Does Farming Have an Effect on Health Status? A Comparison Study in West Greece
Abstract
:1. Introduction
2. Materials and Methods
2.1. Ethics Approval
2.2. Statistical Analysis
3. Results
3.1. Study Population
| Farmers | Non-farmers | ||||||
|---|---|---|---|---|---|---|---|
| Total | Males | Females | Total | Males | Females | ||
| n = 328 | n= 196 | n = 132 | n = 347 | n = 208 | n = 139 | ||
| Age distribution (%) | <40 | 18.6 | 18.4 | 18.9 | 18.4 | 18.3 | 18.7 |
| 40–49 | 25.6 | 25.5 | 25.8 | 25.6 | 25.5 | 25.9 | |
| 50–59 | 27.4 | 27.6 | 27.3 | 27.1 | 26.9 | 27.3 | |
| 60–69 | 18.3 | 18.4 | 18.2 | 19.3 | 19.7 | 18.7 | |
| 70+ | 10.1 | 10.2 | 9.8 | 9.5 | 9.6 | 9.4 | |
| Family Status (%) | Married | 86.6 | 81.6 | 93.9 | 88.1 | 81.6 | 97.7 |
| Single | 10.1 | 14.3 | 3.8 | 11.6 | 17.9 | 2.3 | |
| Other | 3.4 | 4.1 | 2.3 | 0.3 | 0.5 | - | |
| Number of children (%) | 0 | 11.0 | 15.8 | 3.8 | 13.6 | 20.7 | 3.1 |
| 1 | 6.1 | 8.7 | 2.3 | 11.8 | 18.0 | 3.1 | |
| 2 | 27.8 | 28.6 | 26.5 | 29.4 | 25.3 | 34.6 | |
| 3 | 31.4 | 27.0 | 37.9 | 27.1 | 19.3 | 38.5 | |
| >3 | 23.8 | 19.9 | 29.5 | 18.2 | 16.6 | 20.8 | |
| Educational Level (%) | Illiterate 1 | 10.1 | 10.7 | 9.1 | 7.8 | 6.0 | 10.8 |
| Elementary | 83.2 | 82.1 | 84.8 | 60.5 | 51.6 | 75.4 | |
| Secondary | 5.5 | 5.1 | 6.1 | 15.6 | 19.4 | 9.2 | |
| Higher | 1.2 | 2.0 | - | 16.1 | 23.0 | 4.6 | |
| Income Level (%) | <9,000 € | 42.1 | 20.4 | 74.3 | 43.0 | 43.8 | 41.6 |
| 9,000–15,000 € | 31.4 | 38.8 | 20.5 | 35.1 | 32.7 | 39.2 | |
| >15,000 € | 26.5 | 40.8 | 5.3 | 21.9 | 23.5 | 19.2 | |
| Body Mass Index (%) | Normal | 17.4 | 16.8 | 18.2 | 16.7 | 12.9 | 23.1 |
| Overweight | 56.7 | 52.6 | 62.9 | 56.5 | 56.7 | 56.2 | |
| Obese | 25.9 | 30.6 | 18.9 | 26.8 | 30.4 | 20.8 | |
| Smoking (%) | Non smoker | 59.45 | 36.73 | 93.18 | 55.04 | 39.17 | 81.54 |
| Medium | 14.33 | 19.90 | 6.06 | 22.48 | 26.27 | 16.15 | |
| Heavy | 26.22 | 43.37 | 0.76 | 22.48 | 34.56 | 2.31 | |
| Alcohol Consumption (%) | Abstainer | 55.18 | 31.12 | 90.91 | 56.77 | 36.87 | 90.00 |
| Moderate | 8.54 | 11.73 | 3.79 | 11.53 | 14.75 | 6.15 | |
| Heavier | 36.28 | 57.14 | 5.30 | 31.70 | 48.39 | 3.85 | |
| Coffee Consumption (%) | No coffee | 4.57 | 6.12 | 2.27 | 5.19 | 6.45 | 3.08 |
| 1 cup/day | 14.33 | 6.63 | 25.76 | 7.49 | 7.37 | 7.69 | |
| 2 cup/day | 62.80 | 64.80 | 59.85 | 66.28 | 61.29 | 74.62 | |
| ≥3 cup/day | 18.29 | 22.45 | 12.12 | 21.04 | 24.88 | 14.62 | |
