Pesticide Flow Analysis to Assess Human Exposure in Greenhouse Flower Production in Colombia
Abstract
:1. Introduction
- (1)
- How can the material flow analysis methodology be adapted to study human exposure to pesticides in agricultural systems?
- (2)
- What are the advantages and disadvantages of using this methodology in the field of human exposure and risk assessment of pesticide use?
- (3)
- Based on the model outputs, what is the current situation with respect to human exposure to pesticides in the flower crop systems in Colombia, and how can the management of human exposure to pesticides be improved?
2. Methodology
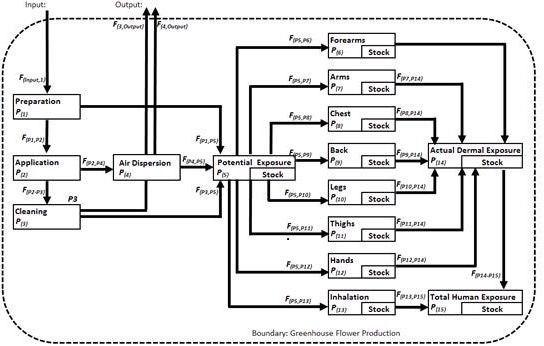
2.1. Material Flow Analysis



2.2. Description of the Study Area

2.3. Data Measurement
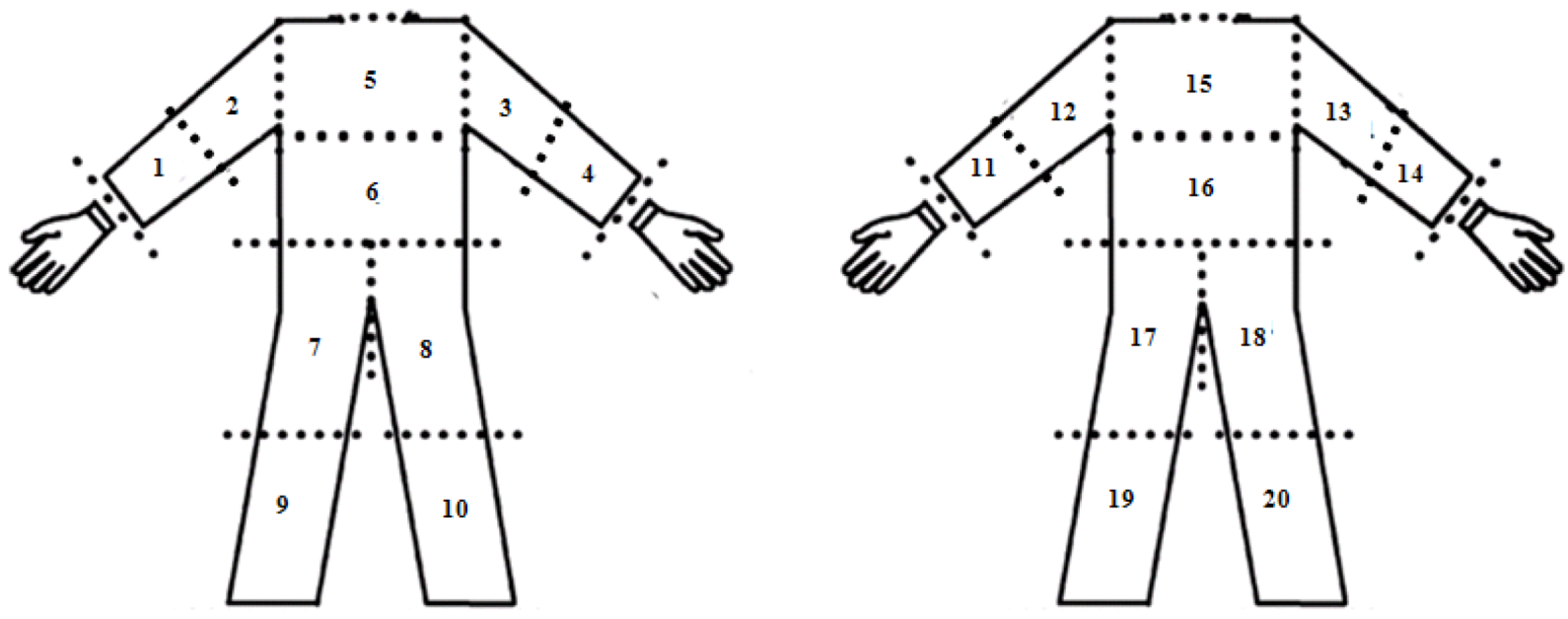
2.3.1. Dermal Exposure Measurement
| Commercial Name | Active Ingredient | Chemical Group | % of Active Ingredient | Dose | Total AI Applied (g/d) | Confirmed Health Effects [58] | Possible Health Effects [58] |
|---|---|---|---|---|---|---|---|
| Bavistin | Carbendazim | Benzimidazole | 50% | 0.6 g/L | 728 | Reproduction/development effects | Endocrine disrupter |
| Carbovax | Carboxin | Oxathiin | 20% | 1 g/L | 447 | Eye irritant | Carcinogen, reproductive/development effects |
| Thiram | Dithiocarbamate | 20% | 1 g/L | 447 | No information available | Carcinogen, mutagen, endocrine disrupter, reproduction/development effects, respiratory tract, eye and skin irritant | |
| Dithane | Mancozeb | Dithiocarbamate | 100% | 2 cc/L | 2400 | Carcinogen, respiratory tract irritant, reproduction/development effects | Mutagen, endocrine disrupter, skin irritant |
| Forum | Dimethomorph | Morpholine | 50% | 0.7 g/L | 878 | Respiratory tract, eye and skin Irritant | Reproductive/development effects |
| Previcur | Propamocarb Hydrochloride | Carbamate | 53% | 1.8 g/L | 2,365 | Skin irritant | Acetyl cholinesterase inhibitor |
| Fosetyl | Organophosphate | 31% | 1.8 g/L | 1,383 | Eye irritant, reproduction/development effects | Carcinogen, acetyl cholinesterase inhibitor, neurotoxicant | |
| Revus | Mandipropamid | Mandelamide | 25% | 0.8 g/L | 480 | Skin irritant | No information available |





2.3.2. Inhalation Exposure Measurement
2.3.3. Exposure Assessment in the Study Region
3. Results
3.1. Pesticide Flow Analysis

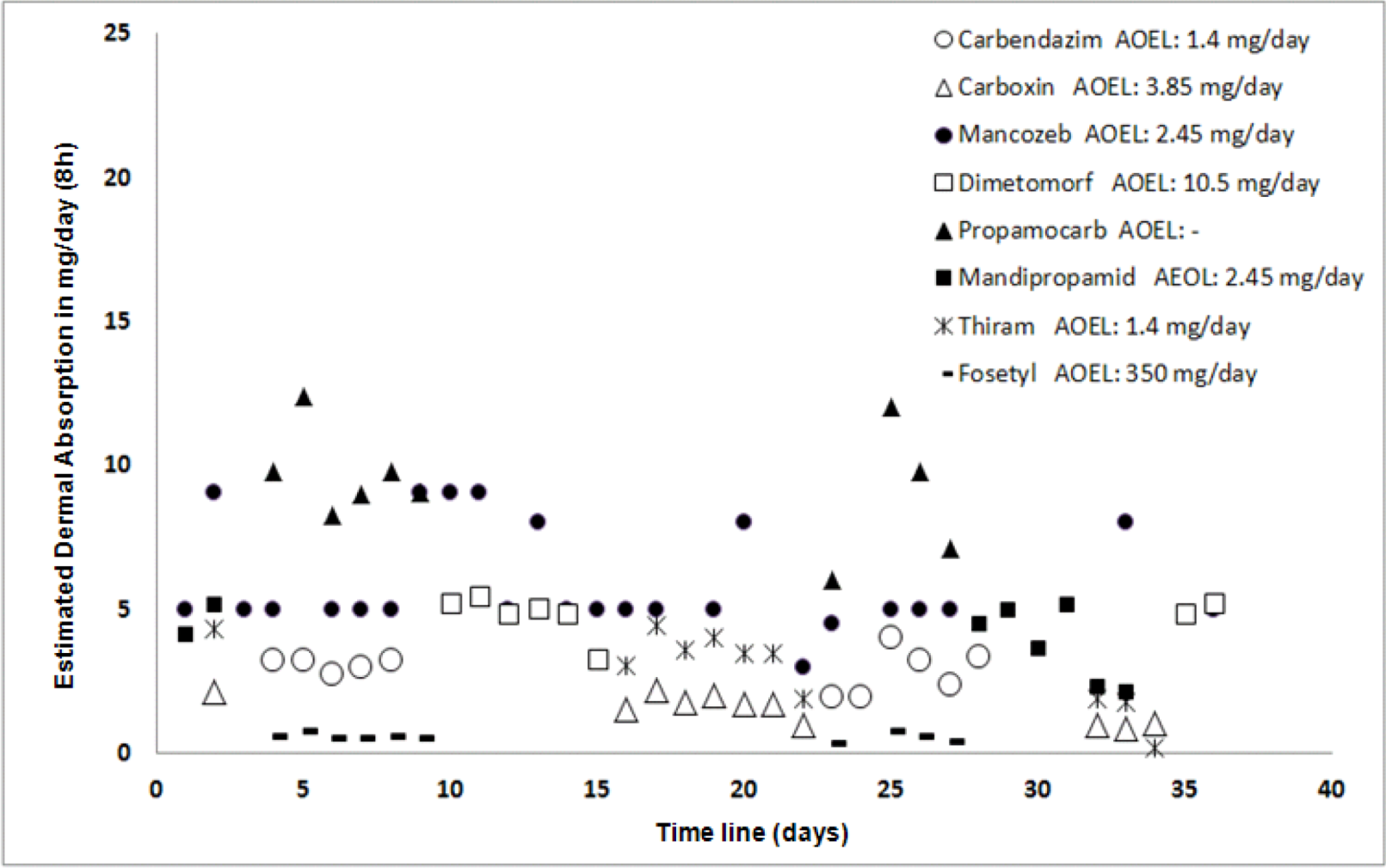
