Assessing Weather Effects on Dengue Disease in Malaysia
Abstract
:1. Introduction
2. Materials and Methods
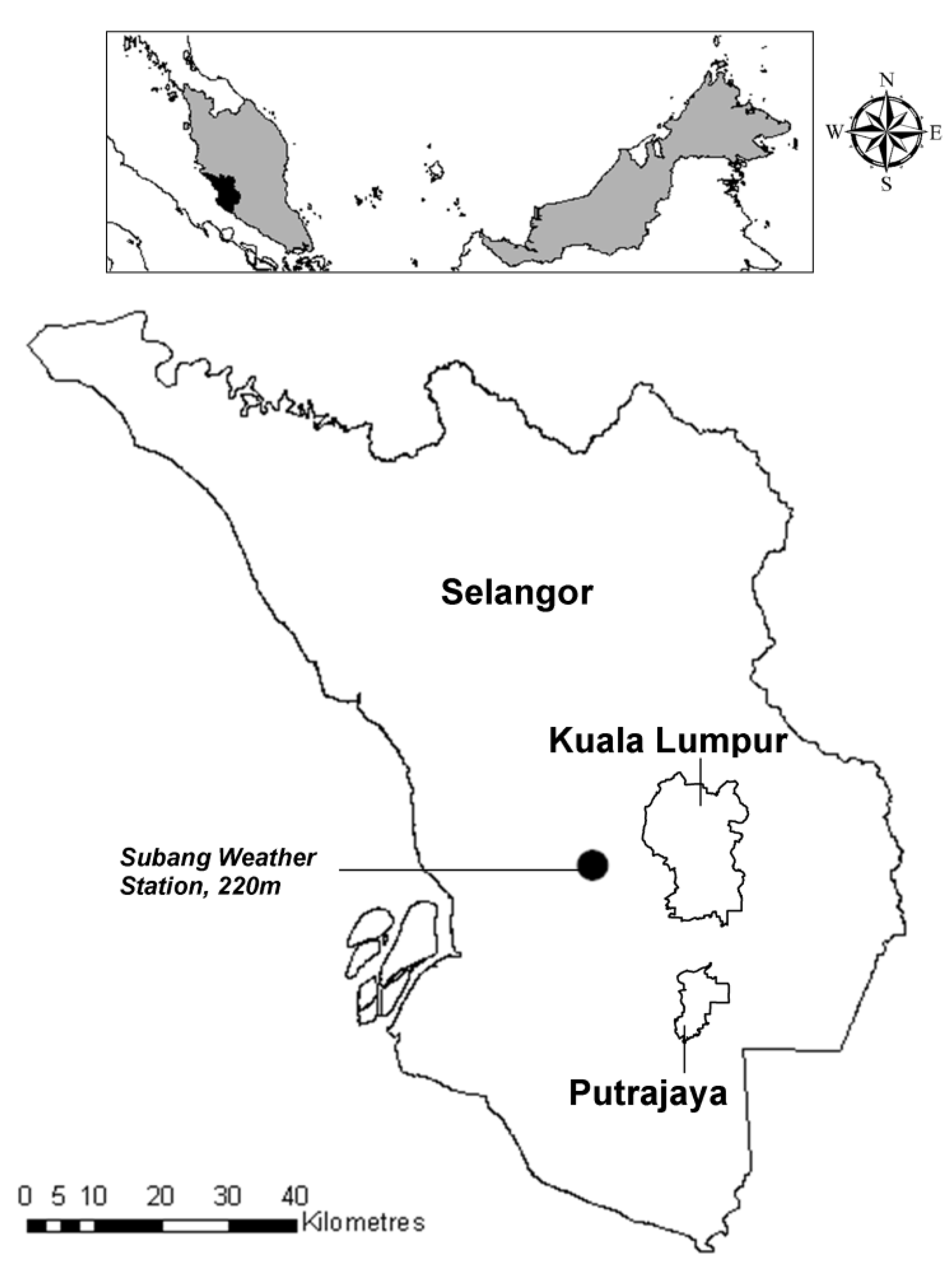
2.1. Study Area
2.2. Data
| Variables (unit) | Mean | Standard deviation | Minimum | Percentiles | |||
|---|---|---|---|---|---|---|---|
| 25th | 50th | 75th | 100th | ||||
| Daily total dengue cases | 29.4 | 13.7 | 1.0 | 20.0 | 28.0 | 38.0 | 117.0 |
| Daily minimum temperature (°C) | 24.2 | 1.0 | 20.4 | 23.5 | 24.0 | 24.9 | 27.0 |
| Daily maximum temperature (°C) | 32.9 | 1.6 | 25.4 | 32.0 | 33.0 | 34.0 | 36.4 |
| Daily mean temperature (°C) | 27.8 | 1.3 | 23.3 | 26.9 | 27.8 | 28.8 | 31.3 |
| Daily relative humidity (%) | 78.0 | 6.0 | 59.9 | 73.6 | 78.5 | 82.6 | 93.5 |
| Cumulative bi-weekly rainfall (mm) | 117.4 | 72.9 | 1.3 | 55.6 | 108.2 | 170.4 | 329.2 |
| Daily mean wind speed (knots) | 2.9 | 0.9 | 1.0 | 2.3 | 2.7 | 3.4 | 6.2 |
2.3. Statistical Analysis
3. Results
3.1. Best Model Selection and Validation
3.2. Association of Temperature and Dengue
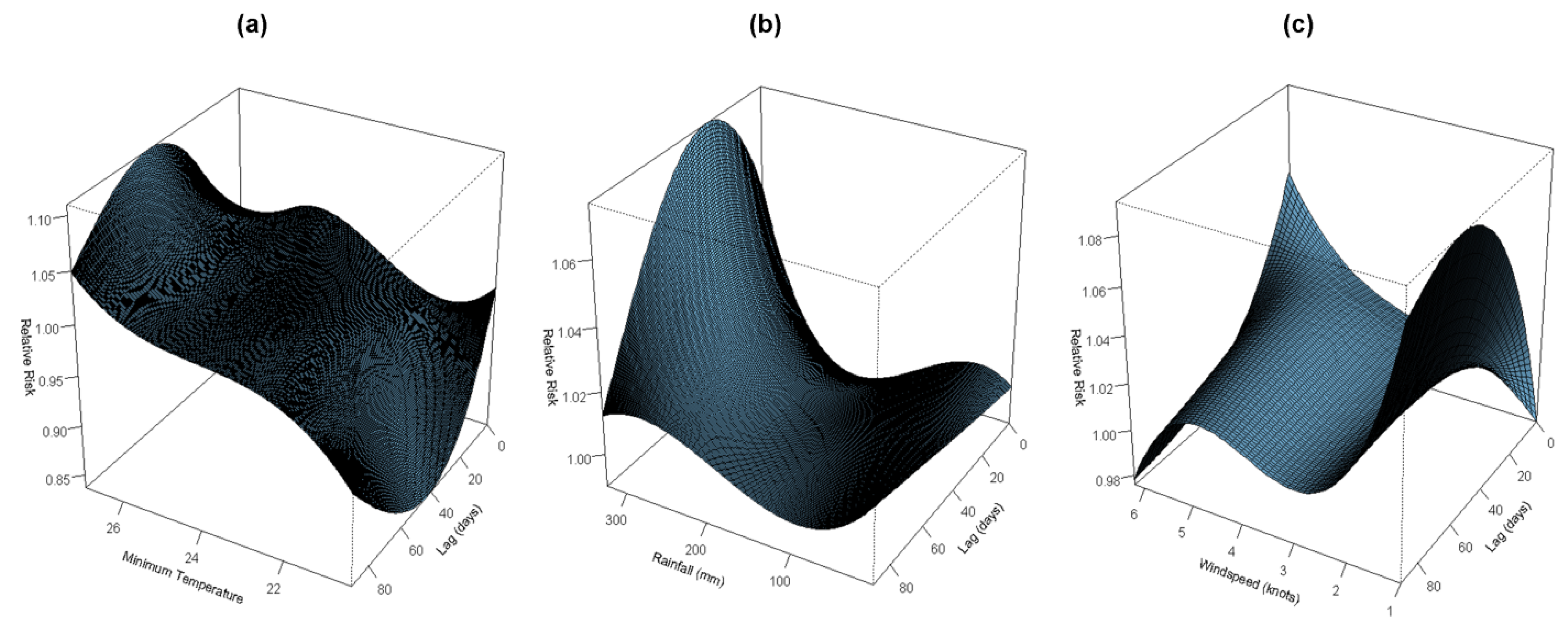
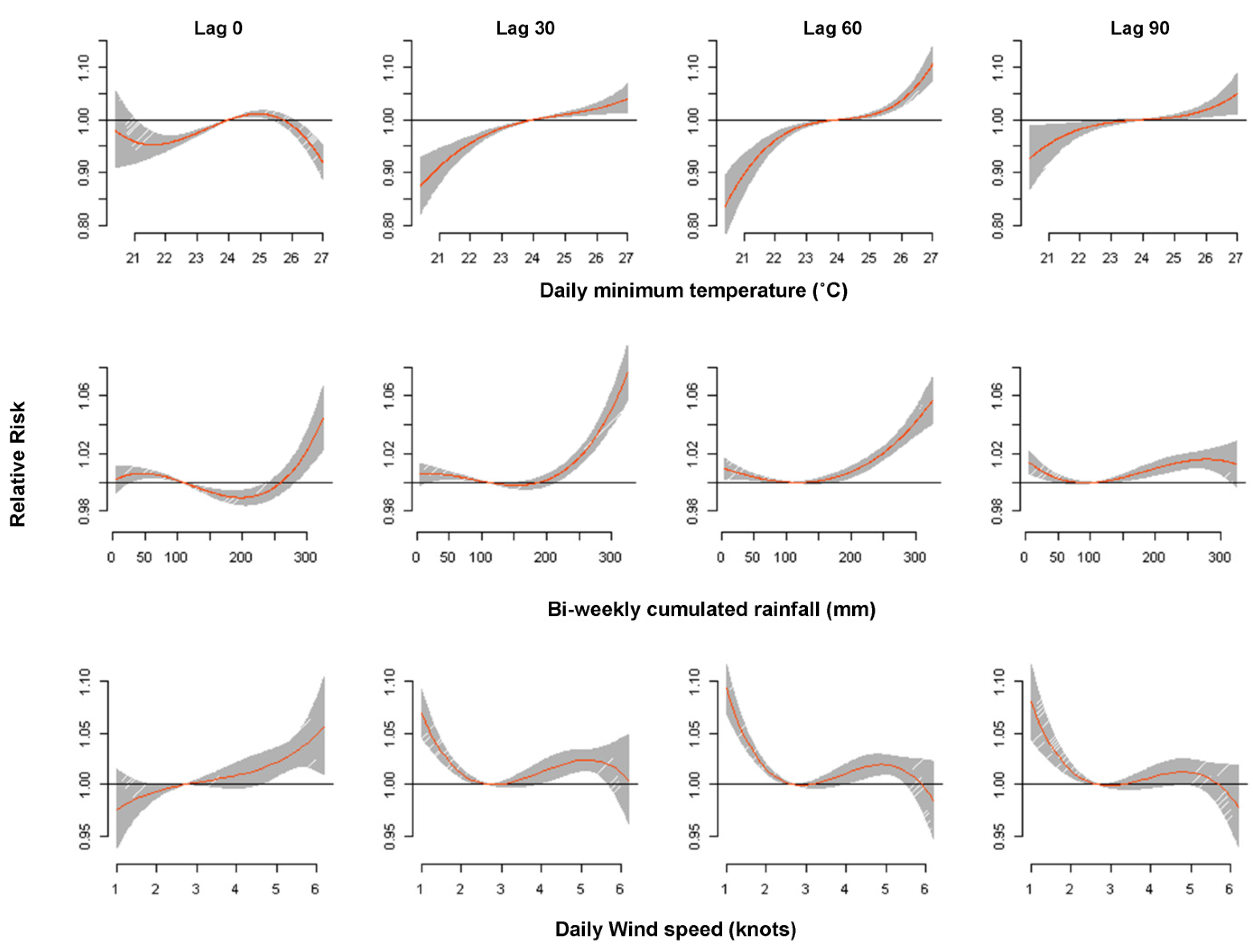
3.3. Association between Rainfall and Dengue
3.4. Association of Wind Speed and Dengue
4. Discussion
