An Environmentally Friendly Method for Testing Photocatalytic Inactivation of Cyanobacterial Propagation on a Hybrid Ag-TiO2 Photocatalyst under Solar Illumination
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation of Ag-TiO2 Film Supported on Diatomite
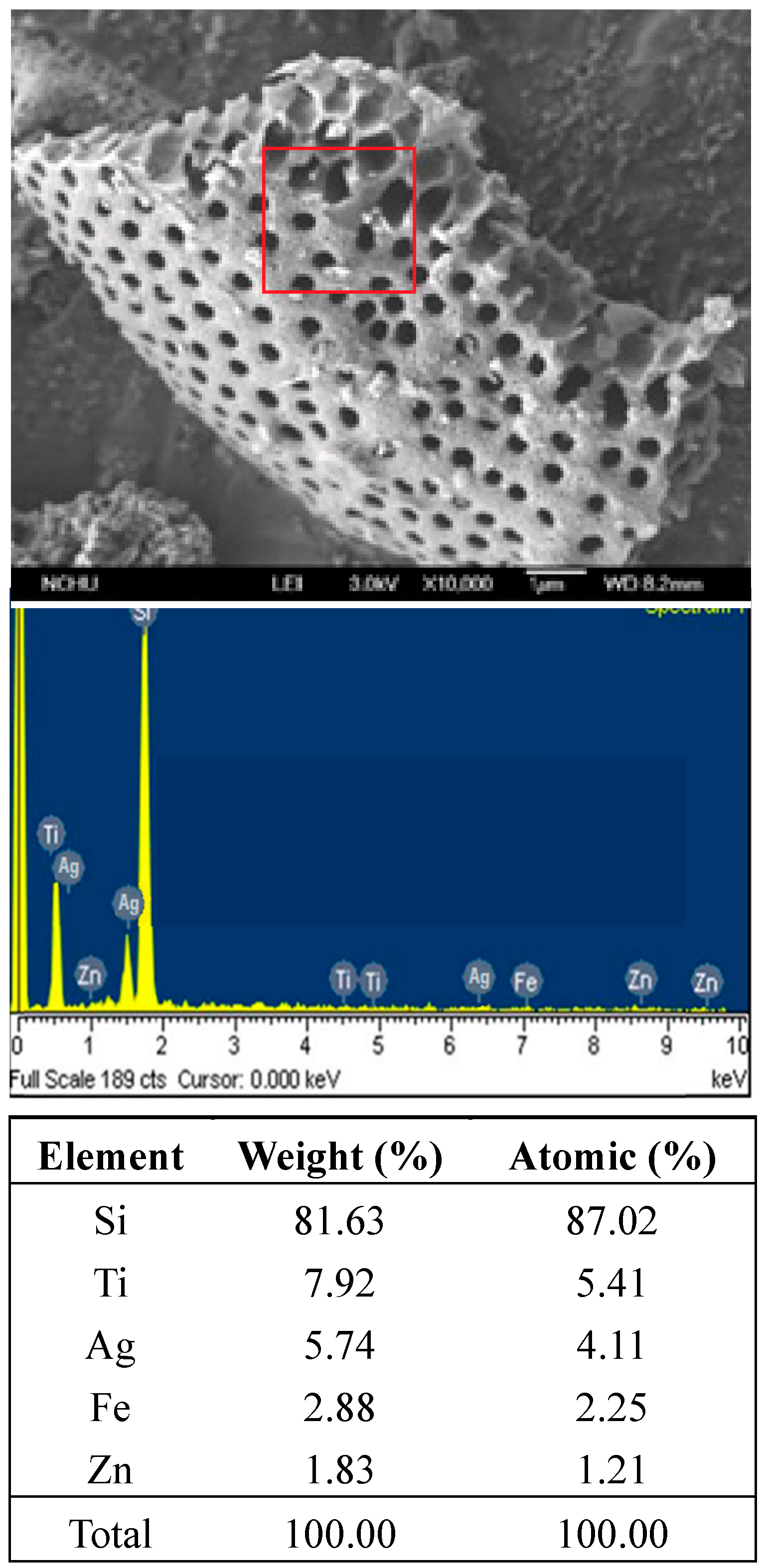
2.2. Cultivation of Cyanobacteria
| Cyanobacteria | Treatment | Nutrients | |||
|---|---|---|---|---|---|
| Light (Μe/m2s) | NO3−1-N (mg/L) | NH3-N (mg/L) | PO4−3-P (mg/L) | ||
| Microcystis aeruginosa | ASM-1 a | 30 | 32.8 ± 2.6 | - | 3.2 ± 0.3 |
| Oscillatoria tenuisa | DY-3 b | 32 | 6.8 ± 0.4 | - | 0.7 ± 0.1 |
2.3. Photocatalytic Inactivation of Cyanobacteria
2.4. Analytical Methods
2.5. Mutagenicity Tests
3. Results and Discussion
3.1. Inhibition of Cyanobacterial Growth
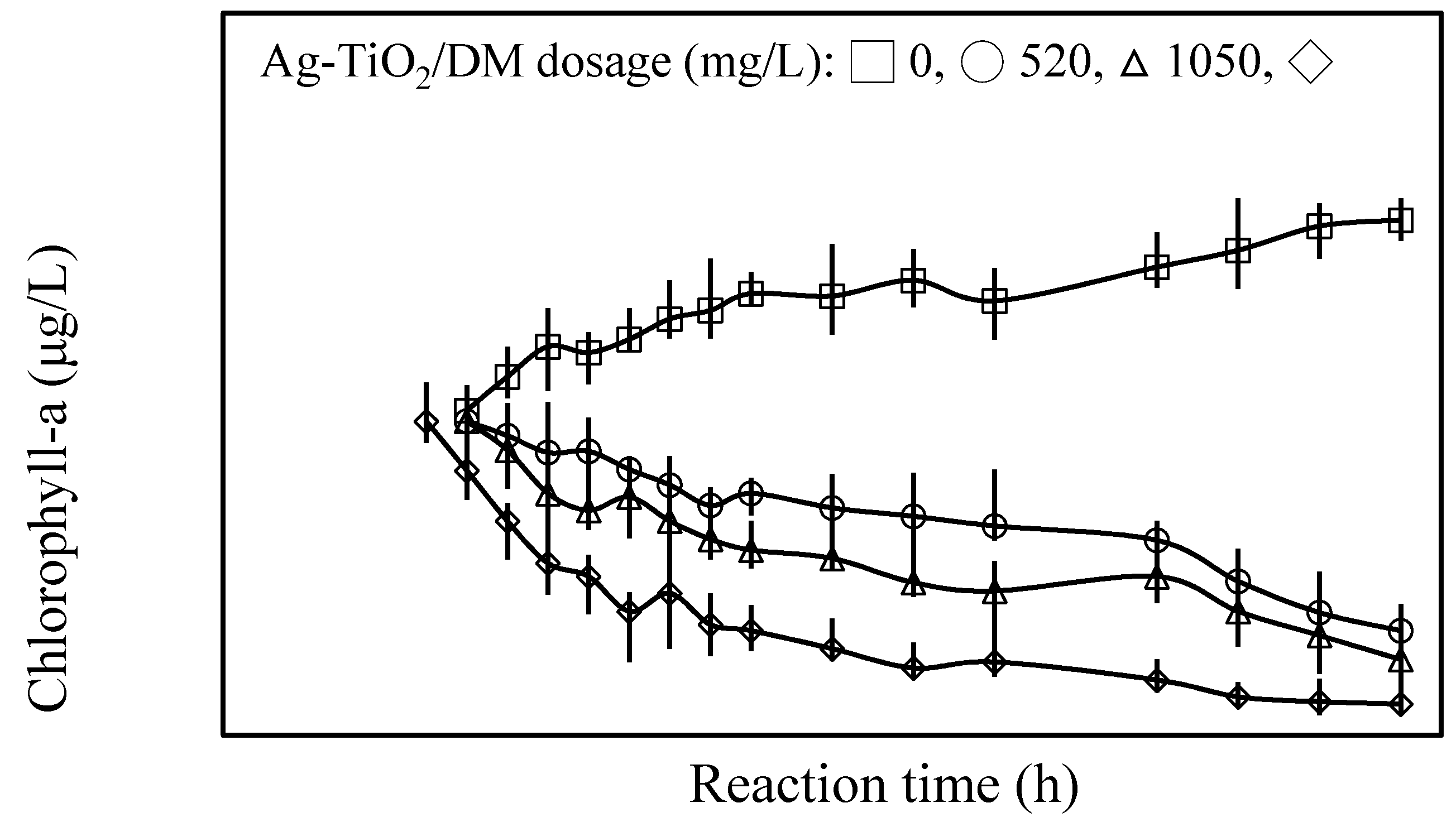
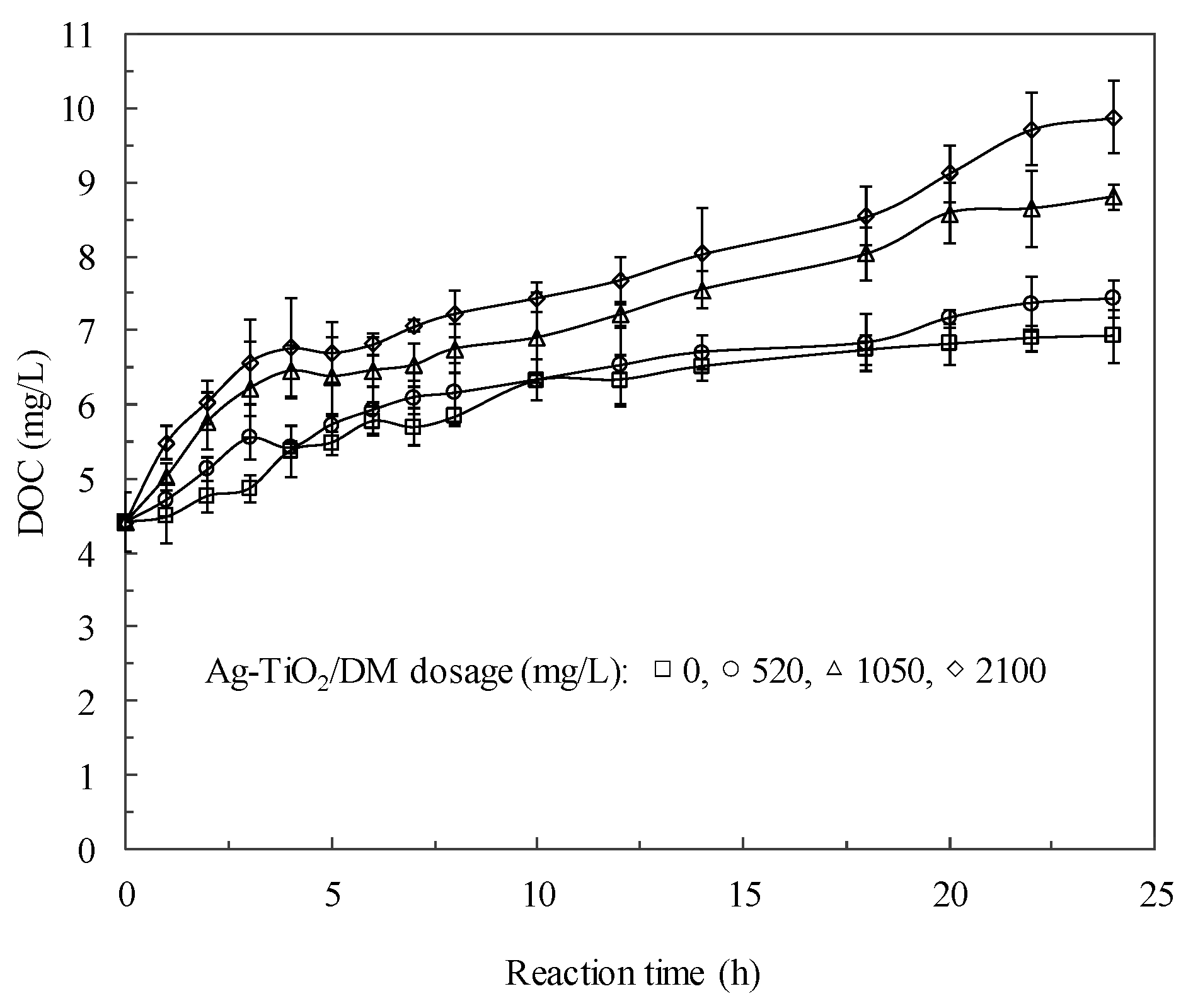
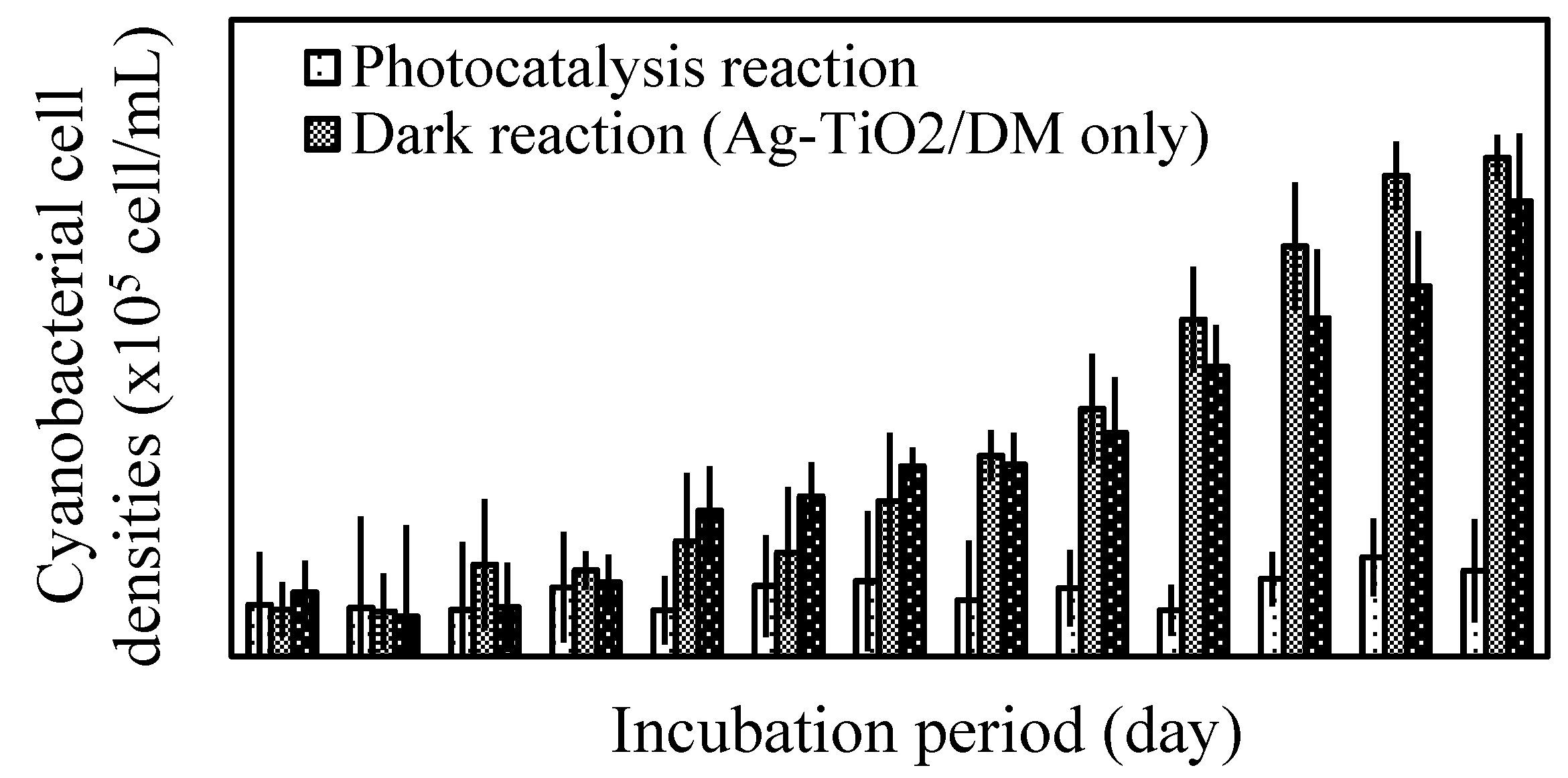
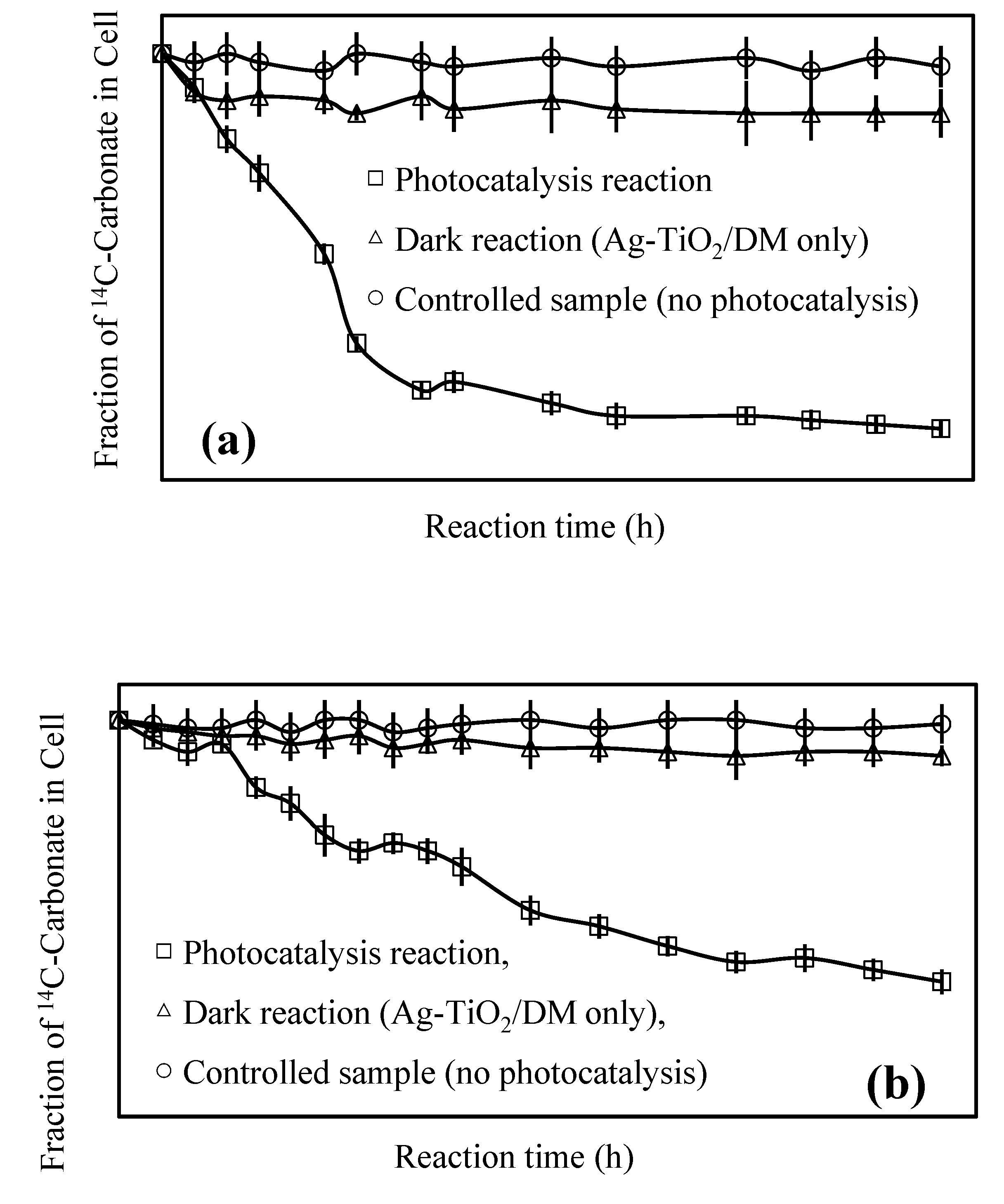

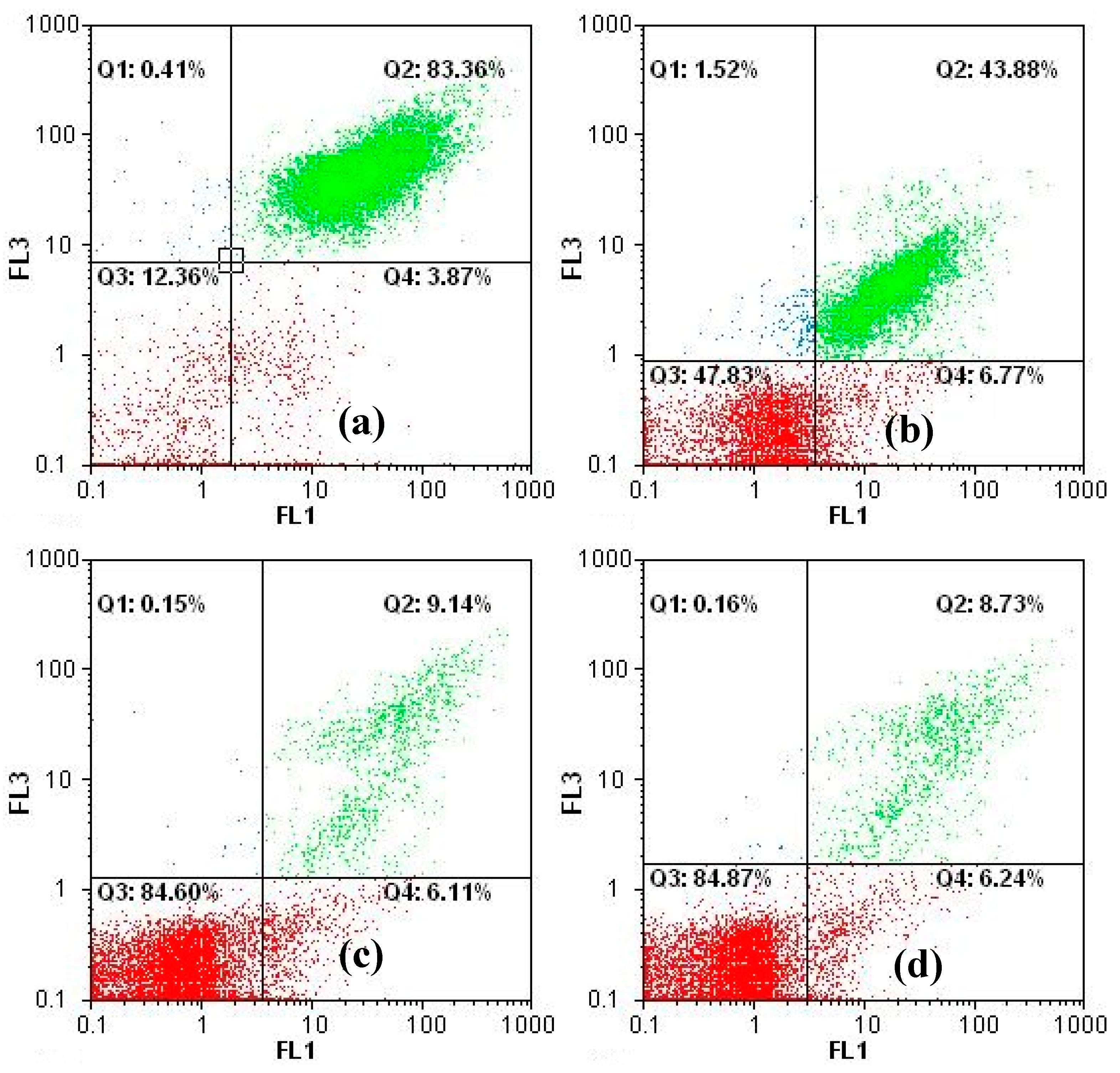
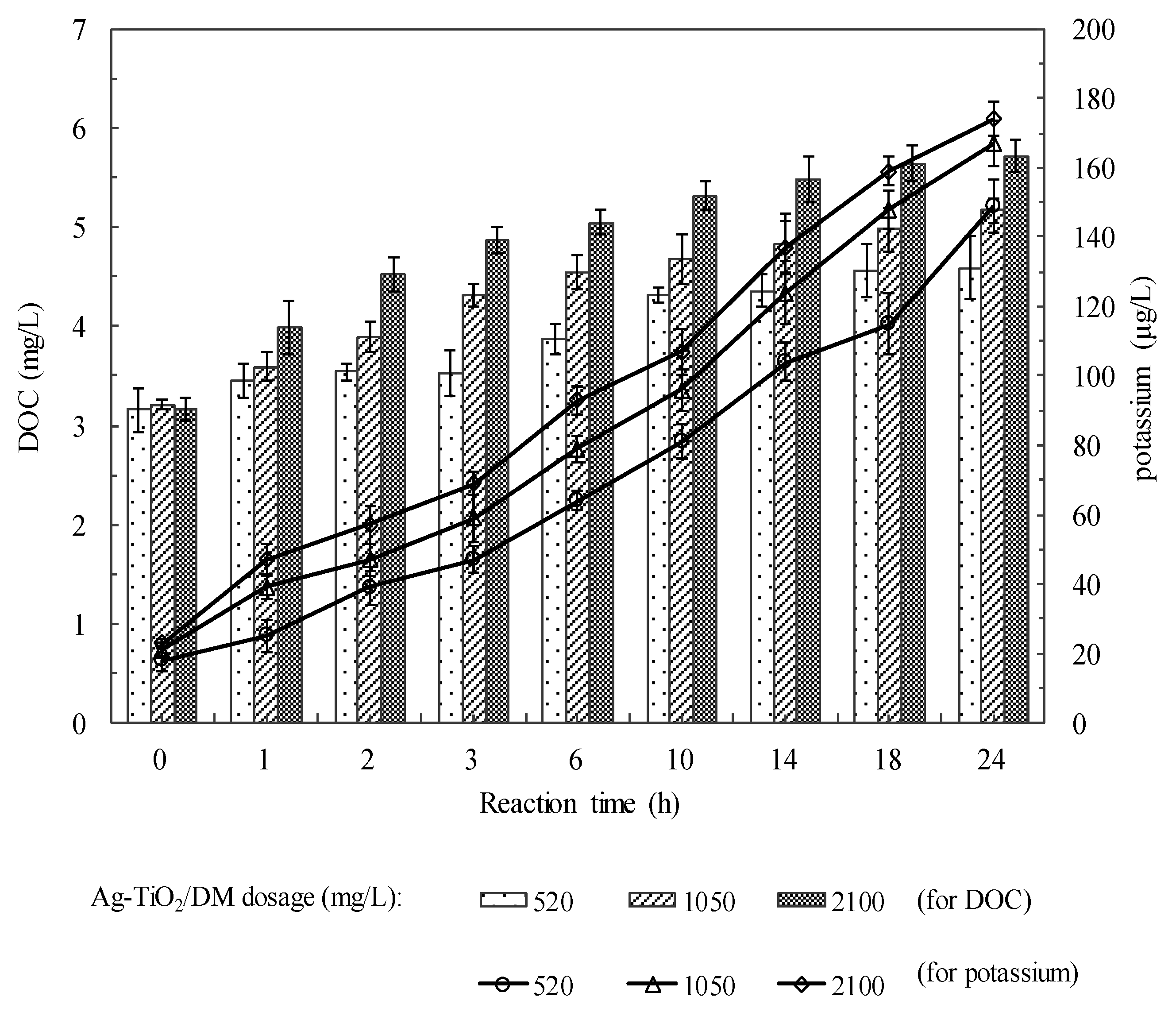
3.2. Fate of Metabolic Products During Photocatalysis
| Samples | Dose (L Per Plate) | Mean Revertant Colonies/Plate | ||||||
|---|---|---|---|---|---|---|---|---|
| TA98 | TA100 | |||||||
| −S9 a | +S9 b | −S9 | +S9 | |||||
| Blank control | 0 | 27 ± 7 c | 51 ± 6 | 184 ± 15 | 207 ± 24 | |||
| Microcystis aeruginosa | 4.0 | 52 ± 8 | 92 ± 12 | 322 ± 37 | 452 ± 30 d | |||
| 2.0 | 42 ± 4 | 76 ± 16 | 274 ± 25 | 436 ± 19 d | ||||
| 1.0 | 37 ± 6 | 68 ± 8 | 245 ± 13 | 365 ± 27 | ||||
| 0.5 | 33 ± 7 | 66 ± 5 | 196 ± 18 | 377 ± 33 | ||||
| 0.25 | 31 ± 5 | 60 ± 7 | 208 ± 26 | 349 ± 25 | ||||
| Oscillatoria tenuisa | 4.0 | 45 ± 3 | 86 ± 11 | 280 ± 16 | 428 ± 35 d | |||
| 2.0 | 48 ± 6 | 79 ± 12 | 286 ± 28 | 386 ± 21 | ||||
| 1.0 | 37 ± 5 | 78 ± 5 | 256 ± 12 | 391 ± 28 | ||||
| 0.5 | 31 ± 6 | 62 ± 4 | 252 ± 8 | 345 ± 52 | ||||
| 0.25 | 34 ± 7 | 55 ± 13 | 197 ± 15 | 293 ± 18 | ||||
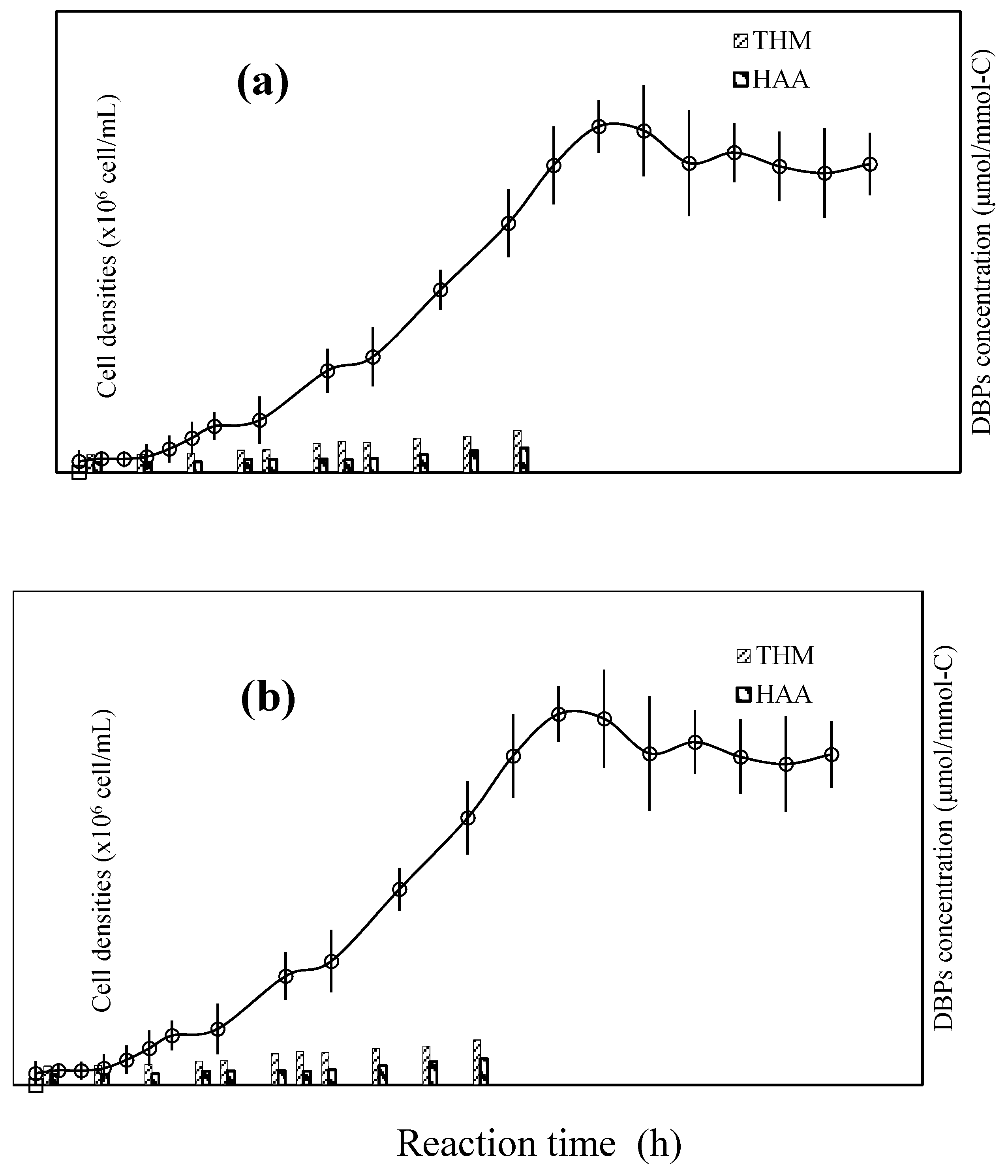
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- WHO. Guidelines for Drinking Water Quality, 3rd ed.; World Health Organization: Geneva, Switzerland, 2004; Volume 1. [Google Scholar]
