Autonomic Nervous System Responses to Viewing Green and Built Settings: Differentiating Between Sympathetic and Parasympathetic Activity
Abstract
:1. Introduction
The Present Research
- (i)
- compared to viewing built scenes, viewing green scenes prior to exposure to a stressor buffers against stress, indicated by a smaller stress response during the stressor (smaller decrease in PEP and/or RSA);
- (ii)
- compared to viewing built scenes, viewing green scenes after exposure to a stressor supports recovery from stress, indicated by a larger post-stressor recovery response (larger increase in PEP and/or RSA).
2. Method
2.1. Participants
2.2. Stimuli

2.3. Stress Task
2.4. Design

2.5. Measures
2.5.1. Autonomic Response Measures: RSA and PEP
2.5.2. Restoration Outcome Scale
2.6. Procedure
2.7. Analysis
3. Results
3.1. Perceived Restoration Outcomes
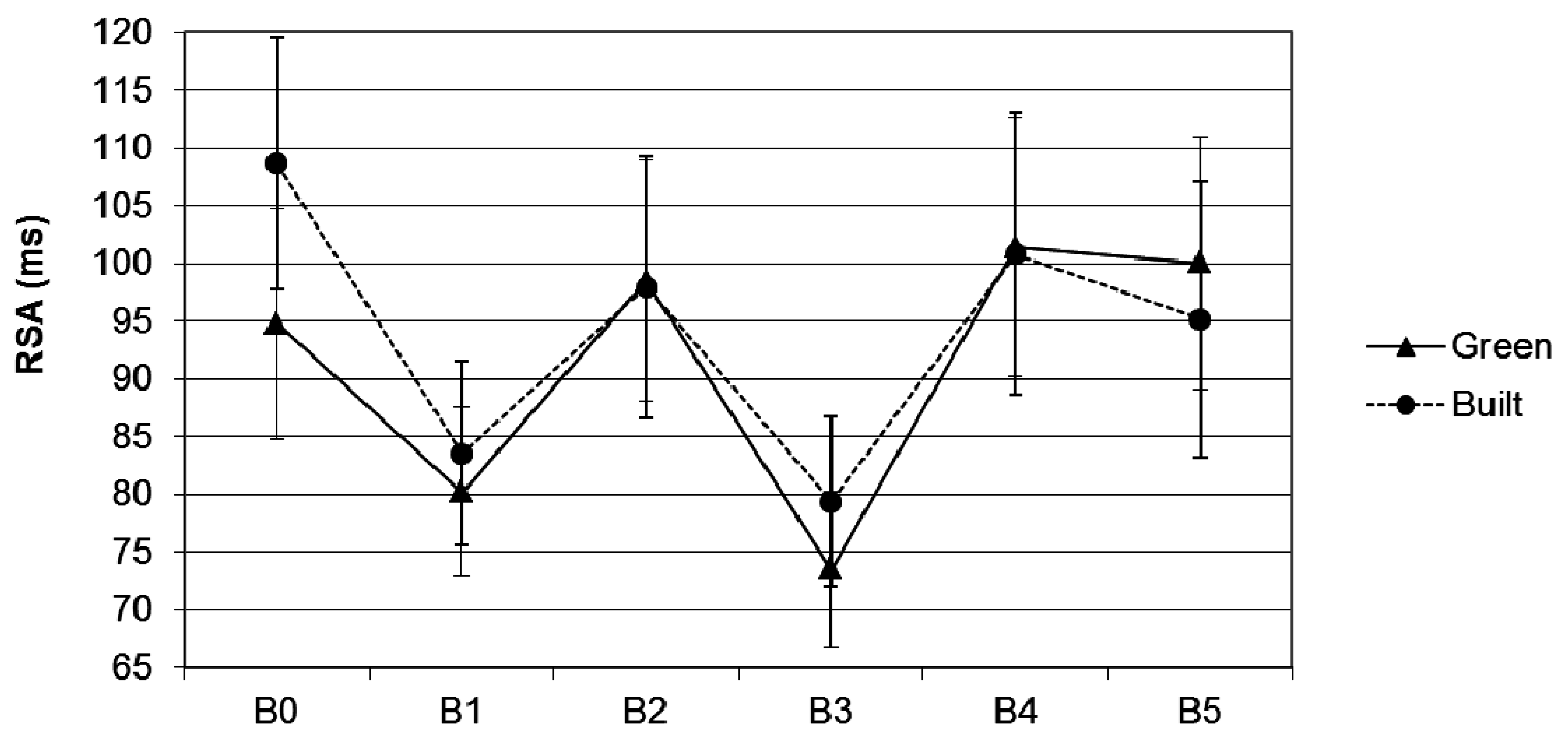
3.2. RSA
3.2.1. Effects of Scene Type on RSA
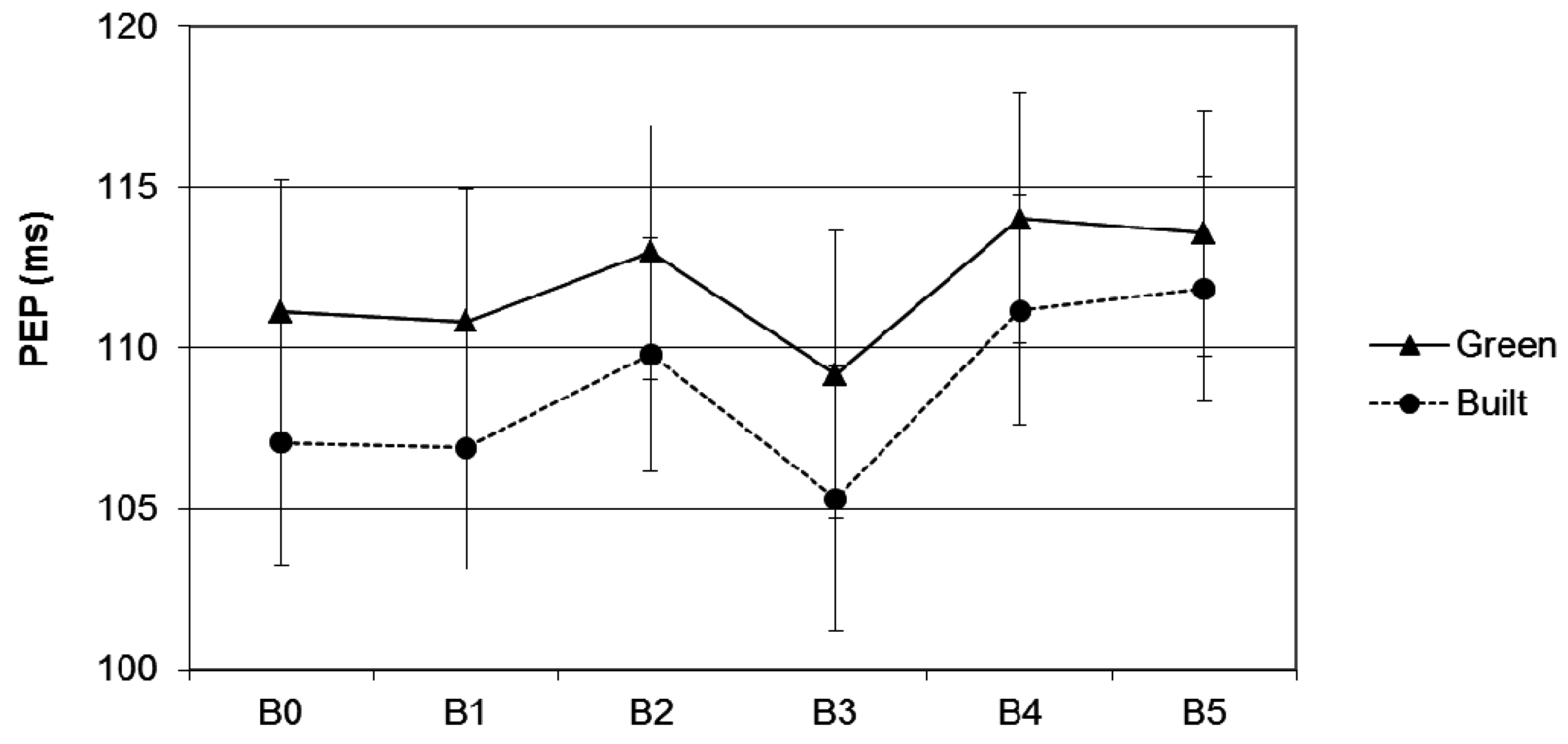
3.3. PEP
3.3.1. Effects of Scene Type on PEP
4. Discussion
4.1. Strengths and Limitations
4.2. Suggestions for Future Research
5. Conclusions
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Krabbendam, L.; van Os, J. Schizophrenia and urbanicity: A major environmental influence—Conditional on genetic risk. Schizophr. Bull. 2005, 31, 795–799. [Google Scholar] [CrossRef] [PubMed]
- Peen, J.; Schoevers, R.; Beekman, A.; Dekker, J. The current status of urban-rural differences in psychiatric disorders. Acta Psychiatr. Scand. 2010, 121, 84–93. [Google Scholar] [CrossRef] [PubMed]
- Van Os, J.; Kenis, G.; Rutten, B.P. The environment and schizophrenia. Nature 2010, 468, 203–212. [Google Scholar] [CrossRef] [PubMed]
- Evans, G.W. The built environment and mental health. J. Urban Health 2003, 80, 536–555. [Google Scholar] [CrossRef] [PubMed]
- Van den Berg, A.E.; Hartig, T.; Staats, H. Preference for nature in urbanized societies: Stress, restoration, and the pursuit of sustainability. J. Soc. Issues 2007, 63, 79–96. [Google Scholar] [CrossRef]
- Health Council of the Netherlands and Dutch Advisory Council for Research on Spatial Planning Nature and the Environment. Nature and Health. The Influence of Nature on Social, Psychological and Physical Well-Being 2004/09; Health Council of the Netherlands: The Hague, the Netherlands, 2004. [Google Scholar]
- Joye, J.; Van den Berg, A.E. Restorative environments. In Environmental Psychology: An Introduction; Steg, E.M., van den Berg, A.E., de Groot, J.I.M., Eds.; Wiley-Blackwell: London, UK, 2012; pp. 57–66. [Google Scholar]