3.2. Health Risk in the Study Area
| Commercial Name | Active Ingredient (AI) | * Average Applied/ Operator (cc/d) | Actual Dermal Exposure (mg/d) | Inhalation Exposure | Dermal Absorption (%) [58] | Estimated Pesticide Absorbed (mg/d) | AOEL (mg/d) |
|---|---|---|---|---|---|---|---|
| Bavistin | Carbendazim | 485 | 20.2 ± 14.2 | 0.03 ± 0.02 | 10 | 2.0 ± 1.4 | 1.4 |
| Carbovax | Carboxin | 716 | 29,2 ± 21.0 | 0.05 ± 0.03 | 5 | 1.5 ± 2.1 | 3.85 |
| Thiram | 745 | 31.1 ± 21.9 | 0.05 ± 0.03 | 10 | 3.1 ± 2.1 | 1.4 | |
| Dithane | Mancozeb | 786 | 32.8 ± 23.1 | 0.05 ± 0.03 | 11 | 3.6 ± 2.5 | 2.45 |
| Forum | Dimethomorph | 585 | 24.4 ± 17.2 | 0.04 ± 0.03 | 20 | 4.8 ± 3.4 | 10.5 |
| Previcur | Propamocarb | 1,480 | 61.9 ± 43.5 | 0.09 ± 0.06 | 10 | 6.1 ± 4.3 | - |
| Fosetyl | 1,488 | 61.9 ± 43.5 | 0.09 ± 0.06 | 1 | 0.6 ± 0.4 | 350 | |
| Revus | Mandipropamide | 640 | 26.7 ± 18.8 | 0.04 ± 0.03 | 10 | 2.6 ± 1.8 | 2.45 |
4. Discussion
4.1. Pesticide Flow Analysis Approach
4.2. Pesticide Management in the Case Study
| Body Parts | PDE (% in Body) | |||
|---|---|---|---|---|
| 1. Spray Sideways with 5 Nozzles | 2. Spray Gun Downward | 3. Spray Lance Forward | 4. Spray Lance Backward | |
| Back | 13.1 | 0.5 | 0.8 | 1.4 |
| Chest | 19.5 | 0.8 | 1.5 | 1.9 |
| Arm | 17.7 | 18.8 | 10.0 | 6.0 |
| Forearm | 15.7 | 13.3 | 7.3 | 10.0 |
| Thighs | 15.2 | 12.6 | 11.3 | 8.1 |
| Legs | 15.9 | 46.7 | 55.1 | 27.0 |
| Hands | 3.0 | 7.3 | 14.0 | 45.6 |
| Total | 100.0 | 100.0 | 100.0 | 100.0 |
4.3. Health Risk in the Study Area
5. Conclusions
Acknowledgements
Appendix
| PDE | ADE | Stock | |||||
|---|---|---|---|---|---|---|---|
| Body Parts | |||||||
| Forearms (n = 9) | 1.84E−05 ± 7.57E−06 | 1.43E−07 ± 8.83E−08 | 1.83E−05 ± 7.48E−06 | ||||
| Arms (n = 9) | 2.07E−05 ± 1.01E−05 | 6.10E−08 ± 4.19E−08 | 2.06E−05 ± 1.00E−05 | ||||
| Chest & Abdomen (n = 9) | 2.28E−05 ± 8.37E−06 | 8.94E−08 ± 5.30E−08 | 2.27E−05 ± 8.32E−06 | ||||
| Back (n = 9) | 1.53E−05 ± 6.24E−06 | 6.47E−08 ± 4.37E−08 | 1.52E−05 ± 6.20E−06 | ||||
| Thighs (n = 9) | 1.77E−05 ± 8.63E−06 | 7.95E−08 ± 5.81E−08 | 1.77E−05 ± 8.57E−06 | ||||
| Legs (n = 9) | 1.86E−05 ± 1.22E−05 | 1.16E−07 ± 6.72E−08 | 1.85E−05 ± 1.21E−05 | ||||
| Hands (n = 9) | 3.48E−06 ± 2.92E−06 | 1.79E−07 ± 1.62E−07 | 3.30E−06 ± 2.76E−06 | ||||
| Total Dermal (n = 9) | 1.17E−04 ± 5.60E−05 | 7.32E−07 ± 5.14E−07 | 1.16E−04 ± 5.55E−05 | ||||
