Effect of Smoking Abstinence and Reduction in Asthmatic Smokers Switching to Electronic Cigarettes: Evidence for Harm Reversal
Abstract
:1. Introduction
2. Methods
2.1. Patient Population
2.2. Study Assessments
2.3. Analyses
3. Results
3.1. Characteristics of the Patients and of Their E-cig Use
| Parameter | Pre-baseline | Baseline | ||
|---|---|---|---|---|
| All Subjects (n = 18) | All Subjects (n = 18) | Single Users (n = 10) | Dual Users (n = 8) | |
| Gender | 11M, 7F | 11M, 7F | 7M, 3F | 4M, 4F |
| Age | 37.8 (±12.3) | 38.8 (±12.3) | 36.1 (±13.5) | 42.3 (±10.6) |
| Asthma Duration | 18.7 (±6.3) | 19.7 (±6.3) | 18.6 (±6.1) | 21.0 (±6.8) |
| Conventional Cigarettes/Day | 23.2 (±5.1) | 21.9 (±4.5) | 21.6 (±3.9) | 22.4 (±5.3) |
| Smoking Pack Years | 20.1 (±9.9) | 21.0 (±10.7) | 16.5 (±8.4) | 26.6 (±11.2) |
| No. Exacerbations (from the previous follow up visit) | 1.06 (±1.0) | 1.17 (±0.9) | 1.20 (±0.8) | 1.13 (±1.0) |
| FEV1 (L) | 3.35 (±0.76) | 3.30 (±0.78) | 3.42 (±0.84) | 3.16 (±0.73) |
| FVC (L) | 4.33 (±0.85) | 4.28 (±0.90) | 4.35 (±0.96) | 4.19 (±0.88) |
| FEV1/FVC (%) | 77.3 (±5.53) | 76.8 (±4.52) | 78.3 (±4.59) | 75.0 (±3.98) |
| FEF25-75% (L/sec.) | 2.86 (±0.69) | 2.75 (±0.72) | 2.95 (±0.53) | 2.49 (±0.88) |
| ACQ | 2.07 (±0.36) | 2.03 (±0.37) | 2.12 (±0.42) | 1.93 (±0.29) |
| PC20 (mg/mL) * | 1.31 (0.55, 1.75) | 1.24 (0.49, 3.27) | 1.10 (0.49, 2.07) | 1.40 (0.82, 3.27) |
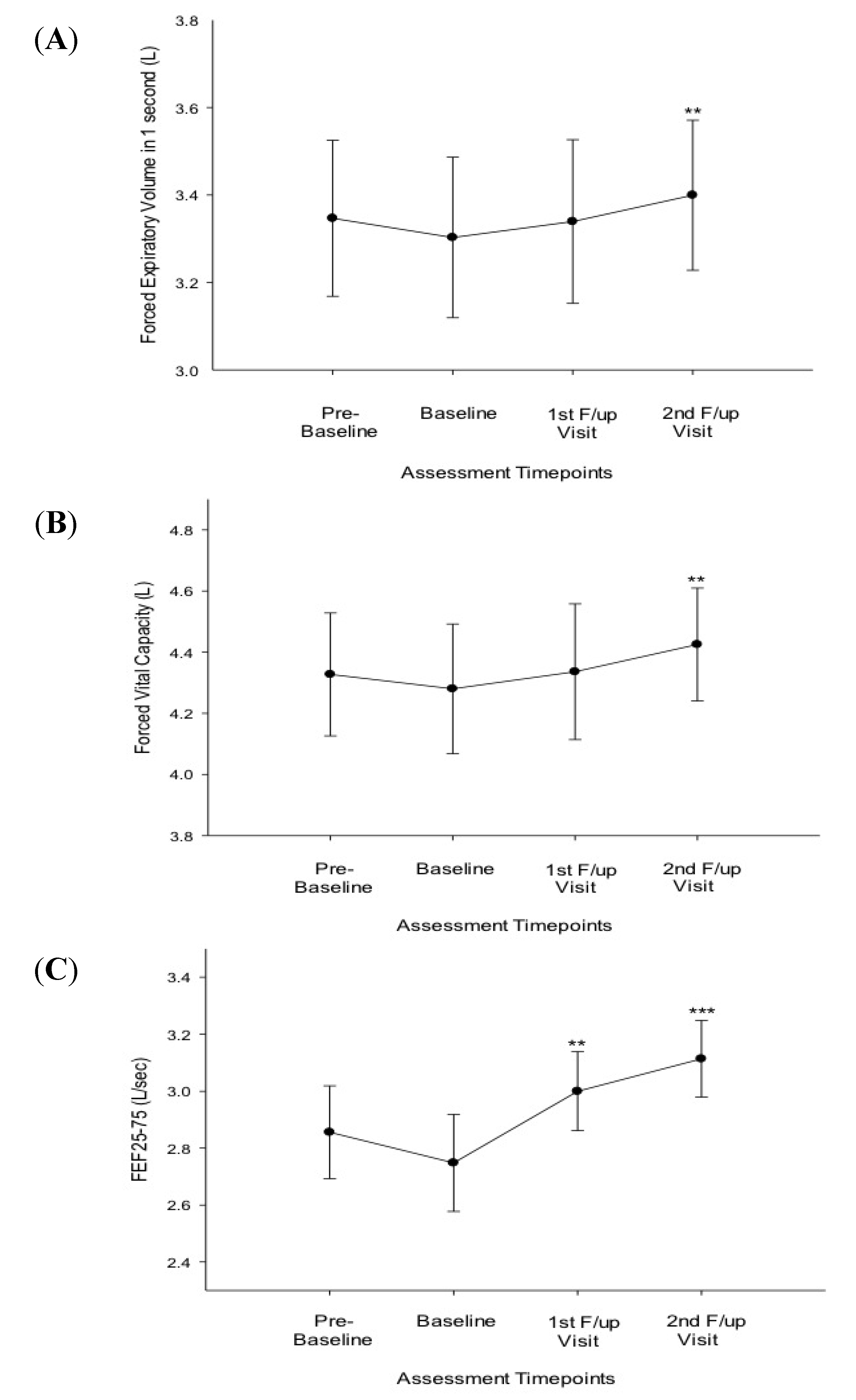
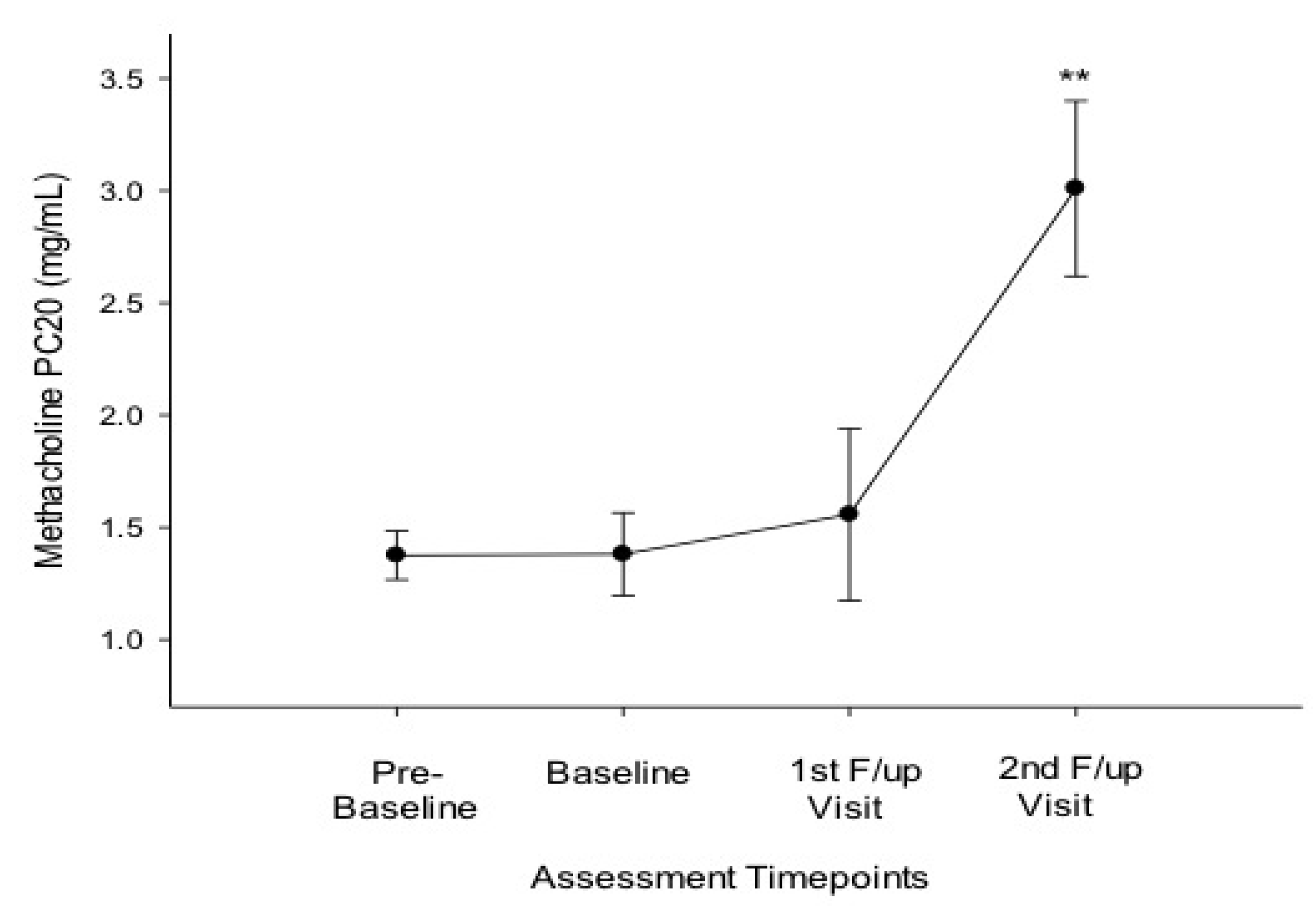
3.2. Efficacy of Smoking Reduction in Lung Function, BHR and ACQ Scores
3.3. Conventional Smoking
3.4. Exacerbations
| Parameter | Baseline | 1st Follow-up Visit (6 months ± 1) | 2nd Follow-up Visit (12 months ± 2) | ||
|---|---|---|---|---|---|
| p value to Baseline | p value to Baseline | ||||
| All patients (n = 18; 11M, 7F) | |||||
| FEV1 (L) | 3.30 (±0.78) | 3.34 (±0.72) | 0.078 | 3.40 (±0.73) | 0.005 |
| FVC (L) | 4.28 (±0.90) | 4.34 (±0.86) | 0.105 | 4.43 (±0.78) | 0.006 |