- Barton, J.; Pretty, J. What is the best dose of nature and green exercise for improving mental health? A multi-study analysis. Environ. Sci. Technol. 2010, 44, 3947–3955. [Google Scholar] [CrossRef] [PubMed]
- Bowler, D.; Buyung-Ali, L.; Knight, T.; Pullin, A. A systematic review of evidence for the added benefits to health of exposure to natural environments. BMC Public Health 2010, 10. [Google Scholar] [CrossRef] [PubMed]
- Thompson Coon, J.; Boddy, K.; Stein, K.; Whear, R.; Barton, J.; Depledge, M.H. Does participating in physical activity in outdoor natural environments have a greater effect on physical and mental wellbeing than physical activity indoors? A systematic review. Environ. Sci. Technol. 2011, 45, 1761–1772. [Google Scholar] [CrossRef] [PubMed]
- Berto, R. The role of nature in coping with psycho-physiological stress: A literature review on restorativeness. Behav. Sci. 2014, 4, 394–409. [Google Scholar] [CrossRef] [PubMed]
- Hartig, T.; Mitchell, R.; de Vries, S.; Frumkin, H. Nature and Health. Annu. Rev. Public Health 2014, 35, 207–228. [Google Scholar] [CrossRef] [PubMed]
- Van den Berg, A.E.; Koole, S.L.; van der Wulp, N.Y. Environmental preference and restoration: (How) are they related? J. Environ. Psychol. 2003, 23, 135–146. [Google Scholar] [CrossRef]
- Hartig, T.; Evans, G.W.; Jamner, L.D.; Davis, D.S.; Gärling, T. Tracking restoration in natural and urban field settings. J. Environ. Psychol. 2003, 23, 109–123. [Google Scholar] [CrossRef]
- Park, B.; Tsunetsugu, Y.; Kasetani, T.; Kagawa, T.; Miyazaki, Y. The physiological effects of Shinrin-yoku (taking in the forest atmosphere or forest bathing): Evidence from field experiments in 24 forests across Japan. Environ. Health Prev. Med. 2010, 15, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Van den Berg, A.E.; Custers, M.H.G. Gardening promotes neuroendocrine and affective restoration from stress. J. Health Psychol. 2011, 16, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Roe, J.J.; Thompson, C.W.; Aspinall, P.A.; Brewer, M.J.; Duff, E.I.; Miller, D.; Mitchell, R.; Clow, A. Green space and stress: Evidence from cortisol measures in deprived urban communities. Int. J. Environ. Res. Public. Health 2013, 10, 4086–4103. [Google Scholar] [CrossRef] [PubMed]
- Parsons, R.; Tassinary, L.G.; Ulrich, R.S.; Hebl, M.R.; Grossman-Alexander, M. The view from the road: Implications for stress recovery and immunization. J. Environ. Psychol. 1998, 18, 113–140. [Google Scholar] [CrossRef]
