Twelve-Months Follow-up of Supervised Exercise after Percutaneous Transluminal Angioplasty for Intermittent Claudication: A Randomised Clinical Trial
Abstract
:1. Introduction
2. Methods
2.1. Study Design
2.2. Sample and Sample Size Calculation
2.3. Ethical Considerations
2.4. Randomisation and Blinding
2.5. PTA and Post-Operative Care
2.6. Intervention
2.7. Assessments at Baseline and Follow-ups
2.8. Statistical Analysis
3. Results
| Intervention group (n = 29) | Control group (n = 21) | |||
|---|---|---|---|---|
| Mean | (SD) | Mean | (SD) | |
| or | or | |||
| % | (n) | % | (n) | |
| Demographics | ||||
| Age (years) | 66.9 | (7.1) | 66 | (8.3) |
| Body mass index (kg/m2) | 27.2 | (5) | 27.4 | (4) |
| Gender (men) | 14 | (48.3) | 10 | (47.6) |
| Marital status (married) | 18 | (62.1) | 10 | (47.6) |
| Years of school (>9 years) | 22 | (75.9) | 16 | (76.2) |
| Blood status | ||||
| Total cholesterol (mmol/L) | 4.8 | (1) | 4.9 | (1.1) |
| HDL 1 (mmol/L) | 1.7 | (0.6) | 1.6 | (0.6) |
| LDL 1 (mmol/L) | 2.6 | (0.7) | 2.6 | (0.8) |
| Triglycerides (mmol/L) | 1.3 | (0.6) | 1.5 | (1) |
| HbA1c 1 (%) | 5.9 | (0.7) | 6.2 | (0.6) |
| Smoking status | ||||
| Have never smoked | 0 | (0) | 4.8 | (1) |
| Used to smoke | 62.1 | (18) | 52.4 | (11) |
| Currently smoke | 37.9 | (11) | 42.8 | (9) |
| Current medication | ||||
| Statins | 96.6 | (28) | 90.5 | (19) |
| Platelet inhibitors | 86.2 | (25) | 95.2 | (20) |
| Anticoagulants | 6.9 | (2) | 0 | (0) |
| Hypertension | 51.7 | (15) | 61.9 | (13) |
| Comorbidity | ||||
| Diabetes | 10.3 | (4) | 19.0 | (4) |
| COPD | 6.9 | (2) | 4.8 | (1) |
