Bioaccumulation of Trace Elements in Ruditapes philippinarum from China: Public Health Risk Assessment Implications
Abstract
:1. Introduction
2. Methods
2.1. Sampling

2.2. Trace Element Analysis
2.3. Human Exposure Assessment
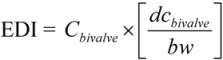
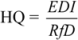
3. Results
3.1. Geographical Distribution of Trace Elements in Clams
3.2. Estimated Exposure and Hazards Quotients of Trace Elements

| Location | Cu | Zn | Mn | Se | Ni | Cd | Cr | Pb | Hg | As | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Qinghuangdao | EDI | 0.002710 | 0.012001 | 0.003872 | 0.000509 | 0.000592 | 0.000046 | 0.000118 | 0.000048 | 0.000016 | 0.000047 |
| HQ | 0.0678 | 0.0400 | 0.0277 | 0.1018 | 0.0118 | 0.0460 | 0.0393 | n.a. | 0.0533 | 0.1175 | |
| Dalian | EDI | 0.001904 | 0.008721 | 0.004849 | 0.000299 | 0.000739 | 0.000053 | 0.000155 | 0.000046 | 0.000011 | 0.000031 |
| HQ | 0.0476 | 0.0291 | 0.0346 | 0.0598 | 0.0148 | 0.0530 | 0.0517 | n.a. | 0.0367 | 0.0775 | |
| Qingdao | EDI | 0.001376 | 0.005441 | 0.004086 | 0.000519 | 0.001021 | 0.000058 | 0.000338 | 0.000061 | 0.000006 | 0.000069 |
| HQ | 0.0344 | 0.0181 | 0.0292 | 0.1038 | 0.0204 | 0.0580 | 0.1127 | n.a. | 0.0200 | 0.1725 | |
| Lianyungang | EDI | 0.001184 | 0.006132 | 0.003922 | 0.000503 | 0.000724 | 0.000070 | 0.000241 | 0.000046 | 0.000005 | 0.000059 |
| HQ | 0.0296 | 0.0204 | 0.0280 | 0.1006 | 0.0145 | 0.0700 | 0.0803 | n.a. | 0.0167 | 0.1475 | |
| Ningbo | EDI | 0.001262 | 0.003530 | 0.004114 | 0.000466 | 0.000530 | 0.000069 | 0.000214 | 0.000127 | 0.000006 | 0.000027 |
| HQ | 0.0316 | 0.0118 | 0.0294 | 0.0932 | 0.0106 | 0.0690 | 0.0713 | n.a. | 0.0200 | 0.0675 | |
| Fuzhou | EDI | 0.001376 | 0.004207 | 0.005398 | 0.000367 | 0.000339 | 0.000056 | 0.000208 | 0.000185 | 0.000006 | 0.000023 |
| HQ | 0.0344 | 0.0140 | 0.0386 | 0.0734 | 0.0068 | 0.0560 | 0.0693 | n.a. | 0.0200 | 0.0575 | |
| Xiamen | EDI | 0.001547 | 0.004613 | 0.003209 | 0.000583 | 0.000944 | 0.000079 | 0.000139 | 0.000171 | 0.000004 | 0.000021 |
| HQ | 0.0387 | 0.0154 | 0.0229 | 0.1166 | 0.0189 | 0.0790 | 0.0463 | n.a. | 0.0133 | 0.0525 | |
| Shenzheng | EDI | 0.001690 | 0.005155 | 0.004613 | 0.000621 | 0.000792 | 0.000053 | 0.000080 | 0.000031 | 0.000004 | 0.000004 |
| HQ | 0.0423 | 0.0172 | 0.0330 | 0.1242 | 0.0158 | 0.0530 | 0.0267 | n.a. | 0.0133 | 0.0100 | |
| Beihai | EDI | 0.000863 | 0.005041 | 0.003872 | 0.000504 | 0.000695 | 0.000073 | 0.000086 | 0.000032 | 0.000004 | 0.000005 |
| HQ | 0.0216 | 0.0168 | 0.0277 | 0.1008 | 0.0139 | 0.0730 | 0.0287 | n.a. | 0.0133 | 0.0125 | |
| PTWI | 3.5 | 1.5 | 0.98 | 0.035 | n.a. | 0.007 | n.a. | 0.025 | 0.005 | 0.015 | |
| ADI | 0.5 | 0.21 | 0.14 | 0.005 | n.a. | 0.001 | n.a. | 0.0036 | 0.0007 | 0.0021 | |
| RfD | 0.04 | 0.3 | 0.14 | 0.005 | 0.05 | 0.001 | 0.003 | n.a. | 0.0003 | 0.0004 | |
4. Discussion and Conclusions
4.1. Geographical Variance
| Region | Cu | Zn | Mn | Se | Ni | Cd | Cr | Pb | Hg | As | Reference |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Bohai Sea, China | 20.0–38.0 | 122.3–168.3 | 47.5–68.0 | 4.19–7.14 | 8.30–12.26 | 0.64–0.75 | 1.66–2.29 | 0.64–0.72 | 0.15–0.23 | 4.31–6.62 | This study |