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- World Health Organization. DengueNet Database and Geographic Information System. Available online: http://www.who.int/globalatlas (accessed on 9 January 2013).
- Gubler, D. Dengue/dengue haemorrhagic fever: History and current status. Novartis Found. Symp. 2006, 277, 3–16. [Google Scholar] [CrossRef]
- Hay, S.I.; Myers, M.F.; Burke, D.S.; Vaughn, D.W.; Endy, T.; Ananda, N.; Shanks, G.D.; Snow, R.W.; Rogers, D.J. Etiology of interepidemic periods of mosquito-borne disease. Proc. Natl. Acad. Sci. USA 2000, 97, 9335–9339. [Google Scholar] [CrossRef]
- Gubler, D.J.; Rosen, L. Quantitative aspects of replication of dengue viruses in Aedes albopictus (Diptera: Culicidae) after oral and parenteral infection. J. Med. Entomol. 1977, 13, 469–472. [Google Scholar]
- Halstead, S.B. Dengue virus-mosquito interactions. Annu. Rev. Entomol. 2008, 53, 273–291. [Google Scholar] [CrossRef]
- Hales, S.; de Wet, N.; Maindonald, J.; Woodward, A. Potential effect of population and climate changes on global distribution of dengue fever: An empirical model. Lancet 2002, 360, 830–834. [Google Scholar] [CrossRef]
- Wardekker, J.; de Jong, A.; van Bree, L.; Turkenburg, W.; van der Sluijs, J. Health risks of climate change: An assessment of uncertainties and its implications for adaptation policies. Environ. Health 2012, 11, 67. [Google Scholar] [CrossRef]