| Previous cardiovascular events | ||||
| Myocardial infarction | 27.6 | (8) | 38.1 | (8) |
| Stroke/TIA 1 | 3.4 | (1) | 0 | (0) |
| Peripheral arterial surgery or endovascular treatment | 17.2 | (5) | 19.0 | (4) |

3.1. Physical Function
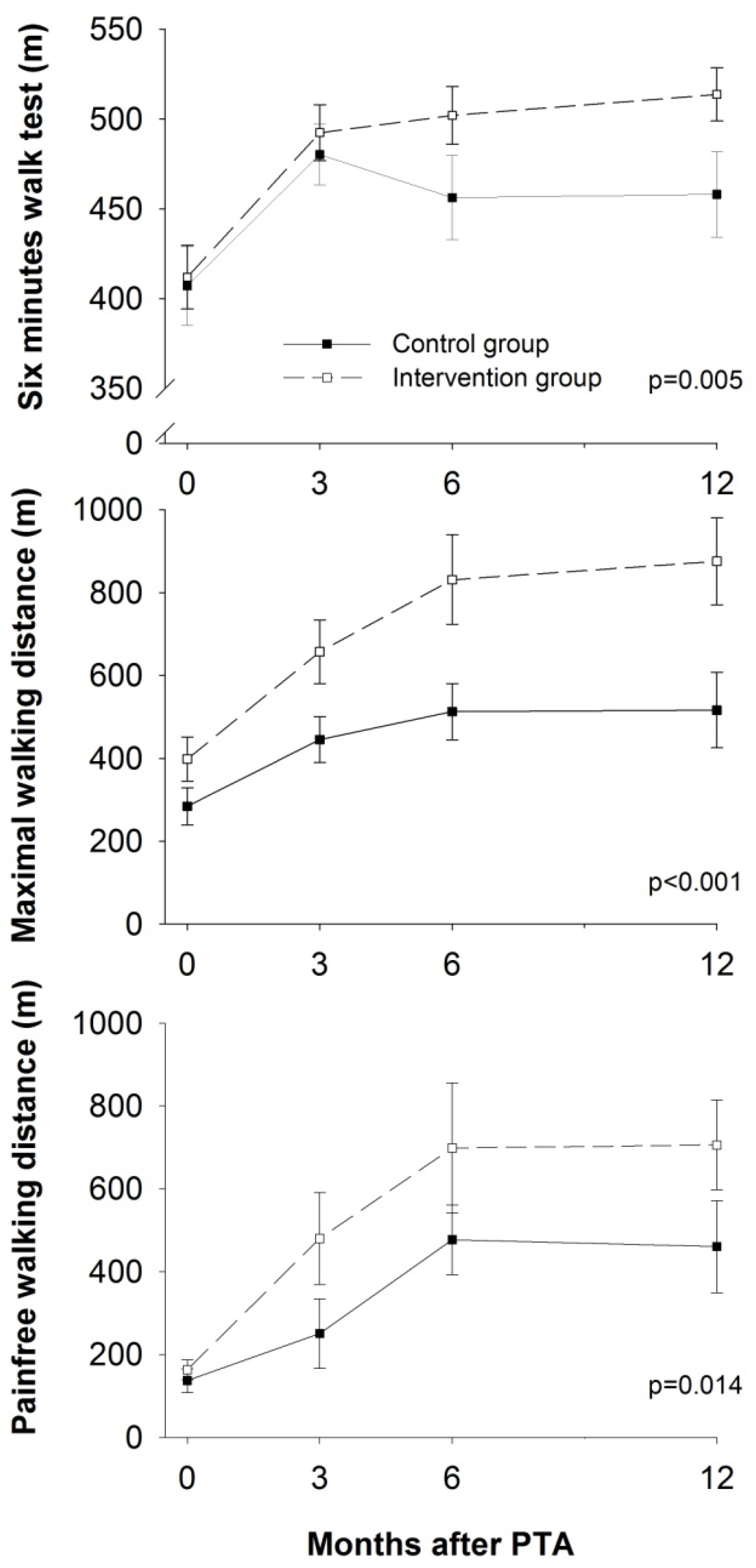
3.2. Limb Hemodynamics

3.3. HRQoL
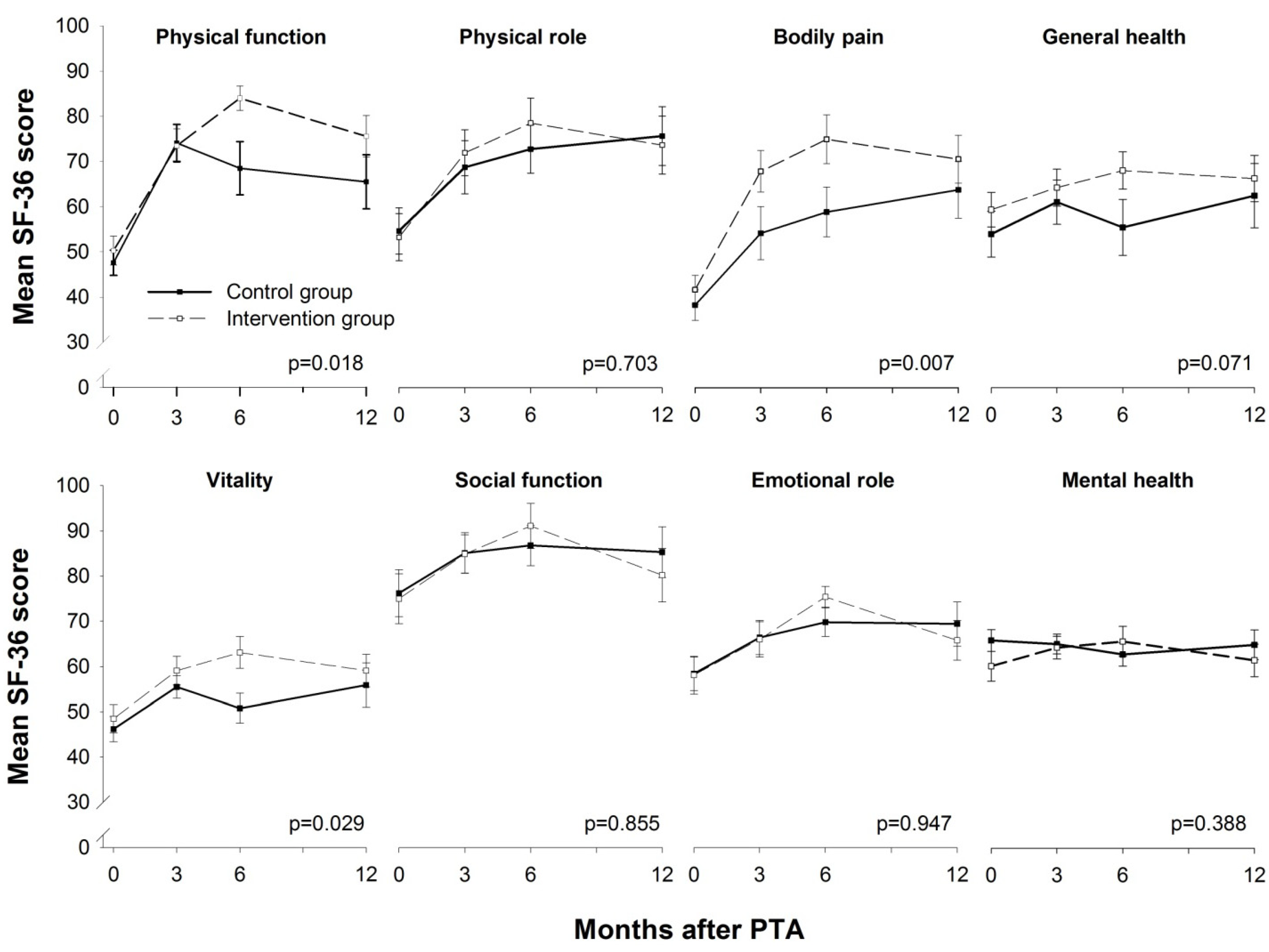
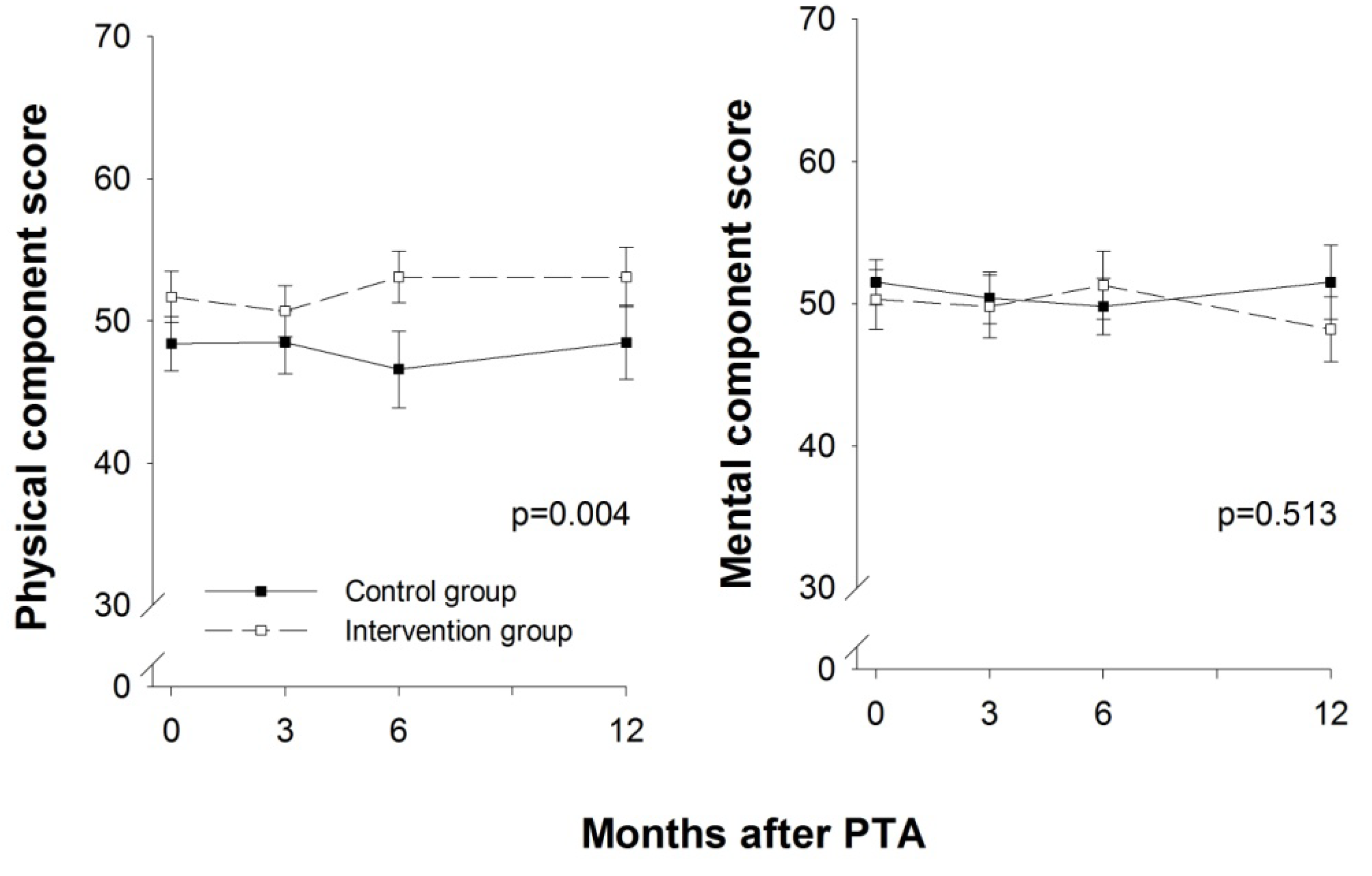
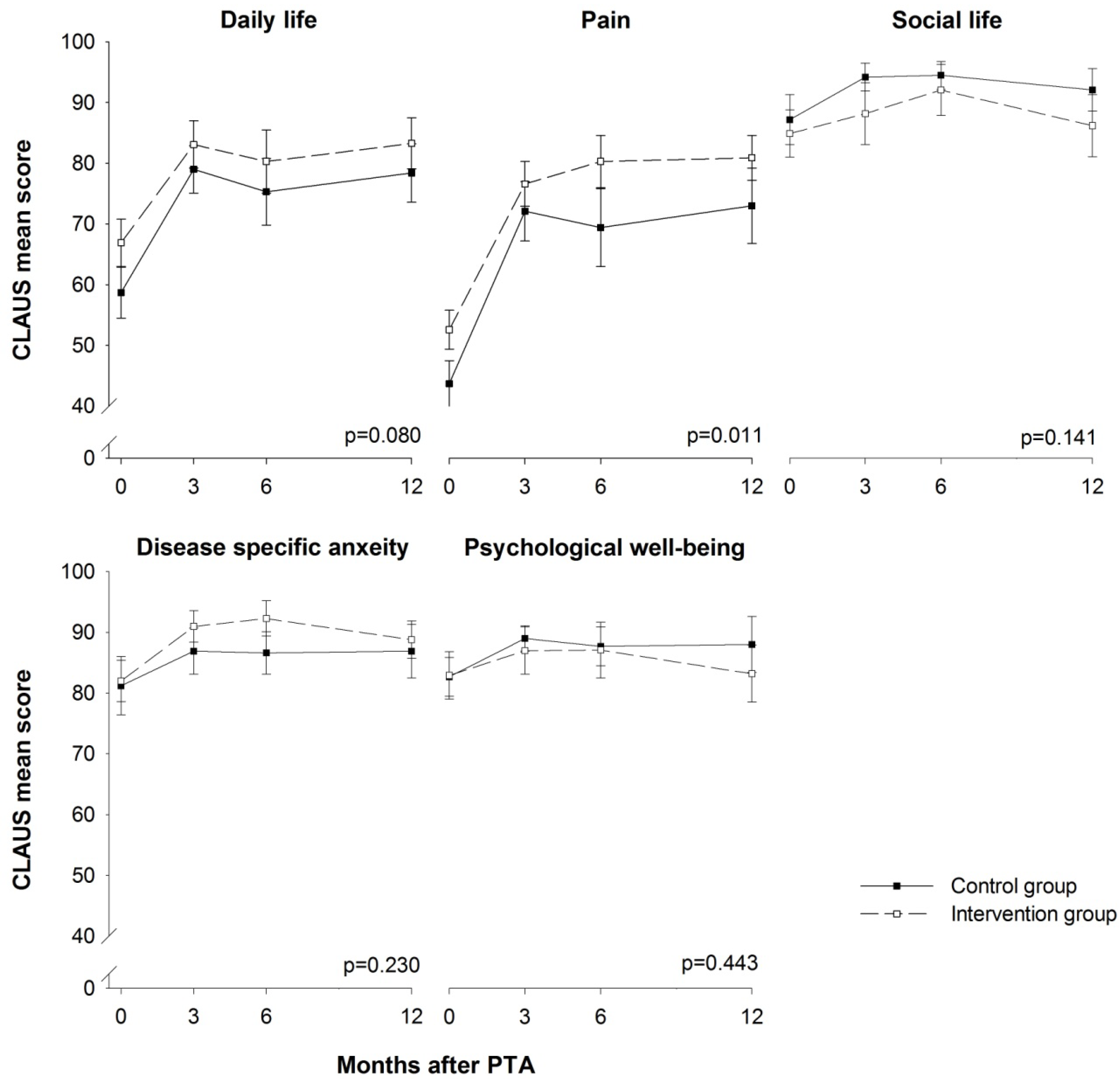
3.4. Medication and Adverse Events
4. Discussion
5. Conclusions
Acknowledgements
Conflicts of Interest
References
- Norgren, L.; Hiatt, W.R.; Dormandy, J.A.; Nehler, M.R.; Harris, K.A.; Fowkes, F.G. Inter-society consensus for the management of peripheral arterial disease (Tasc II). J. Vasc. Surg. 2007, 45, S5–S67. [Google Scholar] [CrossRef]
- Hirsch, A.T.; Criqui, M.H.; Treat-Jacobson, D.; Regensteiner, J.G.; Creager, M.A.; Olin, J.W.; Krook, S.H.; Hunninghake, D.B.; Comerota, A.J.; Walsh, M.E.; et al. Peripheral arterial disease detection, awareness, and treatment in primary care. JAMA 2001, 286, 1317–1324. [Google Scholar] [CrossRef]
- Stroke, O.E.; Tendera, M.; Aboyans, V.; Bartelink, M.L.; Baumgartner, I.; Clement, D.; Collet, J.P.; Cremonesi, A.; de Carlo, M.; Erbel, R.; et al. Esc guidelines on the diagnosis and treatment of peripheral artery diseases. Eur. Heart J. 2011, 32, 2851–2906. [Google Scholar]
