Subsurface Transport Behavior of Micro-Nano Bubbles and Potential Applications for Groundwater Remediation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Set-up
2.1.1. Generation Method
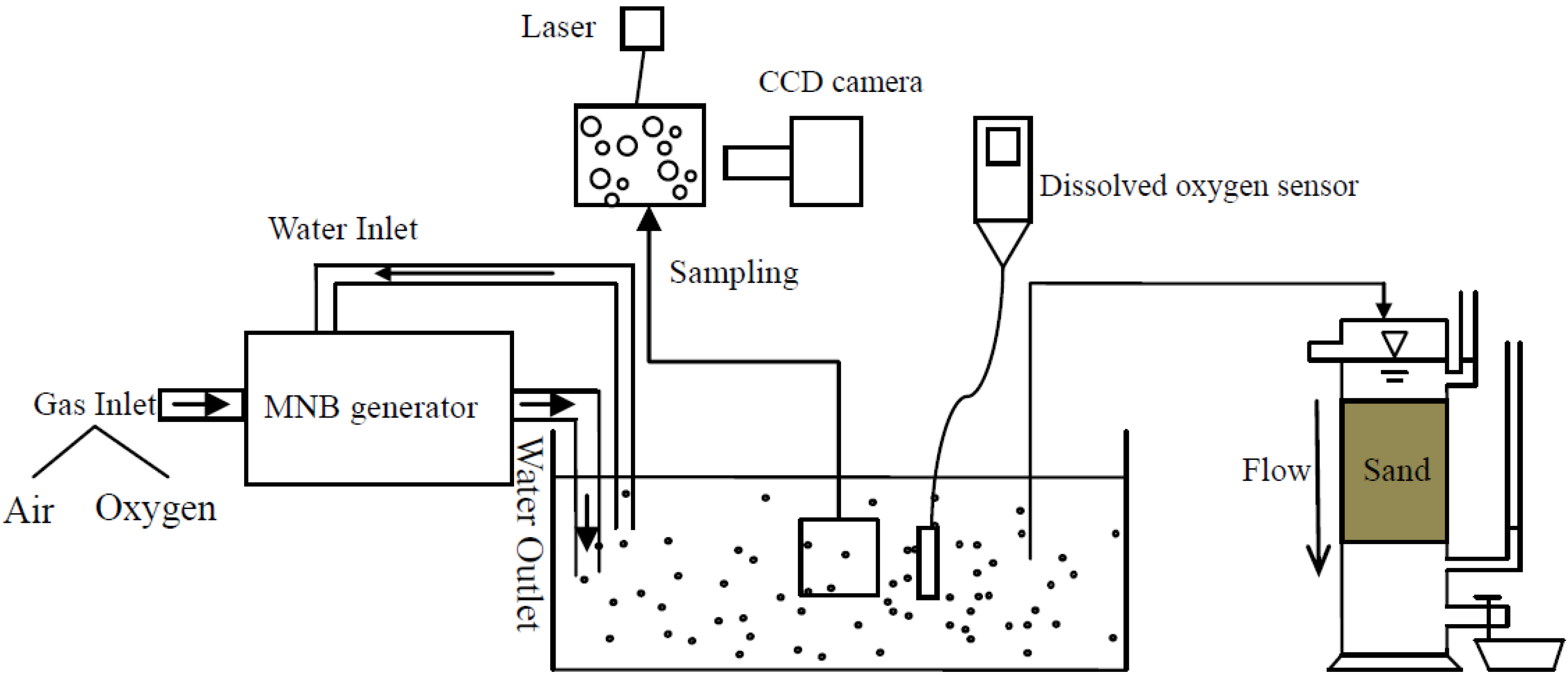
2.1.2. Bubble Size Distribution Analyzer
2.1.3. Dissolved Oxygen Sensor
2.2. Materials
| Material | Properties |
|---|---|
| Water | Deionized water |
| Gases | Air |
| Oxygen (90% concentrated) | |
| Surfactant | Sodium dodecyl sulfate (SDS) |
| Soil | British standard sand (D10 = 0.11 mm, D50 = 0.14 mm) |
2.3. Experimental Procedure
| Description | |
|---|---|
| Group 1 | Air macrobubbles in deionized water |
| Group 2 | Oxygen macrobubbles in deionized water |
| Group 3 | Air micro-nanobubbles in deionized water |
| Group 4 | Oxygen micro-nanobubbles in deionized water |
| Group 5 | Air micro-nanobubbles in deionized water with SDS concentration 5 mg/L |
| Group 6 | Air micro-nanobubbles in deionized water with SDS concentration 10 mg/L |
3. Results and Discussion
3.1. Bubble Size Distribution

| Cases | Deionized Water without Surfactant | Deionized Water with 5 mg/L Surfactant Concentration | Deionized Water with 10 mg/L Surfactant Concentration |
|---|---|---|---|
