1. Introduction
Visceral leishmaniasis (VL), popularly known as kala-azar, is an anthropozoonosis arising in endemic or epidemic form in the populations of peri-urban or rural areas that have a strong tendency to urbanize [
1]. In Brazil, the disease is caused by the protozoan
Leishmania chagasi, transmitted by sandflies of the genus
Lutzomyia [
2,
3]. The Northeast region of Brazil has a high incidence of VL, with 47% of reported human cases in both urban and rural areas [
4]. The state of Rio Grande do Norte has experienced an increase in the area affected by the disease, demonstrated by the increase in the number of municipalities reporting cases in recent years [
5]. The WHO describes VL as an important public health problem in the state of Rio Grande do Norte, with a high incidence rate, high mortality rate and widespread distribution in the state [
6]. Other results suggest that the state of Rio Grande do Norte is an important endemic area for VL [
7,
8].
The increasing number of cases of VL has been associated with the process of urbanization, overcrowded cities, poverty and inadequate sanitation [
9,
10,
11]. The transmission dynamics of VL are associated with a variety of complex eco-epidemiological settings, which offer suitable conditions for interactions between parasite, vector and reservoirs [
12]. For this reason, it is important to characterize areas near rivers and ponds to understand the environment and how it supports the maintenance of the VL vector. A study in Iran showed that the high susceptibility areas for VL outbreaks account for 36.3% of the studied area and occurred mainly in the north (which may affect the neighbouring countries) and south (which is a warning for other provinces in Iran), and these parts of the study area had many nomadic, riverside villages [
13].
The species of the greatest epidemiological importance in the transmission of VL in the Brazilian territory is the sand fly
Lutzomyia longipalpis [
14,
15]. This species easily adapts to urban environments, and its distribution pattern in urban areas can most likely be explained by the degradation of the environment and the presence of humans as a potential source of food [
16]. However, other environmental features, such as the presence of domestic animals, fruit trees and unsanitary conditions can also increase the abundance of sand flies, as they provide feeding and breeding places for these insects [