| FEF25-75% (L/sec.) | 2.75 (±0.72) | 3.00 (±0.54) | 0.006 | 3.11 (±0.57) | 0.001 |
| ACQ | 2.03 (±0.37) | 1.60 (±0.24) | <0.001 | 1.47 (±0.20) | <0.001 |
| PC20 (mg/mL) * | 1.24 (0.49, 3.27) | 1.20 (0.44, 4.23) | 0.594 | 2.56 (0.5, 5.55) | 0.003 |
| Cigarettes/day | 21.9 (±4.5) | 1.9 (±2.9) | <0.001 | 1.7 (±2.1) | <0.001 |
| Exacerbations | 1.17 (±0.9) | 0.87 (±0.7) | 0.296 | 0.78 (±0.7) | 0.153 |
| Single Users (n = 10; 7M, 3F) | |||||
| FEV1 (L) | 3.42 (±0.84) | 3.49 (±0.75) | 0.779 | 3.51 (±0.75) | 0.032 |
| FVC (L) | 4.35 (±0.96) | 4.52 (±0.86) | 0.161 | 4.51 (0.81) | 0.059 |
| FEF25-75% (L/sec.) | 2.95 (±0.53) | 3.17 (±0.39) | 0.093 | 3.20 (±0.54) | 0.047 |
| ACQ | 2.12 (±0.42) | 1.69 (±0.29) | 0.024 | 1.50 (±0.19) | <0.001 |
| PC20 (mg/mL) * | 1.10 (0.49, 2.07) | 1.14 (0.44, 3.55) | 0.463 | 2.06 (0.5, 5.55) | 0.043 |
| Cigarettes/day | 21.6 (±3.9) | - | - | - | - |
| Exacerbations | 1.20 (±0.8) | 1.13 (±0.6) | 0.831 | 1.10 (±0.7) | 0.773 |
| Dual Users (n = 8; 4M, 4F) | |||||
| FEV1 (L) | 3.16 (±0.73) | 3.17 (±0.71) | 0.062 | 3.26 (±0.72) | 0.05 |
| FVC (L) | 4.19 (±0.88) | 4.13 (±0.88) | 0.352 | 4.32 (±0.78) | 0.05 |
| FEF25-75% (L/sec.) | 2.49 (±0.88) | 2.81 (±0.64) | 0.028 | 3.01 (±0.62) | 0.012 |
| ACQ | 1.93 (±0.29) | 1.50 (±0.15) | 0.004 | 1.43 (±0.21) | 0.002 |
| PC20 (mg/mL) * | 1.40 (0.82, 3.27) | 1.28 (0.72, 4.23) | 0.893 | 3.17 (2.39, 4.09) | 0.028 |
| Cigarettes/day | 22.4 (±5.3) | 5.0 (±2.6) | <0.001 | 3.9 (±1.0) | <0.001 |
| Exacerbations | 1.13 (±1.0) | 0.57 (±0.8) | 0.257 | 0.38 (±0.5) | 0.078 |

3.5. Safety and Tolerability
4. Discussion
5. Conclusions
Acknowledgements
Author Contributions
Conflicts of Interest
References
- To, T.; Stanojevic, S.; Moores, G.; Gershon, A.S.; Bateman, E.D.; Cruz, A.A.; Boulet, L.P. Global asthma prevalence in adults: Findings from the cross-sectional world health survey. BMC Public Health 2012, 12. [Google Scholar] [CrossRef]