3.2. Comorbidities
| Farmers (n = 328) | Non-farmers (n=347) | ||||
|---|---|---|---|---|---|
| n | (%) | n | (%) | p | |
| None | 23 | (7.0) | 40 | (11.5) | 0.044 |
| Gastrointestinal diseases 1 | 39 | (11.9) | 44 | (12.7) | 0.755 |
| Rheumatoid arthritis | 3 | (0.9) | 3 | (0.9) | 1.000 |
| Polymyalgia rheumatica 2 | 7 | (2.1) | 9 | (2.6) | 0.695 |
| Osteoporosis | 27 | (8.2) | 24 | (6.9) | 0.518 |
| Thyroid gland disorders 3 | 15 | (4.6) | 23 | (6.6) | 0.247 |
| Diabetes mellitus | 29 | (8.8) | 32 | (9.2) | 0.863 |
| Hypertension | 89 | (27.1) | 44 | (12.7) | <0.001 |
| Other cardiovascular disorders 4 | 29 | (8.8) | 16 | (4.6) | 0.028 |
| Dislipidemia 5 | 45 | (13.7) | 47 | (13.5) | 0.947 |
| Respiratory Diseases 6 | 27 | (8.2) | 27 | (7.8) | 0.829 |
| ENT diseases 7 | 19 | (5.8) | 8 | (2.3) | 0.021 |
| Dermatological diseases 8 | 14 | (4.3) | 20 | (5.8) | 0.375 |
| Ophthalmological diseases 9 | 21 | (6.4) | 17 | (4.9) | 0.397 |
| Various orthopaedic disorders 10 | 95 | (29.0) | 43 | (12.4) | <0.001 |
| Brucellosis | 3 | (0.9) | 1 | (0.3) | 0.360 |
| Renal diseases 11 | 11 | (3.4) | 12 | (3.5) | 0.940 |
| Parkinson’s disease | 3 | (0.9) | 2 | (0.6) | 0.678 |
| Psychosis | 3 | (0.9) | 4 | (1.2) | 1.000 |
| Depression | 24 | (7.3) | 33 | (9.5) | 0.306 |
| Cancer 12 | 7 | (2.1) | 5 | (1.4) | 0.496 |
3.3. Haematological and Biochemical Blood Examinations
| Males | Females | |||||
|---|---|---|---|---|---|---|
| Farmers n = 196 | Non-farmers n = 208 | Farmers n = 132 | Non-farmers n = 139 | |||
| Median (range) | p | Median (range) | p | |||
| RBC 1 (106·μL−1) | 4.61 | 4.57 | 0.588 | 4.55 | 4.58 | 0.275 |
| (3.65–6.75) | (3.74–6.75) | (3.74–6.52) | (3.65–6.14) | |||
| Haematocrit (%) | 42.90 | 44.70 | <0.001 | 38.95 | 41.00 | <0.001 |
| (29.20–59.90) | (33.90–54.60) | (23.40–52.20) | (30.60–49.20) | |||
| Haemoglobin (g·dL−1) | 14.20 | 14.90 | <0.001 | 12.60 | 13.60 | <0.001 |
| (10.40–19.60) | (11.30–18.20) | (7.30–17.90) | (10.20–16.40) | |||
| MCV 2 (fL) | 89.20 | 89.40 | 0.993 | 90.30 | 89.20 | 0.237 |
| (58.00–106.00) | (58.00–106.00) | (8.10–102.00) | (61.00–102.00) | |||
| MCH 3 (pg) | 29.20 | 29.40 | 0.471 | 29.60 | 28.85 | 0.014 |
| (18.40–34.80) | (18.90–35.10) | (18.90–32.50) | (18.20–34.30) | |||
| WBC 4 (103·μL−1) | 7.45 | 7.50 | 0.849 | 7.50 | 6.65 | 0.010 |
| (3.50–17.00) | (3.70–17.00) | (3.10–13.50) | (3.10–13.50) | |||
| Neutrophils (103·μL−1) | 56.30 | 57.70 | 0.253 | 57.85 | 57.05 | 0.174 |