17,
18].
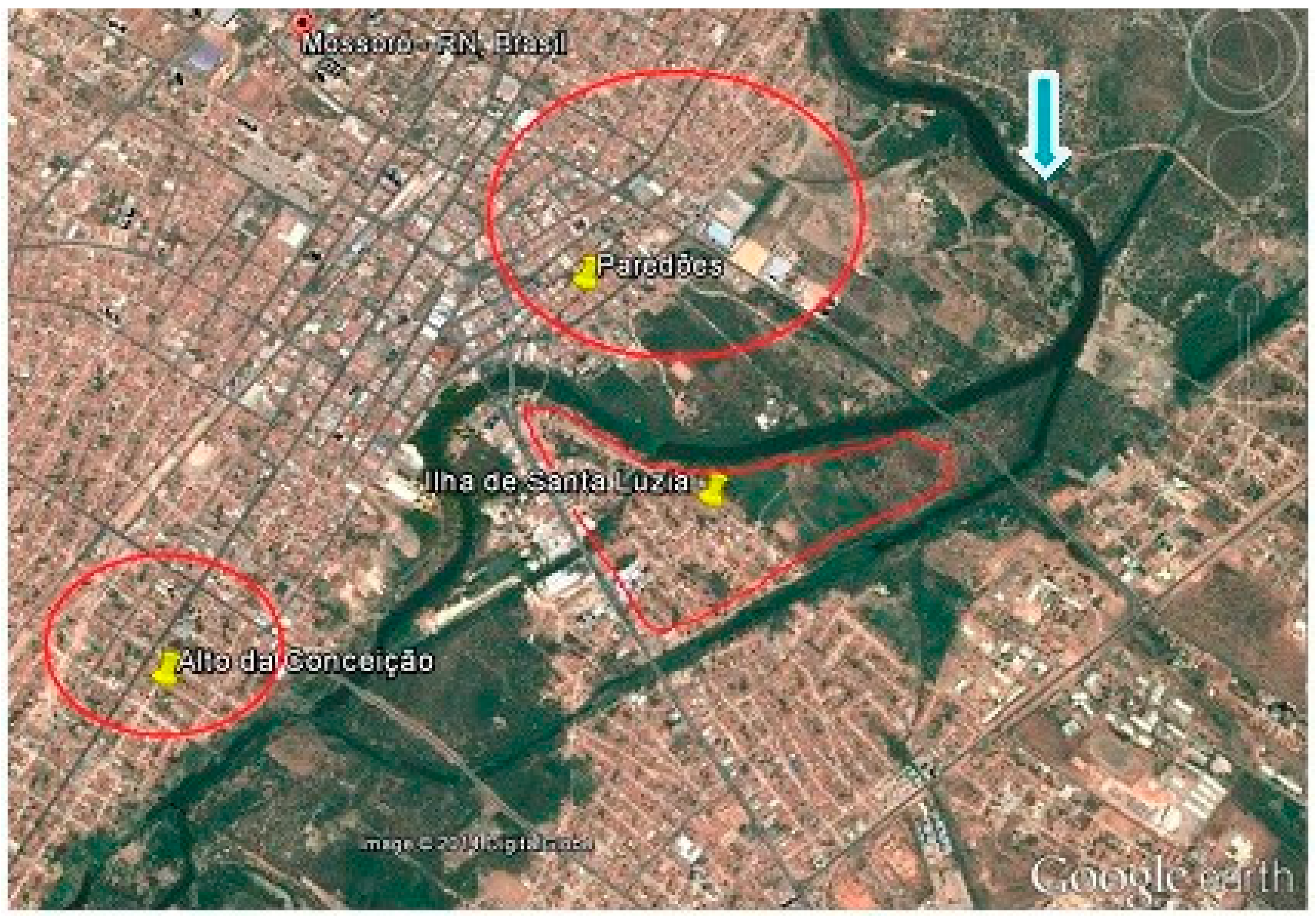
For these reasons, this study aimed to determine whether the people living in areas near the Mossoró River in the city of Mossoró, Rio Grande do Norte, Brazil, have knowledge of VL. It also sought to understand the environmental characteristics of the study area and to examine whether these characteristics favor the presence of the vector in these areas, supporting the transmission cycle of VL and therefore strengthening the endemicity of the disease.
6. Results and Discussion
The demographics of the respondents are summarized in
Table 1. The majority of participants were female, had an age range of 18–40 years of age and had completed some schooling (
p < 0.05).
Table 1.
Details of the social profile of respondents who live in areas near the Mossoró River, in the neighborhoods of Ilha de Santa Luzia, Alto da Conceição and Paredões in the city of Mossoró, Rio Grande do Norte, Brazil, 2013.
Table 1.
Details of the social profile of respondents who live in areas near the Mossoró River, in the neighborhoods of Ilha de Santa Luzia, Alto da Conceição and Paredões in the city of Mossoró, Rio Grande do Norte, Brazil, 2013.
| Variables Separated by Category 1 | Number of Responses/Number Total | Percent of Total (%) |
|---|
| Gender | | |
| Female | 396/478 | 82.8 * |
| Male | 82/478 | 17.2 |
| Age | | |
| 18–40 years | 205/478 | 42.9 * |
| 41–70 years | 204/478 | 42.7 |
| 71–100 years | 45/478 | 9.4 |
| No response | 24/478 | 5.0 |
| Education | | |
| Completed middle school | 257/478 | 53.8 * |
| Completed high school | 188/478 | 39.3 |
| Completed college | 33/478 | 6.9 |
The social profile of those who answered the questionnaire was comparable to that of similar studies, in which the majority of respondents were female and the mean age was 35 years [
20,
21]). Most had completed basic schooling, similar to other studies that discussed the association between low educational level and the risk of VL infection [
10,
20]. Because the present study results corroborate prior research, it is possible to conclude that the low educational level of the population in areas next to the Mossoró River may be susceptible VL infection. The goal was only to characterize the population surveyed, and this analysis was not used for discussion, it was just done a comparison with other similar studies.