- Polosa, R.; Knoke, J.D.; Russo, C.; Piccillo, G.; Caponnetto, P.; Sarva, M.; Proietti, L.; Al-Delaimy, W.K. Cigarette smoking is associated with a greater risk of incident asthma in allergic rhinitis. J. Allerg. Clin. Immunol. 2008, 121, 1428–1434. [Google Scholar] [CrossRef]
- Polosa, R.; Thomson, N.C. Smoking and asthma: Dangerous liaisons. Eur. Respir. J. 2013, 41, 716–726. [Google Scholar] [CrossRef]
- Polosa, R.; Russo, C.; Caponnetto, P.; Bertino, G.; Sarva, M.; Antic, T.; Mancuso, S.; Al-Delaimy, W.K. Greater severity of new onset asthma in allergic subjects who smoke: A 10-year longitudinal study. Respir. Res. 2011, 12. [Google Scholar] [CrossRef]
- Lange, P.; Parner, J.; Vestbo, J.; Schnohr, P.; Jensen, G. A 15-year follow-up study of ventilatory function in adults with asthma. N. Engl. J. Med. 1998, 339, 1194–1200. [Google Scholar] [CrossRef]
- Tomlinson, J.E.; McMahon, A.D.; Chaudhuri, R.; Thompson, J.M.; Wood, S.F.; Thomson, N.C. Efficacy of low and high dose inhaled corticosteroid in smokers versus non-smokers with mild asthma. Thorax 2005, 60, 282–287. [Google Scholar] [CrossRef]
- Chaudhuri, R.; Livingston, E.; McMahon, A.D.; Thomson, L.; Borland, W.; Thomson, N.C. Cigarette smoking impairs the therapeutic response to oral corticosteroids in chronic asthma. Amer. J. Respir. Crit. Care Med. 2003, 168, 1308–1311. [Google Scholar] [CrossRef]
- Tonnesen, P.; Pisinger, C.; Hvidberg, S.; Wennike, P.; Bremann, L.; Westin, A.; Thomsen, C.; Nilsson, F. Effects of smoking cessation and reduction in asthmatics. Nicotine Tob. Res. 2005, 7, 139–148. [Google Scholar] [CrossRef]
- Chaudhuri, R.; Livingston, E.; McMahon, A.D.; Lafferty, J.; Fraser, I.; Spears, M.; McSharry, C.P.; Thomson, N.C. Effects of smoking cessation on lung function and airway inflammation in smokers with asthma. Amer. J. Respir. Crit. Care Med. 2006, 174, 127–133. [Google Scholar] [CrossRef]
- Eisner, M.D.; Yelin, E.H.; Katz, P.P.; Shiboski, S.C.; Henke, J.; Blanc, P.D. Predictors of cigarette smoking and smoking cessation among adults with asthma. Amer. J. Public Health 2000, 90, 1307–1311. [Google Scholar] [CrossRef]
