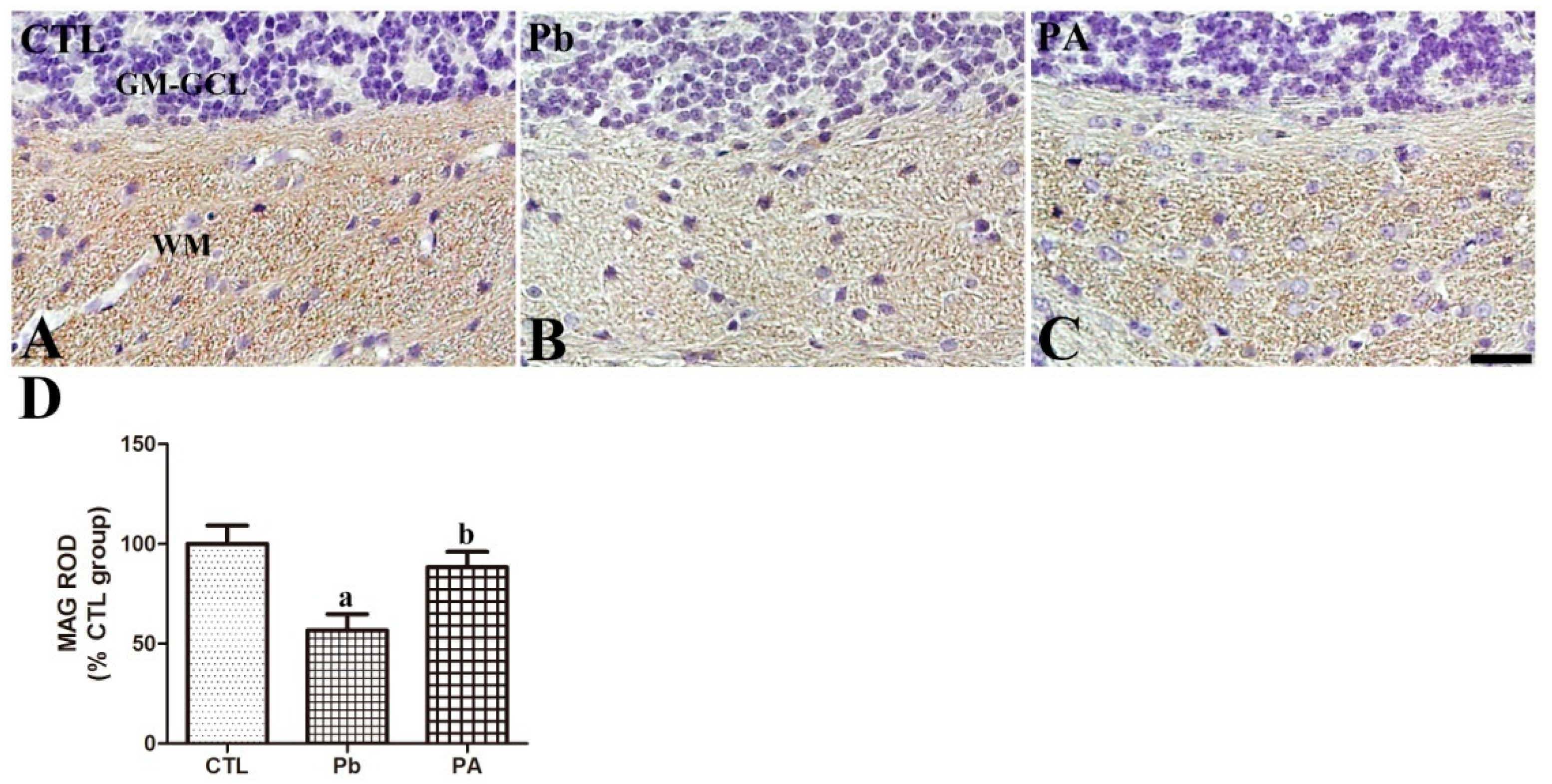
3.5. Effect of Pb Exposure and Ascorbic Acid Treatment on MAG Expression in the Developing Cerebellum
3.6. Effect of Pb Exposure and Ascorbic Acid Treatment on Locomotive Function (Bar Holding Test and Wire Mesh Ascending Test)
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Denhardt, D.T.; Guo, X. Osteopontin: A protein with diverse functions. FASEB J. 1993, 7, 1475–1482. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.X.; Denhardt, D.T. Osteopontin: Role in immune regulation and stress responses. Cytokine Growth Factor Rev. 2008, 19, 333–345. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Holm, E.; Gleberzon, J.S.; Liao, Y.; Sørensen, E.S.; Beier, F.; Hunter, G.K.; Goldberg, H.A. Osteopontin mediates mineralization and not osteogenic cell development in vitro. Biochem. J. 2014, 464, 355–364. [Google Scholar] [CrossRef] [PubMed]
- Chambers, A.F.; Behrend, E.I.; Wilson, S.M.; Denhardt, D.T. Induction of expression of osteopontin (OPN; secreted phosphoprotein) in metastatic, ras-transformed NIH3T3 cells. Anticancer Res. 1992, 12, 43–48. [Google Scholar] [PubMed]
- Murry, C.E.; Giachelli, C.M.; Schwartz, S.M.; Vracko, R. Macrophages express osteopontin during repair of myocardial necrosis. Am. J. Pathol. 1994, 145, 1450–1462. [Google Scholar] [PubMed]
- Goncalves, R.F.; Chapman, D.A.; Bertolla, R.P. Pretreatment of cattle semen or oocyte with purified milk osteopontin affects in vitro fertilization and embryo development. Anim. Reprod. Sci. 2008, 108, 375–383. [Google Scholar] [CrossRef]
- Liu, Y.; Xu, H.; Zhong, W.; Shen, Q.; Zhuang, T.; Huang, K. Organic selenium alleviated the formation of ethylene glycol-induced calcium oxalate renal calculi by improving osteopontin expression and antioxidant capability in dogs. Biol. Trace Elem. Res. 2015, 168, 392–400. [Google Scholar] [CrossRef]
- Moon, C.; Shin, T. Increased expression of osteopontin in the spinal cords of Lewis rats with experimental autoimmune neuritis. J. Vet. Sci. 2004, 5, 289–293. [Google Scholar] [CrossRef]
- Pio, G.M.; Xia, Y.; Piaseczny, M.M.; Chu, J.E.; Allan, A.L. Soluble bone-derived osteopontin promotes migration and stem-like behavior of breast cancer cells. PLoS ONE 2017, 12, e0177640. [Google Scholar] [CrossRef]
