Drug-Induced Nephrotoxicity and Dose Adjustment Recommendations: Agreement Among Four Drug Information Sources
Abstract
:1. Introduction
2. Materials and Methods
2.1. Design
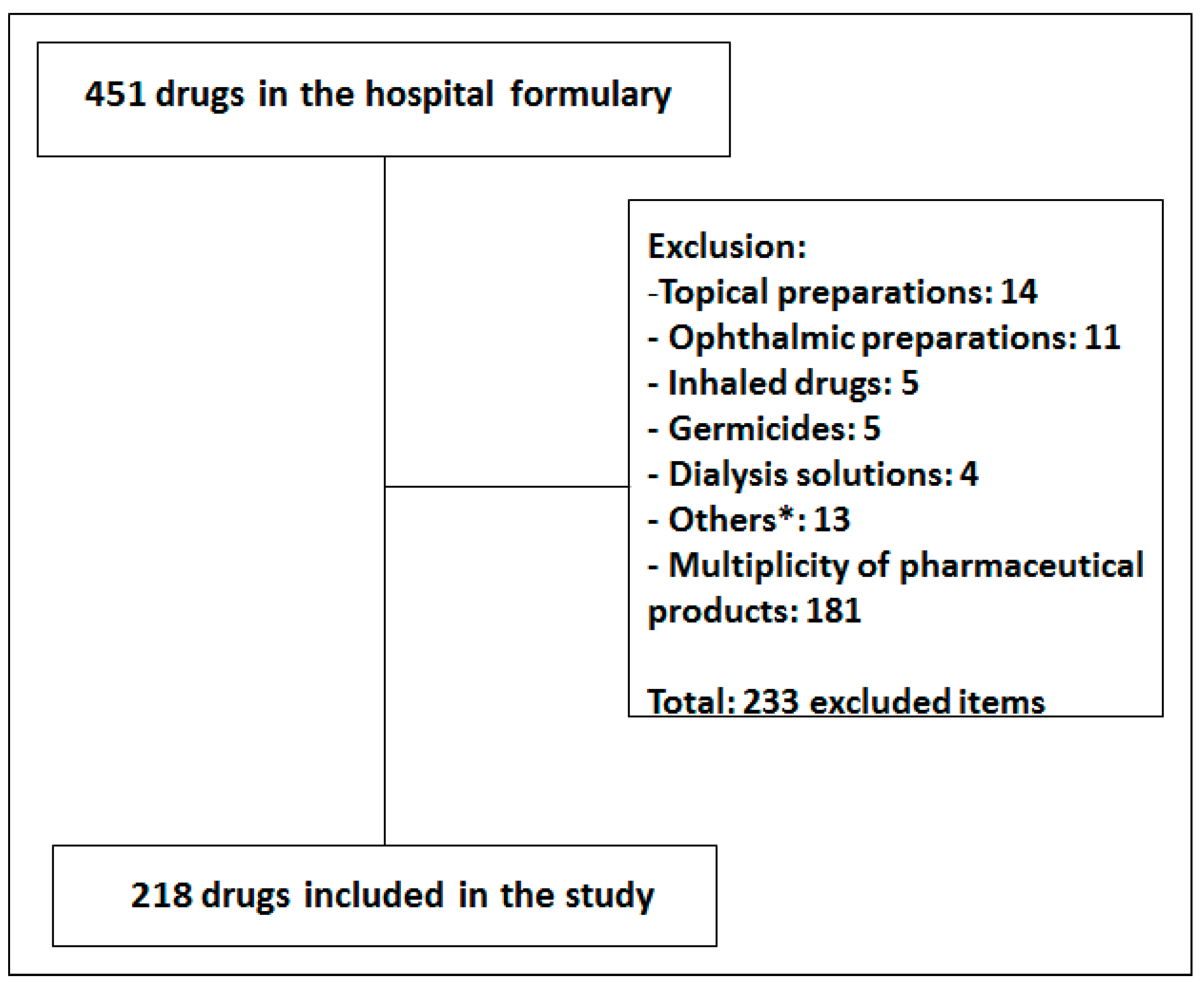
2.2. List of Investigated Drugs
2.3. Procedures for Searching on Drug Information Sources
- (1)
- Contraindicated (CI): This category included drugs that were recommended to be avoided in the presence of renal impairment of any severity.