| D50/μm | 33 ± 13 | 9.7 ± 1.3 | 4.1 ± 1.1 |
| M1P/μm | 10.7 ± 2.4 | 0.71 ± 0.16 | 0.67 ± 0.21 |
| M2P/μm | 43.0 ± 4.4 | 10.8 ± 0.8 | 10.2 ± 1.0 |
3.2. Permeability Results
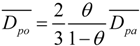
| Hydraulic Conductivity/10−5 m/s (20 °C) | Ratio | ||
|---|---|---|---|
| Air Free Water (kw) | Micro-Nano Bubble Water (kb) | kb/kw | |
| Soil sample 1 (porosity = 0.50) | 8.1 ± 2.1 | 7.7 ± 2.3 | 94% |
| Soil sample 2 (porosity = 0.41) | 6.1 ± 1.3 | 6.1 ± 2.1 | 100% |
| Soil sample 3 (porosity = 0.35) | 4.3 ± 1.4 | 3.8 ± 1.4 | 88% |
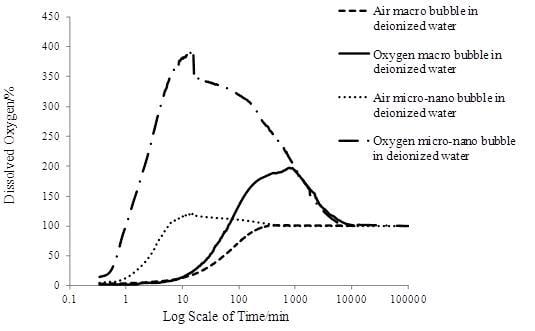
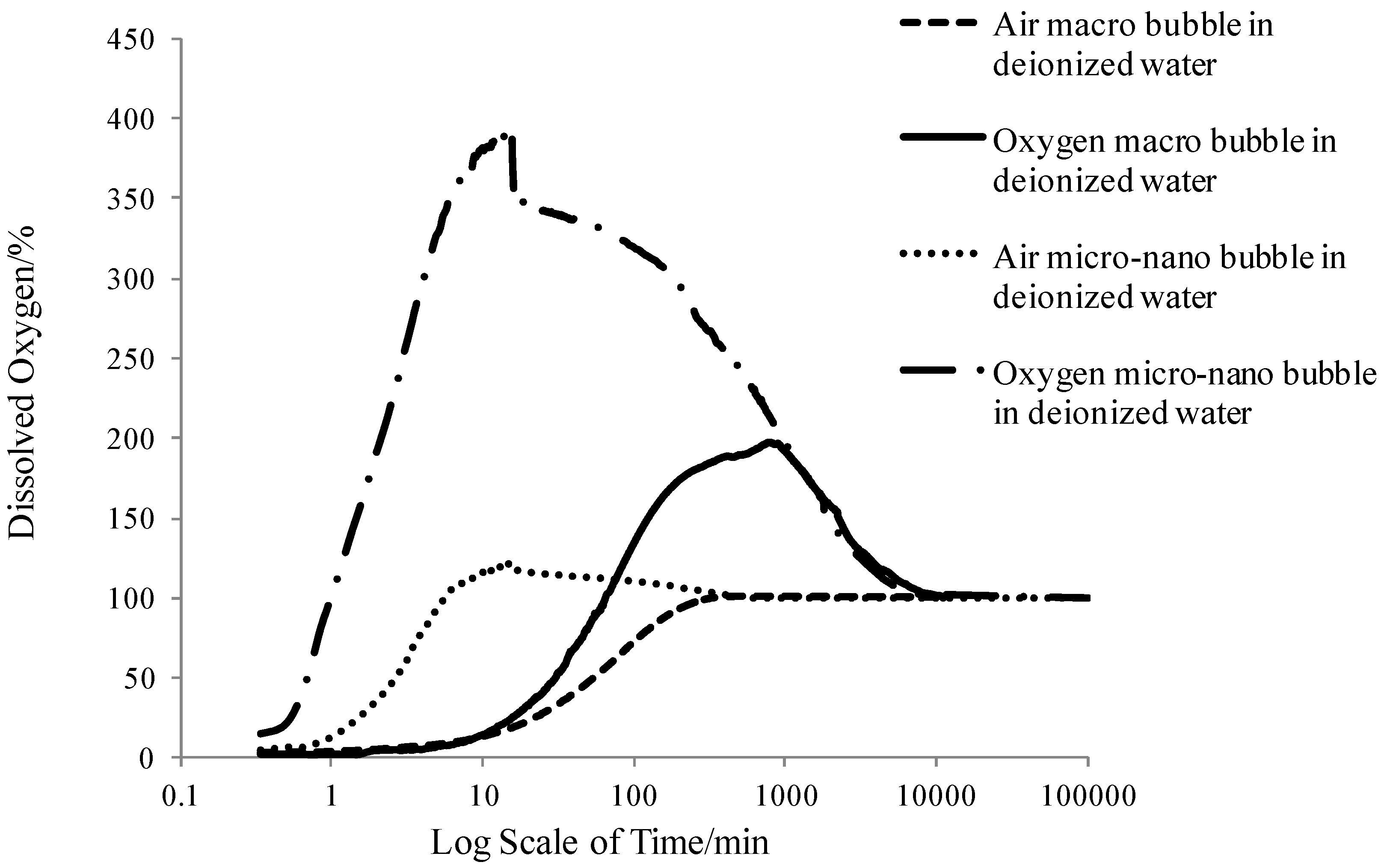
3.3. The Oxygen Transfer Efficiency
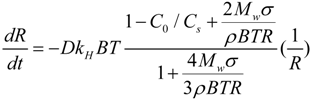
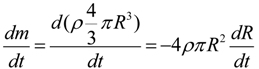
| Parameter | Value | Unit |
|---|---|---|
| D | 1.75 × 10−9 | m2/s |
| kH | 7.44 × 10−6 | mol/N·m |
| B | 8.3144 | J/mol·K |
| T | 293.15 | K |
| Mw | 29 | g/mol |
| σ | 0.072 | N/m |
| AIDOIR (10−2 mg/L/min) | DOPV (mg/L) | ST (103 min) | |
|---|---|---|---|
| Group 1 1 | 2.70 ± 0.20 | 9.89 ± 0.08 | - |
| Group 2 4 | 3.50 ± 0.30 | 19.1 ± 1.2 | 9.30 ± 0.12 |
| Group 3 4 | 94.4 ± 2.1 | 10.4 ± 0.3 | 0.58 ± 0.06 |
| Group 4 4 | 342 ± 19 | 34.2 ± 1.9 | 9.98 ± 0.14 |
| Group 5 4 | 74.1 ± 0.5 | 9.63 ± 0.07 | 1.86 ± 0.08 |
| Group 6 4 | 63.8 ± 0.3 | 9.57 ± 0.05 | 2.58 ± 0.09 |
4. Conclusions
- (1)
- Two peaks were found in the size distribution of MNBs generated by the spiral liquid flow type generator, one was 10 μm and the other was 50 μm. Adding surfactant into water made the bubble size smaller, with two peaks of 700 nm and 10 μm. Higher surfactant concentration led to smaller bubble size;
- (2)
- MNB water had no influence on the hydraulic conductivity of fine sand;
- (3)
- MNBs greatly enhanced the oxygen transfer efficiency compared to the macro bubbles, which would predictably facilitate the aerobic biodegradation process for groundwater contaminant removal. Oxygen MNBs had the fastest DO mass transfer rate which was nearly 125 times faster than air macro bubble, the highest DO peak value which was nearly three times larger than air macro bubble, the longest dissolved oxygen enhancement durability which was 16 times longer than air micro-nanobubbles. Higher surfactant concentration exerted larger mass transfer resistance on the bubble interface resulting in lower oxygen transfer efficiency and longer stagnation time of MNBs in water.