- Olin, J.W.; Allie, D.E.; Belkin, M.; Bonow, R.O.; Casey, D.E., Jr.; Creager, M.A.; Gerber, T.C.; Hirsch, A.T.; Jaff, M.R.; Kaufman, J.A.; et al. Accf/aha/acr/scai/sir/svm/svn/svs 2010 performance measures for adults with peripheral artery disease. A report of the american college of cardiology foundation/american heart association task force on performance measures, the american college of radiology, the society for cardiac angiography and interventions, the society for interventional radiology, the society for vascular medicine, the society for vascular nursing, and the society for vascular surgery (writing committee to develop clinical performance measures for peripheral artery disease). Vasc. Med. 2010, 15, 481–512. [Google Scholar] [CrossRef]
- Ahimastos, A.A.; Pappas, E.P.; Buttner, P.G.; Walker, P.J.; Kingwell, B.A.; Golledge, J. A meta-analysis of the outcome of endovascular and noninvasive therapies in the treatment of intermittent claudication. J. Vasc. Surg. 2011, 54, 1511–1521. [Google Scholar]
- Frans, F.A.; Bipat, S.; Reekers, J.A.; Legemate, D.A.; Koelemay, M.J. Systematic review of exercise training or percutaneous transluminal angioplasty for intermittent claudication. Br. J. Surg. 2011, 99, 16–28. [Google Scholar]
- Guidon, M.; McGee, H. Exercise-based interventions and health-related quality of life in intermittent claudication: A 20-year (1989–2008) review. Eur. J. Cardiovasc. Prev. Rehabil. 2010, 17, 140–154. [Google Scholar] [CrossRef]
- Lauret, G.J.; van Dalen, D.C.; Willigendael, E.M.; Hendriks, E.J.; de Bie, R.A.; Spronk, S.; Teijink, J.A. Supervised exercise therapy for intermittent claudication: Current status and future perspectives. Vascular 2012, 20, 12–19. [Google Scholar] [CrossRef]