| Yellow Sea, China | 14.0–19.3 | 76.3–86.0 | 51.0–57.3 | 6.24–7.28 | 10.15–14.32 | 0.42–0.98 | 3.38–4.74, | 0.65–0.86 | 0.07–0.09 | 8.25–11.95 | This study |
| East China Sea, China | 17.7–21.7 | 49.5–64.7 | 45.0–75.7 | 5.15–8.18 | 4.76–13.24 | 0.79–1.11 | 1.95–3.00 | 1.78–2.59 | 0.06–0.09 | 2.90–3.79 | This study |
| South China Sea, China | 12.1–23.7 | 50.7–72.3 | 42.0–64.7 | 6.21–8.71 | 8.40–9.74 | 0.41–1.03 | 0.94–1.25 | 0.32–0.54 | 0.03–0.06 | 0.46–0.85 | This study |
| Bohai Sea, China | 7.5–25.7 | 58.5–190.5 | 14.1–94.8 | 2.66–23.10 | 9.90–14.25 | 0.82–3.71 | 0.94–19.1 | 0.47–2.00 | 0.08–0.27 | 9.9–14.25 | [15] |
| Bohai Sea, China | 8.96–30.59 | 70.0–140.4 | n.a. | n.a. | 5.04–20.79 | 0.98–4.41 | n.a. | 0.91–2.38 | n.a. | n.a. | [30] |
| Jiaozhou Bay, China | 5.1–26.0 | 52.1–110.3 | 10.3–63.9 | n.a. | 5.26–52.75 | 0.65–3.31 | 9.64–35.47 | 0.89–14.77 | n.a. | n.a. | [18] |
| Jiaozhou Bay, China | 7.7–9.1 | 62.2–87.1 | n.a. | n.a. | n.a. | 0.39–0.68 | 0.96–4.04 | 0.53–1.42 | 0.04–0.07 | 8.32–14.33 | [34] |
| Jiaozhou Bay, China | 8.8–21.5 | n.a. | n.a. | n.a. | n.a. | 0.07–1.05 | 9.03–10.92 | 2.10–3.64 | 0.02–0.21 | 9.80–15.54 | [35] |
| Jiaozhou Bay, China | 6.4–19.8 | 35.5–85.5 | 27.45–67.6 | n.a. | n.a. | 0.51–0.67 | n.a. | 0.31–1.01 | n.a. | n.a. | [36] |
| East China Sea, China | 9.2–14.4 | 26.0–73.5 | n.a. | n.a. | n.a. | 0.48–0.85 | n.a. | 0.23–0.42 | 0.06–0.15 | 2.47–3.45 | [16] |
| South China Sea, China | 6.0–10.0 | 71.0–116.0 | n.a. | n.a. | n.a. | 0.60–5.00 | 0.20–14.50 | 0.80–2.70 | n.a. | n.a. | [6] |
| Pearl River Delta, China | 4.0–10.1 | 39.4–212.9 | n.a. | n.a. | 5.04–5.88 | 0.42–1.19 | 3.85–4.13 | 1.47–1.75 | n.a. | n.a. | [40] |
| Pearl River Delta, China | 5.2–13.9 | 54.9–74.6 | n.a. | n.a. | 9.78–23.72 | 0.30–7.60 | 3.13–12.25 | 1.50–14.99 | n.a. | n.a. | [41] |
| Southern Atlantic, Spanish | 5.1–26.0 | 52.1–110.3 | 10.3–63.9 | n.a. | 5.26–52.75 | 0.47–2.71 | 9.6–35.5 | 0.41–1.94 | 0.13–2.72 | 12.60–32.20 | [9] |
| Gironde Estuary, France | 9.4 ± 1.0 | 97.2 ± 8.0 | n.a. | n.a. | n.a. | 0.52 ± 0.11 | n.a. | n.a. | n.a. | n.a. | [11] |
| Kyeonggi Bay, Korea | 5.5–14.3 | 64.7–162.0 | 20.5–177.0 | n.a. | 2.74–33.50 | 0.53–2.20 | 0.61–2.38 | 0.34–1.72 | n.a. | n.a. | [20] |
| Shihwa Lake, Korea | 6.6–23.2 | 70.0–144.0 | n.a. | n.a. | 3.20–13.4 | 0.56–1.11 | 0.42–1.40 | 0.50–1.82 | n.a. | 12.4–18.3 | [21] |
| Venice lagoon, Italy | 8.2–29.0 | 60.0–122.0 | 8.0–29.1 | n.a. | 0.15–8.37 | 0.26–2.19 | 1.89–5.70 | 0.49–2.54 | 0.25–2.30 | 18.90–64.00 | [22] |
4.2. Public Health Risk Assessment
Acknowledgments
Conflict of Interest
References
- Maanan, M. Heavy metal concentrations in marine mollusks from the Moroccan coastal region. Environ. Pollut. 2008, 153, 176–183. [Google Scholar] [CrossRef]
- Rainbow, P.S. Trace metal concentrations in aquatic invertebrates, why and so what? Environ. Pollut. 2002, 120, 497–507. [Google Scholar] [CrossRef]