- Van den Berg, A.E.; Maas, J.; Verheij, R.A.; Groenewegen, P.P. Green space as a buffer between stressful life events and health. Soc. Sci. Med. 2010, 70, 1203–1210. [Google Scholar] [CrossRef] [PubMed]
- Sapolsky, R.M. Why Zebras Don’t Get Ulcers: A Guide to Stress, Stress-Related Disorders and Coping, 2nd ed.; Freeman: New York, NY, USA, 1998. [Google Scholar]
- Porges, S.W. The polyvagal perspective. Biol. Psychol. 2007, 74, 116–143. [Google Scholar] [CrossRef] [PubMed]
- Thayer, J.F.; Åhs, F.; Fredrikson, M.; Sollers, J.J.; Wager, T.D. A meta-analysis of heart rate variability and neuroimaging studies: Implications for heart rate variability as a marker of stress and health. Neurosci. Biobehav. Rev. 2012, 36, 747–756. [Google Scholar] [CrossRef] [PubMed]
- Buccelletti, E.; Gilardi, E.; Scaini, E.; Galiuto, L.; Persiani, R.; Biondi, A.; Basile, F.; Silveri, N.G. Heart rate variability and myocardial infarction: Systematic literature review and metanalysis. Eur. Rev. Med. Pharmacol. Sci. 2009, 13, 299–307. [Google Scholar] [PubMed]
- Bazhenova, O.V.; Plonskaia, O.; Porges, S.W. Vagal reactivity and affective adjustment in infants during interaction challenges. Child Dev. 2001, 72, 1314–1326. [Google Scholar] [CrossRef] [PubMed]
- Gramzow, R.H.; Willard, G.; Mendes, W.B. Big tales and cool heads: academic exaggeration is related to cardiac vagal reactivity. Emotion 2008, 8, 138–144. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.K.; Barton, J.L.; Gladwell, V.F. Viewing nature scenes positively affects recovery of autonomic function following acute-mental stress. Environ. Sci. Technol. 2013, 47, 5562–5569. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tsunetsugu, Y.; Park, B.; Ishii, H.; Hirano, H.; Kagawa, T.; Miyazaki, Y. Physiological effects of Shinrin-yoku (taking in the atmosphere of the forest) in an old-growth broadleaf forest in Yamagata Prefecture, Japan. J. Physiol. Anthropol. 2007, 26, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Ulrich, R.S.; Simons, R.F.; Losito, B.D.; Fiorito, E.; Miles, M.A.; Zelson, M. Stress recovery during exposure to natural and urban environments. J. Environ. Psychol. 1991, 11, 201–230. [Google Scholar] [CrossRef]
- Laumann, K.; Garling, T.; Stormark, K. Selective attention and heart rate responses to natural and urban environments. J. Environ. Psychol. 2003, 23, 125–134. [Google Scholar] [CrossRef]
