Blood Pressure Control in Smokers with Arterial Hypertension Who Switched to Electronic Cigarettes
Abstract
:1. Introduction
2. Methods
2.1. Patient Population
2.2. Study Design
2.3. Data Management
2.4. Approach to Treatment
2.5. Study Outcomes
2.6. Analyses
3. Results
3.1. Patients’ Characteristics
3.2. Changes in Smoking Behaviour and Patterns of E-Cigarette Use
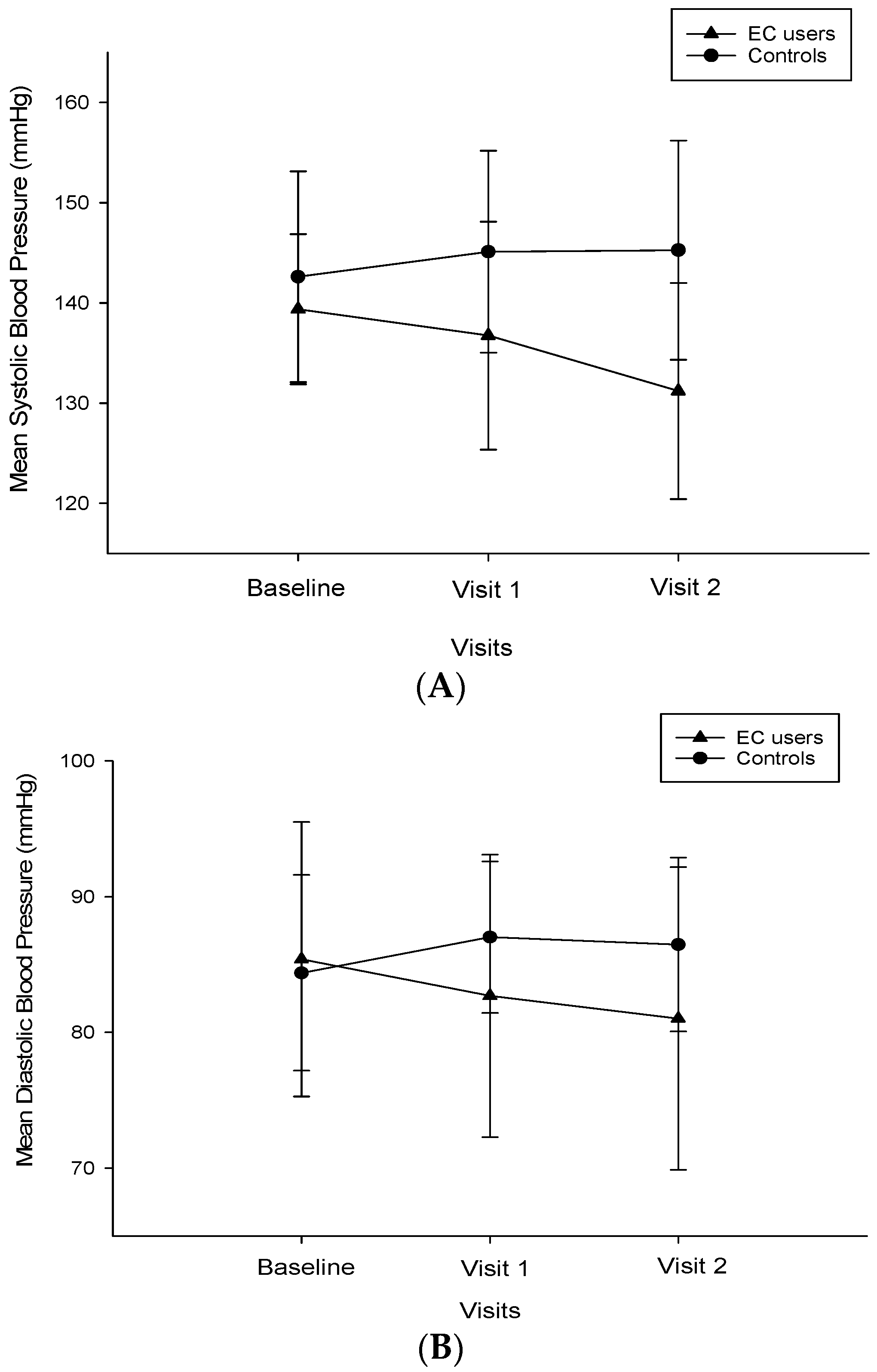
3.3. Changes in BP, HR, and Body Weight
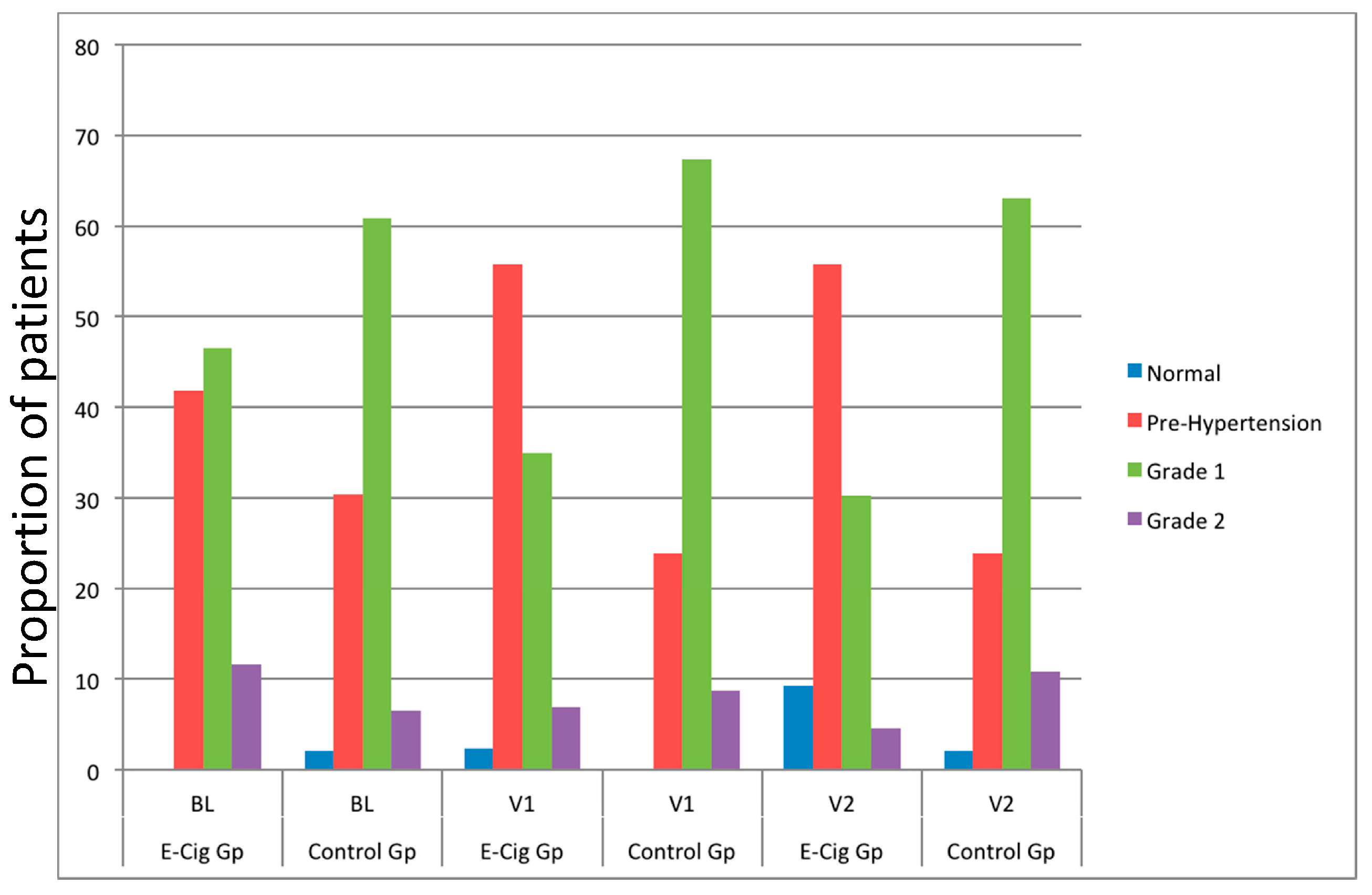
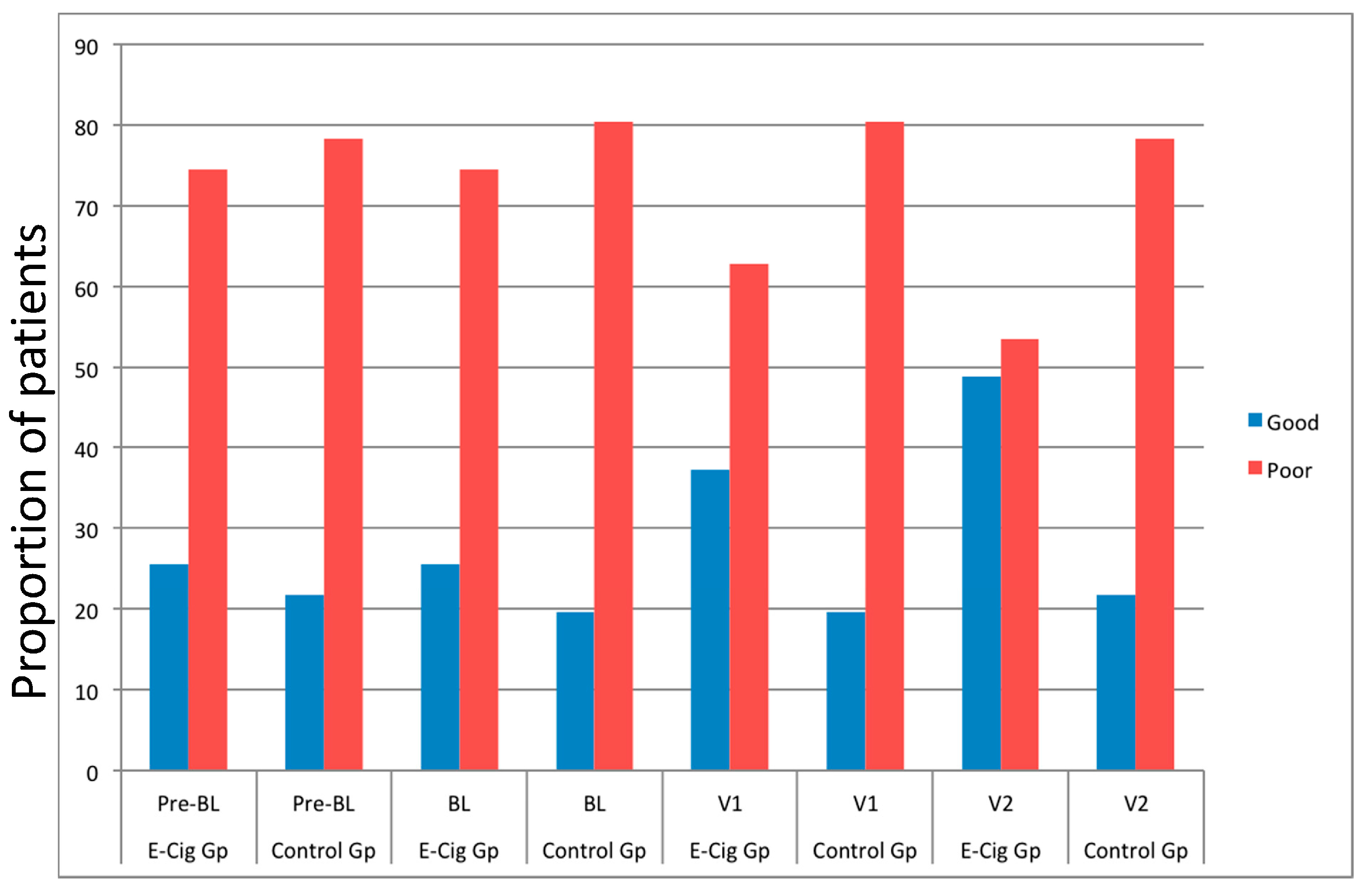
3.4. Changes in BP Control and Anti-Hypertensive Medications
3.5. Safety and Tolerability
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- US Department of Health and Human Services. The Health Consequences of Smoking: 50 Years of Progress: A Report of the Surgeon General; US Department of Health and Human Services, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health: Atlanta, GA, USA, 2014.
- Perk, J.; De Backer, G.; Gohlke, H.; Graham, I.; Reiner, Z.; Verschuren, M.; Albus, C.; Benlian, P.; Boysen, G.; Cifkova, R.; et al. European guidelines on cardiovascular disease prevention in clinical practice (version 2012). The fifth joint task force of the european society of cardiology and other societies on cardiovascular disease prevention in clinical practice (constituted by representatives of nine societies and by invited experts). Eur. Heart J. 2012, 33, 1635–1701. [Google Scholar] [PubMed]
- Prescott, E.; Hippe, M.; Schnohr, P.; Hein, H.O.; Vestbo, J. Smoking and risk of myocardial infarction in women and men: Longitudinal population study. BMJ 1998, 316, 1043–1047. [Google Scholar] [CrossRef] [PubMed]
- Prescott, E.; Scharling, H.; Osler, M.; Schnohr, P. Importance of light smoking and inhalation habits on risk of myocardial infarction and all cause mortality. A 22 year follow up of 12 149 men and women in the copenhagen city heart study. J. Epidemiol. Community Health 2002, 56, 702–706. [Google Scholar] [CrossRef] [PubMed]
- Teo, K.K.; Ounpuu, S.; Hawken, S.; Pandey, M.R.; Valentin, V.; Hunt, D.; Diaz, R.; Rashed, W.; Freeman, R.; Jiang, L.; et al. Tobacco use and risk of myocardial infarction in 52 countries in the interheart study: A case-control study. Lancet 2006, 368, 647–658. [Google Scholar] [CrossRef]
- Tonstad, S.; Cowan, J.L. C-reactive protein as a predictor of disease in smokers and former smokers: A review. Int. J. Clin. Pract. 2009, 63, 1634–1641. [Google Scholar] [CrossRef] [PubMed]