- Rainbow, P.S.; Wolowicz, M.; Fialkowski, W.; Smith, B.D.; Sokolowski, A. Biomonitoring of trace metals in the Gulf of Gdansk, using mussels (Mytilus trossulus) and barnacles (Balanus improvisus). Water. Res. 2000, 34, 1823–1829. [Google Scholar] [CrossRef]
- Szefer, P.; Frelek, K.; Szefer, K.; Lee, C.B.; Kim, B.S.; Warzocha, J.; Zdrojewska, I.; Ciesielski, T. Distribution and relationships of trace metals in soft tissue, byssus and shells of Mytilus edulis trossulus from the southern Baltic. Environ. Pollut. 2002, 120, 423–444. [Google Scholar] [CrossRef]
- Saha, M.; Sarkar, S.K.; Bhattacharya, B. Interspecific variation in heavy metal body concentrations in biota of Sunderban mangrove wetland, northeast India. Environ. Int. 2006, 32, 203–207. [Google Scholar] [CrossRef]
- Pan, K.; Wang, W.X. Trace metal contamination in estuarine and coastal environments in China. Sci. Total. Environ. 2011, 421–422, 3–16. [Google Scholar]
- Zhao, L.; Yang, F.; Yan, X.; Huo, Z.; Zhang, G. Heavy metal concentrations in surface sediments and Manila clams (Ruditapes philippinarum) from the Dalian coast, China after the Dalian Port oil spill. Biol. Trace. Elem. Res. 2012, 149, 241–247. [Google Scholar] [CrossRef]
- Szefer, P.; Kim, B.S.; Kim, C.K.; Kim, E.H.; Lee, C.B. Distribution and coassociations of trace elements in soft tissue and byssus of Mytilus galloprovincialis relative to the surrounding seawater and suspended matter of the southern part of the Korean Peninsula. Environ. Pollut. 2004, 129, 209–228. [Google Scholar] [CrossRef]
- Usero, J.; Morillo, J.; Gracia, I. Heavy metal concentrations in molluscs from the Atlantic coast of southern Spain. Chemosphere 2005, 59, 1175–1181. [Google Scholar] [CrossRef]
- Mertz, W. The essential trace elements. Science 1981, 213, 1332–1338. [Google Scholar]
- Baudrimont, M.; Schäfer, J.; Marie, V.; Maury-Brachet, R.; Bossy, C.; Boudou, A.; Blanc, G. Geochemical survey and metal bioaccumulation of three bivalve species (Crassostrea gigas, Cerastoderma edule and Ruditapes philippinarum) in the Nord Médoc salt marshes (Gironde estuary, France). Sci. Total. Environ. 2005, 337, 265–280. [Google Scholar] [CrossRef]
- Sapkota, A.; Sapkota, A.R.; Kucharski, M.; Burke, J.; Mckenzie, S.; Walker, P.; Lawrence, R. Aquaculture practices and potential human health risks, current knowledge and future priorities. Environ. Int. 2008, 34, 1215–1226. [Google Scholar] [CrossRef]
- Mostafa, A.R.; Al-Alimi, A.K.A.; Barakat, A.O. Metals in surface sediments and marine bivalves of the Hadhramout coastal area, Gulf of Aden, Yemen. Mar. Pollut. Bull. 2009, 58, 308–311. [Google Scholar] [CrossRef]
- Fung, C.N.; Lam, J.C.W.; Zheng, G.J.; Connell, D.W.; Monirith, I.; Tanabe, S. Mussel-based monitoring of trace metal and organic contaminants along the east coast of China using Perna viridis and Mytilus edulis. Environ. Pollut. 2004, 127, 203–216. [Google Scholar] [CrossRef]
- Wang, Y.W.; Liang, L.N.; Shi, J.B.; Jiang, G.B. Study on the contamination of heavy metals and their correlations in mollusks collected from coastal sites along the Chinese Bohai Sea. Environ. Int. 2005, 31, 1103–1113. [Google Scholar] [CrossRef]
