Structure Analysis of Aerobic Granule from a Sequencing Batch Reactor for Organic Matter and Ammonia Nitrogen Removal
Abstract
:1. Introduction
2. Experimental Section
2.1. Experimental Setup
2.2. Analytical Methods
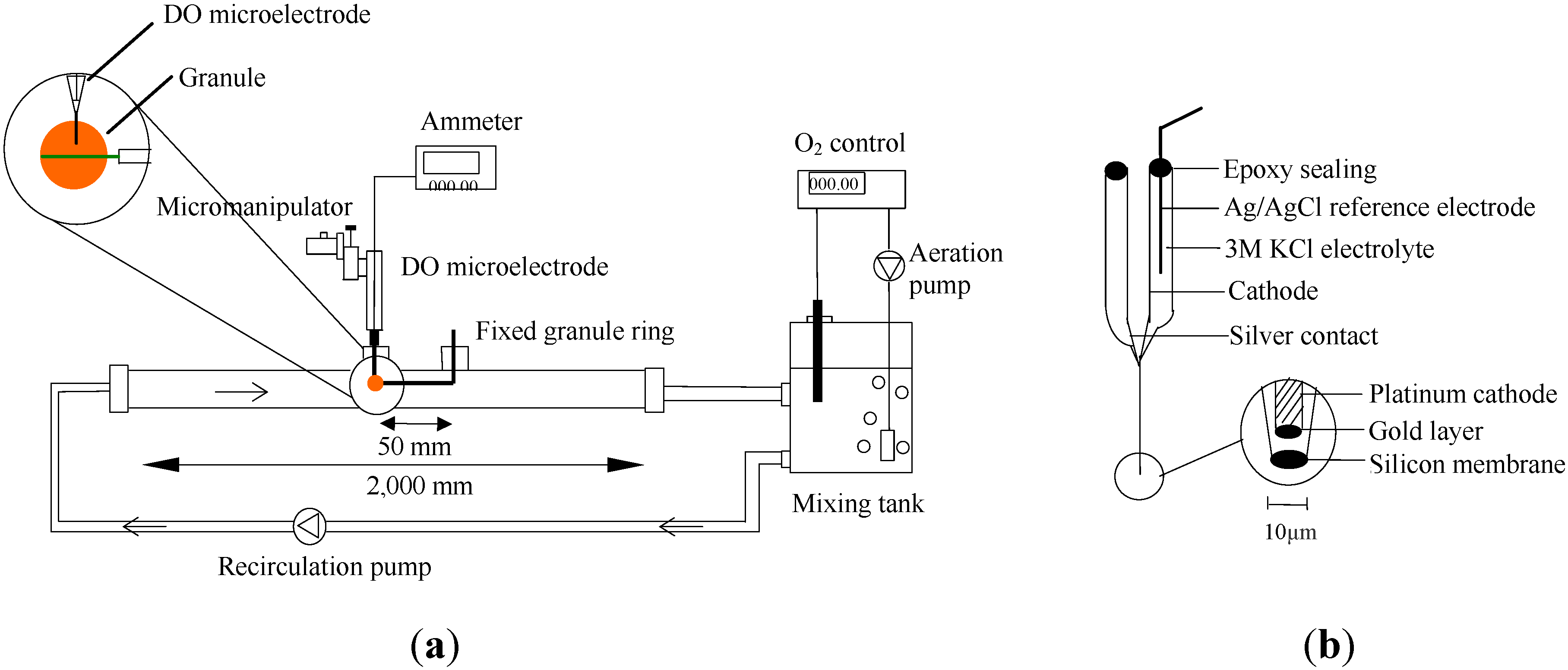
3. Results and Discussion
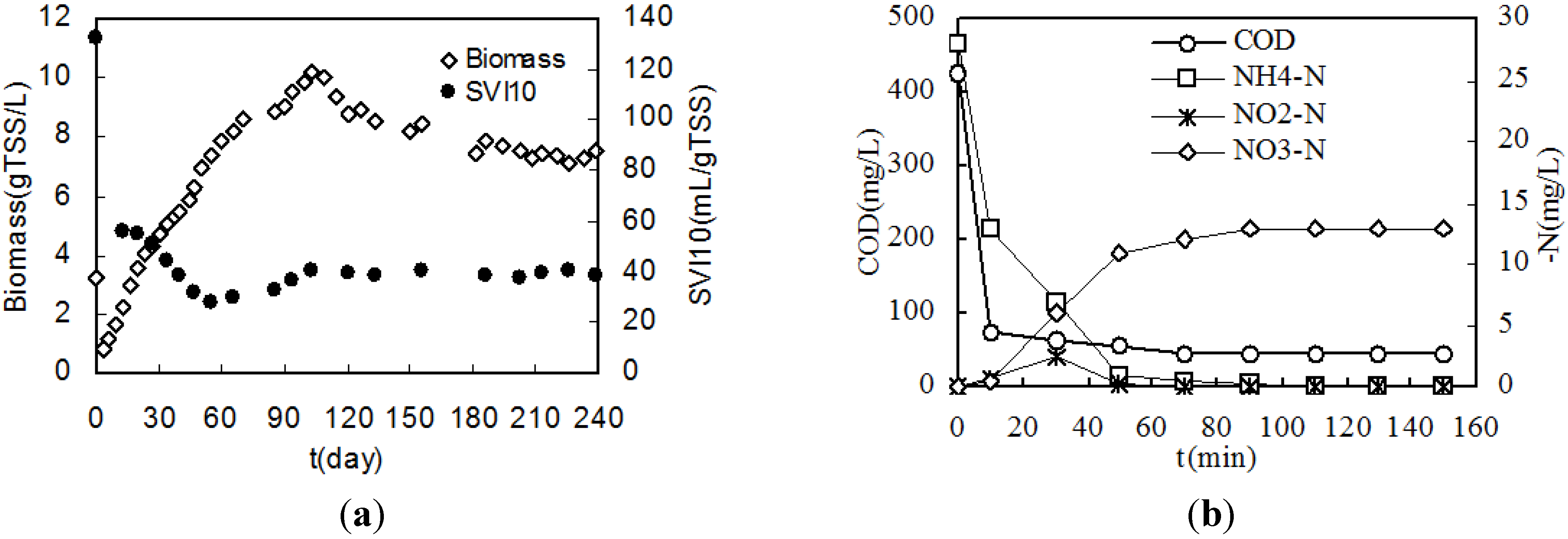
3.1. Formation and Characteristic of Granules

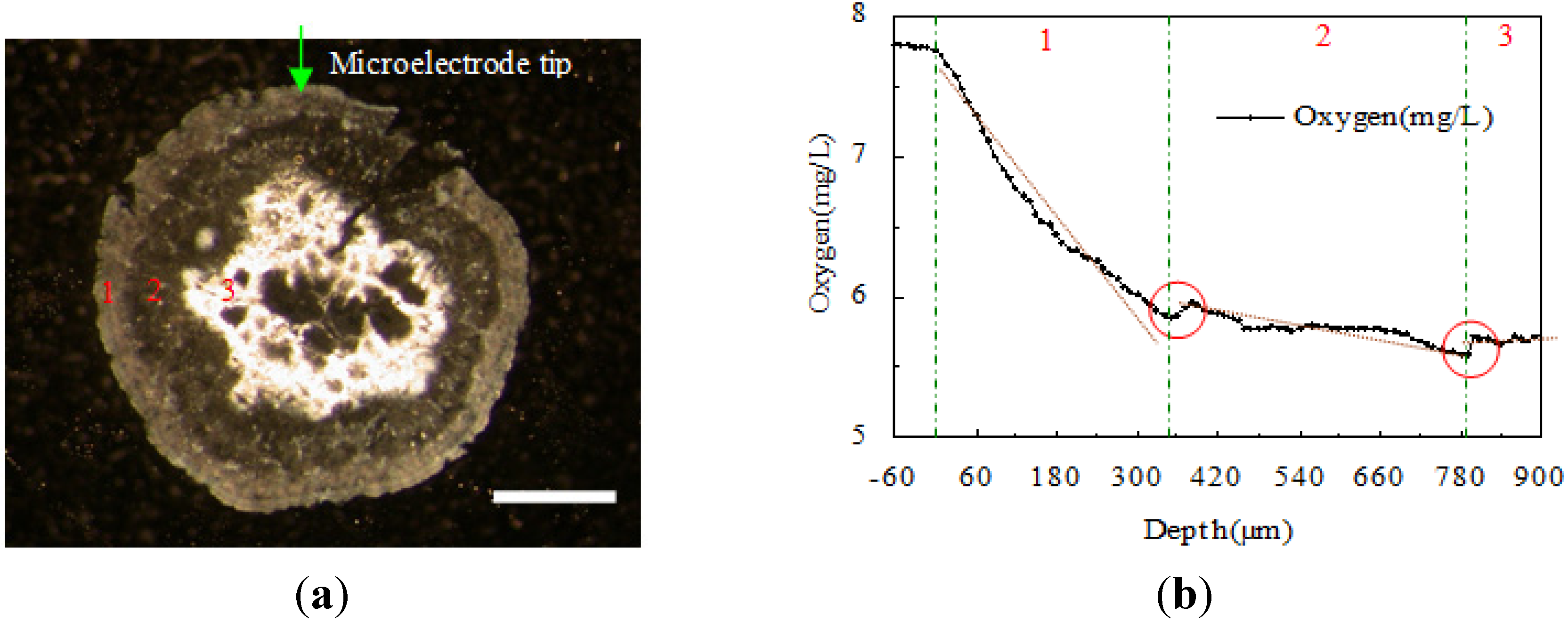
3.2. Structure Measurement by DO Microelectrode

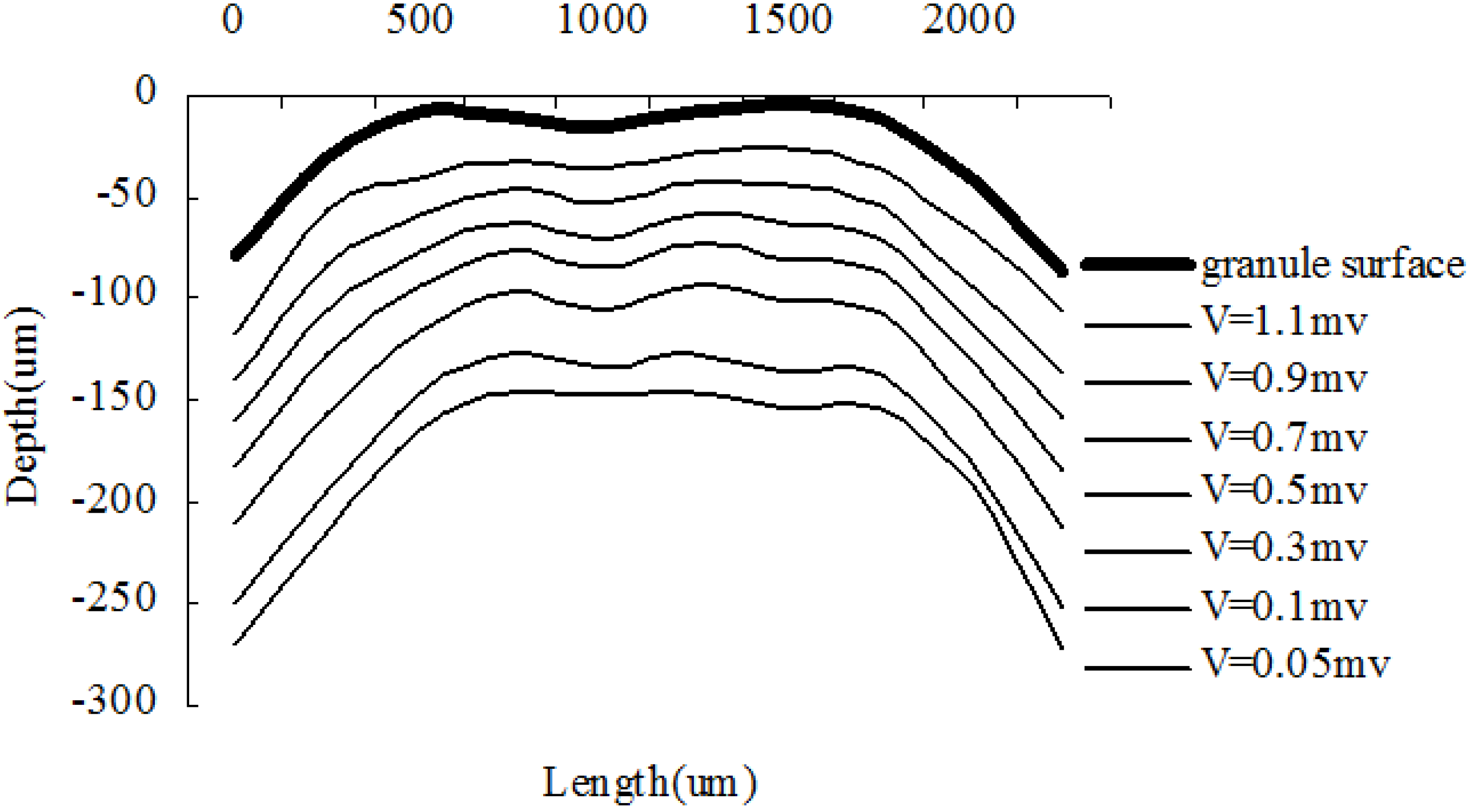
3.3. Structure Measurement by CLSM

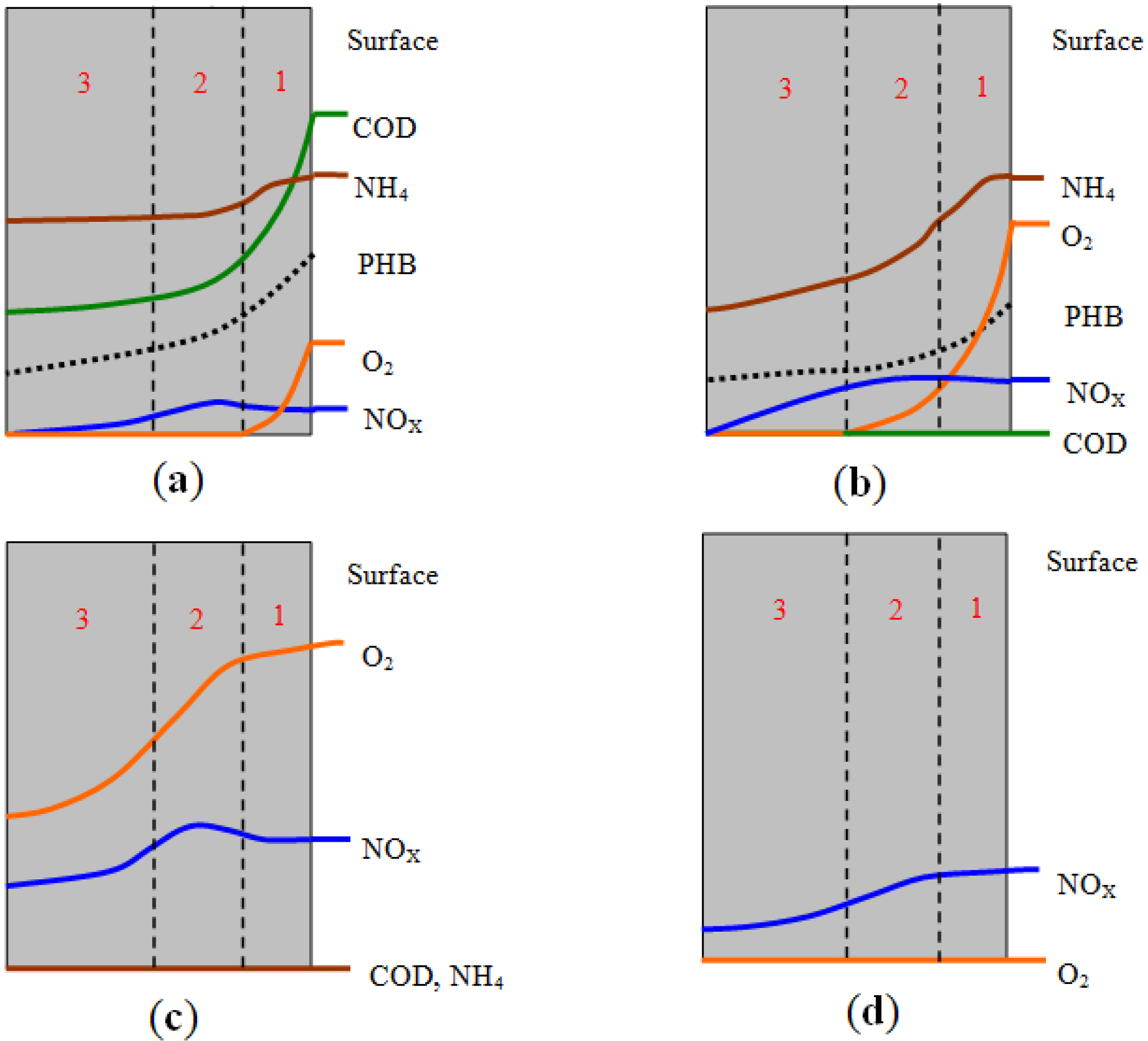
3.4. Analysis and Hypothesis of Granule Structure
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Morgenroth, E.; Sherden, T.; van Loosdrecht, M.C.M.; Heijnen, J.J.; Wilderer, P.A. Aerobic granular sludge in a sequencing batch reactor. Water Res. 1997, 31, 3191–3194. [Google Scholar] [CrossRef]
- Adav, S.S.; Lee, D.J.; Show, K.Y.; Tay, J.H. Aerobic granular sludge: Recent advances. Biotechnol. Adv. 2008, 26, 411–423. [Google Scholar] [CrossRef]