Table 2 shows the environmental characteristics of the neighborhoods under study. With respect to vegetation, the vast majority of respondents (
p < 0.05) stated that there are remnants of forest in the areas around their homes. Respondents also reported the presence of fruit trees in the yards of homes (
p > 0.05), although this result was not significant. Most reported that garbage was collected by garbage collection trucks (
p < 0.05), though many also reported the presence of open-air garbage dumps in these areas (
p < 0.05). The majority of respondents reported that they dumped waste water into the river (
p < 0.05), and the majority of respondents reported a lack of insecticidal or repellent plants in the area around their homes (
p < 0.05).
Regarding the presence of mosquitoes in the house, most respondents reported that the region “has a lot of insects”, (86.2%; p < 0.05), that in the rainy season the number of insects, including mosquitoes (Culex sp.), significantly increases (83.5%; p < 0.05) and that insects are most active in the early evening.
Most respondents reported that they lived near areas of vegetation (
p < 0.05), and it is known that environments with remnants of forest and fruit trees provide suitable breeding sites for sand flies because the insects feed on fallen fruits [
17,
18]. Thus, the presence of this type of vegetation around the homes of residents is a factor enabling the spread of the vector in these areas.
Table 2.
Environmental features around the residences of respondents who live in areas near the Mossoró River, neighborhoods Ilha de Santa Luzia, Alto da Conceição and Paredões in the city of Mossoró, Rio Grande do Norte, 2013.
Table 2.
Environmental features around the residences of respondents who live in areas near the Mossoró River, neighborhoods Ilha de Santa Luzia, Alto da Conceição and Paredões in the city of Mossoró, Rio Grande do Norte, 2013.
| Variables Separated by Category 1 | Number of Responses/Number Total | Number Relative (%) |
|---|
| Lives near areas of vegetation | | |
| Yes | 449/478 | 93.9 * |
| No | 29/478 | 6.1 |
| Destination of Household Waste | | |
| Garbage truck | 288/478 | 60.3 * |
| Local dump site | 6/478 | 1.3 |
| Garbage truck and other destinations | 184/478 | 38.5 |
| Lives near open-air garbage dump | | |
| Yes | 366/478 | 76.6 * |
| No | 112/478 | 23.4 |
| Disposal of domestic sewage | | |
| Mossoró River | 213/478 | 44.6 * |
| Sanitation system | 181/478 | 37.9 |
| Sink | 70/478 | 14.6 |
| Sink and Mossoró River | 8/478 | 1.7 |
| Unknown | 6/478 | 1.3 |
| Vegetation around residence | | |
| Fruit trees | 135/478 | 28.2 |
| Other types of vegetation | 166/478 | 34.7 |
| Does not have vegetation | 177/478 | 37.0 |
| Presence of mosquitoes in house | | |
| Yes | 412/478 | 86.2 * |
| No | 66/478 | 13.8 |
| Increase in mosquitoes in the rainy season | | |
| Yes | 399/478 | 83.5 * |
| No | 79/478 | 16.5 |
| Repellent plants/insecticides used near home | | |
| Yes | 15/478 | 3.1 |
| No | 463/478 | 96.9 * |
The lack of sanitation in the study area is conducive to its maintenance as an environmental vector of VL because the garbage dumps and sewers exposed to the environment provide the type of organic matter that is a food source for immature and adult sand flies [
17,
18]. The presence of poor housing and environmental sanitation conditions are most likely associated with maintaining the endemicity of VL in these areas, given that the disease tends to affect disadvantaged populations [
15]. The areas of study also present an environment rich in sand fly food sources.
The interior of homes also presents favorable conditions for the presence and reproduction of several species of insects that are active from early evening throughout the night. The improper disposal of solid waste and the presence of spaces that collect standing water in the rainy season are conducive to mosquito breeding. Increased populations of disease-carrying insects, such as sand flies, can eventually cause public health problems [
22]. Thus, this population may eventually be exposed to sand flies without knowing the risk to their health.