- Doonan, R.J.; Hausvater, A.; Scallan, C.; Mikhailidis, D.P.; Pilote, L.; Daskalopoulou, S.S. The effect of smoking on arterial stiffness. Hypertens. Res. 2010, 33, 398–410. [Google Scholar] [CrossRef] [PubMed]
- Niskanen, L.; Laaksonen, D.E.; Nyyssonen, K.; Punnonen, K.; Valkonen, V.P.; Fuentes, R.; Tuomainen, T.P.; Salonen, R.; Salonen, J.T. Inflammation, abdominal obesity, and smoking as predictors of hypertension. Hypertension 2004, 44, 859–865. [Google Scholar] [CrossRef] [PubMed]
- Fagard, R.H. Smoking amplifies cardiovascular risk in patients with hypertension and diabetes. Diabetes Care 2009, 32, S429–S431. [Google Scholar] [CrossRef] [PubMed]
- Zanchetti, A.; Hansson, L.; Dahlof, B.; Elmfeldt, D.; Kjeldsen, S.; Kolloch, R.; Larochelle, P.; McInnes, G.T.; Mallion, J.M.; Ruilope, L.; et al. Effects of individual risk factors on the incidence of cardiovascular events in the treated hypertensive patients of the hypertension optimal treatment study. Hot study group. J. Hypertens. 2001, 19, 1149–1159. [Google Scholar] [CrossRef] [PubMed]
- Primatesta, P.; Falaschetti, E.; Gupta, S.; Marmot, M.G.; Poulter, N.R. Association between smoking and blood pressure: Evidence from the health survey for england. Hypertension 2001, 37, 187–193. [Google Scholar] [CrossRef] [PubMed]
- Al-Safi, S.A. Does smoking affect blood pressure and heart rate? Eur. J. Cardiovasc. Nurs. 2005, 4, 286–289. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.H.; Ha, M.H.; Kim, J.R.; Jacobs, D.R., Jr. Effects of smoking cessation on changes in blood pressure and incidence of hypertension: A 4-year follow-up study. Hypertension 2001, 37, 194–198. [Google Scholar] [CrossRef] [PubMed]
- Caponnetto, P.; Russo, C.; Bruno, C.M.; Alamo, A.; Amaradio, M.D.; Polosa, R. Electronic cigarette: A possible substitute for cigarette dependence. Monaldi Arch. Chest Dis. 2013, 79, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Farsalinos, K.E.; Romagna, G.; Tsiapras, D.; Kyrzopoulos, S.; Voudris, V. Characteristics, perceived side effects and benefits of electronic cigarette use: A worldwide survey of more than 19,000 consumers. Int. J. Environ. Res. Public Health 2014, 11, 4356–4373. [Google Scholar] [CrossRef] [PubMed]