| (28.10–78.90) | (5.90–84.70) | (3.00–79.50) | (3.00–77.10) | |||
| Lymphocytes (103·μL−1) | 30.15 | 32.00 | 0.101 | 32.95 | 31.00 | 0.090 |
| (13.70–63.50) | (8.30–54.40) | (17.40–50.50) | (13.70–63.50) | |||
| Monocytes (103·μL−1) | 5.70 | 6.10 | 0.192 | 7.30 | 6.70 | 0.181 |
| (1.80–14.40) | (1.80–13.80) | (3.10–12.60) | (2.50–14.40) | |||
| Platelets (103·μL−1) | 210.00 | 213.00 | 0.426 | 223.00 | 221.00 | 0.591 |
| (10.00–365.00) | (63.00–377.00) | (65.00–377.00) | (121.00–376.00) | |||
| Farmers n = 328 | Non-farmers n = 347 | ||
|---|---|---|---|
| Median (range) | p | ||
| Glucose (mg·dL−1) | 97 | 95 | 0.155 |
| (59–250) | (59–328) | ||
| Urea (mg·dL−1) | 41 | 39 | 0.369 |
| (13–98) | (13–98) | ||
| Creatinine (mg·dL−1) | 0.8 | 0.8 | 0.614 |
| (0.5–3.8) | (0.5–5.1) | ||
| Sodium (mEq·L−1) | 139 | 139 | 0.738 |
| (123–144) | (123–439) | ||
| Potassium (mEq·L−1) | 4.3 | 4.3 | 0.024 |
| (3.6–5.2) | (3.6–5) | ||
| Calcium (mEq·L−1) | 9.3 | 8.9 | <0.001 |
| (8.4–10.2) | (8.4–10.2) | ||
| SGOT 1 (U·L−1) | 40 | 35.0 | <0.001 |
| (9–95) | (9–55) | ||
| SGPT 2 (U·L−1) | 33 | 33 | 0.214 |
| (10–387) | (10–72) | ||
| γ-GT 3 (U·L−1) | 34 | 35 | 0.679 |
| (7–206) | (7–190) | ||
| LDH 4 (U·L−1) | 186 | 169 | <0.001 |
| (112–428) | (112–257) | ||
| Amylase (U·L−1) | 69 | 65 | 0.250 |
| (24–133) | (24–125) | ||
| Alkaline phosphatase (U·L−1) | 67 | 65 | 0.140 |
| (26–137) | (26–116) | ||
| Total bilirubin (mg·dL−1) | 0.8 | 0.7 | 0.001 |
| (0.1–1.7) | (0.1–1.1) | ||
| Cholesterole (mg·dL−1) | 169 | 166 | 0.894 |
| (113–496) | (113–496) | ||
| Triglycerides (mg·dL−1) | 116.5 | 110 | 0.049 |
| (67–286) | (67–228) | ||
| Uric acid (mg·dL−1) | 4.4 | 4.6 | 0.320 |
| (2.6–8.1) | (2.6–8.1) | ||
| Total proteins (g·dL−1) | 6.9 | 6.6 | <0.001 |
| (5.9–8.8) | (5.9–8.8) | ||
| CRP 5 (mg·dL−1) | 0.6 | 0.4 | <0.001 |
| (0.1–1) | (0.1–0.8) | ||
| Pseudocholinesterase (U·mL−1) | 7.7 | 7.7 | 0.018 |
| (4.1–12.4) | (4.1–13.9) | ||
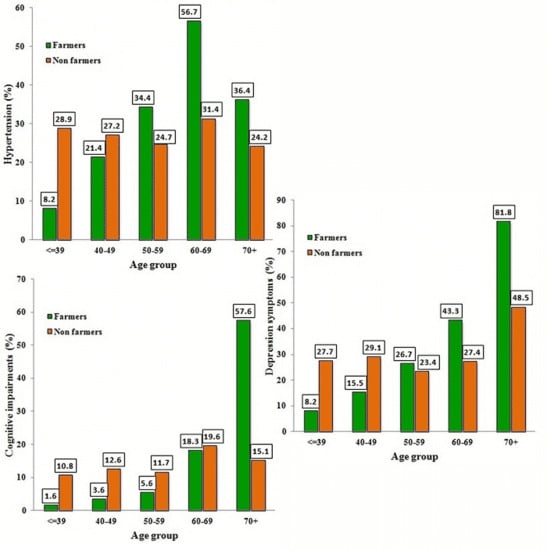
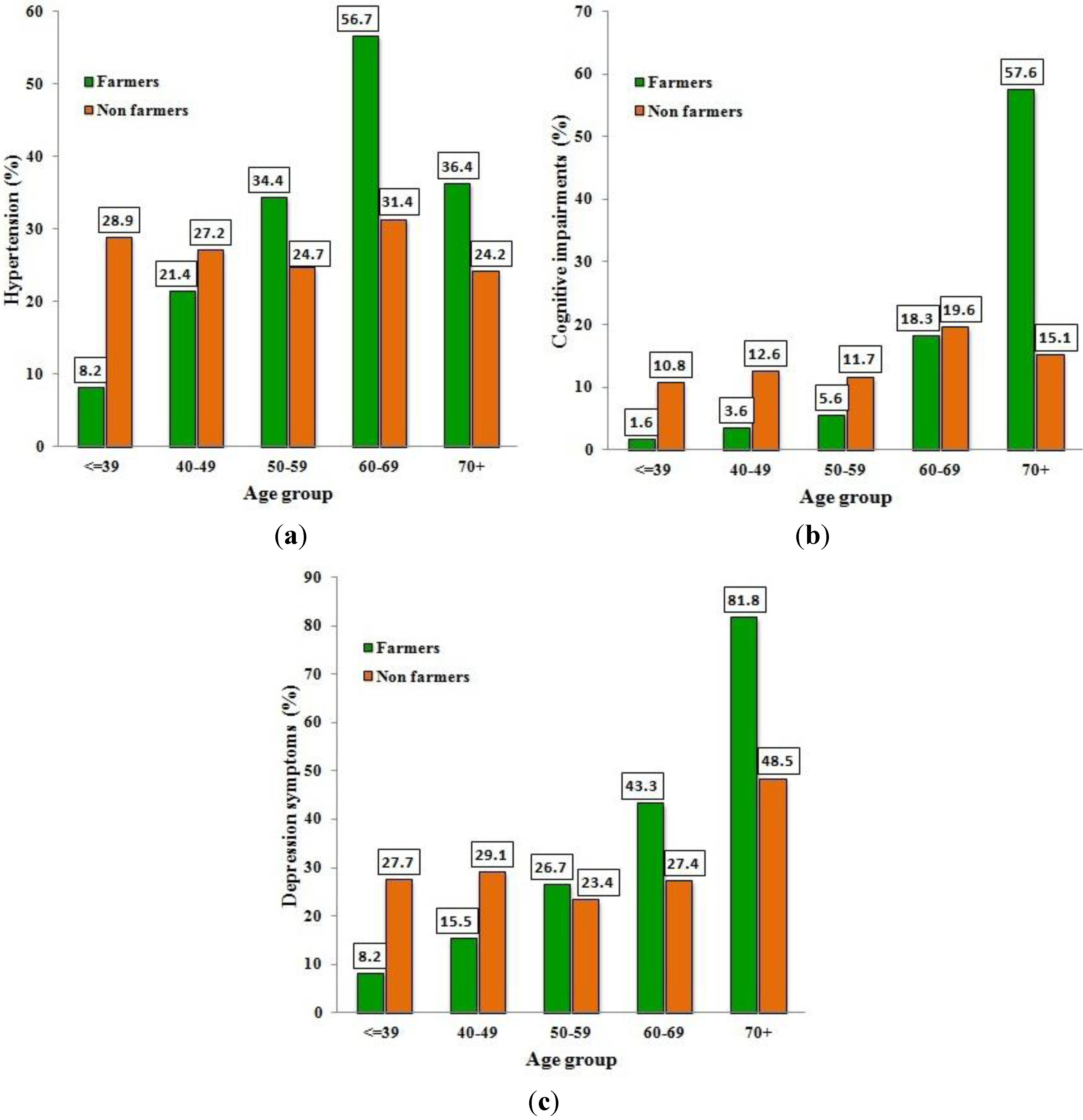
3.5. Blood Pressure and Clinical Tests
| MADRS | MMSE | HYPERTENSION | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| N 1 | Dep. 2 | N 1 | Cog. 3 | No | Yes | |||||||||||
| Factor | Coding | n | n | β | 95% C.I. 4 | p | n | n | β | 95% C.I. | p | n | n | β | 95% C.I. | p |
| Occupation (Occ) | ||||||||||||||||
| Non-farmers | Occ(0) * | 246 | 101 | 301 | 46 | 252 | 95 | |||||||||