- (2)
- Numerical recommendations (N): Dose modifications were recommended based on the creatinine clearance (CrCl) or eGFR/serum creatinine (SCr) value. Dose modifications based on CrCl/eGFR/SCr were not mentioned, but there was a clear recommendation to avoid the drug below a certain range of CrCl/eGFR/SCr values.
- (3)
- Non-numerical recommendations (NN): Recommendations that were ambiguous when describing the required dose for a particular stage of renal impairment, including severe renal impairment, or that were described without an eGFR/CrCl value/severity of renal impairment for which the drug should be avoided or reduced. This category could also include recommendations to use the medication with caution that were given without specific requirements for dose adjustments based on the CrCl/eGFR/SCr value.
- (4)
- Dosage adjustments not required (NR): The information source advised to give the usual drug dose in the presence of renal impairment.
- (5)
- No advice mentioned (NA): The drug monograph was presented in the information source, but there was no information regarding its use in patients with renal impairment.
- (6)
- Missing (M): This category included drugs that were not found in the information source.
2.4. Analysis of Data
3. Results
| DRUGDEX® | UpToDate® | Medscape® |
|---|---|---|
| albumin | domperidone | alfentanil |
| biperiden | doxycycline | chlordiazepoxide |
| calcium carbonate | haloperidol | |
| calcium gluconate | heparin | |
| diltiazem | hydroxyethylstarch | |
| dobutamine | ivermectine | |
| epinephrine | mebendazole | |
| ephedrine | octreotide | |
| ketamin | pralidoxime | |
| magnesium sulfate | promethazine | |
| metaraminol | propranolol | |
| pyridoxine | ||
| rifampicin and isoniazid | ||
| sodium chloride | ||
| sorbitolstreptokinase | ||
| teicoplanin | ||
| tenoxicam | ||
| thiopental | ||
| tiabendazole | ||
| zidovudine and lamivudine |
| Source Compared to DRUGDEX® | Nephrotoxicity | Dose Adjustment Recommendations |
|---|---|---|
| UpToDate® | 0.558 | 0.476 |
| Medscape® | 0.315 | 0.449 |
| National Therapeutic Formulary | 0.135 | 0.382 |
| Category | Sources | |||
|---|---|---|---|---|
| DRUGDEX® | UpToDate® | Medscape® | NTF * | |
| Contraindicated (CI) | 2 | 3 | - | 4 |
| Numerical (N) | 64 | 61 | 50 | 42 |
| Non-numerical (NN) | 15 | 44 | 13 | 34 |
| Not-required (NR) | 38 | 25 | 8 | - |
| No advice (NA) | 96 | 78 | 127 | 4 |
| Missing | 3 | 7 | 20 | 134 |
| Total drugs | 218 | 218 | 218 | 218 |
4. Discussion
| Therapeutical Class | Drug | Sources | |||
|---|---|---|---|---|---|
| DRUGDE® | UpToDate® | Medscape® | NTF * | ||
| Anti-infectives | amikacin | N | N | N | N |
| amoxicillin | N | N | N | N | |
| amphotericin B | N | N | N | N | |
| benzylpenicillin | N | N | N | N | |
| cefotaxime | N | N | NA | N | |
| ceftazidime | N | N | N | N | |
| ceftriaxone | NR | NR | NA | NN | |
| ciprofloxacin | N | N | N | N | |
| clarithromycin | N | N | N | NN | |
| erythromycin | N | NA | NR | NN | |
| gentamicin | N | N | N | N | |
| metronidazole | N | N | NA | N | |
| nitrofurantoin | N | NN | NN | N | |
| oxacillin | NR | NA | NA | NN | |
| pyrazinamide | N | N | NA | N | |
| sulfadiazine | NN | NA | NA | NN | |
| vancomycin | N | N | N | NN | |
| Anti-hypertensives | acetazolamide | N | N | N | N |
| captopril | NN | N | NA | N | |
| furosemide | N | N | N | NN | |
| hydralazine | N | N | NA | N | |
| hydrochlorothiazide | N | NN | N | CI | |
| losartan | NR | NR | NA | N | |
| mannitol | CI | NN | NA | CI | |
| Analgesics and antipyretics | paracetamol | N | N | N | N |