- Benabbou, A.K.; Guillard, C.; Pigeot-Rémy, S.; Cantau, C.; Pigot, T.; Lejeune, P.; Derriche, Z.; Lacombe, S. Water Disinfection Using Photosensitizers Supported on Silica. J. Photochem. Photobiol. A. Chem. 2011, 219, 101–118. [Google Scholar] [CrossRef]
- Baram, N.; Starosvetsky, D.; Starosvetsky, J.; Epshtein, M.; Armon, R.; Ein-Eli, Y. Photocatalytic Inactivation of Microorganisms Using Nanotubular TiO2. Appl. Catal. B Environ. 2011, 101, 212–219. [Google Scholar] [CrossRef]
- Martinez, T.; Bertron, A.; Escadeillas, G.; Ringot, E. Algal Growth Inhibition on Cement Mortar: Efficiency of Water Repellent and Photocatalytic Treatments under UV/VIS Illumination. Int. Biodeterior. Biodegrad. 2014, 89, 115–125. [Google Scholar] [CrossRef]
- van Grieken, R.; Marugán, J.; Sordo, C.; Martínez, P.; Pablos, C. Photocatalytic Inactivation of Bacteria in Water Using Suspended and Immobilized Silver-TiO2. Appl. Catal. B Environ. 2009, 93, 112–118. [Google Scholar] [CrossRef]
- Van Grieken, R.; Marugán, J.; Pablos, C.; Furones, L.; López, A. Comparison between the Photocatalytic Inactivation of Gram-Positive E. faecalis and Gram-Negative E. coli Faecal Contamination Indicator Microorganisms. Appl. Catal. B Environ. 2010, 100, 212–220. [Google Scholar] [CrossRef]
- Zan, L.; Fa, W.; Peng, T.; Gong, Z. Photocatalysis Effect of Nanometer TiO2 and TiO2-Coated Ceramic Plate on Hepatitis B Virus. J. Photochem. Photobiol. B 2007, 86, 165–169. [Google Scholar] [CrossRef] [PubMed]
- Guillard, C.; Bui, T.; Felix, C.; Moules, V.; Lina, B.; Lejeune, P. Microbiological Disinfection of Water and Air by Photocatalysis. C. R. Chim. 2008, 11, 107–113. [Google Scholar] [CrossRef]
- Liga, M.V.; Bryant, E.L.; Colvin, V.L.; Li, Q. Virus Inactivation by Silver Doped Titanium Dioxide Nanoparticles for Drinking Water Treatment. Water Res. 2011, 45, 535–544. [Google Scholar] [CrossRef] [PubMed]
- Cho, M.; Cates, E.L.; Kim, J. Inactivation and Surface Interactions of MS-2 Bacteriophage in a TiO2 Photoelectrocatalytic Reactor. Water Res. 2011, 45, 2104–2110. [Google Scholar] [CrossRef] [PubMed]