| Farmers | Occ (1) | 233 | 95 | −0.755 | −2.209–0.699 | 0.309 | 289 | 39 | −1.857 | −3.971–0.258 | 0.085 | 228 | 100 | −1.591 | −2.633–−0.549 | 0.003 |
| Gender (Gen) | ||||||||||||||||
| Male | Gen (0) * | 311 | 102 | 376 | 37 | 281 | 132 | |||||||||
| Female | Gen (1) | 168 | 94 | −0.217 | −0.781–0.347 | 0.450 | 214 | 48 | 0.539 | −0.002–1.08 | 0.051 | 199 | 63 | −0.822 | −1.227–−0.417 | 0.000 |
| Age | 0.199 | 0.912 | 0.975 | |||||||||||||
| <40 | Age (0) * | 116 | 28 | 134 | 10 | 115 | 29 | |||||||||
| 40–49 | Age (1) | 144 | 43 | −0.226 | −0.910–0.457 | 0.516 | 171 | 16 | 0.114 | −0.838–1.065 | 0.815 | 141 | 46 | 0.009 | −0.652–0.669 | 0.979 |
| 50–59 | Age (2) | 125 | 42 | −0.581 | −1.342–0.180 | 0.135 | 153 | 14 | −0.082 | −1.122–0.958 | 0.877 | 117 | 50 | −0.046 | −0.770–0.678 | 0.900 |
| 60–69 | Age (3) | 71 | 40 | −0.308 | −1.133–0.518 | 0.465 | 90 | 21 | 0.397 | −0.641–1.436 | 0.453 | 61 | 50 | 0.156 | −0.627–0.939 | 0.696 |
| 70+ | Age (4) | 23 | 43 | 0.482 | −0.411–1.374 | 0.290 | 42 | 24 | 0.049 | −1.195–1.293 | 0.938 | 46 | 20 | −0.185 | −1.135–0.764 | 0.702 |
| Gen * Occ | ||||||||||||||||
| Gen (1) by Occ (1) | 168 | 94 | 1.103 | 0.326–1.880 | 0.005 | |||||||||||
| Age * Occ | 0.002 | 0.001 | 0.001 | |||||||||||||
| Age (1) by Occ (1) | 71 | 13 | 0.682 | −0.661–2.026 | 0.320 | 81 | 3 | 0.338 | −2.154–2.831 | 0.790 | 66 | 18 | 1.294 | 0.044–2.544 | 0.043 | |
| Age (2) by Occ (1) | 66 | 24 | 1.500 | 0.139–2.862 | 0.031 | 85 | 5 | 0.791 | −1.625–3.207 | 0.521 | 59 | 31 | 1.992 | 0.740–3.243 | 0.002 | |
| Age (3) by Occ (1) | 34 | 26 | 2.156 | 0.732–3.580 | 0.003 | 49 | 11 | 1.920 | −0.424–4.264 | 0.108 | 26 | 34 | 2.754 | 1.431–4.078 | 0.000 | |
| Age (4) by Occ (1) | 6 | 27 | 2.846 | 1.189–4.503 | 0.001 | 14 | 19 | 3.631 | 1.166–6.096 | 0.004 | 21 | 12 | 2.280 | 0.769–3.791 | 0.003 | |
| Number of children (Child) | ||||||||||||||||
| 0 | Child (0) * | 73 | 12 | 0.010 | 81 | 4 | 0.014 | |||||||||
| 1 | Child (1) | 52 | 11 | 0.166 | −0.979–1.311 | 0.776 | 57 | 6 | 0.356 | −1.009–1.720 | 0.609 | |||||
| 2 | Child (2) | 137 | 54 | 1.449 | 0.482–2.415 | 0.003 | 176 | 15 | 0.225 | −0.993–1.443 | 0.717 | |||||
| 3 | Child (3) | 132 | 63 | 1.109 | 0.121–2.097 | 0.028 | 173 | 22 | 0.285 | −0.923–1.493 | 0.644 | |||||
| ≥4 | Child (4) | 85 | 56 | 0.854 | −0.184–1.892 | 0.107 | 103 | 38 | 1.218 | 0.038–2.399 | 0.043 | |||||
| Ch.N. * Occ | 0.011 | |||||||||||||||