- Lauret, G.J.; Fakhry, F.; Fokkenrood, H.J.P.; Hunink, M.G.; Teijink, J.A.W.; Spronk, S. Modes of exercise training for intermittent claudication (protocol). Cochrane Database Syst. Rev. 2012, 2. [Google Scholar] [CrossRef]
- Muller-Riemenschneider, F.; Reinhold, T.; Nocon, M.; Willich, S.N. Long-term effectiveness of interventions promoting physical activity: A systematic review. Prev. Med. 2008, 47, 354–368. [Google Scholar] [CrossRef]
- Bendermacher, B.L.; Willigendael, E.M.; Teijink, J.A.; Prins, M.H. Supervised exercise therapy versus non-supervised exercise therapy for intermittent claudication. Cochrane Database Syst. Rev. 2006, 2. [Google Scholar] [CrossRef]
- Spronk, S.; Bosch, J.L.; den Hoed, P.T.; Veen, H.F.; Pattynama, P.M.; Hunink, M.G. Intermittent claudication: Clinical effectiveness of endovascular revascularization versus supervised hospital-based exercise training—Randomized controlled trial. Radiology 2009, 250, 586–595. [Google Scholar] [CrossRef]
- Kolh, P. Improving quality of life in patients with peripheral arterial disease: An important goal. Eur. J. Vasc. Endovasc. Surg. 2010, 40, 626–627. [Google Scholar] [CrossRef]
- Kruidenier, L.M.; Nicolai, S.P.; Rouwet, E.V.; Peters, R.J.; Prins, M.H.; Teijink, J.A. Additional supervised exercise therapy after a percutaneous vascular intervention for peripheral arterial disease: A randomized clinical trial. J. Vasc. Interven. Radiol 2011, 22, 961–968. [Google Scholar] [CrossRef]
- Mazari, F.A.; Khan, J.A.; Carradice, D.; Samuel, N.; Rahman, M.N.A.; Gulati, S.; Lee, H.L.; Mehta, T.A.; McCollum, P.T.; Chetter, I.C. Randomized clinical trial of percutaneous transluminal angioplasty, supervised exercise and combined treatment for intermittent claudication due to femoropopliteal arterial disease. Br. J. Surg. 2011, 99, 39–48. [Google Scholar]