- Gladwell, V.; Brown, D.; Barton, J.; Tarvainen, M.; Kuoppa, P.; Pretty, J.; Suddaby, J.; Sandercock, G.R. The effects of views of nature on autonomic control. Eur. J. Appl. Physiol. 2012, 112, 3379–3386. [Google Scholar] [CrossRef] [PubMed]
- Alvarsson, J.J.; Wiens, S.; Nilsson, M.E. Stress recovery during exposure to nature sound and environmental noise. Int. J. Environ. Res. Public. Health 2010, 7, 1036–1046. [Google Scholar] [CrossRef] [PubMed]
- Schächinger, H.; Weinbacher, M.; Kiss, A.; Ritz, R.; Langewitz, W. Cardiovascular indices of peripheral and central sympathetic activation. Psychosom. Med. 2001, 63, 788–796. [Google Scholar] [CrossRef] [PubMed]
- Berntson, G.G.; Bigger, J.T.; Eckberg, D.L.; Grossman, P.; Kaufmann, P.G.; Malik, M.; Nagaraja, H.N.; Porges, S.W.; Saul, J.P.; Stone, P.H. Heart rate variability: origins, methods, and interpretive caveats. Psychophysiology 1997, 34, 623–648. [Google Scholar] [CrossRef] [PubMed]
- De Geus, E.; Van Lien, R.; Neijts, M.; Willemsen, G. Genetics of autonomic nervous system activity. In The Oxford Handbook of Molecular Psychology; Canli, T., Ed.; Oxford University Press: Oxford, UK, 2014; pp. 357–390. [Google Scholar]
- Peschardt, K.K.; Stigsdotter, U.K. Associations between park characteristics and perceived restorativeness of small public urban green spaces. Landsc. Urban Plan. 2013, 112, 26–39. [Google Scholar] [CrossRef]
- White, M.; Smith, A.; Humphryes, K.; Pahl, S.; Snelling, D.; Depledge, M. Blue space: The importance of water for preference, affect, and restorativeness ratings of natural and built scenes. J. Environ. Psychol. 2010, 30, 482–493. [Google Scholar] [CrossRef]
- Herzog, T.R.; Maguire, C.P.; Nebel, M.B. Assessing the restorative components of environments. J. Environ. Psychol. 2003, 23, 159–170. [Google Scholar] [CrossRef]
- Dedovic, K.; Duchesne, A.; Andrews, J.; Engert, V.; Pruessner, J.C. The brain and the stress axis: The neural correlates of cortisol regulation in response to stress. Neuroimage 2009, 47, 864–871. [Google Scholar] [CrossRef] [PubMed]
- Jones, A.; Steeden, J.A.; Pruessner, J.C.; Deanfield, J.E.; Taylor, A.M.; Muthurangu, V. Detailed assessment of the hemodynamic response to psychosocial stress using real-time MRI. J. Magn. Reson. Imaging 2011, 33, 448–454. [Google Scholar] [CrossRef] [PubMed]
- Hernaus, D.; Collip, D.; Lataster, J.; Ceccarini, J.; Kenis, G.; Booij, L.; Pruessner, J.; van Laere, K.; van Winkel, R.; van Os, J. COMT Val158Met genotype selectively alters prefrontal [18F] fallypride displacement and subjective feelings of stress in response to a psychosocial stress challenge. PLoS ONE 2013, 8. [Google Scholar] [CrossRef]