| Child (1) by Occ (1) | 17 | 3 | −0.426 | −2.448–1.597 | 0.680 | |||||||||||
| Child (2) by Occ (1) | 76 | 15 | −2.145 | −3.712–−0.578 | 0.007 | |||||||||||
| Child (3) by Occ (1) | 69 | 34 | −1.417 | −2.979–0.144 | 0.075 | |||||||||||
| Child (4) by Occ (1) | 40 | 38 | −0.562 | −2.175–1.050 | 0.494 | |||||||||||
| Income (Inc) | 0.043 | |||||||||||||||
| <9,000 € | Inc (0) * | 233 | 54 | 0.010 | 195 | 92 | ||||||||||
| 9,000–15,000 € | Inc (1) | 203 | 22 | −0.710 | −1.296–−0.125 | 0.017 | 168 | 57 | −0.519 | −0.941–−0.097 | 0.016 | |||||
| >15,000 € | Inc (2) | 154 | 9 | −1.001 | −1.793–−0.208 | 0.013 | 117 | 46 | −0.393 | −0.865–0.079 | 0.103 | |||||
| Smoking (Smok) | 0.092 | |||||||||||||||
| Smok (0) * | Νο | 257 | 129 | |||||||||||||
| Smok (1) | Moderate | 105 | 20 | −0.575 | −1.157–0.006 | 0.052 | ||||||||||
| Smok (2) | Heavy | 117 | 47 | 0.091 | −0.428–0.611 | 0.730 | ||||||||||
| Coffee (Coff) | 0.122 | |||||||||||||||
| No Coffee | Coff (0) * | 28 | 5 | |||||||||||||
| 1 cup /day | Coff (1) | 44 | 29 | 1.226 | 0.110–2.342 | 0.031 | ||||||||||
| 2 cups/day | Coff (2) | 311 | 125 | 0.802 | −0.207–1.812 | 0.119 | ||||||||||
| ≥3 cups/day | Coff (3) | 97 | 36 | 0.625 | −0.435–1.684 | 0.248 | ||||||||||
4. Discussion
Conflict of Interest
References
- Stiernström, E.L.; Holmberg, S.; Thelin, A.; Svärdsudd, K. Reported health status among farmers and non-farmers in nine rural districts. J. Occup. Environ. Med. 1998, 40, 917–924. [Google Scholar] [CrossRef]
- Fleming, L.E.; Bean, J.A.; Rudolph, M.; Hamilton, K. Mortality in a cohort of licensed pesticide applicators in Florida. Occup. Environ. Med. 1999, 56, 14–21. [Google Scholar] [CrossRef]
- Stiernström, E.L.; Holmberg, S.; Thelin, A.; Svärdsudd, K. A prospective study of morbidity and mortality rates among farmers and rural and urban nonfarmers. J. Clin. Epidemiol. 2001, 54, 121–126. [Google Scholar] [CrossRef]
- Wang, Y.; Hwang, S.A.; Lewis-Michl, E.L.; Fitzgerald, E.F.; Stark, A.D. Mortality among a cohort of female farm residents in New York state. Arch. Environ. Health 2003, 58, 642–648. [Google Scholar] [CrossRef]
- Thelin, N.; Holmberg, S.; Nettelbladt, P.; Thelin, A. Mortality and morbidity among farmers, nonfarming rural men, and urban referents: A prospective population-based study. Int. J. Occup. Environ. Health 2009, 15, 21–28. [Google Scholar] [CrossRef]