- Wakefield, M.; Ruffin, R.; Campbell, D.; Roberts, L.; Wilson, D. Smoking-related beliefs and behaviour among adults with asthma in a representative population sample. Aust. N. Z. J. Med. 1995, 25, 12–17. [Google Scholar] [CrossRef]
- Caponnetto, P.; Russo, C.; Bruno, C.M.; Alamo, A.; Amaradio, M.D.; Polosa, R. Electronic cigarette: A possible substitute for cigarette dependence. Monaldi Arch. Chest Dis. 2013, 79, 12–19. [Google Scholar]
- Polosa, R.; Rodu, B.; Caponnetto, P.; Maglia, M.; Raciti, C. A fresh look at tobacco harm reduction: The case for the electronic cigarette. Harm Reduct. J. 2013, 10. [Google Scholar] [CrossRef]
- Caponnetto, P.; Campagna, D.; Cibella, F.; Morjaria, J.B.; Caruso, M.; Russo, C.; Polosa, R. EffiCiency and Safety of an eLectronic cigAreTte (ECLAT) as tobacco cigarettes substitute: A prospective 12-month randomized control design study. PLoS One 2013, 8. [Google Scholar] [CrossRef]
- Bullen, C.; Howe, C.; Laugesen, M.; McRobbie, H.; Parag, V.; Williman, J.; Walker, N. Electronic cigarettes for smoking cessation: A randomised controlled trial. Lancet 2013, 382, 1629–1637. [Google Scholar] [CrossRef]
- Global Initiative for Asthma (GINA). From the Global Strategy for Asthma Management and Prevention. 2012. Available online: http://www.ginasthma.org/ (accessed on 17 February 2014).
- Juniper, E.F.; Svensson, K.; Mork, A.-C.; Stahl, E. Measurement properties and interpretation of three shortened versions of the asthma control questionnaire. Respir. Med. 2005, 99, 553–558. [Google Scholar] [CrossRef]
- Caponnetto, P.; Cibella, F.; Mancuso, S.; Campagna, D.; Arcidiacono, G.; Polosa, R. Effect of a nicotine-free inhalator as part of a smoking-cessation programme. Eur. Respir. J. 2011, 38, 1005–1011. [Google Scholar] [CrossRef]
- Chalmers, G.W.; MacLeod, K.J.; Thomson, L.; Little, S.A.; McSharry, C.; Thomson, N.C. Smoking and airway inflammation in patients with mild asthma. Chest 2001, 120, 1917–1922. [Google Scholar]