| Inhalation (n = 12) | 2.31E−08 ± 1.80E−08 | 1.10E−09 ± 8.50E−10 | 2.20E−08 ± 1.72E−08 | ||||
| Pesticide Management Activities | |||||||
| Preparation (n = 3) | 4.67E−06 ± 3.21E−06 | ||||||
| Application (n = 9) | 1.10E−04 ± 5.16E−05 | ||||||
| Cleaning (n = 3) | 1.92E−06 ± 1.18E−06 | ||||||
References
- De Roos, A.J.; Zahm, S.H.; Cantor, K.P.; Weisenburger, D.D.; Holmes, F.F.; Burmeister, L.F.; Blair, A. Integrative assessment of multiple pesticides as risk factors for non-Hodgkin’s lymphoma among men. Occup. Environ. Med. 2003, 60. [Google Scholar] [CrossRef]
- Hardell, L.; Eriksson, M.; Nordström, M. Exposure to pesticides as risk factor for Non-Hodgkin’s lymphoma and hairy cell leukemia: Pooled analysis of two Swedish case-control studies. Leuk. Lymphoma 2002, 43, 1043–1049. [Google Scholar]
- Infante-Rivard, C.; Sinnett, D. Preconceptional paternal exposure to pesticides and increased risk of childhood leukaemia. Lancet 1999, 354, 1819. [Google Scholar]
- Richter, E.D.; Chlamtac, N. Ames, pesticides, and cancer revisited. Int. J. Occup. Environ. Health 2002, 8, 63–72. [Google Scholar]
- Baldi, I.; Cantagrel, A.; Lebailly, P.; Tison, F.; Dubroca, B.; Chrysostome, V.; Dartigues, J.F.; Brochard, P. Association between Parkinson’s disease and exposure to pesticides in southwestern France. Neuroepidemiology 2003, 22, 305–310. [Google Scholar]
- Baldi, I.; Lebailly, P.; Mohammed-Brahim, B.; Letenneur, L.; Dartigues, J.F.; Brochard, P. Neurodegenerative diseases and exposure to pesticides in the elderly. Am. J. Epidemiol. 2003, 157, 409–414. [Google Scholar]
- Elbaz, A.; Levecque, C.; Clavel, J.; Vidal, J.S.; Richard, F.; Amouyel, P.; Alpérovitch, A.; Chartier-Harlin, M.C.; Tzourio, C. CYP2D6 polymorphism, pesticide exposure, and Parkinson’s Disease. Ann. Neurol. 2004, 55, 430–434. [Google Scholar]
- Salameh, P.R.; Baldi, I.; Brochard, P.; Raherison, C.; Abi Saleh, B.; Salamon, R. Respiratory symptoms in children and exposure to pesticides. Eur. Respir. J. 2003, 22, 507–512. [Google Scholar]
- Weidner, I.S.; Møller, H.; Jensen, T.K.; Skakkebæk, N.E. Cryptorchidism and hypospadias in sons of gardeners and farmers. Environ. Health Perspect. 1998, 106, 793–796. [Google Scholar]
- Bell, E.M.; Hertz-Picciotto, I.; Beaumont, J.J. Case-cohort analysis of agricultural pesticide applications near maternal residence and selected causes of fetal death. Am. J. Epidemiol. 2001, 154, 702–710. [Google Scholar]