- Pomrehn, P.R.; Wallace, R.B.; Burmeister, L.F. Ischemic heart disease mortality in Iowa farmers: The influence of life-style. JAMA 1982, 248, 1073–1076. [Google Scholar] [CrossRef]
- Garcia-Palmieri, M.R.; Sorlie, P.D.; Havlik, R.J.; Costas, R.; Cruz-Vidal, M. Urban-rural differences in 12 year coronary heart disease mortality: The Puerto-Rico heart health program. J. Clin. Epidemiol. 1988, 41, 285–292. [Google Scholar] [CrossRef]
- Frank, A.L.; McKnight, R.; Kirkhorn, S.R.; Gunderson, P. Issues of agricultural safety and health. Annu. Rev. Publ. Health 2004, 25, 225–245. [Google Scholar] [CrossRef]
- Brumby, S.; Chandrasekara, A.; McCoombe, S.; Kremer, P.; Lewandowski, P. Cardiovascular risk factors and psychological distress in Australian farming communities. Aust. J. Rural Health 2012, 20, 131–137. [Google Scholar] [CrossRef]
- Lee, W.J.; Cha, E.S.; Moon, E.K. Disease prevalence and mortality among agricultural workers in Korea. J. Korean Med. Sci. 2010, 25, S112–S118. [Google Scholar] [CrossRef]
- Waggoner, J.K.; Kullman, G.J.; Henneberger, P.K.; Umbach, D.M.; Blair, A.; Alavanja, M.C.; Kamel, F.; Lynch, C.F.; Knott, C.; London, S.J.; Hines, C.J; Thomas, K.W.; Sandler, D.P; Lubin, J.H.; Beane Freeman, L.E.; Hoppin, J.A. Mortality in the agricultural health study, 1993–2007. Am. J. Epidemiol. 2011, 173, 71–83. [Google Scholar] [CrossRef]
- Arcury, T.; Quandt, S.A. Living and working safely: Challenges for migrant and seasonal farmworkers. N. C. Med. J. 2011, 72, 466–470. [Google Scholar]
- Nguyen, H.T.; Quandt, S.A.; Grzywacz, J.G.; Chen, H.; Galván, L.; Kitner-Triolo, M.H.; Arcury, T.A. Stress and cognitive function in Latino farmworkers. Am. J. Ind. Med. 2012, 55, 707–713. [Google Scholar] [CrossRef]
- Rural Health Series No. 9. Cat. No. PHE 97; Australian Institute of Health and Welfare (AIHW). Rural, Regional and Remote Health Indicators of Health Status and Determinants of Health; Canberra, Australia, 2008. Available online: http://www.aihw.gov.au/publication-detail/?id=6442468076 (accessed on 18 January 2013).
- Hounsome, B.; Edwards, R.T.; Hounsome, N.; Edwards-Jones, G. Psychological morbidity of farmers and non-farming population: Results from a UK survey. Community Ment. Health J. 2012, 48, 503–510. [Google Scholar] [CrossRef]
- Hellenic Republic. National Statistical Service of Greece. Available online: http://www.statistics.gr/portal/page/portal/ESYE/PAGE-themes?p_param=A1604 (accessed on 18 January 2013).