| Anticoagulants | warfarin | NR | NR | NA | NN |
| Antipsychotics | risperidone | N | N | N | NN |
| Hypoglycemic | glibenclamide | NR | NA | N | NN |
| Drugs | DRUGDEX® | UpToDate® | Medscape® | NTF |
|---|---|---|---|---|
| amphotericin B | ClCr > 10 mL/min: no adjustment required. ClCr < 10 mL/min: 20–50 mg every 24–36 hours. | If renal dysfunction is drug-induced, the total daily dose can be reduced in 50% or given in alternate days. | ClCr < 10 mL/min: 0.5–0.7 mg/kg IV every 24–48 hours. Consider other antifungal agents with less nephrotoxicity. | ClCr 10–50 mL/min: doses every 24 hours. ClCr < 10 mL/min: doses every 24 or 36 hours. |
| bumetanide | No adjustment required. | Use is contraindicated in anuria. Use with caution in renal impairment. | No advice. | Missing. |
| carbamazepine | No adjustment required. | ClCr < 10 mL/min: administrate 75% of dose. | ClCr < 10 mL/min: administrate 75% of dose and monitor. | Missing. |
| fluoxetine | No adjustment required. | Use with caution. Drug accumulation may occur. | Use with caution. Drug accumulation may occur. | Missing. |
| heparin | No adjustment required. | No dose adjustment provided in the label of the manufacturer. | No advice. | Severe renal impairment: contraindicated. Renal impairment: reduce dose due to increased risk of bleeding. |
| losartan | No adjustment required. | No adjustment required. | No advice. | ClCr < 20 mL/min: reduce dose. Recommended: 25mg. |
| methylene blue | Dose adjustment should be considered (specific recommendations unavailable) | Be cautious in case of severe renal impairment. | No adjustment required. Be cautious in case of severe renal impairment. | Missing. |
| neostigmine | No adjustment required. | ClCr > 50 ml/min: no adjustment required. ClCr 10–50 mL/min: administrate 50% of normal dose. ClCr < 10 mL/min: administrate 25% of normal dose. | No advice. | Adjust dose in renal impairment. |
| rifampicin | ClCr ≤ 50 mL/min: 50%–100% of total dose. | No adjustment required. | No advice. | ClCr ≥ 50 mL/min: 100% of dose. ClCr < 49 mL/min: 50% of recommended dose. No need to adjust the dose if it does not exceed 600 mg/day and liver function is normal. |
| verapamil | No adjustment required. | Use with caution. | Use with caution. Manufacturer recommends 100 mg at bedtime. ClCr < 10 mL/min: reduce the dose in 25%–50%. | Missing. |
5. Conclusions
Acknowledgements
Author Contributions
Conflicts of Interest
References
- Nasri, H. World kidney day. Chronic kidney disease and aging: A global health alert. Iran. J. Public Health 2014, 43, 126–127. [Google Scholar] [PubMed]
- Schieppati, A.; Remuzzi, G. Chronic renal diseases as a public health problem: Epidemiology, social, and economic implications. Kidney Int. Suppl. 2005. [Google Scholar] [CrossRef] [PubMed]
- Vlagopoulos, P.T.; Sarnak, M.J. Traditional and nontraditional cardiovascular risk factors in chronic kidney disease. Med. Clin. N. Am. 2005, 89, 587–611. [Google Scholar] [CrossRef] [PubMed]
- Marcen, R. Cardiovascular risk factors in renal transplantation—Current controversies. Nephrol. Dial. Transplant. 2006, 21, 3–8. [Google Scholar] [CrossRef] [PubMed]
- McLaughlin, K.; Manns, B.; Culleton, B.; Donaldson, C.; Taub, K. An economic evaluation of early versus late referral of patients with progressive renal insufficiency. Am. J. Kidney Dis. 2001, 38, 1122–1128. [Google Scholar] [CrossRef] [PubMed]
- National Kidney Foundation (NKF). Acute Kidney Injury (AKI). 2015. Available online: https://www.kidney.org/atoz/content/AcuteKidneyInjury (accessed on 24 June 2015).