Acknowledgments
Conflicts of Interest
References
- Attard, P.; Moody, M.P.; Tyrrell, J.W.G. Nanobubbles: The big picture. Physica A: Stat. Mech. Appl. 2002, 314, 696–705. [Google Scholar] [CrossRef]
- Chu, L.-B.; Xing, X.-H.; Yu, A.-F.; Zhou, Y.-N.; Sun, X.-L.; Jurcik, B. Enhanced ozonation of simulated dyestuff wastewater by microbubbles. Chemosphere 2007, 68, 1854–1860. [Google Scholar] [CrossRef]
- Chu, L.-B.; Xing, X.-H.; Yu, A.-F.; Sun, X.-L.; Jurcik, B. Enhanced treatment of practical textile wastewater by microbubble ozonation. Process Saf. Environ. Prot. 2008, 86, 389–393. [Google Scholar] [CrossRef]
- Xu, Q.; Nakajima, M.; Ichikawa, S.; Nakamura, N.; Shiina, T. A comparative study of microbubble generation by mechanical agitation and sonication. Innov. Food Sci. Emerg. Technol. 2008, 9, 489–494. [Google Scholar] [CrossRef]
- Li, P.; Takahashi, M.; Chiba, K. Degradation of phenol by the collapse of microbubbles. Chemosphere 2009, 75, 1371–1375. [Google Scholar] [CrossRef]
- Li, P.; Takahashi, M.; Chiba, K. Enhanced free-radical generation by shrinking microbubbles using a copper catalyst. Chemosphere 2009, 77, 1157–1160. [Google Scholar] [CrossRef]
- Takahashi, M. Base and technological application of micro-bubble and nano-bubble. Mater. Integr. 2009, 22, 2–19. [Google Scholar]
- Agarwal, A.; Ng, W.J.; Liu, Y. Principle and applications of microbubble and nanobubble technology for water treatment. Chemosphere 2011, 84, 1175–1180. [Google Scholar] [CrossRef]
- Zimmerman, W.B.; Tesař, V.; Bandulasena, H. Towards energy efficient nanobubble generation with fluidic oscillation. Curr. Opin. Colloid Interface Sci. 2011, 16, 350–356. [Google Scholar] [CrossRef]
- Li, H.; Hu, L.; Xia, Z. Impact of groundwater salinity on bioremediation enhanced by micro-nano bubbles. Materials 2013, 6, 3676–3687. [Google Scholar] [CrossRef]
- Hu, L.; Meegoda, J.N.; Du, J.; Gao, S.; Wu, X. Centrifugal study of zone of influence during air-sparging. J. Environ. Monit. 2011, 13, 2443–2449. [Google Scholar] [CrossRef]
- Li, H.; Hu, L.; Wang, J.; Wu, X.; Liu, P. 3D numerical simulation of air sparging remediation process. Chin. J. Environ. Sci. 2012, 33, 1532–1539. [Google Scholar]
- Hu, L.; Wu, X.; Liu, Y.; Meegoda, J.N.; Gao, S. Physical modeling of air flow during air sparging remediation. Environ. Sci. Technol. 2010, 44, 3883–3888. [Google Scholar] [CrossRef]
- Meegoda, J.N.; Hu, L. A review of centrifugal testing of gasoline contamination and remediation. Int. J. Environ. Res. Public Health 2011, 8, 3496–3513. [Google Scholar] [CrossRef]
- Turner, W. Microbubble persistence in fresh water. J. Acoust. Soc. Am. 1961, 33, 1223–1233. [Google Scholar] [CrossRef]
- Yoshida, A.; Takahashi, O.; Ishii, Y.; Sekimoto, Y.; Kurata, Y. Water purification using the adsorption characteristics of microbubbles. Jpn. J. Appl. Phys. 2008, 47. [Google Scholar] [CrossRef]
- Tasaki, T.; Wada, T.; Baba, Y.; Kukizaki, M. Degradation of surfactants by an integrated nanobubbles/VUV irradiation technique. Ind. Eng. Chem. Res. 2009, 48, 4237–4244. [Google Scholar] [CrossRef]
- Ohnari, H.; Saga, T.; Watanabe, K.; Maeda, K.; Matsuo, K. High functional characteristics of micro-bubbles and water purification. Res. Process. 1999, 46, 238–244. [Google Scholar] [CrossRef]
- Couto, H.J.B.; Nunes, D.G.; Neumann, R.; Franca, S.C.A. Micro-bubble size distribution measurements by laser diffraction technique. Miner. Eng. 2009, 22, 330–335. [Google Scholar] [CrossRef]
- Keitel, G.; Onken, U. Inhibition of bubble coalescence by solutes in air/water dispersions. Chem. Eng. Sci. 1982, 37, 1635–1638. [Google Scholar] [CrossRef]
- Malard, F.; Hervant, F. Oxygen supply and the adaptations of animals in groundwater. Freshw. Biol. 1999, 41, 1–30. [Google Scholar] [CrossRef]