It is possible that the absence of plants with repellent or insecticidal qualities around homes can be a factor that maintains the presence of insects near the home when associated with the other environmental factors. According to a study, of insecticidal plants, oil from the seeds of
Acalypha indica, popularly known as neem, has an insecticidal effect on the three stages of development of
L. longipalpis, and this plant is easily grown in tropical regions [
23]. An Ethiopian study of the neem plant concluded that the application of neem oil may be safe for personal use as a low-cost protective measure against sand fly bites for people living in endemic areas [
24]. However, a lack of knowledge about the action of these plants most likely prevents them from being used to control the population of VL vectors in specific regions. Encouragement of research in this area may lead to a decrease in the density of the sand fly population.
As shown in
Table 3, respondents were asked whether they performed fumigation in their homes, and 163 (34.1%) reported that they used chemicals to prevent insect infestation. The frequency of fumigation was around one to three times per year (
p > 0.05), and generally, respondents did not know the active ingredient or trade name of the product used (
p > 0.05). When the population was asked if the fumigation was conducted in homes, was obtained as a response all the possibilities of alternatives included in the questionnaire, including the daily fumigation. However, it was realized that this practice was done randomly and with different types of chemicals, including poisons to kill ants and cockroaches.
Table 3.
Information on home fumigation from respondents who live in areas near the Mossoró River, in the neighborhoods of Ilha de Santa Luzia, Alto da Conceição and Paredões, in the city of Mossoró, Rio Grande do Norte, 2013.
Table 3.
Information on home fumigation from respondents who live in areas near the Mossoró River, in the neighborhoods of Ilha de Santa Luzia, Alto da Conceição and Paredões, in the city of Mossoró, Rio Grande do Norte, 2013.
| Variables Separated by Category 1 | Number of Responses/Number Total | Percent of Total (%) |
|---|
| Frequency of fumigation | | |
| Daily | 5/163 | 3.1 |
| 1 to 3× per week | 29/163 | 17.8 |
| 1 to 3× per month | 61/163 | 37.4 |
| 1 to 3× per a year | 68/163 | 41.7 * |
| Chemical products used | | |
| Pyrethroids | 6/163 | 3.7 |
| Others | 76/163 | 46.6 |
| Unknown | 81/163 | 49.7 * |
This demonstrates the ignorance of the people about the products used specifically to control sand flies such as pyrethroids. This ignorance, therefore, may increase the risk of this population being bitten by females of these vectors. Adult sand fly control measures include the use of spray insecticides in homes and animal shelters [
9]. Chemical treatment using insecticides with residual action is a recommended strategy for vector control; however, it only affects the adult insect and aims to prevent and/or reduce the risk of disease transmission [
2]. Vector control actions are still the most powerful tools for the prevention of vector-borne diseases [
25]. Area residents interviewed for this study were unaware of this information and ultimately did not use this strategy to control the vector of VL, which may cause the persistence of sandflies in the area.
Few respondents reported using synthetic pyrethroids for pest control. These products have been evaluated for the control of VL vectors and are used by health services as a control measure in disease transmission areas [
9]. Although residents interviewed for this study used pyrethroids, they do not specifically use these products to control the VL vector because they were unaware of this benefit.
Regarding cleaning the area around the home, 440 (92.1%) of the respondents reported performing this activity (
Table 4). It was observed that the cleaning of backyards was performed daily with brooms (
p < 0.05). Most respondents reported that they did not use chemicals to clean their homes (
p < 0.05).
Table 4.
Information on cleaning around the homes of the residences of respondents who live in areas near the Mossoró River, in the neighborhoods of Ilha de Santa Luzia, Alto da Conceição and Paredões in the city of Mossoró, Rio Grande do Norte, 2013.
Table 4.