- Schulz, K.F.; Altman, D.G.; Moher, D.; Group, C. Consort 2010 statement: Updated guidelines for reporting parallel group randomised trials. BMJ 2010, 340. [Google Scholar] [CrossRef]
- Perera, S.; Mody, S.H.; Woodman, R.C.; Studenski, S.A. Meaningful change and responsiveness in common physical performance measures in older adults. J. Amer. Geriat. Soc. 2006, 54, 743–749. [Google Scholar] [CrossRef]
- Nilsson, B.B.; Hellesnes, B.; Westheim, A.; Risberg, M.A. Group-based aerobic interval training in patients with chronic heart failure: Norwegian ullevaal model. Phys. Ther. 2008, 88, 523–535. [Google Scholar] [CrossRef]
- Borg, G.A. Psychophysical bases of perceived exertion. Med. Sci. Sport. Exercise 1982, 14, 377–381. [Google Scholar]
- Guyatt, G.H.; Sullivan, M.J.; Thompson, P.J.; Fallen, E.L.; Pugsley, S.O.; Taylor, D.W.; Berman, L.B. The 6-minute walk: A new measure of exercise capacity in patients with chronic heart failure. Can. Med. Assn. J. 1985, 132, 919–923. [Google Scholar]
- McDermott, M.M.; Ades, P.A.; Dyer, A.; Guralnik, J.M.; Kibbe, M.; Criqui, M.H. Corridor-based functional performance measures correlate better with physical activity during daily life than treadmill measures in persons with peripheral arterial disease. J. Vasc. Surg. 2008, 48, 1231–1237. [Google Scholar] [CrossRef]
- Montgomery, P.S.; Gardner, A.W. The clinical utility of a six-minute walk test in peripheral arterial occlusive disease patients. J. Amer. Geriat. Soc. 1998, 46, 706–711. [Google Scholar]