- Huang, H.; Wu, J.Y.; Wu, J.H. Heavy metal monitoring using bivalved shellfish from Zhejiang Coastal Waters, East China Sea. Environ. Monit. Assess. 2007, 129, 315–320. [Google Scholar] [CrossRef]
- Zhang, G.; Yan, X.; Yang, F. A new three-phase culture method for Manila clam, Ruditapes philippinarum, farming in northern China. Aquaculture 2006, 258, 452–461. [Google Scholar] [CrossRef]
- Wang, X.Y.; Zhou, Y.; Yang, H.S.; Wang, Q.; Liu, S. Investigation of heavy metals in sediments and Manila clams Ruditapes philippinarum from Jiaozhou Bay, China. Environ. Monit. Assess. 2010, 170, 631–643. [Google Scholar] [CrossRef]
- Usero, J.; Gonzalez-Regalado, E.; Gracia, I. Trace metals in the bivalve molluscs Ruditapes decussatus and Ruditapes philippinarum from the Atlantic Coast of Southern Spain. Environ. Int. 1997, 23, 291–298. [Google Scholar] [CrossRef]
- Ji, J.Y.; Choi, H.J.; Ahn, I.Y. Evaluation of Manila clam Ruditapes philippinarum as a sentinel species for metal pollution monitoring in estuarine tidal flats of Korea: Effects of size, sex, and spawning on baseline accumulation. Mar. Pollut. Bull. 2006, 52, 447–453. [Google Scholar] [CrossRef]
- Ra, K.; Kim, K.T.; Bang, J.H.; Lee, J.M.; Kim, E.S.; Cho, S.R. A preliminary study of environmental impact assessment of tidal power plant in Shihwa Lake, Korea: Heavy metal accumulation in the transplanted Manila clam (Ruditapes philippinarum). J. Coast. Res. 2011, 64, 932–936. [Google Scholar]
- Sfriso, A.; Argese, E.; Bettiol, C.; Facca, C. Tapes philippinarum seed exposure to metals in polluted areas of the Venice lagoon. Estuar. Coast. Shelf. Sci. 2008, 79, 581–590. [Google Scholar] [CrossRef]
- Onsanit, S.; Ke, C.H.; Wang, X.H.; Wang, K.J.; Wang, W.X. Trace elements in two marine fish cultured in fish cages in Fujian province, China. Environ. Pollut. 2010, 158, 1334–1342. [Google Scholar] [CrossRef]
- Joint Food and Agriculture Organization/World Health Organization Expert Committee on Food Additives. Summary and Conclusions of the 61st Meeting of the Joint FAO/WHO Expert Committee on Food Additives. In Proceedings of JECFA/61/Sc, Rome, Italy, 10–19 June 2003; pp. 1–22.
- Risk-based Concentration Table; United States Environmental Protection Agency: Philadelphia, PA, USA, 2011.
- FAO, Food Security Statistics, Food Consumption; Statistics Division, Food and Agricultural Organization of the United Nations, Rome, Italy, 2008.
- Gu, Y.; He, J.; Duan, X.F.; Reynolds, K.; Wu, X.; Chen, J.; Huang, G.; Chen, C.S.; Whelton, P.K. Body weight and mortality among men and women in China. J. Am. Med. Assoc. 2006, 295, 776–783. [Google Scholar] [CrossRef]
- Ng, J.C. Environmental contamination of arsenic and its toxicological impact on humans. Environ. Chem. 2005, 2, 146–160. [Google Scholar] [CrossRef]
- United States Food and Drug Administration, Guidance Documents for Trace Elements in Seafood; Center for Food Safety and Applied Nutrition: Washington, DC, USA, 1993.