- Garry, V.F.; Harkins, M.E.; Erickson, L.L.; Long-Simpson, L.K.; Holland, S.E.; Burroughs, B.L. Birth defects, season of conception, and sex of children born to pesticide applicators living in the Red River Valley of Minnesota, USA. Environ. Health Perspect. 2002, 110, 441–449. [Google Scholar]
- Garry, V.F.; Holland, S.E.; Erickson, L.L.; Burroughs, B.L. Male reproductive hormones and thyroid function in pesticide applicators in the Red River Valley of Minnesota. J. Toxicol. Environ. Health Part A 2003, 66, 965–986. [Google Scholar]
- Hanke, W.; Jurewicz, J. The risk of adverse reproductive and developmental disorders due to occupational pesticide exposure: An overview of current epidemiological evidence. Int. J. Occup. Med. Environ. Health 2004, 17, 223–243. [Google Scholar]
- Glass, C.R.; Machera, K. Evaluating the risks of occupational pesticide exposure. Hell. Plant Prot. J. 2009, 2, 1–9. [Google Scholar]
- Illing, H.P.A. Is working in greenhouses healthy? Evidence concerning the toxic risks that might affect greenhouse workers. Occup. Med. 1997, 47, 281–293. [Google Scholar] [CrossRef]
- Ribeiro, M.G.; Colasso, C.G.; Monteiro, P.P.; Filho, W.R.P.; Yonamine, M. Occupational safety and health practices among flower greenhouses workers from Alto Tietê region (Brazil). Sci. Total Environ. 2012, 416, 121–126. [Google Scholar] [CrossRef]
- ASOCOFLORES, Colombian Florriculture Report. In Colombian Association of Flower Exporters; Santafé de Bogotá, Colombia, 2010.
- Restrepo, M.; Munoz, N.; Day, N.E.; Parra, J.E.; de Romero, L.; Nguyen-Dinh, X. Prevalence of adverse reproductive outcomes in a population occupationally exposed to pesticides in Colombia. Scand. J. Work Environ. Health 1990, 16, 232–238. [Google Scholar] [CrossRef]
- Restrepo, M.; Muñoz, N.; Day, N.; Hernandez, C.; Blettner, M.; Giraldo, A. Birth defects among children born to a population occupationally exposed to pesticides in Colombia. Scand. J. Work Environ. Health 1990, 16, 239–246. [Google Scholar] [CrossRef]
- Cerrillo, I.; Olea-Serrano, M.F.; Ibarluzea, J.; Exposito, J.; Torne, P.; Laguna, J.; Pedraza, V.; Olea, N. Environmental and lifestyle factors for organochlorine exposure among women living in Southern Spain. Chemosphere 2006, 62, 1917–1924. [Google Scholar] [CrossRef]