- VU-AMS Ambulatory Monitoring System. Available online: www.vu-ams.nl (accessed on 9 April 2013).
- Willemsen, G.H.; DeGeus, E.J.; Klaver, C.H.; VanDoornen, L.J.; Carrofl, D. Ambulatory monitoring of the impedance cardiogram. Psychophysiology 1996, 33, 184–193. [Google Scholar] [CrossRef] [PubMed]
- Grossman, P. Respiration, stress, and cardiovascular function. Psychophysiology 1983, 20, 284–300. [Google Scholar] [CrossRef] [PubMed]
- Katona, P.G.; Jih, F. Respiratory sinus arrhythmia: Noninvasive measure of parasympathetic cardiac control. J. Appl. Physiol. 1975, 39, 801–805. [Google Scholar] [PubMed]
- Porges, S.W.; Doussard-Roosevelt, J.A.; Portales, A.L.; Greenspan, S.I. Infant regulation of the vagal “brake” predicts child behavior problems: A psychobiological model of social behavior. Dev. Psychobiol. 1996, 29, 697–712. [Google Scholar] [CrossRef]
- Van Lien, R.; Schutte, N.M.; Meijer, J.H.; de Geus, E.J. Estimated preejection period (PEP) based on the detection of the R-wave and dZ/dt-min peaks does not adequately reflect the actual PEP across a wide range of laboratory and ambulatory conditions. Int. J. Psychophysiol. 2013, 87, 60–69. [Google Scholar] [CrossRef] [PubMed]
- Riese, H.; Groot, P.F.; van den Berg, M.; Kupper, N.H.; Magnee, E.H.; Rohaan, E.J.; Vrijkotte, T.G.; Willemsen, G.; de Geus, E.J. Large-scale ensemble averaging of ambulatory impedance cardiograms. Behav. Res. Meth. Instrum. Comput. 2003, 35, 467–477. [Google Scholar] [CrossRef]
- Korpela, K.M.; Ylén, M.; Tyrväinen, L.; Silvennoinen, H. Determinants of restorative experiences in everyday favorite places. Health Place 2008, 14, 636–652. [Google Scholar] [CrossRef] [PubMed]
- Korpela, K.M.; Ylén, M.; Tyrväinen, L.; Silvennoinen, H. Favorite green, waterside and urban environments, restorative experiences and perceived health in Finland. Health Promot. Int. 2010, 25, 200–209. [Google Scholar] [CrossRef] [PubMed]
- Houtveen, J.H.; de Geus, E.J. Noninvasive psychophysiological ambulatory recordings: Study design and data analysis strategies. Eur. Psychol. 2009, 14, 132–141. [Google Scholar] [CrossRef]
- Bratman, G.N.; Hamilton, J.P.; Daily, G.C. The impacts of nature experience on human cognitive function and mental health. Ann. N. Y. Acad. Sci. 2012, 1249, 118–136. [Google Scholar] [CrossRef] [PubMed]
- Task Force of the European Society of Cardiology. Heart rate variability standards of measurement, physiological interpretation, and clinical use. Eur. Heart J. 1996, 17, 354–381. [Google Scholar]