- Liang, L.N.; He, B.; Jiang, G.B.; Chen, D.Y.; Yao, Z.W. Evaluation of mollusks as biomonitors to investigate heavy metal contaminations along the Chinese Bohai Sea. Sci. Total. Environ. 2005, 324, 105–113. [Google Scholar]
- Zheng, N.; Liu, J.; Wang, Q.; Liang, Z. Health risk assessment of heavy metal exposure to street dust in the zinc smelting district, Northeast of China. Sci. Total. Environ. 2010, 408, 726–733. [Google Scholar] [CrossRef]
- Zheng, N.; Wang, Q.; Zheng, D. Health risk of Hg, Pb, Cd, Zn, and Cu to the inhabitants around Huludao Zinc Plant in China via consumption of vegetables. Sci. Total. Environ. 2007, 383, 81–89. [Google Scholar] [CrossRef]
- Wang, Y.W.; Liang, L.N.; Shi, J.B.; Jiang, G.B. Chemometrics methods for the investigation of methylmercury and total mercury contamination in mollusks samples collected from coastal sites along the Chinese Bohai Sea. Environ. Pollut. 2005, 135, 457–467. [Google Scholar] [CrossRef]
- Ma, H.; Song, Q.; Wang, X. Accumulation of petroleum hydrocarbons and heavy metals in clams (Ruditapes philippinarum) in Jiaozhou Bay, China. Chin. J. Oceanol. Limnol. 2009, 27, 887–897. [Google Scholar] [CrossRef]
- Zhu, X.; Xu, W.; Wang, X.; Huang, X.; Deng, L.; Kang, X. Research on heavy metals in Ruditapes philippinarum and soda industry wastes. Chin. J. Oceanol. Limnol. 2005, 23, 39–42. [Google Scholar] [CrossRef]
- Li, Y.; Yu, Z.; Song, X.; Mu, Q. Trace metal concentrations in suspended particles, sediments and clams (Ruditapes philillinarum) from Jiaozhou Bay of China. Environ. Monit. Assess. 2006, 121, 491–501. [Google Scholar]
- Asante, K.A.; Agusa, T.; Mochizuki, H.; Ramu, K.; Inoue, S.; Kubodera, T.; Takahashi, S.; Subramanian, A.; Tanabe, S. Trace elements and stable isotopes (δ13C and δ15N) in shallow and deep-water organisms from the East China Sea. Environ. Pollut. 2008, 156, 862–873. [Google Scholar] [CrossRef]
- Müller, B.; Berg, M.; Yao, Z.P.; Zhang, X.F.; Wang, D.; Pfluger, A. How polluted is the Yangtze river? Water quality downstream from the Three Gorges Dam. Sci. Total. Environ. 2008, 402, 232–247. [Google Scholar] [CrossRef]
- Lin, F.J.; Hsu, S.C.; Jeng, W.L. Lead in the southern East China Sea. Mar. Environ. Res. 2000, 49, 329–342. [Google Scholar] [CrossRef]
- Fang, Z.Q.; Cheung, R.Y.H.; Wong, M.H. Heavy metal concentrations in edible bivalves and gastropods available in major markets of the Pearl River Delta. J. Environ. Sci. China 2001, 13, 210–217. [Google Scholar]
- Fang, Z.Q.; Cheung, R.Y.H.; Wong, M.H. Heavy metals in oysters, mussels and clams collected from coastal sites along the Pearl River Delta, South China. J. Environ. Sci. China 2003, 15, 9–24. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yang, F.; Zhao, L.; Yan, X.; Wang, Y. Bioaccumulation of Trace Elements in Ruditapes philippinarum from China: Public Health Risk Assessment Implications. Int. J. Environ. Res. Public Health 2013, 10, 1392-1405. https://doi.org/10.3390/ijerph10041392
Yang F, Zhao L, Yan X, Wang Y. Bioaccumulation of Trace Elements in Ruditapes philippinarum from China: Public Health Risk Assessment Implications. International Journal of Environmental Research and Public Health. 2013; 10(4):1392-1405. https://doi.org/10.3390/ijerph10041392
Chicago/Turabian StyleYang, Feng, Liqiang Zhao, Xiwu Yan, and Yuan Wang. 2013. "Bioaccumulation of Trace Elements in Ruditapes philippinarum from China: Public Health Risk Assessment Implications" International Journal of Environmental Research and Public Health 10, no. 4: 1392-1405. https://doi.org/10.3390/ijerph10041392