- Sichel, C.; de Cara, M.; Tello, J.; Blanco, J.; Fernández-Ibáñez, P. Solar Photocatalytic Disinfection of Agricultural Pathogenic Fungi: Fusarium Species. Appl. Catal. B Environ. 2007, 74, 152–160. [Google Scholar] [CrossRef]
- Sichel, C.; Tello, J.; de Cara, M.; Fernández-Ibánez, P. Effect of UV Solar Intensity and Dose on the Photocatalytic Disinfection of Bacteria and Fungi. Catal. Today 2007, 129, 152–160. [Google Scholar] [CrossRef]
- Fernández-Ibánez, P.; Sichel, C.; Polo-López, M.I.; de Cara-García, M.; Tello, J.C. Photocatalytic Disinfection of Natural Well Water Contaminated by Fusarium solani Using TiO2 Slurry in Solar CPC Photo-Reactors. Catal. Today 2009, 144, 62–68. [Google Scholar] [CrossRef]
- Sontakke, S.; Modak, J.; Madras, G. Photocatalytic Inactivation of Escherichia coli and Pichia pastoris with Combustion Synthesized Titanium Dioxide. Chem. Eng. J. 2010, 165, 225–233. [Google Scholar] [CrossRef]
- Mitoraj, D.; Jańczyk, A.; Strus, M.; Kisch, H.; Stochel, G.; Heczko, P.B.; Macyk, W. Visible Light Inactivation of Bacteria and Fungi by Modified Titanium Dioxide. Photochem. Photobiol. Sci. 2007, 6, 642–648. [Google Scholar] [CrossRef] [PubMed]
- Sökmen, M.; Değerli, S.; Aslan, A. Photocatalytic Disinfection of Giardia intestinalis and Acanthamoeba castellani Cysts in Water. Exp. Parasitol. 2008, 119, 44–48. [Google Scholar] [CrossRef] [PubMed]
- Ryu, H.; Gerrity, D.; Crittenden, J.C.; Abbaszadegan, M. Photocatalytic Inactivation of Cryptosporidium parvum with TiO2 and Low-Pressure Ultraviolet Irradiation. Water Res. 2008, 42, 1523–1530. [Google Scholar] [CrossRef] [PubMed]
- Méndez-Hermida, F.; Ares-Mazás, E.; McGuigan, K.G.; Boyle, M.; Sichel, C.; Fernández-Ibánez, P. Disinfection of Drinking Water Contaminated with Cryptosporidium parvum Oocysts under Natural Sunlight and Using the Photocatalyst TiO2. J. Photochem. Photobiol. B 2007, 88, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Benabbou, A.K.; Derriche, Z.; Felix, C.; Lejeune, P.; Guillard, C. Photocatalytic Inactivation of Escherichia coli: Effect of Concentration of TiO2 and Microorganism, Nature, and Intensity of UV Irradiation. Appl. Catal. B Environ. 2007, 76, 257–263. [Google Scholar] [CrossRef]