- Pedersen, B.; Dahl, R.; Karlstrom, R.; Peterson, C.G.; Venge, P. Eosinophil and neutrophil activity in asthma in a one-year trial with inhaled budesonide: The impact of smoking. Amer. J. Respir. Crit. Care Med. 1996, 153, 1519–1529. [Google Scholar] [CrossRef]
- Lazarus, S.C.; Chinchilli, V.M.; Rollings, N.J.; Boushey, H.A.; Cherniack, R.; Craig, T.J.; Deykin, A.; di Mango, E.; Fish, J.E.; Ford, J.G.; et al. Smoking affects response to inhaled corticosteroids or leukotriene receptor antagonists in asthma. Amer. J. Respir. Crit. Care Med. 2007, 175, 783–790. [Google Scholar] [CrossRef]
- Piccillo, G.; Caponnetto, P.; Barton, S.; Russo, C.; Origlio, A.; Bonaccorsi, A.; Maria, A.D.; Oliveri, C.; Polosa, R. Changes in airway hyperresponsiveness following smoking cessation: Comparisons between Mch and AMP. Respir. Med. 2008, 102, 256–265. [Google Scholar] [CrossRef]
- Sparrow, D.; O’Connor, G.; Colton, T.; Barry, C.L.; Weiss, S.T. The relationship of nonspecific bronchial responsiveness to the occurrence of respiratory symptoms and decreased levels of pulmonary function: The normative aging study. Amer. Rev. Respir. Dis. 1987, 135, 1255–1260. [Google Scholar]
- Tashkin, D.P.; Altose, M.D.; Connett, J.E.; Kanner, R.E.; Lee, W.W.; Wise, R.A. Methacholine reactivity predicts changes in lung function over time in smokers with early chronic obstructive pulmonary disease. The lung health study research group. Amer. J. Respir. Crit. Care Med. 1996, 153, 1802–1811. [Google Scholar] [CrossRef]
- Laforest, L.; van Ganse, E.; Devouassoux, G.; Bousquet, J.; Chretin, S.; Bauguil, G.; Pacheco, Y.; Chamba, G. Influence of patients’ characteristics and disease management on asthma control. J. Allerg. Clin. Immunol. 2006, 117, 1404–1410. [Google Scholar]
- Schatz, M.; Zeiger, R.S.; Vollmer, W.M.; Mosen, D.; Cook, E.F. Determinants of future long-term asthma control. J. Allerg. Clin. Immunol. 2006, 118, 1048–1053. [Google Scholar] [CrossRef]