- Sodek, J.; Ganss, B.; McKee, M.D. Osteopontin. Crit. Rev. Oral. Biol. Med. 2000, 11, 279–303. [Google Scholar] [CrossRef] [Green Version]
- Kim, M.D.; Cho, H.J.; Shin, T. Expression of osteopontin and its ligand, CD44, in the spinal cords of Lewis rats with experimental autoimmune encephalomyelitis. J. Neuroimmunol. 2004, 151, 78–84. [Google Scholar] [CrossRef]
- Niino, M.; Kikuchi, S. Osteopontin and multiple sclerosis: An update. Clin. Exp. Neuroimmunol. 2011, 2, 33–40. [Google Scholar] [CrossRef] [Green Version]
- Comi, C.; Carecchio, M.; Chiocchetti, A.; Nicola, S.; Galimberti, D.; Fenoglio, C.; Cappellano, G.; Monaco, F.; Scarpini, E.; Dianzani, U. Osteopontin is increased in the cerebrospinal fluid of patients with Alzheimer’s disease and its levels correlate with cognitive decline. J. Alzheimers Dis. 2010, 19, 1143–1148. [Google Scholar] [CrossRef]
- Mattson, N.; Rüetschi, U.; Pijnenburg, Y.A.; Blankenstein, M.A.; Podust, V.N.; Li, S.; Fagerberg, I.; Rosengren, L.; Blennow, K.; Zetterberg, H. Novel cerebrospinal fluid biomarkers of axonal degeneration in frontotemporal dementia. Mol. Med. Rep. 2008, 1, 757–761. [Google Scholar] [CrossRef] [Green Version]
- Jiang, R.; Prell, C.; Lönnerdal, B. Milk osteopontin promotes brain development by up-regulating osteopontin in the brain in early life. FASEB J. 2018, 33, 1681–1694. [Google Scholar] [CrossRef]
- Chang, B.J.; Jang, B.J.; Son, T.G.; Cho, I.H.; Quan, F.S.; Choe, N.H.; Nahm, S.S.; Lee, J.H. Ascorbic acid ameliorates oxidative damage induced by maternal low-level lead exposure in the hippocampus of rat pups during gestation and lactation. Food Chem. Toxicol. 2012, 50, 104–108. [Google Scholar] [CrossRef]
- Nam, S.M.; Chang, B.J.; Kim, J.H.; Nahm, S.S.; Lee, J.H. Ascorbic acid ameliorates lead-induced apoptosis in the cerebellar cortex of developing rats. Brain Res. 2018, 1686, 10–18. [Google Scholar] [CrossRef]
- World Health Organization. Childhood Lead Poisoning. 2010. Available online: http://www.who.int/ceh/publications/childhoodpoisoning/en/ (accessed on 1 January 2019).