- Focks, D.A.; Haile, D.G.; Daniels, E.; Mount, G.A. Dynamic life table model for Aedes aegypti (Diptera: Culicidae): Analysis of the literature and model development. J. Med. Entomol. 1993, 30, 1003–1017. [Google Scholar]
- Patz, J.; Martens, W.; Focks, A.; Jetten, T. Dengue fever epidemic potential as projected by general circulation models of global climate change. Environ. Health Perspect. 1998, 6, 147–153. [Google Scholar]
- Delatte, H.; Gimonneau, G.; Triboire, A.; Fontenille, D. Influence of temperature on immature development, survival, longevity, fecundity, and gonotrophic cycles of aedes albopictus, vector of chikungunya and dengue in the Indian Ocean. J. Med. Entomol. 2009, 46, 33–41. [Google Scholar] [CrossRef]
- Focks, D.A.; Haile, D.G.; Daniels, E.; Mount, G.A. Dynamic life table model for Aedes aegypti (diptera: Culicidae): Simulation results and validation. J. Med. Entomol. 1993, 30, 1018–1028. [Google Scholar]
- Centers for Disease Control and Prevention. Dengue and the Aedes Aegypti Mosquito. Available online: http://www.cdc.gov/dengue/resources/30Jan2012/aegyptifactsheet.pdf (accessed on 2 November 2012).