- Li, Q.; Otsuka, T.; Kobayashi, M.; Wakayama, Y.; Inagaki, H.; Katsumata, M.; Hirata, Y.; Li, Y.; Hirata, K.; Shimizu, T. Acute effects of walking in forest environments on cardiovascular and metabolic parameters. Eur. J. Appl. Physiol. 2011, 111, 2845–2853. [Google Scholar] [CrossRef] [PubMed]
- Annerstedt, M.; Jönsson, P.; Wallergård, M.; Johansson, G.; Karlson, B.; Grahn, P.; Hansen, Å.M.; Währborg, P. Inducing physiological stress recovery with sounds of nature in a virtual reality forest—Results from a pilot study. Physiol. Behav. 2013, 118, 240–250. [Google Scholar] [CrossRef] [PubMed]
- Van den Berg, A.E.; Jorgensen, A.; Wilson, E.R. Evaluating restoration in urban green spaces: Does setting type make a difference? Landscape Urban Plan. 2014, 127, 173–181. [Google Scholar] [CrossRef]
- Van den Berg, M.; Wendel-Vos, W.; van Poppel, M.; Kemper, H.; van Mechelen, W.; Maas, J. Health benefits of green spaces in the living environment: A systematic review of epidemiological studies. Urban For. Urban Gree. 2015, 14, 806–816. [Google Scholar] [CrossRef]
- Kaplan, R. The nature of the view from home psychological benefits. Environ. Behav. 2001, 33, 507–542. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Van den Berg, M.M.H.E.; Maas, J.; Muller, R.; Braun, A.; Kaandorp, W.; Van Lien, R.; Van Poppel, M.N.M.; Van Mechelen, W.; Van den Berg, A.E. Autonomic Nervous System Responses to Viewing Green and Built Settings: Differentiating Between Sympathetic and Parasympathetic Activity. Int. J. Environ. Res. Public Health 2015, 12, 15860-15874. https://doi.org/10.3390/ijerph121215026
Van den Berg MMHE, Maas J, Muller R, Braun A, Kaandorp W, Van Lien R, Van Poppel MNM, Van Mechelen W, Van den Berg AE. Autonomic Nervous System Responses to Viewing Green and Built Settings: Differentiating Between Sympathetic and Parasympathetic Activity. International Journal of Environmental Research and Public Health. 2015; 12(12):15860-15874. https://doi.org/10.3390/ijerph121215026
Chicago/Turabian StyleVan den Berg, Magdalena M.H.E., Jolanda Maas, Rianne Muller, Anoek Braun, Wendy Kaandorp, René Van Lien, Mireille N.M. Van Poppel, Willem Van Mechelen, and Agnes E. Van den Berg. 2015. "Autonomic Nervous System Responses to Viewing Green and Built Settings: Differentiating Between Sympathetic and Parasympathetic Activity" International Journal of Environmental Research and Public Health 12, no. 12: 15860-15874. https://doi.org/10.3390/ijerph121215026