- Lee, M.Y.; Choi, J.S.; Lim, S.W.; Cha, J.H.; Chun, M.H.; Chung, J.W. Expression of osteopontin mRNA in developing rat brainstem and cerebellum. Cell Tissue Res. 2001, 306, 179–185. [Google Scholar] [CrossRef]
- Nam, S.M.; Cho, I.S.; Seo, J.S.; Go, T.H.; Kim, J.H.; Nahm, S.S.; Chang, B.J.; Lee, J.H. Ascorbic acid attenuates lead-induced alterations in the synapses in the developing rat cerebellum. Biol. Trace Elem. Res. 2019, 187, 142–150. [Google Scholar] [CrossRef]
- Lee, J.Y.; Chang, M.Y.; Park, C.H.; Kim, H.Y.; Kim, J.H.; Son, H.; Lee, Y.S.; Lee, S.H. Ascorbate-induced differentiation of embryonic cortical precursors into neurons and astrocytes. J. Neurosci. Res. 2003, 73, 156–165. [Google Scholar] [CrossRef]
- Nam, S.M.; Seo, J.S.; Go, T.H.; Nahm, S.S.; Chang, B.J. Ascorbic acid supplementation prevents the detrimental effects of prenatal and postnatal lead exposure on the Purkinje cell and related proteins in the cerebellum of developing rats. Biol. Trace Elem. Res. 2018. [Google Scholar] [CrossRef]
- Perez-Polo, J.; Rea, H.C.; Infante, S.K. Locomotor coordination assay in rats. Bio-protocol 2015, 5, e1655. [Google Scholar] [CrossRef]
- Nam, S.M.; Seo, M.; Seo, J.-S.; Rhim, H.; Nahm, S.-S.; Cho, I.-H.; Chang, B.-J.; Kim, H.-J.; Choi, S.-H.; Nah, S.-Y. Ascorbic acid mitigates D-galactose-induced brain aging by increasing hippocampal neurogenesis and improving memory function. Nutrients 2019, 11, 176. [Google Scholar] [CrossRef]
- Volk, B. Cerebellar histogenesis and synaptic maturation following pre-and postnatal alcohol administration. Acta Neuropathol. 1984, 63, 57–65. [Google Scholar] [CrossRef]
- Quarles, R.H. Myelin-associated glycoprotein (MAG): Past, present and beyond. J. Neurochem. 2007, 100, 1431–1448. [Google Scholar] [CrossRef]
- Lanphear, B.P. The impact of toxins on the developing brain. Annu. Rev. Public Health 2015, 36, 211–230. [Google Scholar] [CrossRef]
- Wang, Q.; Luo, W.; Zheng, W.; Liu, Y.; Xu, H.; Zheng, G.; Dai, Z.; Zhang, W.; Chen, Y.; Chen, J. Iron supplement prevents lead-induced disruption of the blood-brain barrier during rat development. Toxicol. Appl. Pharmacol. 2007, 219, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.L.; Chang, B.J.; Nam, S.M.; Nahm, S.S.; Lee, J.H. Increased osteopontin expression and mitochondrial swelling in 3-nitropropionic acid-injured rat brains. Rom. J. Morphol. Embryol. 2017, 58, 1249–1256. [Google Scholar]
- Maetzler, W.; Berg, D.; Schalamberidze, N.; Melms, A.; Schott, K.; Mueller, J.C.; Liaw, L.; Gasser, T.; Nitsch, C. Osteopontin is elevated in Parkinson’s disease and its absence leads to reduced neurodegeneration in the MPTP model. Neurobiol. Dis. 2007, 25, 473–482. [Google Scholar] [CrossRef]
- Scatena, M.; Liaw, L.; Giachelli, C.M. Osteopontin: A multifunctional molecule regulating chronic inflammation and vascular disease. Arterioscler. Thromb. Vasc. Biol. 2007, 27, 2302–2309. [Google Scholar] [CrossRef]
- Choi, J.S.; Park, H.J.; Cha, J.H.; Chung, J.W.; Chun, M.H.; Lee, M.Y. Induction and temporal changes of osteopontin mRNA and protein in the brain following systemic lipopolysaccharide injection. J. Neuroimmunol. 2003, 141, 65–73. [Google Scholar] [CrossRef]