- Rizzo, L. Inactivation and Injury of Total Coliform Bacteria after Primary Disinfection of Drinking Water by TiO2 Photocatalysis. J. Hazard. Mater. 2009, 165, 48–51. [Google Scholar] [CrossRef] [PubMed]
- Lydakis-Simantiris, N.; Riga, D.; Katsivela, E.; Mantzavinos, D.; Xekoukoulotakis, N.P. Disinfection of Spring Water and Secondary Treated Municipal Wastewater by TiO2 Photocatalysis. Desalination 2010, 250, 351–355. [Google Scholar] [CrossRef]
- Marugán, J.; van Grieken, R.; Pablos, C.; Sordo, C. Analogies and Differences between Photocatalytic Oxidation of Chemicals and Photocatalytic Inactivation of Microorganisms. Water Res. 2010, 44, 789–796. [Google Scholar] [CrossRef] [PubMed]
- Malato, S.; Fernández-Ibáñez, P.; Maldonado, M.I.; Blanco, J.; Gernjak, W. Decontamination and Disinfection of Water by Solar Photocatalysis: Recent Overview and Trends. Catal. Today 2009, 147, 1–59. [Google Scholar] [CrossRef]
- McCullagh, C.; Robertson, J.M.C.; Bahnemann, D.W.; Robertson, P.K.J. The application of TiO2 photocatalysis for disinfection of water contaminated with pathogenic micro-organisms: A review. Res. Chem. Intermed. 2007, 33, 359–375. [Google Scholar] [CrossRef]
- Matsunaga, T.; Tomoda, R.; Nakajima, T.; Wake, H. Photoelectrochemical sterilization of microbial cells by semiconductor powders. FEMS Microbiol. Lett. 1985, 29, 211–214. [Google Scholar] [CrossRef]
- Peller, J.R.; Whitman, R.L.; Griffith, S.; Harris, P.; Peller, C.; Scalzitti, J. TiO2 as a Photocatalyst for Control of the Aquatic Invasive Alga, Cladophora, under Natural and Artificial Light. J. Photochem. Photobiol. A Chem. 2007, 186, 212–217. [Google Scholar] [CrossRef]
- Cai, R.; Hashimoto, K.; Kubota, Y.; Fujishima, A. Increment of photocatalytic killing of cancer cells using titanium dioxide with the aid of superoxide dismutase. Chem. Lett. 1992, 427–430. [Google Scholar] [CrossRef]