- Farsalinos, K.E.; Polosa, R. Safety evaluation and risk assessment of electronic cigarettes as tobacco cigarette substitutes: A systematic review. Ther. Adv. Drug Saf. 2014, 5, 67–86. [Google Scholar] [CrossRef] [PubMed]
- Polosa, R.; Rodu, B.; Caponnetto, P.; Maglia, M.; Raciti, C. A fresh look at tobacco harm reduction: The case for the electronic cigarette. Harm Reduct. J. 2013, 10, 19. [Google Scholar] [CrossRef] [PubMed]
- Caponnetto, P.; Campagna, D.; Cibella, F.; Morjaria, J.B.; Caruso, M.; Russo, C.; Polosa, R. Efficiency and safety of an electronic cigarette (eclat) as tobacco cigarettes substitute: A prospective 12-month randomized control design study. PLoS ONE 2013, 8, e66317. [Google Scholar] [CrossRef] [PubMed]
- Polosa, R.; Morjaria, J.B.; Caponnetto, P.; Campagna, D.; Russo, C.; Alamo, A.; Amaradio, M.; Fisichella, A. Effectiveness and tolerability of electronic cigarette in real-life: A 24-month prospective observational study. Intern. Emerg. Med. 2014, 9, 537–546. [Google Scholar] [CrossRef] [PubMed]
- Bullen, C.; Howe, C.; Laugesen, M.; McRobbie, H.; Parag, V.; Williman, J.; Walker, N. Electronic cigarettes for smoking cessation: A randomised controlled trial. Lancet 2013, 382, 1629–1637. [Google Scholar] [CrossRef]
- Caponnetto, P.; Auditore, R.; Russo, C.; Cappello, G.C.; Polosa, R. Impact of an electronic cigarette on smoking reduction and cessation in schizophrenic smokers: A prospective 12-month pilot study. Int. J. Environ. Res. Public Health 2013, 10, 446–461. [Google Scholar] [CrossRef] [PubMed]
- Polosa, R.; Morjaria, J.; Caponnetto, P.; Caruso, M.; Strano, S.; Battaglia, E.; Russo, C. Effect of smoking abstinence and reduction in asthmatic smokers switching to electronic cigarettes: Evidence for harm reversal. Int. J. Environ. Res. Public Health 2014, 11, 4965–4977. [Google Scholar] [CrossRef] [PubMed]
- Chobanian, A.V.; Bakris, G.L.; Black, H.R.; Cushman, W.C.; Green, L.A.; Izzo, J.L., Jr.; Jones, D.W.; Materson, B.J.; Oparil, S.; Wright, J.T., Jr.; et al. Seventh report of the joint national committee on prevention, detection, evaluation, and treatment of high blood pressure. Hypertension 2003, 42, 1206–1252. [Google Scholar] [CrossRef] [PubMed]
- Mancia, G.; Fagard, R.; Narkiewicz, K.; Redon, J.; Zanchetti, A.; Bohm, M.; Christiaens, T.; Cifkova, R.; De Backer, G.; Dominiczak, A.; et al. 2013 esh/esc guidelines for the management of arterial hypertension: The task force for the management of arterial hypertension of the european society of hypertension (esh) and of the european society of cardiology (esc). Eur. Heart J. 2013, 34, 2159–2219. [Google Scholar] [CrossRef] [PubMed]