- Gubler, D.J. Dengue and dengue hemorrhagic fever. Clin. Microbiol. Rev. 1998, 11, 480–496. [Google Scholar]
- Chan, M.; Johansson, M.A. The incubation periods of dengue viruses. PLoS One 2012, 7, e50972. [Google Scholar] [CrossRef]
- World Health Organization. Dengue Guidelines for Diagnosis, Treatment, Prevention and Control; World Health Organization: Geneva, Switzerland, 2009; Volume I, pp. 1–14. [Google Scholar]
- Yang, H.M.; Macoris, M.L.G.; Galvani, K.C.; Andrighetti, M.T.M.; Wanderley, D.M.V. Assessing the effects of temperature on the population of Aedes aegypti, the vector of dengue. Epidemiol. Infect. 2009, 137, 1188–1202. [Google Scholar]
- Hales, S.; Weinstein, P.; Souares, Y.; Woodward, A. El Nino and the dynamics of vectorborne disease transmission. Environ. Health Perspect. 1999, 107, 99–102. [Google Scholar]
- Gagnon, A.S.; Bush, A.B.G.; Smoyer-Tomic, K.E. Dengue epidemics and the El Niño Southern Oscillation. Climate Res. 2001, 19, 35–43. [Google Scholar] [CrossRef]
- Cazelles, B.; Chavez, M.; McMichael, A.J.; Hales, S. Nonstationary influence of El Niño on the synchronous dengue epidemics in Thailand. PLoS Med. 2005, 2, e106. [Google Scholar] [CrossRef] [Green Version]
- Garcia, C.; Garcia, L.; Espinosa-Carreon, L.; Ley, C. Abundance and distribution of Aedes aegypti (Diptera: Culicidae), and dengue dispersion in Guasave Sinaloa, Mexico. Rev. Biol. Trop. 2011, 59, 1609–1619. [Google Scholar]
- Gomes, A.F.; Nobre, A.A.; Cruz, O.G. Temporal analysis of the relationship between dengue and meteorological variables in the city of Rio de Janeiro, Brazil, 2001–2009. Cad. Saúde Pública 2012, 28, 2189–2197. [Google Scholar] [CrossRef]
- Gharbi, M.; Quenel, P.; Gustave, J.; Cassadou, S.; Ruche, G.; Girdary, L.; Marrama, L. Time series analysis of dengue incidence in Guadeloupe, French West Indies: Forecasting models using climate variables as predictors. BMC Infect. Dis. 2011, 11, 166. [Google Scholar] [CrossRef]
- Depradine, C.A.; Lovell, E.H. Climatological variables and the incidence of Dengue fever in Barbados. Int. J. Environ. Health Res. 2004, 14, 429–441. [Google Scholar] [CrossRef]
- Bi, P.; Tong, S.; Donald, K.; Parton, K.A.; Hobbs, J. Climate Variability and the Dengue Outbreak in Townsville, Queensland, 1992–1993. Environ. Health 2001, 1, 54–60. [Google Scholar]
- Earnest, A.; Tan, S.B.; Wilder-Smith, A. Meteorological factors and El Niño Southern Oscillation are independently associated with dengue infections. Epidemiol. Infect. 2012, 140, 1244–1251. [Google Scholar] [CrossRef]
- Arcari, P.; Tapper, N.; Pfueller, S. Regional variability in relationships between climate and dengue/DHF in Indonesia. Singap. J. Trop. Geogr. 2007, 28, 251–272. [Google Scholar] [CrossRef]
- Brunkard, J.M.; Cifuentes, E.; Rothenberg, S.J. Assessing the roles of temperature, precipitation, and ENSO in dengue re-emergence on the Texas-Mexico border region. Salud Publica Mexico 2008, 50, 227–234. [Google Scholar]
- Jeefoo, P.; Tripathi, N.K.; Souris, M. Spatio-temporal diffusion pattern and hotspot detection of dengue in Chachoengsao Province, Thailand. Int. J. Environ. Res. Public Health 2010, 8, 51–74. [Google Scholar] [CrossRef]
- Chen, M.J.; Lin, C.Y.; Wu, Y.T.; Wu, P.C.; Lung, S.C.; Su, H.J. Effects of extreme precipitation to the distribution of infectious diseases in Taiwan, 1994–2008. PLoS One 2012, 7, e34651. [Google Scholar] [CrossRef]