- Fujishima, A.; Zhang, X.; Tryk, D.A. TiO2 photocatalysis and related surface phenomena. Surf. Sci. Rep. 2008, 63, 515–582. [Google Scholar] [CrossRef]
- Sunada, K.; Watanabe, T.; Hashimoto, K. Studies on photokilling of bacteria on TiO2 thin film. J. Photochem. Photobiol. A Chem. 2003, 156, 227–233. [Google Scholar] [CrossRef]
- Nair, R.G.; Paul, S.; Samdarshi, S.K. High UV/Visible Light Activity of Mixed Phase Titania: A Generic Mechanism. Sol. Energ. Mater. Sol. Cells 2011, 95, 1901–1907. [Google Scholar] [CrossRef]
- Hurum, D.C.; Agrios, A.G.; Gray, K.A.; Rajh, T.; Thurnauer, M.C. Explaining the Enhanced Photocatalytic Activity of Degussa P25 Mixed-Phase TiO2 Using EPR. J. Phys. Chem. B 2003, 107, 4545–4549. [Google Scholar] [CrossRef]
- Ochiai, T.; Fujishima, A. Photoelectrochemical properties of TiO2 photocatalyst and its applications for environmental purification. Photochem. Photobiol. C Photochem. Rev. 2012, 13, 247–262. [Google Scholar] [CrossRef]
- Sakthivel, S.; Shankar, M.V.; Palanichamy, M.; Arabindoo, B.; Bahnemann, D.W.; Murugesan, V. Enhancement of photocatalytic activity by metal deposition: Characterisation and photonic efficiency of Pt, Au and Pd deposited on TiO2 catalyst. Water Res. 2004, 38, 3001–3008. [Google Scholar] [CrossRef] [PubMed]
- He, C.; Yu, Y.; Hu, X.; Larbot, A. Influence of Silver Doping on the Photocatalytic Activity of Titania Films. Appl. Surf. Sci. 2002, 200, 239–247. [Google Scholar] [CrossRef]
- Rengaraj, S.; Li, X.Z. Enhanced Photocatalytic Activity of TiO2 by Doping with Ag for Degradation of 2,4,6-Trichlorophenol in Aqueous Suspension. J. Mol. Catal. A. Chem. 2006, 243, 60–67. [Google Scholar] [CrossRef]
- Li, Y.; Ding, H.; Mao, K.; Wang, D.; Wang, Q.; Zhang, W. Bactericidal Activity of Ag Nanoparticles Decorated TiO2 Microspheres and Effects of Water Composition and Extracellular Polymeric Substances. Clean Soil Air Water 2015, 43, 512–520. [Google Scholar] [CrossRef]