- Costa, C.; Silva, S.; Coelho, P.; Roma-Torres, J.; Teixeira, J.P.; Mayan, O. Micronucleus analysis in a Portuguese population exposed to pesticides: Preliminary survey. Int. J. Hyg. Environ. Health 2007, 210, 415–418. [Google Scholar] [CrossRef]
- Herńndez, A.F.; Mackness, B.; Rodrigo, L.; López, O.; Pla, A.; Gil, F.; Durrington, P.N.; Pena, G.; Parrón, T.; Serrano, J.L.; Mackness, M.I. Paraoxonase activity and genetic polymorphisms in greenhouse workers with long term pesticide exposure. Hum. Exp. Toxicol. 2003, 22, 565–574. [Google Scholar] [CrossRef]
- Jurewicz, J.; Hanke, W.; Sobala, W.; Ligocka, D. Dermal exposure to pesticides among women working in Polish greenhouses using cotton patches (Ekspozycja dermalna na pestycydy kobiet pracujacych w gospodarstwach ogrodniczych - Wyniki badań z wykorzystaniem próbników bawełnianych). Med Pr. 2008, 59, 197–202. (in Polish). [Google Scholar]
- Machera, K.; Goumenou, M.; Kapetanakis, E.; Kalamarakis, A.; Glass, C.R. Determination of potential dermal and inhalation operator exposure to malathion in greenhouses with the whole body dosimetry method. Ann. Occup. Hyg. 2003, 47, 61–70. [Google Scholar] [CrossRef]
- Monsó, E.; Magarolas, R.; Badorrey, I.; Radon, K.; Nowak, D.; Morera, J. Occupational asthma in greenhouse flower and ornamental plant growers. Am. J. Respir. Crit. Care Med. 2002, 165, 954–960. [Google Scholar]
- Rosano, A.; Gemelli, V.; Giovannelli, C.; Paciotti, G.; Sabatucci, A.; Spagnolo, A. Fertility changes in women working in greenhouses (Alterazione della fertilità nelle lavoratrici in serra). Med Lav. 2009, 100, 448–454. (in Italian). [Google Scholar]
- Gerth van Wijk, R.; Patiwael, J.A.; de Jong, N.W.; de Groot, H.; Burdorf, A. Occupational rhinitis in bell pepper greenhouse workers: Determinants of leaving work and the effects of subsequent allergen avoidance on health-related quality of life. Allergy Eur. J. Allergy Clin. Immunol. 2011, 66, 903–908. [Google Scholar] [CrossRef]
- Flores, A.P.; Berenstein, G.A.; Hughes, E.A.; Zalts, A.; Montserrat, J.M. Pesticide risk assessment in flower greenhouses in Argentina: The importance of manipulating concentrated products. J. Hazard. Mater. 2011, 189, 222–228. [Google Scholar] [CrossRef]
- Nuyttens, D.; Braekman, P.; Windey, S.; Sonck, B. Potential dermal pesticide exposure affected by greenhouse spray application technique. Pest Manag. Sci. 2009, 65, 781–790. [Google Scholar] [CrossRef]