- Gardner, A.W.; Skinner, J.S.; Cantwell, B.W.; Smith, L.K. Progressive vs single-stage treadmill tests for evaluation of claudication. Med. Sci Sport Exercise 1991, 23, 402–408. [Google Scholar]
- Hiatt, W.R.; Hirsch, A.T.; Regensteiner, J.G.; Brass, E.P. Clinical trials for claudication. Assessment of exercise performance, functional status, and clinical end points. Vascular clinical trialists. Circulation 1995, 92, 614–621. [Google Scholar] [CrossRef]
- Nicolai, S.P.; Viechtbauer, W.; Kruidenier, L.M.; Candel, M.J.; Prins, M.H.; Teijink, J.A. Reliability of treadmill testing in peripheral arterial disease: A meta-regression analysis. J. Vasc. Surg. 2009, 50, 322–329. [Google Scholar] [CrossRef]
- Ware, J.E., Jr.; Sherbourne, C.D. The mos 36-item short-form health survey (sf-36). I. Conceptual framework and item selection. Med. Care 1992, 30, 473–483. [Google Scholar] [CrossRef]
- Spengel, F.A.; Brown, T.M.; Dietze, S.; Kirchberger, I.; Comte, S. The claudication scale (Clau-s). A new disease-specific quality-of-life instrument in intermittent claudication. Dis. Manag. Health Outcomes 1997, 2, 65–70. [Google Scholar]
- Beattie, D.K.; Golledge, J.; Greenhalgh, R.M.; Davies, A.H. Quality of life assessment in vascular disease: Towards a consensus. Eur. J. Vasc. Endovasc. Surg. 1997, 13, 9–13. [Google Scholar] [CrossRef]
- Chetter, I.C.; Scott, D.J.; Kester, R.C. An introduction to quality of life analysis: The new outcome measure in vascular surgery. Eur. J. Vasc. Endovasc. Surg. 1998, 15, 4–6. [Google Scholar] [CrossRef]