- Finlay, S.; Bray, B.; Lewington, A.J.; Hunter-Rowe, C.T.; Banerjee, A.; Atkinson, J.M.; Jones, M.C. Identification of risk factors associated with acute kidney injury in patients admitted to acute medical units. Clin. Med. 2013, 13, 233–238. [Google Scholar] [CrossRef] [PubMed]
- Taber, S.S.; Mueller, B.A. Drug-associated renal dysfunction. Crit. Care Clin. 2006, 22, 357–374. [Google Scholar] [CrossRef] [PubMed]
- Mehta, R.L.; Pascual, M.T.; Soroko, S.; Savage, B.R.; Himmelfarb, J.; Ikizler, T.A.; Paganini, E.P.; Chertow, G.M. Spectrum of acute renal failure in the intensive care unit: The PICARD experience. Kidney Int. 2004, 66, 1613–1621. [Google Scholar] [CrossRef] [PubMed]
- Uchino, S.; Kellum, J.A.; Bellomo, R.; Doig, G.S.; Morimatsu, H.; Morgera, S.; Schetz, M.; Tan, I.; Bouman, C.; Macedo, E.; et al. Acute renal failure in critically ill patients: A multinational, multicenter study. JAMA 2005, 294, 813–818. [Google Scholar] [CrossRef] [PubMed]
- Naughton, C.A. Drug-induced nephrotoxicity. Am. Fam. Phys. 2008, 78, 743–750. [Google Scholar]
- Singh, N.P.; Ganguli, A.; Prakash, A. Drug-induced kidney diseases. J. Assoc. Phys. India 2003, 51, 970–979. [Google Scholar]
- Nolin, T.D.; Himmelfarb, J. Mechanisms of drug-induced nephrotoxicity. Handb. Exp. Pharmacol. 2010. [Google Scholar] [CrossRef]
- Guo, X.; Nzerue, C. How to prevent, recognize, and treat drug-induced nephrotoxicity. Clevel. Clin. J. Med. 2002, 69, 289–296. [Google Scholar] [CrossRef]
- Khanal, A.; Castelino, R.L.; Peterson, G.M.; Jose, M.D. Dose adjustment guidelines for medications in patients with renal impairment: How consistent are drug information sources? Intern. Med. J. 2014, 44, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Vidal, L.; Shavit, M.; Fraser, A.; Paul, M.; Leibovici, L. Systematic comparison of four sources of drug information regarding adjustment of dose for renal function. BMJ 2005. [Google Scholar] [CrossRef] [PubMed]
- Abarca, J.; Malone, D.C.; Armstrong, E.P.; Grizzle, A.J.; Hansten, P.D.; van Bergen, R.C.; Lipton, R.B. Concordance of severity ratings provided in four drug interaction compendia. J. Am. Pharm. Assoc. 2004, 44, 136–141. [Google Scholar]
- Martins, M.A.P.; Carlos, P.P.S.; Ribeiro, D.D.; Nobre, V.A.; Cesar, C.C.; Rocha, M.O.C.; Ribeiro, A.L.P. Warfarin drug interactions: A comparative evaluation of the lists provided by five information sources. Eur. J. Clin. Pharmacol. 2011, 67, 1301–1308. [Google Scholar] [CrossRef] [PubMed]
- Micromedex. DRUG-REAX® System (Electronic Version); Truven Health Analytics: Greenwood Village, CO, USA, 2014. Available online: http://www-micromedexsolutions-com.ez27.periodicos.capes.gov.br/ (accessed on 8 March 2014).
- UpToDate. Available online: http://www.UpToDate.com/home/index.html (accessed on 24 March 2014).
- MEDSCAPE (Internet). Copyright© 1994–2014. 2014. Available online: http//reference.medscape.com/pharmacists (accessed on 13 March 2014).
- National Therapeutic Formulary. In Formulário Terapêutico Nacional–FTN. 2010. Rename; Ministry of Health. [Ministério da Saúde]: Brasília, Brazil, 2010; p. 1135.