- Dufour, M.C. What is moderate drinking? Defining “drinks” and drinking levels. Alcohol Res. Health 1999, 23, 5–14. [Google Scholar]
- Folstein, M.; Folstein, S.; McHugh, P. “Mini-mental state.” A practical method for grading the cognitive state of patients for the clinician. J. Psychiatr. Res. 1975, 12, 189–198. [Google Scholar] [CrossRef]
- Pezzotti, P.; Scalmana, S.; Mastromattei, A.; Di Lallo, D. The accuracy of the MMSE in detecting cognitive impairment when administered by general practitioners: A prospective observational study. BMC Fam. Pract. 2008, 9. [Google Scholar] [CrossRef]
- Montgomery, S.A.; Åsberg, M. A new depression scale designed to be sensitive to change. Br. J. Psychiatr. 1979, 134, 382–389. [Google Scholar] [CrossRef]
- McDowell, I. Measuring Health: A Guide to Rating Scales and Questionnaires, 3rd ed; Oxford University Press: New York, NY, USA, 2006; pp. 382–387. [Google Scholar]
- Rosales, C.; Ortega, M.I.; De Zapien, J.G.; Contreras Paniagua, A.D.; Zapien, A.; Ingram, M.; Aranda, P. The US/Mexico border: A binational approach to framing challenges and constructing solutions for improving farmworkers’ lives. Int. J. Environ. Res. Public Health 2012, 9, 2159–2174. [Google Scholar] [CrossRef]
- Stark, A.; Chang, H.G.; Fitzgerald, E.; Riccardi, K.; Stone, R. A retrospective cohort study of mortality among New York State farm bureau members. Arch. Environ. Health 1987, 42, 204–212. [Google Scholar]
- Bener, A.; Lestringant, G.G.; Beshwari, M.M.; Pasha, M.A.H. Respiratory symptoms, skin disorders and serum IgE levels in farm workers. Allerg. Immunol. 1999, 31, 52–56. [Google Scholar]
- Hwang, S.A.; Gomez, M.I.; Sobotova, L.; Stark, A.D.; May, J.J.; Hallman, E.M. Predictors of hearing loss in New York farmers. Am. J. Ind. Med. 2001, 40, 23–31. [Google Scholar] [CrossRef]
- Garry, V.F.; Harkins, M.; Lyubimov, A.; Erickson, L.; Long, L. Reproductive outcomes in the women of the Red River Valley of the north. I. The spouses of pesticide applicators: pregnancy loss, age at menarche, and exposures to pesticides. J. Toxicol Environ. Health A 2002, 65, 769–786. [Google Scholar] [CrossRef]
- Arbuckle, T.E.; Lin, Z.; Mery, L.S. An exploratory analysis of the effect of pesticide exposure on the risk of spontaneous abortion in an Ontario farm population. Environ. Health Perspect. 2002, 109, 851–857. [Google Scholar] [CrossRef]
- Gerhard, I.; Daniel, V.; Link, S.; Monga, B.; Runnebaum, B. Chlorinated hydrocarbons in women with repeated miscarriages. Environ. Health Perspect. 1998, 106, 675–681. [Google Scholar] [CrossRef]
- Patil, J.A.; Patil, A.J.; Govindwar, S.P. Biochemical effects of various pesticides on sprayers of grape gardens. Indian J. Clin. Biochem. 2003, 18, 16–22. [Google Scholar] [CrossRef]
- Patil, J.A.; Patil, A.J.; Sontakke, A.V.; Govindwar, S.P. Occupational pesticides exposure of sprayers of grape gardens in western Maharashtra (India): Effects on liver and kidney function. J. Basic Clin. Physiol. Pharmacol. 2009, 20, 335–355. [Google Scholar]
- Ismail, A.A.; Rohlman, D.S.; Abdel Rasoul, G.M.; Abou Salem, M.E.; Hendy, O.M. Clinical and Biochemical Parameters of Children and Adolescents Applying Pesticides. Available online: http://www.theijoem.com/ijoem/index.php/ijoem/article/view/41/100 (accessed on 18 January 2013).