- Yasuoka, J.; Levins, R. Ecology of vector mosquitoes in Sri Lanka--suggestions for future mosquito control in rice ecosystems. Southeast Asian J. Trop. Med. Public Health 2007, 38, 646–657. [Google Scholar]
- Lu, L.; Lin, H.; Tian, L.; Yang, W.; Sun, J.; Liu, Q. Time series analysis of dengue fever and weather in Guangzhou, China. BMC Public Health 2009, 9, 395–400. [Google Scholar] [CrossRef]
- Tipayamongkholgul, M.; Fang, C.T.; Klinchan, S.; Liu, C.M.; King, C.C. Effects of the El Niño-Southern Oscillation on dengue epidemics in Thailand, 1996–2005. BMC Public Health 2009, 9, 422. [Google Scholar] [CrossRef]
- Shang, C.S.; Fang, C.T.; Liu, C.M.; Wen, T.H.; Tsai, K.H.; King, C.C. The role of imported cases and favorable meteorological conditions in the onset of dengue epidemics. PLoS Negl. Trop. Dis. 2010, 4, e775:1–e775:9. [Google Scholar]
- Chew, M.H.; Rahman, M.M.; Jelip, J.; Hassan, M.R.; Isahak, I. All serotypes of dengue viruses circulating in Kuala Lumpur, Malaysia. Curr. Res. J. Biol. Sci. 2012, 4, 229–234. [Google Scholar]
- Chen, C.D.; Nazni, W.A.; Lee, H.L.; Seleena, B.; Mohd Masri, S.; Chiang, Y.F.; Sofian-Azirun, M. Mixed breeding of Aedes aegypti (L.) and Aedes albopictus Skuse in four dengue endemic areas in Kuala Lumpur and Selangor, Malaysia. Trop. Biomed. 2006, 23, 224–227. [Google Scholar]
- Saleeza, S.N.R.; Norma-Rashid, Y.; Sofian-Azirun, M. Mosquitoes larval breeding habitat in Urban and Suburban Areas, Peninsular Malaysia. World Acad. Sci. Eng. Technol. 2011, 58, 569–573. [Google Scholar]
- Rozilawati, H.; Zairi, J.; Adanan, C.R. Seasonal abundance of Aedes albopictus in selected urban and suburban areas in Penang, Malaysia. Trop. Biomed. 2007, 24, 83–94. [Google Scholar]
- Rohani, A.; Suzilah, I.; Malinda, M.; Anuar, I.; Mazlan, I.M.; Maszaitun, M.S.; Topek, O.; Tanrang, Y.; Ooi, S.C.; Rozilawati, H.; et al. Aedes larval population dynamics and risk for dengue epidemics in Malaysia. Trop. Biomed. 2011, 28, 237–248. [Google Scholar]
- Saifur, R.G.M.; Hassan, A.A.; Dieng, H.; Ahmad, H.; Salmah, M.R.C.; Satho, T.; Saad, A.R.; Vargas, R.E.M. Update on temporal and spatial abundance of dengue vectors in Penang, Malaysia. J Amer Mosquito Contr Assn. 2012, 28, 84–92. [Google Scholar] [CrossRef]
- Dieng, H.; Saifur, R.G.M.; Abu Hassan, A.; Salmah, M.R.C.; Boots, M.; Satho, T.; Jaal, Z.; AbuBakar, S. Indoor-breeding of Aedes albopictus in Northern Peninsular Malaysia and its potential epidemiological implications. PLoS One 2010, 5, e11790. [Google Scholar] [CrossRef]
- Li, C.F.; Lim, T.W.; Han, L.L.; Fang, R. Rainfall, abundance of Aedes aegypti and dengue infection in Selangor, Malaysia. Southeast Asian J. Trop. Med. Public Health 1985, 16, 560–568. [Google Scholar]
- Shafie, A. Evaluation of the spatial risk factors for high incidence of dengue fever and dengue hemorrhagic fever using GIS application. Sains Malays. 2011, 40, 937–943. [Google Scholar]
- Olaniyi, A.O.; Abdullah, A.M.; Ramli, M.F.; Sood, A.M. Factors affecting agricultural land use for vegetables production—A case study of the state of Selangor, Malaysia. Afr. J. Agr. Res. 2012, 7, 5939–5948. [Google Scholar]
- Ministry of Health Malaysia, Academy of Medicine of Malaysia. Clinical Practice Guidelines on Management of Dengue Infection in Adults, 2nd ed.Ministry of Health Malaysia: Putrajaya, Malaysia, 2010; pp. 11–12.