- Yusuf, S.; Hawken, S.; Ounpuu, S.; Dans, T.; Avezum, A.; Lanas, F.; McQueen, M.; Budaj, A.; Pais, P.; Varigos, J.; et al. Effect of potentially modifiable risk factors associated with myocardial infarction in 52 countries (the interheart study): Case-control study. Lancet 2004, 364, 937–952. [Google Scholar] [CrossRef]
- Virdis, A.; Giannarelli, C.; Neves, M.F.; Taddei, S.; Ghiadoni, L. Cigarette smoking and hypertension. Curr. Pharm. Des. 2010, 16, 2518–2525. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. Quitting Smoking among Adults—United States, 2001–2010. MMWR Morb. Mortal. Wkly. Rep. 2011, 60, 1513–1519. [Google Scholar]
- Zhu, S.H.; Lee, M.; Zhuang, Y.L.; Gamst, A.; Wolfson, T. Interventions to increase smoking cessation at the population level: How much progress has been made in the last two decades? Tob. Control 2012, 21, 110–118. [Google Scholar] [CrossRef] [PubMed]
- Caponnetto, P.; Maglia, M.; Polosa, R. Commentary on dawkins et al. (2015): Electronic cigarettes—From smoking cessation to smoking sensation and back. Addiction 2015, 110, 678–679. [Google Scholar] [CrossRef] [PubMed]
- Farsalinos, K.; Cibella, F.; Caponnetto, P.; Campagna, D.; Morjaria, J.B.; Battaglia, E.; Caruso, M.; Russo, C.; Polosa, R. Effect of continuous smoking reduction and abstinence on blood pressure and heart rate in smokers switching to electronic cigarettes. Intern. Emerg. Med. 2016, 11, 85–94. [Google Scholar] [CrossRef] [PubMed]
- Cryer, P.E.; Haymond, M.W.; Santiago, J.V.; Shah, S.D. Norepinephrine and epinephrine release and adrenergic mediation of smoking-associated hemodynamic and metabolic events. N. Engl. J. Med. 1976, 295, 573–577. [Google Scholar] [CrossRef] [PubMed]
- Grassi, G.; Seravalle, G.; Calhoun, D.A.; Bolla, G.B.; Giannattasio, C.; Marabini, M.; Del Bo, A.; Mancia, G. Mechanisms responsible for sympathetic activation by cigarette smoking in humans. Circulation 1994, 90, 248–253. [Google Scholar] [CrossRef] [PubMed]
- Scallan, C.; Doonan, R.J.; Daskalopoulou, S.S. The combined effect of hypertension and smoking on arterial stiffness. Clin. Exp. Hypertens. 2010, 32, 319–328. [Google Scholar] [CrossRef] [PubMed]