Information on cleaning around the homes of the residences of respondents who live in areas near the Mossoró River, in the neighborhoods of Ilha de Santa Luzia, Alto da Conceição and Paredões in the city of Mossoró, Rio Grande do Norte, 2013.
| Variables Separated by Category 1 | Number of Responses/Number Total | Percent of Total (%) |
|---|
| Method of cleaning | | |
| Sweep | 389/440 | 88.4 * |
| Sweep and wash | 50/440 | 11.4 |
| Weed | 1/440 | 0.2 |
| Frequency of cleaning | | |
| Daily | 266/440 | 60.5 * |
| 1 to 3× per week | 145/440 | 33.0 |
| 1 to 2× per month | 21/440 | 4.8 |
| 2 to 6× per a year | 8/440 | 1.8 |
| Chemical products used | | |
| General cleaning products | 50/440 | 11.4 |
| None used | 390/440 | 88.6 * |
We can see that most residents were concerned about maintaining clean yards, though not with the intention of eliminating sand fly habitats. It is known that this is a way to avoid the maintenance of environments that support sand fly populations because, preventive measures for environmental management include the cleaning of backyards, land and public squares to eliminate favorable habitats for the breeding of immature forms of the vector [
2].
Respondents were asked whether they raised domestic pets and/or livestock in their backyards, and 265 (55.4%) said yes (
p < 0.05).
L. longipalpis is the most common sand fly in the area under study and is found throughout the year in different parts of the city in question [
5,
26]. These insects are well adapted to environments modified by human activity where chickens and other domestic animals are present, and these factors can increase the abundance of sandflies by providing suitable breeding sites [
17].
One finding that merits attention, although it is not statistically significant, is the 68 (25.6%) households that raise chickens. Although chickens are not a confirmed reservoir of Leishmania, chickens are common sources of blood for
L. longipalpis, and their shelters are known habitats and breeding locations for sand flies, which makes chickens important in the maintenance of vector populations in a given area [
27,
28]. Thus, the presence of chickens in these areas of study is a hazard to residents.
On the topic of domestic animals in homes, when asked which animals lived with their families in the home environment, 163 (61.5%) respondents reported pet dogs (
p < 0.05), a fact which confirms the epidemiology of the disease in the country, given that dogs are the main reservoir of
L. chagasi in urban areas [
29,
30,
31]. In a study conducted in Iran, it was concluded that the low prevalence of VL in humans could be explained by the scarcity of infected domestic dogs [
32]. In the study area, residents commonly have contact with dogs, and there is no effective strategy against canine VL infection, which may cause the increased susceptibility of contamination by VL.
Respondents who raised dogs were asked questions about which strategies they used to protect their dogs from sand fly bites. The majority of respondents did not use repellent collars on their dogs (
p < 0.05). When asked if they walked their dogs at night, most said they did not (
p > 0.05), although this figure was not statistically significant (
Table 5).
Table 5.
Methods of protecting dogs against sand fly bites in areas near the Mossoró River, in the neighborhoods of Ilha de Santa Luzia, Alto da Conceição and Paredões in the city of Mossoró, Rio Grande do Norte, 2013.
Table 5.
Methods of protecting dogs against sand fly bites in areas near the Mossoró River, in the neighborhoods of Ilha de Santa Luzia, Alto da Conceição and Paredões in the city of Mossoró, Rio Grande do Norte, 2013.
| Variables Separated by Category 1 | Number of Responses/Number Total | Percent of Total (%) |
|---|
| Use of repellent collars | | |
| Yes | 3/163 | 1.8 |
| No | 160/163 | 98.2 * |
| The dog is walked between 6 pm and 6 am | | |
| Yes | 69/163 | 42.3 |
| No | 94/163 | 57.7 |
The literature states that the use of insecticide-impregnated collars on dogs is one of the measures used to control the population of adult sand flies [
9] and that avoiding possible canine infection is a way to maintain distance from the vector. A study carried out in Italy suggests that the mass use of insecticide-impregnated collars led to a reduction of movement of
L. chagasi in a region [
33]. Because few dogs living in the study areas wear collars with repellent action, dogs in the study area may be at risk of infection by VL. Another factor that draws attention is the habit that some have to walk their dogs at night, which is a risk to animals because
L. longipalpis activity occurs from dusk to dawn [
34]. Dogs that are walked when vector activity is high have a higher susceptibility of infection due to the possibility of contact with sandflies in the environment.