- Chadwick, D.; Arch, B.; Wilder-Smith, A.; Paton, N. Distinguishing dengue fever from other infections on the basis of simple clinical and laboratory features: Application of logistic regression analysis. J. Clin. Virol. 2006, 35, 147–153. [Google Scholar] [CrossRef]
- Krishnan, N.; Purswani, M.; Hagmann, S. Severe dengue virus infection in pediatric travelers visiting friends and relatives after travel to the Caribbean. Amer. J. Trop. Med. Hyg. 2012, 86, 474–476. [Google Scholar] [CrossRef]
- Barrera, R.; Amador, M.; MacKay, A.J. Population dynamics of Aedes aegypti and dengue as influenced by weather and human behavior in San Juan, Puerto Rico. PLoS Negl. Trop. Dis. 2011, 5, e1378. [Google Scholar] [CrossRef]
- Guo, Y.M.; Barnett, A.G.; Tong, S.L. Spatiotemporal model or time series model for assessing city-wide temperature effects on mortality? Environ. Res. 2013, 120, 55–62. [Google Scholar] [CrossRef]
- National Climatic Data Center (NCDC). Global Summary of the Day (GSOD). Available online: http://www.ncdc.noaa.gov (accessed on 15 September 2012).
- Hastie, T.; Tibshirani, R. Generalized Additive Models, 2nd ed.; Chapman & Hall/CRC Press: Boca Raton, FL, USA, 1990; pp. 1–22. [Google Scholar]
- R Development Core Team. A Language and Environment for Statistical Computing. R Foundation for Statistical Computing: Vienna, Austria, 2013. Available online: http://www.r-project.org/ (accessed on 15 April 2013).
- Wood, S.N. Fast stable direct fitting and smoothness selection for generalized additive models. J. Roy. Stat. Soc. Ser. B-Stat. Met. 2008, 70, 495–518. [Google Scholar] [CrossRef] [Green Version]
- Wood, S. Generalized Additive Mmodels: An Introduction with R; Chapman & Hall/CRC Press: Boca Raton, FL, USA, 2006; pp. 119–265. [Google Scholar]
- Ferrier, S.; Watson, G.; Pearce, J.; Drielsma, M. Extended statistical approaches to modelling spatial pattern in biodiversity in northeast New South Wales. I. Species-level modelling. Biodivers. Conserv. 2002, 11, 2275–2307. [Google Scholar] [CrossRef]
- Alimadad, A.; Salibian-Barrera, M. An outlier-robust fit for generalized additive models with applications to disease outbreak detection. J. Amer. Statist. Assn. 2011, 106, 719–731. [Google Scholar]
- Burnham, K.P.; Anderson, D.R. Multimodel inference—Understanding AIC and BIC in model selection. Sociol. Method. Res. 2004, 33, 261–304. [Google Scholar] [CrossRef]
- Armstrong, B. Models for the relationship between ambient temperature and daily mortality. Epidemiology 2006, 17, 624–631. [Google Scholar] [CrossRef]
- Gasparrini, A. Distributed lag linear and non-linear models in R: The package dlnm. J. Stat. Softw. 2011, 43, 1–20. [Google Scholar]
- Iñiguez, C.; Ballester, F.; Ferrandiz, J.; Pérez-Hoyos, S.; Sáez, M.; López, A. Relation between temperature and mortality in Thirteen Spanish cities. Int. J. Environ. Res. Public Health 2010, 7, 3196–3210. [Google Scholar] [CrossRef]
- Thach, T.-Q.; Wong, C.-M.; Chan, K.-P.; Chau, Y.-K.; Chung, Y.-N.; Ou, C.-Q.; Yang, L.; Hedley, A.J. Daily visibility and mortality: Assessment of health benefits from improved visibility in Hong Kong. Environ. Res. 2010, 110, 617–623. [Google Scholar] [CrossRef]
- Tian, Z.; Li, S.; Zhang, J.; Jaakkola, J.; Guo, Y. Ambient temperature and coronary heart disease mortality in Beijing, China: A time series study. Environ. Health 2012, 11, 56. [Google Scholar] [CrossRef]
- Zhang, F.; Krafft, T.; Ye, B.; Zhang, J.; Luo, H.; Li, M.; Zhang, Y.; Zhou, L.; Xu, J.; Wang, W. The lag effects and seasonal differences of air pollutants on allergic rhinitis in Beijing. Sci. Total Environ. 2013, 442, 172–176. [Google Scholar] [CrossRef]
- Gasparrini, A.; Armstrong, B.; Kenward, M.G. Distributed lag non-linear models. Stat. Med. 2010, 29, 2224–2234. [Google Scholar] [CrossRef]