- Ramos, L.M.; Querejeta, G.A.; Flores, A.P.; Hughes, E.A.; Zalts, A.; Montserrat, J.M. Potential Dermal Exposure in greenhouses for manual sprayers: Analysis of the mix/load, application and re-entry stages. Sci. Total Environ. 2010, 408, 4062–4068. [Google Scholar] [CrossRef]
- Lu, J.L. Risk factors to pesticide exposure and associated health symptoms among cut-flower farmers. Int. J. Environ. Health Res. 2005, 15, 161–169. [Google Scholar] [CrossRef]
- Munnia, A.; Puntoni, R.; Merlo, F.; Parodi, S.; Peluso, M. Exposure to agrochemicals and DNA adducts in Western Liguria, Italy. Environ. Mol. Mutagen. 1999, 34, 52–56. [Google Scholar] [CrossRef]
- Esechie, J.O.; Ibitayo, O.O. Pesticide use and related health problems among greenhouse workers in Batinah Coastal Region of Oman. J. Forensic Leg. Med. 2011, 18, 198–203. [Google Scholar] [CrossRef]
- Cherrie, J.W.; Tickner, J.; Friar, J. Evaluation and Further Development of the EASE Model 2.0; HSE Books: Edinburgh, United Kingdom, 2003. [Google Scholar]
- van Hemmen, J.J. EUROPOEM, a predictive occupational exposure database for registration purposes of pesticides. Appl. Occup. Environ. Hyg. 2001, 16, 246–250. [Google Scholar] [CrossRef]
- Dosemeci, M.; Alavanja, M.C.R.; Rowland, A.S.; Mage, D.; Hoar Zahm, S.; Rothman, N.; Lubin, J.H.; Hoppin, J.A.; Sandler, D.P.; Blair, A. A quantitative approach for estimating exposure to pesticides in the agricultural health study. Ann. Occup. Hyg. 2002, 46, 245–260. [Google Scholar] [CrossRef]
- van Hemmen, J.J.; Auffarth, J.; Evans, P.G.; Rajan-Sithamparanadarajah, B.; Marquart, H.; Oppl, R. RISKOFDERM: Risk assessment of occupational dermal exposure to chemicals. An introduction to a series of papers on the development of a toolkit. Ann. Occup. Hyg. 2003, 47, 595–598. [Google Scholar] [CrossRef]
- Garrod, A.N.I.; Rajan-Sithamparanadarajah, R. Developing COSHH essentials: Dermal exposure, personal protective equipment and first aid. Ann. Occup. Hyg. 2003, 47, 577–588. [Google Scholar]
- Marquart, H.; Heussen, H.; Le Feber, M.; Noy, D.; Tielemans, E.; Schinkel, J.; West, J.; van der Schaaf, D. ‘Stoffenmanager’, a web-based control banding tool using an exposure process model. Ann. Occup. Hyg. 2008, 52, 429–441. [Google Scholar] [CrossRef]
- U.S. EPA, Dermal Exposure Assessment: A Summary of EPA Approaches; EPA/600/R-07/040F; National Center for Environmental Assessment: Washington, DC, USA, 2007.