- Choi, J.S.; Kim, H.Y.; Cha, J.H.; Choi, J.Y.; Lee, M.Y. Transient microglial and prolonged astroglial upregulation of osteopontin following transient forebrain ischemia in rats. Brain Res. 2007, 1151, 195–202. [Google Scholar] [CrossRef]
- Eltony, S.A.; Othman, M.A.; Mohamed, A.A. Histological study on the effect of low level perinatal lead exposure on the cerebellar cortex of adult male albino rat. Egypt J. Histol. 2010, 33, 781–797. [Google Scholar]
- Mustafa, H.N.; Hussein, A.M. Does allicin combined with vitamin B-complex have superior potentials than alpha-tocopherol alone in ameliorating lead acetate-induced Purkinje cell alterations in rats? An immunohistochemical and ultrastructural study. Folia Morphol. 2016, 75, 76–86. [Google Scholar] [CrossRef]
- Saleh, H.A.; Abdel El-Aziz, G.S.; Mustafa, H.N.; Saleh, A.H.A.; Mal, A.O.; Deifalla, A.H.S.; Abo Rass, M. Protective effect of garlic extract against maternal and fetal cerebellar damage induced by lead administration during pregnancy in rats. Folia Morphol. (Warsz) 2018, 77, 1–15. [Google Scholar] [CrossRef]
- El-Sokkary, G.H.; Awadalla, E.A. The protective role of vitamin C against cerebral and pulmonary damage induced by cadmium chloride in male adult albino rat. Open Neuroendocrinol. J. 2011, 4, 1–8. [Google Scholar] [CrossRef]
- Ibegbu, A.O.; Abdulrazaq, A.A.; Micheal, A.; Daniel, B.; Sadeeq, A.A.; Peter, A.; Hamman, W.O.; Umana, U.E.; Musa, S.A. Histomorphological effect of ascorbic acid on mercury chloride-induced changes on the cerebellum of adult wistar rats. J. Morphol. Sci. 2014, 31, 219–224. [Google Scholar] [Green Version]
- Rabenstein, M.; Hucklenbroich, J.; Willuweit, A.; Ladwig, A.; Fink, G.R.; Schroeter, M.; Langen, K.J.; Rueger, M.A. Osteopontin mediates survival, proliferation and migration of neural stem cells through the chemokine receptor CXCR4. Stem Cell Res. Ther. 2015, 6, 99. [Google Scholar] [CrossRef] [Green Version]
- Ruzafa, N.; Pereiro, X.; Aspichueta, P.; Araiz, J.; Vecino, E. The retina of osteopontin deficient mice in aging. Mol. Neurobiol. 2018, 55, 213–221. [Google Scholar] [CrossRef]
- Toews, A.D.; Krigman, M.R.; Thomas, D.J.; Morell, P. Effect of inorganic lead exposure on myelination in the rat. Neurochem. Res. 1980, 5, 605–616. [Google Scholar] [CrossRef]
- Selvaraju, R.; Bernasconi, L.; Losberger, C.; Graber, P.; Kadi, L.; Avellana-Adalid, V.; Picard-Riera, N.; Baron Van Evercooren, A.; Cirillo, R.; Kosco-Vilbois, M.; et al. Osteopontin is upregulated during in vivo demyelination and remyelination and enhances myelin formation in vitro. Mol. Cell. Neurosci. 2004, 25, 707–721. [Google Scholar] [CrossRef]
- Demmelmair, H.; Prell, C.; Timby, N.; Lönnerdal, B. Benefits of lactoferrin, osteopontin and milk fat globule membranes for infants. Nutrients 2017, 9, 817. [Google Scholar] [CrossRef]
- Christensen, B.; Sorensen, E.S. Structure, function and nutritional potential of milk osteopontin. Int. Dairy J. 2016, 57, 1. [Google Scholar] [CrossRef]
- Smith, K.J.; McDonald, W.I. The pathophysiology of multiple sclerosis: The mechanisms underlying the production of symptoms and the natural history of the disease. Philos. Trans. R. Soc. Lond. B. Biol. Sci. 1999, 354, 1649–1673. [Google Scholar] [CrossRef]