- Luo, Y.; Zhang, Y.; Liu, T.; Rutherford, S.; Xu, Y.; Xu, X.; Wu, W.; Xiao, J.; Zeng, W.; Chu, C.; et al. Lagged effect of diurnal temperature range on mortality in a subtropical megacity of China. PLoS One 2013, 8, e55280. [Google Scholar] [CrossRef]
- Gubler, D.J.; Clark, G.G. Dengue/dengue hemorrhagic fever: The emergence of a global health problem. Emerg. Infect. Dis. 1995, 1, 55–57. [Google Scholar]
- Eisen, L.; Lozano-Fuentes, S. Use of mapping and spatial and space-time modeling approaches in operational control of aedes aegypti and dengue. PLoS Negl. Trop. Dis. 2009, 3, e411. [Google Scholar] [CrossRef]
- Adger, W.N.; Eakin, H.; Winkels, A. Nested and teleconnected vulnerabilities to environmental change. Front. Ecol. Environ. 2008, 7, 150–157. [Google Scholar] [CrossRef]
- Chen, S.C.; Liao, C.M.; Chio, C.P.; Chou, H.H.; You, S.H.; Cheng, Y.H. Lagged temperature effect with mosquito transmission potential explains dengue variability in southern Taiwan: Insights from a statistical analysis. Sci. Total Environ. 2010, 408, 4069–4075. [Google Scholar] [CrossRef]
- Wu, P.C.; Guo, H.R.; Lung, S.C.; Lin, C.Y.; Su, H.J. Weather as an effective predictor for occurrence of dengue fever in Taiwan. Acta Trop. 2007, 103, 50–57. [Google Scholar] [CrossRef]
- Pant, C.P.; Yasuno, M. Field studies on the gonotrophic cycle of Aedes aegypti in Bangkok, Thailand. J. Med. Entomol. 1973, 10, 219–223. [Google Scholar]
- Focks, D.; Daniels, E.; Haile, D.; Keesling, J. A simulation model of the epidemiology of urban dengue fever: Literature analysis, model development, preliminary validation, and samples of simulation results. Amer. J. Trop. Med. Hyg. 1995, 53, 489–506. [Google Scholar]
- Scott, T.W.; Morrison, A.C. Vector dynamics and transmission of dengue virus: Implications for dengue surveillance and prevention strategies: Vector dynamics and dengue prevention. Curr. Top. Microbiol. Immunol. 2010, 338, 115–128. [Google Scholar] [CrossRef]
- Watts, D.M.; Burke, D.S.; Harrison, B.A.; Whitmire, R.E.; Nisalak, A. Effect of temperature on the vector efficiency of Aedes aegypti for dengue 2 virus. Amer. J. Trop. Med. Hyg. 1987, 36, 143–152. [Google Scholar]
- Rowley, W.; Graham, C. The effect of temperature and relative humidity on the flight performance of female Aedes aegypti. J. Insect Physiol. 1968, 14, 1251–1257. [Google Scholar] [CrossRef]
- Moore, C.G.; Cline, B.L.; Ruiz-Tiben, E.; Lee, D.; Romney-Joseph, H.; Rivera-Correa, E. Aedes aegypti in Puerto Rico: Environmental determinants of larval abundance and relation to dengue virus transmission. Amer. J. Trop. Med. Hyg. 1978, 27, 1225–1231. [Google Scholar]
- Pham, H.; Doan, H.; Phan, T.; Minh, N. Ecological factors associated with dengue fever in a central highlands Province, Vietnam. BMC Infect. Dis. 2011, 11, 172. [Google Scholar] [CrossRef]
- Hurtado-Diaz, M.; Riojas-Rodriguez, H.; Rothenberg, S.; Gomez-Dantes, H.; Cifuentes, E. Short communication: Impact of climate variability on the incidence of dengue in Mexico. Trop. Med. Int. Health 2007, 12, 1327–1337. [Google Scholar] [CrossRef]
- Lowe, R.; Bailey, T.C.; Stephenson, D.B.; Graham, R.J.; Coelho, C.A.S.; Carvalho, M.S.; Barcellos, C. Spatio-temporal modelling of climate-sensitive disease risk: Towards an early warning system for dengue in Brazil. Comput. Geosci. 2011, 37, 371–381. [Google Scholar] [CrossRef]
- Hashizume, M.; Dewan, A.; Sunahara, T.; Rahman, M.; Yamamoto, T. Hydroclimatological variability and dengue transmission in Dhaka, Bangladesh: A time-series study. BMC Infect. Dis. 2012, 12, 98. [Google Scholar] [CrossRef]
- Bich, T.H.; Quang, L.N.; Ha le, T.T.; Hanh, T.T.; Guha-Sapir, D. Impacts of flood on health: Epidemiologic evidence from Hanoi, Vietnam. Glob. Health Action 2011, 4. [Google Scholar] [CrossRef]
- Wan-Norafikah, O.; Nazni, W.A.; Noramiza, S.; Shafa’ar-Ko’Ohar, S.; Heah, S.K.; Nor-Azlina, A.H.; Khairuh-Asuad, M.; Lee, H.L. Distribution of aedes mosquitoes in three selected localities in Malaysia. Sains Malays. 2012, 41, 1309–1313. [Google Scholar]