- Mehta, T.; Subramaniam, A.V.; Chetter, I.; McCollum, P. Assessing the validity and responsiveness of disease-specific quality of life instruments in intermittent claudication. Eur. J. Vasc. Endovasc. Surg. 2006, 31, 46–52. [Google Scholar] [CrossRef]
- McHorney, C.A.; Tarlov, A.R. Individual-patient monitoring in clinical practice: Are available health status surveys adequate? Qual. Life Res. 1995, 4, 293–307. [Google Scholar] [CrossRef]
- Bø, E.; Hisdal, J.; Stranden, E.; Jørgensen, J.J.; Sandbaek, G.; Grøtta, O.J.; Bergland, A. Effects of 12 weeks of supervised exercise after endovascular treatment: A randomised clinical trial. Phys. Res. Int. 2013. resubmitted. [Google Scholar]
- Gardner, A.W.; Katzel, L.I.; Sorkin, J.D.; Bradham, D.D.; Hochberg, M.C.; Flinn, W.R.; Goldberg, A.P. Exercise rehabilitation improves functional outcomes and peripheral circulation in patients with intermittent claudication: A randomized controlled trial. J. Amer. Geriat. Soc. 2001, 49, 755–762. [Google Scholar] [CrossRef]
- Tsai, J.C.; Chan, P.; Wang, C.H.; Jeng, C.; Hsieh, M.H.; Kao, P.F.; Chen, Y.J.; Liu, J.C. The effects of exercise training on walking function and perception of health status in elderly patients with peripheral arterial occlusive disease. J. Intern. med. 2002, 252, 448–455. [Google Scholar] [CrossRef]
- Galea, M.N.; Weinman, J.A.; White, C.; Bearne, L.M. Do behaviour-change techniques contribute to the effectiveness of exercise therapy in patients with intermittent claudication? A systematic review. Eur. J. Vasc. Endovasc. Surg 1, 132–141.