- Fleiss, J.L. Measuring nominal scale agreement among many raters. Psychol. Bull. 1971, 76, 378–382. [Google Scholar] [CrossRef]
- Cohen, J. Weighted kappa: Nominal scale agreement with provision for scaled disagreement or partial credit. Psychol. Bull. 1968, 70, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Landis, J.R.; Koch, G.G. An application of hierarchical kappa-type statistics in the assessment of majority agreement among multiple observers. Biometrics 1977, 33, 363–374. [Google Scholar] [CrossRef] [PubMed]
- Khanal, A.; Peterson, G.M.; Castelino, R.L.; Jose, M.D. Renal drug dosing recommendations: Evaluation of product information for brands of the same drug. Intern. Med. J. 2014, 44, 591–596. [Google Scholar] [CrossRef] [PubMed]
- Fulda, T.R.; Valuck, R.J.; Zanden, J.V.; Parker, S.; Byrns, P.J. Disagreement among drug compendia on inclusion and ratings of drug-drug interactions. Curr. Ther. Res. 2000, 61, 540–548. [Google Scholar] [CrossRef]
- Guo, J.J.; Wigle, P.R.; Lammers, K.; Vu, O. Comparison of potentially hepatotoxic drugs among major US drug compendia. Res. Soc. Adm. Pharm. 2005, 1, 460–479. [Google Scholar] [CrossRef] [PubMed]
- Salgado, T.M.; Arguello, B.; Martinez-Martinez, F.; Benrimoj, S.I.; Fernandez-Llimos, F. Clinical relevance of information in the Summaries of Product Characteristics for dose adjustment in renal impairment. Eur. J. Clin. Pharmacol. 2013, 69, 1973–1979. [Google Scholar] [CrossRef] [PubMed]
- Salgado, T.M.; Arguello, B.; Martinez-Martinez, F.; Benrimoj, S.I.; Fernandez-Llimos, F. Lack of harmonisation in the classification of renal impairment in European Summaries of Product Characteristics. Intern. Med. J. 2015, 45, 686–687. [Google Scholar] [CrossRef] [PubMed]
- D’Angio, R.G. Nonsteroidal antiinflammatory drug-induced renal dysfunction related to inhibition of renal prostaglandins. Drug Intell. Clin. Pharm. 1987, 21, 954–960. [Google Scholar] [PubMed]
- Briguori, C.; Tavano, D.; Colombo, A. Contrast agent—Associated nephrotoxicity. Prog. Cardiovasc. Dis. 2003, 45, 493–503. [Google Scholar] [CrossRef] [PubMed]
- Choudhury, D.; Ahmed, Z. Drug-associated renal dysfunction and injury. Nat. Clin. Pract. Nephrol. 2006, 2, 80–91. [Google Scholar] [CrossRef] [PubMed]
- Markowitz, G.S.; Perazella, M.A. Drug-induced renal failure: A focus on tubulointerstitial disease. Clin. Chim. Acta 2005, 351, 31–47. [Google Scholar] [CrossRef] [PubMed]
- Pannu, N.; Nadim, M.K. An overview of drug-induced acute kidney injury. Crit. Care Med. 2008, 36, S216–S223. [Google Scholar] [CrossRef] [PubMed]
- Rossert, J. Drug-induced acute interstitial nephritis. Kidney Int. 2001, 60, 804–817. [Google Scholar] [CrossRef] [PubMed]
- St Peter, W.L.; Clark, J.L.; Levos, O.M. Drug therapy in haemodialysis patients. Special considerations in the elderly. Drugs Aging 1998, 12, 441–459. [Google Scholar] [CrossRef] [PubMed]
- Jick, H. Adverse drug effects in relation to renal function. Am. J. Med. 1977, 62, 514–517. [Google Scholar] [CrossRef]
- Martin-Facklam, M.; Rengelshausen, J.; Tayrouz, Y.; Ketabi-Kiyanvash, N.; Lindenmaier, H.; Schneider, V.; Bergk, V.; Haefeli, W.E. Dose individualisation in patients with renal insufficiency: Does drug labelling support optimal management? Eur. J. Clin. Pharmacol. 2005, 60, 807–811. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bicalho, M.D.; Soares, D.B.; Botoni, F.A.; Reis, A.M.M.; Martins, M.A.P. Drug-Induced Nephrotoxicity and Dose Adjustment Recommendations: Agreement Among Four Drug Information Sources. Int. J. Environ. Res. Public Health 2015, 12, 11227-11240. https://doi.org/10.3390/ijerph120911227
Bicalho MD, Soares DB, Botoni FA, Reis AMM, Martins MAP. Drug-Induced Nephrotoxicity and Dose Adjustment Recommendations: Agreement Among Four Drug Information Sources. International Journal of Environmental Research and Public Health. 2015; 12(9):11227-11240. https://doi.org/10.3390/ijerph120911227
Chicago/Turabian StyleBicalho, Millena Drumond, Danielly Botelho Soares, Fernando Antonio Botoni, Adriano Max Moreira Reis, and Maria Auxiliadora Parreiras Martins. 2015. "Drug-Induced Nephrotoxicity and Dose Adjustment Recommendations: Agreement Among Four Drug Information Sources" International Journal of Environmental Research and Public Health 12, no. 9: 11227-11240. https://doi.org/10.3390/ijerph120911227