- Tay, J.H.; Pan, S.; He, Y.X.; Tay, S.T.L. Effect of organic loading rate on aerobic granulation: I Reactor performance. J. Environ. Eng. 2004, 130, 1094–1101. [Google Scholar] [CrossRef]
- Schwarzenbeck, N.; Erley, R.; Wilderer, P.A. Aerobic granular sludge in an SBR-system treating wastewater rich in particulate matter. Water Sci. Tech. 2004, 49, 21–46. [Google Scholar]
- Arrojo, B.; Mosquera-Corral, A.; Garrido, J.M.; Mendez, R.R. Aerobic granulation with industrial wastewater in sequencing batch reactors. Water Res. 2004, 38, 3389–3399. [Google Scholar] [CrossRef]
- Verawaty, M.; Pijuan, M.; Yuan, Z.; Bond, P. Determining the mechanisms for aerobic granulation from mixed seed of floccular and crushed granules in activated sludge wastewater treatment. Water Res. 2012, 46, 761–771. [Google Scholar] [CrossRef]
- Dirk, B.; Paul, S.; Frank, R.; Zigniew, L. Effects of biofilm structures on oxygen distribution and mass transport. Biotechnol. Bioeng. 1994, 43, 1131–1138. [Google Scholar] [CrossRef]