- van-Wendel-De-Joode, B.; Brouwer, D.H.; Vermeulen, R.; van Hemmen, J.J.; Heederik, D.; Kromhout, H. DREAM: A method for semi-quantitative dermal exposure assessment. Ann. Occup. Hyg. 2003, 47, 71–87. [Google Scholar] [CrossRef]
- Blanco, L.E.; Aragón, A.; Lundberg, I.; Wesseling, C.; Nise, G. The Determinants of Dermal Exposure Ranking Method (DERM): A pesticide exposure assessment approach for developing countries. Ann. Occup. Hyg. 2008, 52, 535–544. [Google Scholar] [CrossRef]
- Kromhout, H.; van Wendel de Joode, B.; van Hemmen, J. The accuracy of DERM may be a self-fulfilling DREAM. Ann. Occup. Hyg. 2008, 52, 783–784. [Google Scholar] [CrossRef]
- Teubl, S.K.; Lesmes-Fabian, C.; Binder, C.R. Evaluation of Models for Dermal Exposure Assessment in Farming Systems in Developing Countries; Karl-Franzens-University: Graz, Austria, 2012. [Google Scholar]
- Binder, C.R. Material flow analysis. In Berkshire Encyclopedia of Sustainability; Anderson, R., Ed.; Berkshire Publishing Group, LLC: Great Barrington, MA, USA, 2012; Volume 6. [Google Scholar]
- Binder, C.; Bader, H.P.; Scheidegger, R.; Baccini, P. Dynamic models for managing durables using a stratified approach: The case of Tunja, Colombia. Ecol. Econ. 2001, 38, 191–207. [Google Scholar] [CrossRef]
- Binder, C.; Schertenleib, R.; Diaz, J.; Bader, H.P.; Baccini, P. Regional water balance as a tool for water management in developing countries. Int. J. Water Resour. Dev. 1997, 13, 5–20. [Google Scholar] [CrossRef]
- Bergbäck, B.; Anderberg, S.; Lohm, U. Accumulated environmental impact: The case of cadmium in Sweden. Sci. Total Environ. 1994, 145, 13–28. [Google Scholar] [CrossRef]
- van der Voet, E.; van Egmond, L.; Kleijn, R.; Huppes, G. Cadmium in the European Community: A policy-oriented analysis. Waste Manag. Res. 1994, 12, 507–526. [Google Scholar]
- Kleijn, R.; van der Voet, E.; Udo de Haes, H.A. Controlling substance flows: The case of chlorine. Environ. Manag. 1994, 18, 523–542. [Google Scholar] [CrossRef]
- Spatari, S.; Bertram, M.; Fuse, K.; Graedel, T.E.; Shelov, E. The contemporary European zinc cycle: 1-year stocks and flows. Resour. Conserv. Recycl. 2003, 39, 137–160. [Google Scholar] [CrossRef]
- Frosch, R.A.; Clark, W.C.; Crawford, J.; Sagar, A.; Tschang, F.T.; Webber, A.; Wilson, W.R. The industrial ecology of metals: A reconnaissance [and discussion]. Philos. Trans. Math. Phys. Eng. Sci. 1997, 355, 1335–1347. [Google Scholar] [CrossRef]
- Graedel, T.E.; Bertram, M.; Fuse, K.; Gordon, R.B.; Lifset, R.; Rechberger, H.; Spatari, S. The contemporary European copper cycle: The characterization of technological copper cycles. Ecol. Econ. 2002, 42, 9–26. [Google Scholar] [CrossRef]
- Gordon, R.B.; Graedel, T.E.; Bertram, M.; Fuse, K.; Lifset, R.; Rechberger, H.; Spatari, S. The characterization of technological zinc cycles. Resour. Conser. Recycl. 2003, 39, 107–135. [Google Scholar] [CrossRef]
- Brunner, P.; Rechberger, H. Practical Handbook of Material Flow Analysis; Lewis Publishers: Boca Raton, Florida, United States of America, 2004. [Google Scholar]
- Baccini, P.; Brunner, P.H. Metabolism of the Anthroposphere—Analysis, Evaluation and Design, 2nd ed; Massachusetts Institute of Technology: Massachusstts, United States of America, 2012; pp. 81–172. [Google Scholar]
- Rajan-Sithamparanadarajah, R.; Roff, M.; Delgado, P.; Eriksson, K.; Fransman, W.; Gijsbers, J.H.J.; Hughson, G.; Mäkinen, M.; van Hemmen, J.J. Patterns of dermal exposure to hazardous substances in European Union Workplaces. Ann. Occup. Hyg. 2004, 48, 285–297. [Google Scholar] [CrossRef]
- AERU. Pesticide Properties Database. 2011. Available online: http://sitem.herts.ac.uk/aeru/footprint/en/index.htm (accessed on 1 December 2012).
