Recently Confirmed Apoptosis-Inducing Lead Compounds Isolated from Marine Sponge of Potential Relevance in Cancer Treatment
Abstract
:1. Introduction
‘marine sponge’ AND (programmed cell death OR apoptosis OR antitumor* OR anti-tumor* OR anticancer* OR anti-cancer* OR antineoplasm* OR anti-neoplasm*)
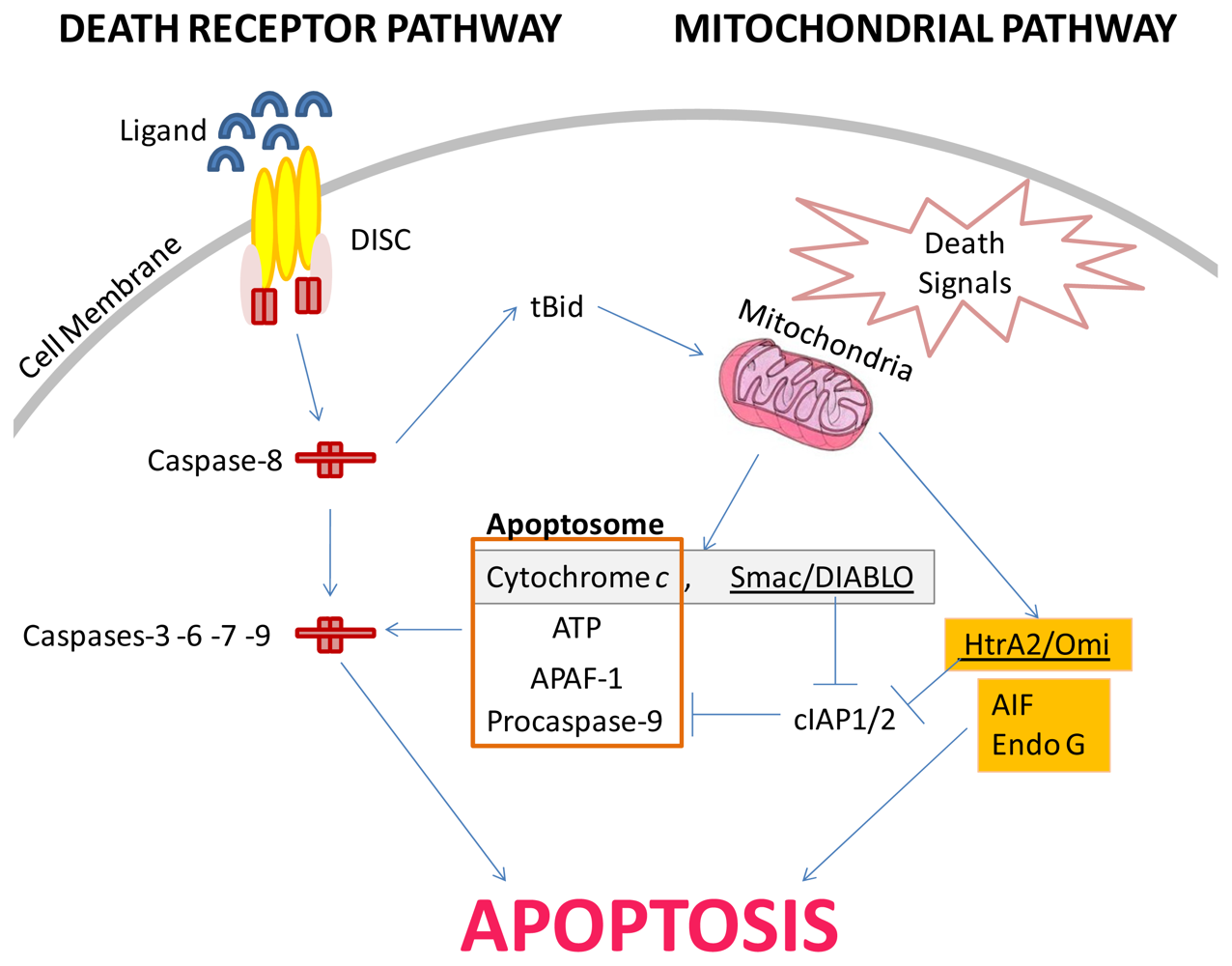
2. Extensively Studied Apoptotic Pathways
2.1. The Death Receptor or “Extrinsic” Apoptosis Pathway of Type I and Type II
2.2. The Mitochondrial or “Intrinsic” Apoptosis Pathway
3. Apoptosis-Inducing Lead Compounds
3.1. Alkaloids