- Halimi, J.M.; Giraudeau, B.; Vol, S.; Caces, E.; Nivet, H.; Tichet, J. The risk of hypertension in men: Direct and indirect effects of chronic smoking. J. Hypertens. 2002, 20, 187–193. [Google Scholar] [CrossRef] [PubMed]
- Perkins, K.A.; Epstein, L.H.; Marks, B.L.; Stiller, R.L.; Jacob, R.G. The effect of nicotine on energy expenditure during light physical activity. N. Engl. J. Med. 1989, 320, 898–903. [Google Scholar] [CrossRef] [PubMed]
- Poulter, N.R. Independent effects of smoking on risk of hypertension: Small, if present. J. Hypertens. 2002, 20, 171–172. [Google Scholar] [CrossRef] [PubMed]
- Russo, C.; Cibella, F.; Caponnetto, P.; Campagna, D.; Maglia, M.; Frazzetto, E.; Mondati, E.; Caruso, M.; Polosa, R. Evaluation of post cessation weight gain in a 1-year randomized smoking cessation trial of electronic cigarettes. Sci. Rep. 2016. [Google Scholar] [CrossRef] [PubMed]
- Hatsukami, D.K.; Kotlyar, M.; Allen, S.; Jensen, J.; Li, S.; Le, C.; Murphy, S. Effects of cigarette reduction on cardiovascular risk factors and subjective measures. Chest 2005, 128, 2528–2537. [Google Scholar] [CrossRef] [PubMed]
- Bolliger, C.T.; Zellweger, J.P.; Danielsson, T.; van Biljon, X.; Robidou, A.; Westin, A.; Perruchoud, A.P.; Sawe, U. Influence of long-term smoking reduction on health risk markers and quality of life. Nicotine Tob. Res. 2002, 4, 433–439. [Google Scholar] [CrossRef] [PubMed]
- Wong, S.L.; Shields, M.; Leatherdale, S.; Malaison, E.; Hammond, D. Assessment of validity of self-reported smoking status. Health Rep. 2012, 23, 47–53. [Google Scholar] [PubMed]
- Polosa, R.; Caponnetto, P. E-cigarettes and smoking cessation: A critique of a New England Journal Medicine-commissioned case study. Intern. Emerg. Med. 2016. [Google Scholar] [CrossRef] [PubMed]
| Parameter | Pre-Baseline | Baseline | p Value between Pre-Baseline and Baseline | p Value between Groups at Baseline |
|---|---|---|---|---|
| E-Cigarette Group (n = 43) | ||||
| Gender | 26 M, 17 F | - | - | - |
| Age (years) § | 53.5 (±6.3) | - | - | 0.623 |
| Smoking pack years § | 37.4 (±14.0) | - | - | 0.718 |
| FTND § | 5.6 (±2.2) | - | - | 0.49 |
| Weight (Kg) § | 73.0 (±12.2) | 73.4 (±12.2) | 0.053 | 0.942 |
| Conventional Cigarettes/day § | 21.5 (±6.9) | 20.2 (±5) | 0.143 | 0.274 |
| SBP (mmHg) | 142 (134, 145.5) | 140 (134.5, 144) | 0.754 | 0.1 |
| DBP (mmHg) | 86 (79.5, 91.5) | 86 (78, 90) | 0.338 | 0.563 |