With regard to knowledge about the transmission of disease, 471 (98.5%) respondents said that they had heard of “kala-azar” (
p < 0.05). Of these, 229 (48.6%) said they did not know the agent of transmission (
p < 0.05). Residents who responded that mosquitoes transmitted VL (170; 36.1%) were asked other questions about the vector, as shown in
Table 6. The association between knowledge of respondents about the transmission of VL and social profile was significant for: the knowledge of “transmitting agent” was associated with age and education of respondents, knowledge of “reported seeing the vector” was associated with the gender of the respondent.
Most respondents knew that mosquitoes transmitted VL, but they reported that they have never seen this insect, did not know its features, were unaware of the environments preferred by the vector and did not know the hours of insect activity (
p < 0.05), which exposes them to the susceptibility of VL infection. Lack of knowledge on sandfly causes the population to maintain favorable environments for the spread of VL, leaving family and pets exposed to the bite of the vector and the possibility of this sandfly contain the infective forms of the parasite, thus being able to infect people and dogs. Increasing the knowledge of sand fly reproduction habitats may prove useful to vector control efforts, leading to a reduction in the density of vectors and thus decrease disease incidence [
35,
36].
Research conducted in the state of Belo Horizonte, Brazil, noted the general lack of VL knowledge among the population of the region, indicating an urgent need for the implementation of educational practices [
20]. The low knowledge in the study population about the VL vector prevents them from seeking effective control measures for vector-borne disease.
VL researchers in Ethiopia reported that the most important prerequisite for the success of prevention and control of any disease is community participation because the cooperation of the affected population is essential for the implementation and use of program activities [
21]. Therefore, it is important that the study population knows about ways to control the VL vector because then they are able to eliminate vector breeding habitats, blocking the chain of transmission.
The lack of knowledge about basic aspects of VL shows a lack of access to information. A study reports that the preventive measures of VL are impeded during implementation by the absence of a public informed about the basics characteristics of the disease [
20]. Services that include measures to control VL should be deployed in health education activities with the population and require the active inclusion of multidisciplinary teams, a practice that so far has not been observed [
2]. It is also known that knowledge about sand fly reproduction habitats provides useful information for guiding biological control efforts, leading to a reduction in the density of vectors and thus a reduction of disease incidence [
36].
Table 6.
Respondents knowledge about the vector of visceral leishmaniasis in Mossoró near the river e associação com perfil social, in the neighborhoods of Ilha de Santa Luzia, Alto da Conceição and Paredões in the city of Mossoró, Rio Grande do Norte areas, 2013.
Table 6.
Respondents knowledge about the vector of visceral leishmaniasis in Mossoró near the river e associação com perfil social, in the neighborhoods of Ilha de Santa Luzia, Alto da Conceição and Paredões in the city of Mossoró, Rio Grande do Norte areas, 2013.