- Tay, J.H.; Liu, Q.S.; Liu, Y. Microscopic observation of aerobic granulation in sequential aerobic sludge blanket reactor. J. Appl. Microbiol. 2001, 91, 168–175. [Google Scholar] [CrossRef]
- Kreuk, M.K.; Heijnen, J.J.; van Loosdrecht, M.C.M. Simultaneous COD, nitrogen and phosphate removal by aerobic granular sludge. Biotechnol. Bioeng. 2005, 90, 761–769. [Google Scholar] [CrossRef]
- Schramm, A.; Larsen, L.H.; Revsbech, N.P.; Amann, R.T. Structure and function of a nitrifying biofilm as determined by microelectrodes and fluorescent oligonucleotide probes. Water Sci. Tech. 1997, 36, 263–270. [Google Scholar]
- Mosquera-Corral, A.; Kreuk, M.K.; Heijnen, J.J.; van Loosdrecht, M.C.M. Effect of oxygen concentration on N-removal in an aerobic granular sludge reactor. Water Res. 2005, 39, 2676–2686. [Google Scholar] [CrossRef]
- Liu, X.W.; Sheng, G.P.; Yu, H.Q. Physicochemical characteristics of microbial granules. Biotechnol. Adv. 2009, 27, 1061–1070. [Google Scholar] [CrossRef]