| Hypertension stage # | ||||
| Normal | 0 | 0 | - | - |
| Pre-hypertension | 16 | 18 | - | - |
| Stage I | 21 | 20 | - | - |
| Stage II | 6 | 5 | - | - |
| HR (bpm) | 80 (67.5, 88) | 77 (69.5, 84.5) | 0.588 | 0.911 |
| Control Group (n = 46) | ||||
| Gender | 24 M, 22 F | - | - | |
| Age (years) § | 54.2 (±7.5) | - | - | |
| Smoking pack years § | 38.4 (±11.6) | - | - | |
| FTND § | 5.9 (±2.0) | - | - | |
| Weight (Kg) § | 72.9 (±12.4) | 73.2 (±12.4) | 0.217 | |
| Conventional Cigarettes/day § | 21.5 (±5.8) | 20.5 (±5.2) | 0.096 | |
| SBP (mmHg) | 143 (135.8, 149.3) | 143 (135, 151.8) | 0.553 | |
| DBP (mmHg) | 85.5 (80, 90) | 85 (80, 90) | 0.239 | |
| Hypertension stage # | ||||
| Normal | 0 | 1 | ||
| Pre-hypertension | 15 | 14 | ||
| Stage I | 28 | 28 | ||
| Stage II | 3 | 3 | ||
| HR (bpm) | 80 (69, 84.8) | 77.5 (72, 85) | 0.709 | |
| Parameter | Baseline (BL) | 6 Months | 12 Months | p Value between Groups Baseline to 12 Months ψ | ||
|---|---|---|---|---|---|---|
| E-Cigarette Group (n = 43; 26 M, 17 F) | p Value ¥ | p Value § | ||||
| Weight (Kg) ¶ | 73.4 (±12.2) | 75.1 (±13.7) | 0.012 | 74.6 (±13.5) | 0.046 | 0.455 |
| Cigarettes/day ¶ | 20.2 (±5.0) | 2.6 (±2.9) | <0.001 | 1.8 (±2.0) | <0.001 | <0.001 |
| SBP (mmHg) | 140 (134.5, 144) | 137 (130, 142.5) | 0.141 | 130 (123.5, 138.5) | <0.001 | <0.001 |
| DBP (mmHg) | 86 (78, 90) | 81 (75, 89) | 0.053 | 80 (74.5, 90) | 0.006 | <0.001 |
| HR (bpm) | 77 (69.5, 84.5) | 78 (71, 86) | 0.414 | 78 (72.5, 89.5) | 0.745 | 0.705 |
| Control Group (n = 46; 24 M, 22 F) | p Value ¥ | p Value § | ||||
| Weight (Kg) ¶ | 73.2 (±12.4) | 73.8 (±12.4) | 0.074 | 73.9 (±12.6) | 0.043 | |
| Cigarettes/day ¶ | 20.5 (±5.2) | 20.8 (±5.5) | 0.532 | 21.4 (±5.6) | 0.223 | |
| SBP (mmHg) | 143 (135, 151.8) | 145 (137.3, 151.8) | 0.105 | 145 (136.3, 150) | 0.095 | |
| DBP (mmHg) | 85 (80, 90) | 87 (85, 90) | 0.007 | 85 (85, 90) | 0.042 | |
| HR (bpm) | 77.5 (72, 85) | 78 (72, 83.5) | 0.791 | 78.5 (72, 83.8) | 0.857 | |
| Parameter | Baseline | 1st Follow-up Visit (6 Months ± 1) | 2nd Follow-up Visit (12 Months ± 2) | ||
|---|---|---|---|---|---|
| All Patients (n = 43; 26 M, 17 F) | |||||
| Weight (Kg) ¶ | 73.4 (±12.2) | 75.1 (±13.7) | 74.6 (±13.5) | ||
| Conventional Cigarettes/day ¶ | 20.2 (±6.9) | 2.6 (±2.9) | 1.8 (±2.0) | ||
| SBP (mmHg) | 140 (134.5, 144) | 137 (130, 142.5) | 130 (123.5, 138.5) | ||