- WHO, Field Surveys of Exposure to Pesticides. Standard Protocol VBC/82.1; World Health Organization of the United Nations: Geneva, Switzerland, 1982.
- Chester, G. Evaluation of agricultural worker exposure to, and absorption of, pesticides. Ann. Occup. Hyg. 1993, 37, 509–523. [Google Scholar] [CrossRef]
- Hughes, E.A.; Zalts, A.; Ojeda, J.J.; Flores, A.P.; Glass, R.C.; Montserrat, J.M. Analytical method for assessing potential dermal exposure to captan, using whole body dosimetry, in small vegetable production units in Argentina. Pest Manag. Sci. 2006, 62, 811–818. [Google Scholar] [CrossRef]
- Akesson, N.B.; Yates, W.E. Problems relating to application of agricultural chemicals and resulting drift residues. Ann. Rev. Entomol. 1964, 9, 285–318. [Google Scholar] [CrossRef]
- Lesmes-Fabian, C.; Garcia-Santos, G.; Leuenberger, F.; Nuyttens, D.; Binder, C.R. Dermal exposure assessment of pesticide use: The case of sprayers in potato farms in the colombian highlands. Sci. Total Environ. 2012, 430, 2002–2008. [Google Scholar]
- García-Santos, G.; Scheiben, D.; Binder, C.R. The weight method: A new screening method for estimating pesticide deposition from knapsack sprayers in developing countries. Chemosphere 2011, 82, 1571–1577. [Google Scholar] [CrossRef]
- Chen, M.R.; Tsai, P.J.; Wang, Y.F. Assessing inhalatory and dermal exposures and their resultant health-risks for workers exposed to polycyclic aromatic hydrocarbons (PAHs) contained in oil mists in a fastener manufacturing industry. Environ. Int. 2008, 34, 971–975. [Google Scholar] [CrossRef]
- De Schampheleire, M.; Spanoghe, P.; Brusselman, E.; Sonck, S. Risk assessment of pesticide spray drift damage in Belgium. Crop Prot. 2007, 26, 602–611. [Google Scholar] [CrossRef]
- Witschger, O.; Grinshpun, S.A.; Fauvel, S.; Basso, G. Performance of personal inhalable aerosol samplers in very slowly moving air when facing the aerosol source. Ann. Occup. Hyg. 2004, 48, 351–368. [Google Scholar] [CrossRef]
- Vitali, M.; Protano, C.; Monte, A.; Ensabella, F.; Guidotti, M. Operative modalities and exposure to pesticides during open field treatments among a group of agricultural subcontractors. Arch. Environ. Contam. Toxicol. 2009, 57, 193–202. [Google Scholar]
- Aprea, C.; Centi, L.; Santini, S.; Lunghini, L.; Banchi, B.; Sciarra, G. Exposure to Omethoate during stapling of ornamental plants in intensive cultivation tunnels: Influence of environmental conditions on absorption of the pesticide. Arch. Environ. Contam. Toxicol. 2005, 49, 577–588. [Google Scholar] [CrossRef]
- Varona, M.; Tolosa, J.; Cardenas, O.; Torres, C.; Pardo, D.; Carrasquilla, G.; Frumkin, H. Descripcion del uso y manejo de plaguicidas en las empresas de flores afiliadas a asocoflores. Biomedica 2005, 25, 377–389. (in Spanish). [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lesmes-Fabian, C.; Binder, C.R. Pesticide Flow Analysis to Assess Human Exposure in Greenhouse Flower Production in Colombia. Int. J. Environ. Res. Public Health 2013, 10, 1168-1185. https://doi.org/10.3390/ijerph10041168
Lesmes-Fabian C, Binder CR. Pesticide Flow Analysis to Assess Human Exposure in Greenhouse Flower Production in Colombia. International Journal of Environmental Research and Public Health. 2013; 10(4):1168-1185. https://doi.org/10.3390/ijerph10041168
Chicago/Turabian StyleLesmes-Fabian, Camilo, and Claudia R. Binder. 2013. "Pesticide Flow Analysis to Assess Human Exposure in Greenhouse Flower Production in Colombia" International Journal of Environmental Research and Public Health 10, no. 4: 1168-1185. https://doi.org/10.3390/ijerph10041168