- Felsky, D.; Voineskos, A.N.; Lerch, J.P.; Nazeri, A.; Shaikh, S.A.; Rajji, T.K.; Mulsant, B.H.; Kennedy, J.L. Myelin-associated glycoprotein gene and brain morphometry in schizophrenia. Front. Psychiatry 2012, 3, 40. [Google Scholar] [CrossRef]
- Barron, T.; Saifetiarova, J.; Bhat, M.A.; Kim, J.H. Myelination of Purkinje axons is critical for resilient synaptic transmission in the deep cerebellar nucleus. Sci. Rep. 2018, 8, 1022. [Google Scholar] [CrossRef] [Green Version]
- Krigman, M.R.; Druse, M.J.; Traylor, T.D.; Wilson, M.H.; Newell, L.R.; Hogan, E.L. Lead encephalopathy in the developing rat: Effect upon myelination. J. Neuropathol. Exp. Neurol. 1974, 33, 58–73. [Google Scholar] [CrossRef]
- Eldridge, C.F.; Bunge, M.B.; Bunge, R.P.; Wood, P.M. Differentiation of axon-related Schwann cells in vitro. I. Ascorbic acid regulates basal lamina assembly and myelin formation. J. Cell Biol. 1987, 105, 1023–1034. [Google Scholar] [CrossRef] [Green Version]
- Kim, J.Y.; Choi, C.S.; Hong, S.K. Coculture of Schwann cells and neuronal cells for myelination in rat. Rapid Commun. Photosci. 2014, 3, 48–49. [Google Scholar] [CrossRef]
- Kelamangalath, L.; Smith, G.M. Neurotrophin treatment to promote regeneration after traumatic CNS injury. Front. Biol. 2013, 8, 486–495. [Google Scholar] [CrossRef] [Green Version]
- Fletcher, J.L.; Murray, S.S.; Xiao, J. Brain-derived neurotrophic factor in central nervous system myelination: A new mechanism to promote myelin plasticity and repair. Int. J. Mol. Sci. 2018, 19, 4131. [Google Scholar] [CrossRef]
- Zhao, H.; Chen, Q.; Alam, A.; Cui, J.; Suen, K.C.; Soo, A.P.; Eguchi, S.; Gu, J.; Ma, D. The role of osteopontin in the progression of solid organ tumour. Cell Death Dis. 2018, 9, 356. [Google Scholar] [CrossRef]
- Travica, N.; Ried, K.; Sali, A.; Scholey, A.; Hudson, I.; Pipingas, A. Vitamin C status and cognitive function: A systematic review. Nutrients 2017, 9, 960. [Google Scholar] [CrossRef]
- Ford, T.C.; Downey, L.A.; Simpson, T.; McPhee, G.; Oliver, C.; Stough, C. The effect of a high-dose vitamin B multivitamin supplement on the relationship between brain metabolism and blood biomarkers of oxidative stress: A randomized control trial. Nutrients 2018, 10, 1860. [Google Scholar] [CrossRef]
- Franklin, R.J.; Ffrench-Constant, C. Remyelination in the CNS: From biology to therapy. Nat. Rev. Neurosci. 2008, 9, 839–855. [Google Scholar] [CrossRef]




© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nam, S.M.; Seo, J.S.; Nahm, S.-S.; Chang, B.-J. Effects of Ascorbic Acid on Osteopontin Expression and Axonal Myelination in the Developing Cerebellum of Lead-Exposed Rat Pups. Int. J. Environ. Res. Public Health 2019, 16, 983. https://doi.org/10.3390/ijerph16060983
Nam SM, Seo JS, Nahm S-S, Chang B-J. Effects of Ascorbic Acid on Osteopontin Expression and Axonal Myelination in the Developing Cerebellum of Lead-Exposed Rat Pups. International Journal of Environmental Research and Public Health. 2019; 16(6):983. https://doi.org/10.3390/ijerph16060983
Chicago/Turabian StyleNam, Sung Min, Jin Seok Seo, Sang-Soep Nahm, and Byung-Joon Chang. 2019. "Effects of Ascorbic Acid on Osteopontin Expression and Axonal Myelination in the Developing Cerebellum of Lead-Exposed Rat Pups" International Journal of Environmental Research and Public Health 16, no. 6: 983. https://doi.org/10.3390/ijerph16060983