| DBP (mmHg) | 86 (78, 90) | 81 (75, 89) | 80 (74.5, 90) | ||
| HR (bpm) | 77 (69.5, 84.5) | 78 (71, 86) | 78 (72.5, 89.5) | ||
| Single Users | |||||
| n = 20 (11 M; 9 F) | n = 21 (11 M; 10 F) | ||||
| p Value § | p Value Ω | ||||
| Weight (Kg) ¶ | 71.0 (±11.8) | 75.0 (±14.6) | <0.001 | 74.2 (±14.8) | 0.003 |
| Conventional Cigarettes/day ¶ | 20.4 (±4.5) | - | - | - | |
| SBP (mmHg) | 140 (135, 146.3) | 134 (130, 142.3) | 0.010 | 130 (123, 138) | <0.001 |
| DBP (mmHg) | 85.6 (75.8, 92) | 80.5 (73.8, 84.8) | 0.019 | 80 (75, 87) | 0.030 |
| HR (bpm) | 76 (71.5, 81.3) | 79.5 (75, 86) | 0.523 | 80 (76, 90) | 0.151 |
| Dual Users | |||||
| n = 23 (15 M; 8 F) | n = 22 (15 M; 7 F) | ||||
| p Value § | p Value Ω | ||||
| Weight (Kg) ¶ | 75.6 (±12.4) | 75.2 (±13.1) | 0.544 | 76.7 (±11.3) | 0.382 |
| Conventional Cigarettes/day ¶ | 20.1 (±5.4) | 4.8 (±2.3) | <0.001 | 3.7 (±1.1) | <0.001 |
| SBP (mmHg) | 138 (133.5, 140.5) | 137 (132, 143.5) | 0.750 | 130 (120.8, 139.8) | 0.011 |
| DBP (mmHg) | 87 (78.5, 90) | 83 (80, 91.5) | 0.691 | 80 (70.8, 90) | 0.109 |
| HR (bpm) | 83 (68.5, 86) | 77 (70, 83) | 0.095 | 76 (70.5, 92.3) | 0.874 |
| Visits | Pre-Baseline | Baseline | Visit 1 | Visit 2 | ||||
|---|---|---|---|---|---|---|---|---|
| Hypertension Control | E-Cig. Group (%) | Control Group (%) | E-Cig. Group (%) | Control Group (%) | E-Cig. Group (%) | Control Group (%) | E-Cig. Group (%) | Control Group (%) |
| Patients with BP Good Control | 11 | 10 | 11 | 9 | 16 | 9 | 21 | 10 |
| (<140/90 mmHg in subjects with no co-morbidities; <130/80 mmHg in subjects with co-morbidities) | (25.6) | (21.7) | (25.6) | (19.6) | (37.2) | (19.6) | (48.8) | (21.7) |
| Patients with BP Poor Control | 32 | 36 | 32 | 37 | 27 | 37 | 22 | 36 |
| (≥140/90 mmHg in subjects with no co-morbidities; ≥130/80 mmHg in subjects with co-morbidities) | (74.4) | (78.3) | (74.4) | (80.4) | (62.8) | (80.4) | (51.2) | (78.3) |
| Parameter | Pre-Baseline | Baseline | 6 Months | 12 Months |
|---|---|---|---|---|
| E-Cigarette Group (n = 43; 26 M, 17 F) | ||||
| Number of drugs ¶ | 3.7 (±1.3) | 3.6 (±1.1) | 3.5 (±1.3) | 3.3 (±1.5) |
| Drugs’ class (%) | ||||
| - Diuretics | 75% | 78% | 75% | 72% |
| - Calcium channel blockers | 43% | 41% | 33% | 30% |
| - Beta-blockers | 31% | 28% | 25% | 23% |
| - ACE inhibitors | 90% | 92% | 90% | 90% |
| - Angiotensin receptor blockers | 75% | 75% | 73% | 73% |