- Garber, C.E.; Blissmer, B.; Deschenes, M.R.; Franklin, B.A.; Lamonte, M.J.; Lee, I.M.; Nieman, D.C.; Swain, D.P. American college of sports medicine position stand. Quantity and quality of exercise for developing and maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: Guidance for prescribing exercise. Med. Sci. Sport. Exercise 2011, 43, 1334–1359. [Google Scholar] [CrossRef]
Supplementary Files
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bø, E.; Hisdal, J.; Cvancarova, M.; Stranden, E.; Jørgensen, J.J.; Sandbæk, G.; Grøtta, O.J.; Bergland, A. Twelve-Months Follow-up of Supervised Exercise after Percutaneous Transluminal Angioplasty for Intermittent Claudication: A Randomised Clinical Trial. Int. J. Environ. Res. Public Health 2013, 10, 5998-6014. https://doi.org/10.3390/ijerph10115998
Bø E, Hisdal J, Cvancarova M, Stranden E, Jørgensen JJ, Sandbæk G, Grøtta OJ, Bergland A. Twelve-Months Follow-up of Supervised Exercise after Percutaneous Transluminal Angioplasty for Intermittent Claudication: A Randomised Clinical Trial. International Journal of Environmental Research and Public Health. 2013; 10(11):5998-6014. https://doi.org/10.3390/ijerph10115998
Chicago/Turabian StyleBø, Elisabeth, Jonny Hisdal, Milada Cvancarova, Einar Stranden, Jørgen J. Jørgensen, Gunnar Sandbæk, Ole J. Grøtta, and Astrid Bergland. 2013. "Twelve-Months Follow-up of Supervised Exercise after Percutaneous Transluminal Angioplasty for Intermittent Claudication: A Randomised Clinical Trial" International Journal of Environmental Research and Public Health 10, no. 11: 5998-6014. https://doi.org/10.3390/ijerph10115998