- Bowley, W.W.; Hammond, G.L. Controlling factors for oxygen transfer through bubbles. Ind. Eng. Chem. Process Des. Dev. 1978, 17, 2–8. [Google Scholar] [CrossRef]
- Bredwell, M.D.; Worden, R.M. Mass transfer properties of microbubbles. 1. Experimental studies. Biotechnol. Prog. 1998, 14, 31–38. [Google Scholar] [CrossRef]
- Wan, J.; Veerapaneni, S.; Gadelle, F.; Tokunaga, T.K. Generation of stable microbubbles and their transport through porous media. Water Resour. Res. 2001, 37, 1173–1182. [Google Scholar] [CrossRef]
- Terasaka, K.; Hirabayashi, A.; Nishino, T.; Fujioka, S.; Kobayashi, D. Development of microbubble aerator for waste water treatment using aerobic activated sludge. Chem. Eng. Sci. 2011, 66, 3172–3179. [Google Scholar] [CrossRef]
- Kukizaki, M.; Goto, M. Size control of nanobubbles generated from shirasu-porous-glass (SPG) membranes. J. Membr. Sci. 2006, 281, 386–396. [Google Scholar] [CrossRef]
- Tasaki, T.; Wada, T.; Fujimoto, K.; Kai, S.; Ohe, K.; Oshima, T.; Baba, Y.; Kukizaki, M. Degradation of methyl orange using short-wavelength UV irradiation with oxygen microbubbles. J. Hazard. Mater. 2009, 162, 1103–1110. [Google Scholar] [CrossRef]
- Takahashi, M.; Chiba, K.; Li, P. Free-radical generation from collapsing microbubbles in the absence of a dynamic stimulus. J. Phys. Chem. B 2007, 111, 1343–1347. [Google Scholar] [CrossRef]
- Young, T. An essay on the cohesion of fluids. Philos. Trans. Royal Soc. London 1805, 95, 65–87. [Google Scholar] [CrossRef]
- Comte de Laplace, P.S. Traité De Mécanique céleste; Crapelet: Paris, France, 1825. [Google Scholar]
- Skorokhod, V.; Get’man, O.; Zuev, A.; Rakitin, S. Correlation between the particle size, pore size, and porous structure of sintered tungsten. Sov. Powder Metall. Met. Ceram. 1988, 27, 941–947. [Google Scholar]
- Fick, A. Ueber diffusion. Annalen der Physik 1855, 170, 59–86. [Google Scholar] [CrossRef]
- Henry, W. Experiments on the quantity of gases absorbed by water, at different temperatures, and under different pressures. Philos. Trans. Royal Soc. London 1803, 93, 29–276. [Google Scholar] [CrossRef]
- Duncan, P.B.; Needham, D. Test of the epstein-plesset model for gas microparticle dissolution in aqueous media: Effect of surface tension and gas undersaturation in solution. Langmuir 2004, 20, 2567–2578. [Google Scholar] [CrossRef]
- Clapeyron, E. Mémoire sur la puissance motrice de la chaleur; Jacques Gabay: Paris, France, 1834. [Google Scholar]
- Haynes, W.M.; Lide, D.R.; Bruno, T.J. CRC Handbook of Chemistry and Physics 2012–2013; CRC Press: Boca Raton, FL, USA, 2012. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Li, H.; Hu, L.; Song, D.; Al-Tabbaa, A. Subsurface Transport Behavior of Micro-Nano Bubbles and Potential Applications for Groundwater Remediation. Int. J. Environ. Res. Public Health 2014, 11, 473-486. https://doi.org/10.3390/ijerph110100473
Li H, Hu L, Song D, Al-Tabbaa A. Subsurface Transport Behavior of Micro-Nano Bubbles and Potential Applications for Groundwater Remediation. International Journal of Environmental Research and Public Health. 2014; 11(1):473-486. https://doi.org/10.3390/ijerph110100473
Chicago/Turabian StyleLi, Hengzhen, Liming Hu, Dejun Song, and Abir Al-Tabbaa. 2014. "Subsurface Transport Behavior of Micro-Nano Bubbles and Potential Applications for Groundwater Remediation" International Journal of Environmental Research and Public Health 11, no. 1: 473-486. https://doi.org/10.3390/ijerph110100473
APA StyleLi, H., Hu, L., Song, D., & Al-Tabbaa, A. (2014). Subsurface Transport Behavior of Micro-Nano Bubbles and Potential Applications for Groundwater Remediation. International Journal of Environmental Research and Public Health, 11(1), 473-486. https://doi.org/10.3390/ijerph110100473