- Chaudhuri, R.; McSharry, C.; McCoard, A.; Livingston, E.; Hothersall, E.; Spears, M.; Lafferty, J.; Thomson, N.C. Role of symptoms and lung function in determining asthma control in smokers with asthma. Allergy 2008, 63, 132–135. [Google Scholar]
- Farsalinos, K.E.; Polosa, R. Safety evaluation and risk assessment of electronic cigarettes as tobacco cigarettes substitutes: A systematic review. Ther. Adv. Drug Safety 2014, 5, 67–86. [Google Scholar] [CrossRef]
- Wong, S.L.; Shields, M.; Leatherdale, S.; Malaison, E.; Hammond, D. Assessment of validity of self-reported smoking status. Health Rep. 2012, 23, 47–53. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Polosa, R.; Morjaria, J.; Caponnetto, P.; Caruso, M.; Strano, S.; Battaglia, E.; Russo, C. Effect of Smoking Abstinence and Reduction in Asthmatic Smokers Switching to Electronic Cigarettes: Evidence for Harm Reversal. Int. J. Environ. Res. Public Health 2014, 11, 4965-4977. https://doi.org/10.3390/ijerph110504965
Polosa R, Morjaria J, Caponnetto P, Caruso M, Strano S, Battaglia E, Russo C. Effect of Smoking Abstinence and Reduction in Asthmatic Smokers Switching to Electronic Cigarettes: Evidence for Harm Reversal. International Journal of Environmental Research and Public Health. 2014; 11(5):4965-4977. https://doi.org/10.3390/ijerph110504965
Chicago/Turabian StylePolosa, Riccardo, Jaymin Morjaria, Pasquale Caponnetto, Massimo Caruso, Simona Strano, Eliana Battaglia, and Cristina Russo. 2014. "Effect of Smoking Abstinence and Reduction in Asthmatic Smokers Switching to Electronic Cigarettes: Evidence for Harm Reversal" International Journal of Environmental Research and Public Health 11, no. 5: 4965-4977. https://doi.org/10.3390/ijerph110504965
APA StylePolosa, R., Morjaria, J., Caponnetto, P., Caruso, M., Strano, S., Battaglia, E., & Russo, C. (2014). Effect of Smoking Abstinence and Reduction in Asthmatic Smokers Switching to Electronic Cigarettes: Evidence for Harm Reversal. International Journal of Environmental Research and Public Health, 11(5), 4965-4977. https://doi.org/10.3390/ijerph110504965