| Variables Separated by Category | Total Freq. (%) | Gender Freq. (%) | Age a Freq. (%) | Education Freq. (%) |
|---|
| Female | Male | p-Value | 18–40 years | 41–70 years | 71–100 years | p-Value τ | Completed Middle School | Completed High School | Completed College | p-Value |
|---|
| Heard about Kala-azar | | | | | | | | | | | | |
| Yes | 471 (98.5) | 391 (83.0) | 80 (17.0) | 0.344 | 203 (44.8) | 206 (45.5) | 44 (9.7) | 0.401 | 251 (53.3) | 187 (39.7) | 33 (7.0) | 0.226 |
| No | 07 (1.5) | 05 (71.4) | 02 (28.6) | 02 (28.6) | 04 (57.1) | 01 (14.3) | 06 (85.7) | 01 (14.3) | 0 (0.0) |
| Total | 478 (100) | 396 (82.8) | 82 (17.2) | | 205 (44.6) | 210 (45.7) | 45 (9.8) | | 257 (53.8) | 188 (39.3) | 33 (6.9) | |
| Transmitting agent | | | | | | | | | | | | |
| Mosquito | 170 (36.1) | 145 (85.3) | 25 (14.7) | 0.585 | 82 (50.9) | 65 (40.4) | 14 (8.7) | 0.046 * | 65 (38.2) | 87 (51.2) | 18 (10.6) | <0.001 * |
| Incorrectly answered | 72 (15.3) | 58 (80.6) | 14 (19.4) | 30 (42.3) | 38 (53.5) | 03 (4.2) | 45 (62.5) | 23 (31.9) | 04 (5.6) |
| Did not know | 229 (48.6) | 188 (82.1) | 41 (17.9) | 91 (41.2) | 103 (46.6) | 27 (12.2) | 141 (61.6) | 77 (33.6) | 11 (4.8) |
| Total b | 471 (100) | 391 (83.0) | 80 (17.0) | | 203 (44.8) | 206 (45.5) | 44 (9.7) | | 251 (53.3) | 187 (39.7) | 33 (7.0) | |
| Reported seeing the vector | | | | | | | | | | | | |
| Yes | 09 (5.3) | 05 (55.6) | 04 (44.4) | 0.028 * | 06 (66.7) | 03 (33.3) | 0 (0.0) | 0.494 | 04 (44.4) | 04 (44.4) | 01 (11.1) | 0.913 |
| No | 161 (94.7) | 140 (87.0) | 21 (13.0) | 76 (50.0) | 62 (40.8) | 14 (9.2) | 61 (37.9) | 83 (51.6) | 17 (10.6) |
| Total b | 170 (100) | 145 (85.3) | 25 (14.7) | | 82 (50.9) | 65 (40.4) | 14 (8.7) | | 65 (38.2) | 87 (51.2) | 18 (10.6) | |
| Characteristics of the vector | | | | | | | | | | | | |
| Known | 07 (4.1) | 04 (57.1) | 03 (42.9) | 0.066 | 04 (57.1) | 03 (42.9) | 0 (0.0) | 0.704 | 04 (57.1) | 02 (28.6) | 01 (14.3) | 0.471 |
| Unknown | 163 (95.9) | 141 (86.5) | 22 (13.5) | 78 (50.6) | 62 (40.3) | 14 (9.1) | 61 (37.4) | 85 (52.1) | 17 (10.4) |
| Total b | 170 (100) | 145 (85.3) | 25 (14.7) | | 82 (50.9) | 65 (40.4) | 14 (8.7) | | 65 (38.2) | 87 (51.2) | 18 (10.6) | |
| Preferred habitats of vector | | | | | | | | | | | | |
| Known | 28 (16.5) | 25 (89.3) | 03 (10.7) | 0.770 | 12 (46.2) | 12 (46.2) | 02 (7.7) | 0.806 | 06 (21.4) | 18 (64.3) | 04 (14.3) | 0.133 |
| Unknown | 142 (83.5) | 120 (84.5) | 22 (15.5) | 70 (51.9) | 53 (39.3) | 12 (8.9) | 59 (41.5) | 69 (48.6) | 14 (9.9) |
| Total b | 170 (100) | 145 (85.3) | 25 (14.7) | | 82 (50.9) | 65 (40.4) | 14 (8.7) | | 65 (38.2) | 87 (51.2) | 18 (10.6) | |
| Vector activity times | | | | | | | | | | | | |
| Known | 25 (14.7) | 24 (96.0) | 01 (4.0) | 0.131 | 11 (55.0) | 09 (45.0) | 0 (0.0) | 0.336 | 08 (32.0) | 15 (60.0) | 02 (8.0) | 0.628 |
| Unknown | 145 (85.3) | 121 (83.4) | 24 (16.6) | 71 (50.4) | 56 (39.7) | 14 (9.9) | 57 (39.3) | 72 (49.7) | 16 (11.0) |
| Total b | 170 (100) | 145 (85.3) | 25 (14.7) | | 82 (50.9) | 65 (40.4) | 14 (8.7) | | 65 (38.2) | 87 (51.2) | 18 (10.6) | |