- Chang, M.S.; Christophel, E.M.; Gopinath, D.; Abdur, R.M. Challenges and future perspective for dengue vector control in the Western Pacific Region. West. Pac. Surveill. Response J. 2011, 2, 9–16. [Google Scholar]
- Adams, B.; Boots, M. How important is vertical transmission in mosquitoes for the persistence of dengue? Insights from a mathematical model. Epidemics 2010, 2, 1–10. [Google Scholar] [CrossRef]
- Hoffmann, E.J.; Miller, J.R. Reduction of mosquito (Diptera: Culicidae) attacks on a human subject by combination of wind and vapor-phase DEET repellent. J. Med. Entomol. 2002, 39, 935–938. [Google Scholar] [CrossRef]
- Kay, B.H.; Ryan, P.A.; Lyons, S.A.; Foley, P.N.; Pandeya, N.; Purdie, D. Winter intervention against aedes aegypti (Diptera: Culicidae) larvae in subterranean habitats slows surface recolonization in summer. J. Med. Entomol. 2002, 39, 356–361. [Google Scholar] [CrossRef]
- Clements, A.N. The Biology of Mosquitoes: Sensory Reception and Behaviour; CABI: Wallingford, UK, 1999; Volume 2, pp. 281–284. [Google Scholar]
- Grimstad, P.R.; DeFoliart, G.R. Mosquito nectar feeding in Wisconsin in relation to twilight and microclimate. J. Med. Entomol. 1975, 11, 691–698. [Google Scholar]
- Service, M.W. Mosquito (Diptera: Culicidae) dispersal—The long and short of it. J. Med. Entomol. 1997, 34, 579–588. [Google Scholar]
- Turner, L.R.; Connell, D.; Tong, S. Exposure to hot and cold temperatures and ambulance attendances in Brisbane, Australia: A time-series study. BMJ Open 2012, 2, e001074. [Google Scholar] [CrossRef] [Green Version]
- Guo, Y.; Barnett, A.G.; Pan, X.; Yu, W.; Tong, S. The impact of temperature on mortality in Tianjin, China: A case-crossover design with a distributed lag nonlinear model. Environ. Health Perspect. 2011, 119, 1719–1725. [Google Scholar] [CrossRef] [Green Version]
- Nazni, W.A.; Lee, H.L.; Rozita, W.M.W.; Lian, A.C.; Chen, C.D.; Azahari, A.H.; Sadiyah, I. Oviposition behaviour of Aedes albopictus in temephos and Bacillus thuringiensis israelensis-treated ovitraps. Dengue Bull. 2009, 33, 209–217. [Google Scholar]
- Rozhan, S.; Jamsiah, M.; Rahimah, A.; Ang, K.T. The COMBI (Communication for Behavioural Impact) in the prevention and control of dengue-the hulu langat experience. J. Commun. Health 2006, 12, 19–32. [Google Scholar]
- Lowe, R.; Bailey, T.C.; Stephenson, D.B.; Jupp, T.E.; Graham, R.J.; Barcellos, C.; Carvalho, M.S. The development of an early warning system for climate-sensitive disease risk with a focus on dengue epidemics in Southeast Brazil. Stat. Med. 2013, 32, 864–883. [Google Scholar] [CrossRef]
- Shepard, D.S.; Undurraga, E.A.; Lees, R.S.; Halasa, Y.; Lum, L.C.S.; Ng, C.W. Use of multiple data sources to estimate the economic cost of dengue illness in Malaysia. Amer. J. Trop. Med. Hyg. 2012, 87, 796–805. [Google Scholar] [CrossRef]
- Kuno, G. Review of the factors modulating dengue transmission. Epidemiol. Rev. 1995, 17, 321–335. [Google Scholar]
Supplementary Files
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Cheong, Y.L.; Burkart, K.; Leitão, P.J.; Lakes, T. Assessing Weather Effects on Dengue Disease in Malaysia. Int. J. Environ. Res. Public Health 2013, 10, 6319-6334. https://doi.org/10.3390/ijerph10126319
Cheong YL, Burkart K, Leitão PJ, Lakes T. Assessing Weather Effects on Dengue Disease in Malaysia. International Journal of Environmental Research and Public Health. 2013; 10(12):6319-6334. https://doi.org/10.3390/ijerph10126319
Chicago/Turabian StyleCheong, Yoon Ling, Katrin Burkart, Pedro J. Leitão, and Tobia Lakes. 2013. "Assessing Weather Effects on Dengue Disease in Malaysia" International Journal of Environmental Research and Public Health 10, no. 12: 6319-6334. https://doi.org/10.3390/ijerph10126319