- Araoud, M.; Neffeti, F.; Douki, W.; Hfaiedh, H.B.; Akrout, M.; Hassine, M.; Najjar, M.F.; Kenani, A. Adverse effects of pesticides on biochemical and haematological parameters in Tunisian agricultural workers. J. Expo. Sci. Environ. Epidemiol. 2012, 22, 243–247. [Google Scholar] [CrossRef]
- Payán-Rentería, R.; Garibay-Chávez, G.; Rangel-Ascencio, R.; Preciado-Martínez, V.; Muñoz-Islas, L.; Beltrán-Miranda, C.; Mena-Munguía, S.; Jave-Suárez, L.; Feria-Velasco, A.; De Celis, R. Effect of chronic pesticide exposure in farm workers of a Mexico community. Arch. Environ. Occup. Health 2012, 67, 22–30. [Google Scholar] [CrossRef]
- Al-Sarar, A.S.; Abo Bakr, Y.; Al-Erimah, G.S.; Hussein, H.I.; Bayoumi, A.E. Hematological and biochemical alterations in occupationally pesticides-exposed workers of Riyadh municipality, Kingdom of Saudi Arabia. Res. J. Environ. Toxicol. 2009, 3, 179–185. [Google Scholar] [CrossRef]
- Khan, D.A.; Hashmi, I.; Mahjabeen, W.; Naqvi, T.A. Monitoring health implications of pesticide exposure in factory workers in Pakistan. Environ. Monit. Assess. 2010, 168, 231–240. [Google Scholar] [CrossRef]
- Hernandez, A.F.; Gomez, M.A.; Perez, V.; García-Lario, J.V.; Pena, G.; Gil, F.; López, O.; Rodrigo, L.; Pino, G.; Pla, A. Influence of exposure to pesticides on serum components and enzyme activities of cytotoxicity among intensive agriculture farmers. Environ. Res. 2006, 102, 70–76. [Google Scholar] [CrossRef]
- Koenig, W.; Sund, M.; Frohlich, M.; Fischer, H.G.; Löwel, H.; Döring, A.; Hutchinson, W.L.; Pepys, M.B. C-reactive protein, a sensitive marker of inflammation, predicts future risk of coronary heart disease in initially healthy middle-aged men: Results from the MONICA (monitoring trends and determinants in cardiovascular disease) Augsburg cohort study, 1984 to 1992. Circulation 1999, 99, 237–242. [Google Scholar] [CrossRef]
- Rohde, L.E.; Hennekens, C.H.; Ridker, P.M. Survey of C-reactive protein and cardiovascular risk factors in apparently healthy men. Am. J. Cardiol. 1999, 84, 1018–1022. [Google Scholar] [CrossRef]
- Sesso, H.D.; Buring, J.E.; Rifai, N.; Blake, G.J.; Gaziano, J.M.; Ridker, P.M. C-reactive protein and the risk of developing hypertension. JAMA 2003, 290, 2945–2951. [Google Scholar] [CrossRef]
- Khan, D.A.; Bhatti, M.M.; Khan, F.A.; Naqvi, S.T. Evaluation of Pesticides Induced Toxicity by Oxidative Stress and Inflammatory Biomarkers. Available online: http://www.pafmj.org/showdetails.php?id=208&t=o (accessed on 18 January 2013).
- Brega, S.M.; Vassilieff, I.; Almeida, A.; Mercadante, A.; Bissacot, D.; Cury, P.R.; Freire-Maia, D.V. Clinical, cytogenetic and toxicological studies in rural workers exposed to pesticides in Botucatu, Sao Paulo, Brazil. Cad. Saude Publica 1998, 14, 109–115. [Google Scholar]
- Ciesielski, S.; Loomis, D.P.; Mims, S.R.; Auer, A. Pesticide exposures, cholinesterase depression, and symptoms among North Carolina migrant farmworkers. Am. J. Public Health 1994, 84, 446–451. [Google Scholar]
- McLaren, S.; Challis, C. Resilience among men farmers: The protective roles of social support and sense of belonging in the depression-suicidal ideation relation. Death Stud. 2009, 33, 262–276. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Demos, K.; Sazakli, E.; Jelastopulu, E.; Charokopos, N.; Ellul, J.; Leotsinidis, M. Does Farming Have an Effect on Health Status? A Comparison Study in West Greece. Int. J. Environ. Res. Public Health 2013, 10, 776-792. https://doi.org/10.3390/ijerph10030776
Demos K, Sazakli E, Jelastopulu E, Charokopos N, Ellul J, Leotsinidis M. Does Farming Have an Effect on Health Status? A Comparison Study in West Greece. International Journal of Environmental Research and Public Health. 2013; 10(3):776-792. https://doi.org/10.3390/ijerph10030776
Chicago/Turabian StyleDemos, Konstantinos, Eleni Sazakli, Eleni Jelastopulu, Nikolaos Charokopos, John Ellul, and Michalis Leotsinidis. 2013. "Does Farming Have an Effect on Health Status? A Comparison Study in West Greece" International Journal of Environmental Research and Public Health 10, no. 3: 776-792. https://doi.org/10.3390/ijerph10030776