| Change of drugs’ class from baseline (%) | - | - | 5 (11.6%) | 5 (11.6%) |
| Same dose as from baseline (%) | - | - | 35 (81.4%) | 33 (76.7%) |
| Reduction in dose from baseline (%) | - | - | 5 (11.6%) | 7 (16.3%) |
| Increase in dose from baseline (%) | - | - | 3 (7.0%) | 3 (7.0%) |
| Control Group (n = 46; 24 M, 22 F) | ||||
| Number of drugs ¶ | 3.5 (±1.3) | 3.7 (±1.4) | 3.5 (±1.4) | 3.6 (±1.3) |
| Drugs’ class (%) | ||||
| - Diuretics | 74% | 72% | 70% | 74% |
| - Calcium channel blockers | 40% | 37% | 35% | 31% |
| - Beta-blockers | 27% | 25% | 25% | 26% |
| - ACE inhibitors | 88% | 90% | 92% | 93% |
| - Angiotensin receptor blockers | 80% | 82% | 83% | 86% |
| Change of drugs’ class from baseline (%) | - | - | 8 (17.4%) | 9 (19.6%) |
| Same dose as from baseline (%) | - | - | 37 (80.4%) | 35 (76.1%) |
| Reduction in dose from baseline (%) | - | - | 4 (8.7%) | 5 (10.9%) |
| Increase in dose from baseline (%) | - | - | 5 (10.9%) | 6 (13.0%) |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Polosa, R.; Morjaria, J.B.; Caponnetto, P.; Battaglia, E.; Russo, C.; Ciampi, C.; Adams, G.; Bruno, C.M. Blood Pressure Control in Smokers with Arterial Hypertension Who Switched to Electronic Cigarettes. Int. J. Environ. Res. Public Health 2016, 13, 1123. https://doi.org/10.3390/ijerph13111123
Polosa R, Morjaria JB, Caponnetto P, Battaglia E, Russo C, Ciampi C, Adams G, Bruno CM. Blood Pressure Control in Smokers with Arterial Hypertension Who Switched to Electronic Cigarettes. International Journal of Environmental Research and Public Health. 2016; 13(11):1123. https://doi.org/10.3390/ijerph13111123
Chicago/Turabian StylePolosa, Riccardo, Jaymin B. Morjaria, Pasquale Caponnetto, Eliana Battaglia, Cristina Russo, Claudio Ciampi, George Adams, and Cosimo M. Bruno. 2016. "Blood Pressure Control in Smokers with Arterial Hypertension Who Switched to Electronic Cigarettes" International Journal of Environmental Research and Public Health 13, no. 11: 1123. https://doi.org/10.3390/ijerph13111123
APA StylePolosa, R., Morjaria, J. B., Caponnetto, P., Battaglia, E., Russo, C., Ciampi, C., Adams, G., & Bruno, C. M. (2016). Blood Pressure Control in Smokers with Arterial Hypertension Who Switched to Electronic Cigarettes. International Journal of Environmental Research and Public Health, 13(11), 1123. https://doi.org/10.3390/ijerph13111123