- Seery, M.K.; George, R.; Floris, P.; Pillai, S.C. Silver doped titanium dioxide nanomaterials for enhanced visible light photocatalysis. J. Photochem. Photobiol. A Chem. 2007, 189, 258–263. [Google Scholar] [CrossRef]
- Harikishore, M.; Sandhyarani, M.; Venkateswarlu, K.; Nellaippan, T.A.; Rameshbabu, N. Effect of Ag doping on antibacterial and photocatalytic activity of nanocrystalline TiO2. Procedia Mater. Sci. 2014, 6, 557–566. [Google Scholar] [CrossRef]
- Zhao, C.; Krall, A.; Zhao, H.; Zhang, Q.; Li, Y. Ultrasonic Spray Pyrolysis Synthesis of Ag/TiO2 Nanocomposite Photocatalysts for Simultaneous H2 Production and CO2 Reduction. Int. J. Hydrog. Energy 2012, 37, 9967–9976. [Google Scholar] [CrossRef]
- Logar, M.; Jančar, B.; Šturm, S.; Suvorov, D. Weak Polyion Multilayer-Assisted in Situ Synthesis as a Route Toward a Plasmonic Ag/TiO2 Photocatalyst. Langmuir 2010, 26, 12215–12224. [Google Scholar] [CrossRef] [PubMed]
- Xin, B.; Jing, L.; Ren, Z.; Wang, B.; Fu, H. Effects of Simultaneously Doped and Deposited Ag on the Photocatalytic Activity and Surface States of TiO2. J. Phys. Chem. B 2005, 109, 2805–2809. [Google Scholar] [CrossRef] [PubMed]
- Sobana, N.; Muruganadham, M.; Swaminathan, M. Nano-Ag Particles Doped TiO2 for Efficient Photodegradation of Direct Azo Dyes. J. Mol. Catal. A. Chem. 2006, 258, 124–132. [Google Scholar] [CrossRef]
- Rodríguez-González, V.; Morán-Pineda, F.M.; del Angel, P.; Vazquez-Cuchillo, O.; Gómez, R. Sol–Gel and Impregnated Prepared Silver TiO2 Semiconductors as Photocatalysts for the UV Decomposition of 2,4-D: A Comparative Study of the Preparation Method. J. Sol Gel Sci. Technol. 2011, 59, 57–62. [Google Scholar] [CrossRef]
- Zhao, Y.; Yang, B.; Xu, J.; Fu, Z.; Wu, M.; Li, F. Facile Synthesis of Ag Nanoparticles Supported on TiO2 Inverse Opal with Enhanced Visible-Light Photocatalytic Activity. Thin Solid Films 2012, 520, 3515–3522. [Google Scholar] [CrossRef]
- Li, H.; Lu, W.; Tian, J.; Luo, Y.; Asiri, A.M.; Al-Youbi, A.O.; Sun, X. Synthesis and Study of Plasmon-Induced Carrier Behavior at Ag/TiO2 Nanowires. Chem. Eur. J. 2012, 18, 8508–8514. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Dong, X.; Li, G.; Su, X.; Zhu, Z. Synthesis of C, Ag Co-Modified TiO2 Photocatalyst and Its Application in Waste Water Purification. Appl. Surf. Sci. 2013, 271, 276–283. [Google Scholar] [CrossRef]