- Lee, D.J.; Chen, Y.Y.; Show, K.Y.; Whiteley, C.G.; Tay, J.H. Advances in aerobic granule formation and granule stability in the course of storage and reactor operation. Biotechnol. Adv. 2010, 28, 919–934. [Google Scholar] [CrossRef]
- Lu, R.; Yu, T. Fabrication and evaluation of an oxygen microelectrode applicable to environmental engineering and science. J. Environ. Eng. Sci. 2002, 1, 25–235. [Google Scholar]
- Binzer, T.; Borum, J.; Pedersen, O. Flow velocity affects internal oxygen conditions in the seagrassCymodoceanodosa. Aquat. Bot. 2005, 83, 239–247. [Google Scholar] [CrossRef]
- Van, T.T.; Gundersen, J.K.; Nielsen, M.S. The microdistribufion of oxygen in Danablu cheese measured by a microsensor during ripening. Int. J. Food Microbiol. 2002, 75, 157–161. [Google Scholar] [CrossRef]
- Jeffrey, K.T.; Mattew, R.P.; Ralph, D.F. Single-Neuron activity and tissue oxygenation in the cerebral cortex. Science 2003, 299, 1069–1072. [Google Scholar]
- Li, J.; Garny, K.; Neu, T.; He, M.; Lindenblatt, C.; Horn, H. Comparison of some characteristics of aerobic granules and sludge flocs from sequencing batch reactors. Water Sci. Technol. 2007, 55, 403–411. [Google Scholar] [CrossRef]
- APHA. Standard Methods for the Examination of Water and Wastewater, 19th ed.; APHA: Washington, DC, USA, 1995. [Google Scholar]
- Horn, H.; Hempel, D.C. Substrate utilization and mass transfer in an autotrophic Biofilm system: experimental results and numerical simulation. Biotechnol. Bioeng. 1997, 53, 363–371. [Google Scholar] [CrossRef]
- Boessmann, M.; Staudt, C.; Neu, T.R.; Horn, H.; Hempel, D.C. Investigation and modeling of growth, structure and oxygen penetration in particle supported biofilms. Chem. Eng. Technol. 2003, 26, 219–222. [Google Scholar] [CrossRef]
- Staudt, C.; Horn, H.; Hempel, D.C.; Neu, T.R. Volumetric measurements of bacterial cells and EPS glycoconjugates in biofilms. Biotechnol. Bioeng. 2004, 88, 585–592. [Google Scholar]
- Neu, T.R.; Lawrence, J.R. One-photon versus two-photon laser scanning microscopy and digital image analysis of microbial biofilms. Methods Microbiol. 2005, 34, 89–136. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Li, J.; Cai, A.; Wang, D.; Chen, C.; Ni, Y. Structure Analysis of Aerobic Granule from a Sequencing Batch Reactor for Organic Matter and Ammonia Nitrogen Removal. Int. J. Environ. Res. Public Health 2014, 11, 2427-2436. https://doi.org/10.3390/ijerph110302427
Li J, Cai A, Wang D, Chen C, Ni Y. Structure Analysis of Aerobic Granule from a Sequencing Batch Reactor for Organic Matter and Ammonia Nitrogen Removal. International Journal of Environmental Research and Public Health. 2014; 11(3):2427-2436. https://doi.org/10.3390/ijerph110302427
Chicago/Turabian StyleLi, Jun, Ang Cai, Danjun Wang, Chao Chen, and Yongjiong Ni. 2014. "Structure Analysis of Aerobic Granule from a Sequencing Batch Reactor for Organic Matter and Ammonia Nitrogen Removal" International Journal of Environmental Research and Public Health 11, no. 3: 2427-2436. https://doi.org/10.3390/ijerph110302427