- Lehman, J.T. Ecological and Nutritional Studies on Dinobryon Ehrenb: Seasonal Periodicity and the Phosphate Toxicity Problem. Limnol. Oceanogr. 1976, 21, 646–658. [Google Scholar] [CrossRef]
- Gorham, P.R.; McLachlan, J.; Hammer, U.T.; Kim, W.K. Isolation and Culture of Toxic Strains of Anabaena flos-aquae (Lyngb.) de Bréb. Int. Assoc. Theo. Appl. Limno 1964, 15, 796–804. [Google Scholar]
- McConnaughey, T.A.; Burdett, J.; Whelan, J.F.; Paull, C.K. Carbon Isotopes in Biological Carbonates: Respiration and Photosynthesis. Geochim. Cosmochim. Acta 1997, 61, 611–622. [Google Scholar] [CrossRef]
- APHA; AWWA; WPCF. Standard Methods for Examination of Water and Wastewater, 21th ed.; American Public Health Association: Washington, DC, USA, 2005. [Google Scholar]
- Maron, D.M.; Ames, B.N. Revised Methods for the Salmonella Mutagenicity Test. Mutat. Res. 1983, 113, 173–215. [Google Scholar] [CrossRef]
- Liang, Y.; Beardall, J.; Heraud, P. Effects of Nitrogen Source and UV Radiation on the Growth, Chlorophyll Fluorescence and Fatty Acid Composition of Phaeodactylum tricornutum and Chaetoceros muelleri (Bacillariophyceae). J. Photochem. Photobiol. B 2006, 82, 161–172. [Google Scholar] [CrossRef] [PubMed]
- Hong, J.; Otaki, M. Effect of Thin Film Photocatalysis on Light Penetration by Control of Algal and Photosynthetic Bacterial Adhesion. Environ. Eng. Res. 2003, 40, 573–578. [Google Scholar]
- Kumar, A.; Tyagi, M.B.; Singh, N.; Tyagi, R.; Jha, P.N.; Sinha, R.P.; Häder, D. Role of White Light in Reversing UV-B-Mediated Effects in the N2-Fixing Cyanobacterium Anabaena BT2. J. Photochem. Photobiol. B 2003, 71, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Andreasson, K.I.M.; Wängberg, S. Biological Weighting Functions as a Tool for Evaluating Two Ways to Measure UVB Radiation Inhibition on Photosynthesis. J. Photochem. Photobiol. B 2006, 84, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Falconer, I.R.; Yeung, D.S.K. Cytoskeletal Changes in Hepatocytes Induced by Microcystis Toxins and Their Relation to Hyperphosphorylation of Cell Proteins. Chem. Biol. Interact 1992, 81, 181–196. [Google Scholar] [CrossRef]
- Matsunaga, T.; Tomada, R.; Nakajima, Y.; Nakamura, N.; Komine, T. Continuous-Sterilization System That Uses Photosemiconductor Powders. Appl. Environ. Microbiol. 1988, 54, 1330–1333. [Google Scholar] [PubMed]
- Aliotta, G.; Greca, M.D.; Monaco, P.; Pinto, G.; Pollio, A.; Previtera, L. In Vitro Algal Growth Inhibition by Phytotoxins of Typha Latifolia. J. Chem. Ecol. 1990, 16, 2637–2646. [Google Scholar] [CrossRef] [PubMed]
- Schmitt, C.; Meysick, K.C.; O’Brien, A.D. Bacterial Toxins: Friends or Foes? Emerg. Infect. Dis. 1999, 5, 224–234. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chang, S.-Y.; Huang, W.-J.; Lu, B.-R.; Fang, G.-C.; Chen, Y.; Chen, H.-L.; Chang, M.-C.; Hsu, C.-F. An Environmentally Friendly Method for Testing Photocatalytic Inactivation of Cyanobacterial Propagation on a Hybrid Ag-TiO2 Photocatalyst under Solar Illumination. Int. J. Environ. Res. Public Health 2015, 12, 15819-15833. https://doi.org/10.3390/ijerph121215023
Chang S-Y, Huang W-J, Lu B-R, Fang G-C, Chen Y, Chen H-L, Chang M-C, Hsu C-F. An Environmentally Friendly Method for Testing Photocatalytic Inactivation of Cyanobacterial Propagation on a Hybrid Ag-TiO2 Photocatalyst under Solar Illumination. International Journal of Environmental Research and Public Health. 2015; 12(12):15819-15833. https://doi.org/10.3390/ijerph121215023
Chicago/Turabian StyleChang, Shu-Yu, Winn-Jung Huang, Ben-Ren Lu, Guor-Cheng Fang, Yeah Chen, Hsiu-Lin Chen, Ming-Chin Chang, and Cheng-Feng Hsu. 2015. "An Environmentally Friendly Method for Testing Photocatalytic Inactivation of Cyanobacterial Propagation on a Hybrid Ag-TiO2 Photocatalyst under Solar Illumination" International Journal of Environmental Research and Public Health 12, no. 12: 15819-15833. https://doi.org/10.3390/ijerph121215023