3.1.1. Renieramycin M
3.1.2. Naamidine A
3.1.3. Psammaplysene A
3.1.4. Monanchocidin
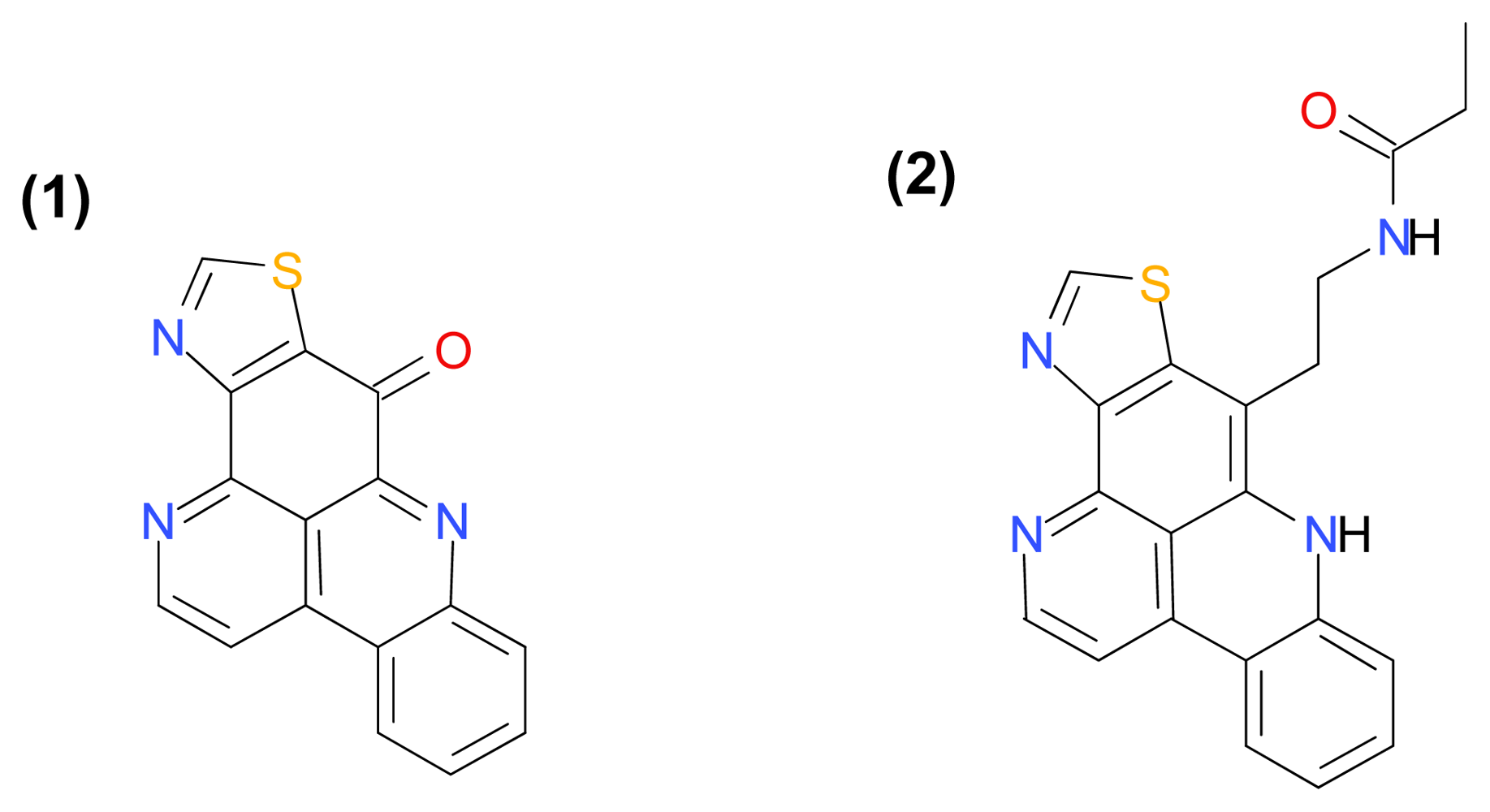
3.1.5. Kuanoniamines A and C
3.2. Terpenoids
3.2.1. Rhabdastrellic Acid-A
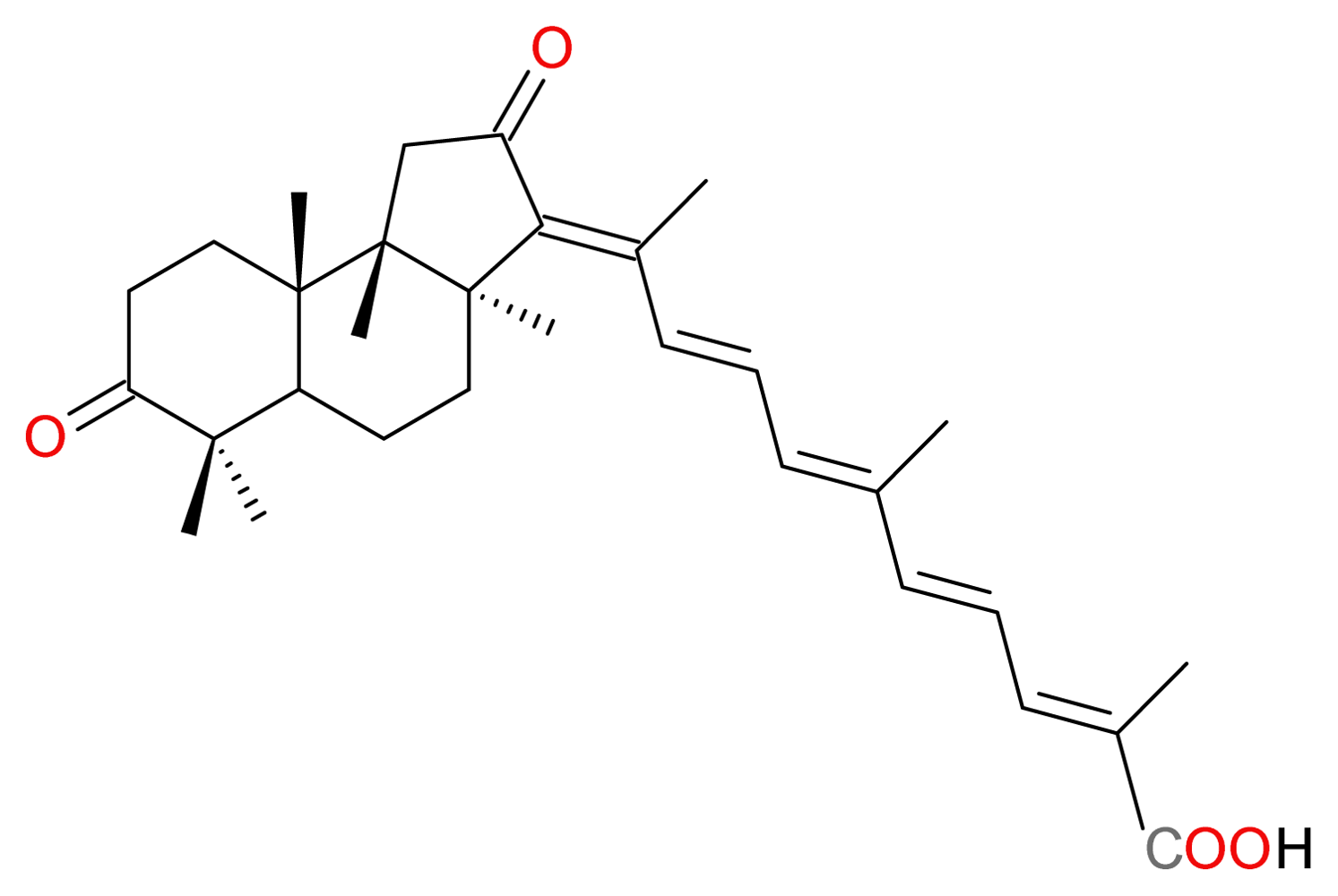
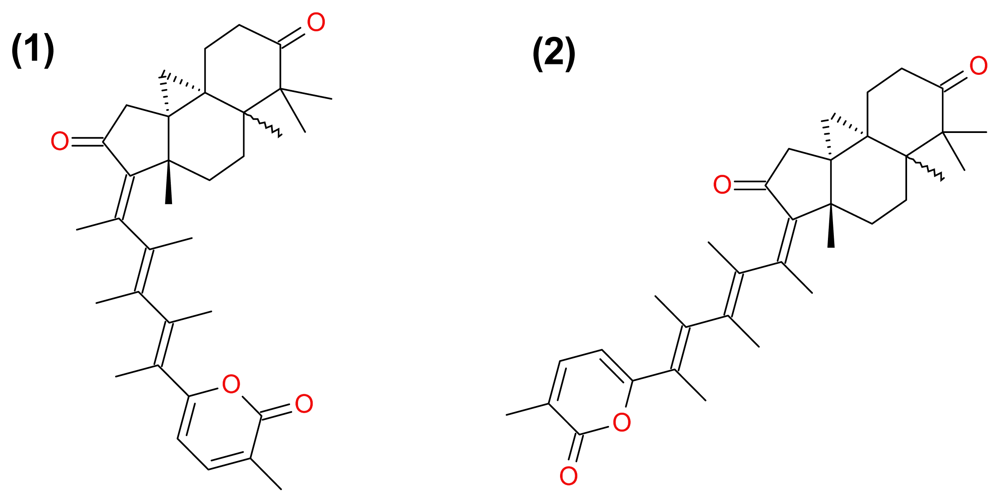
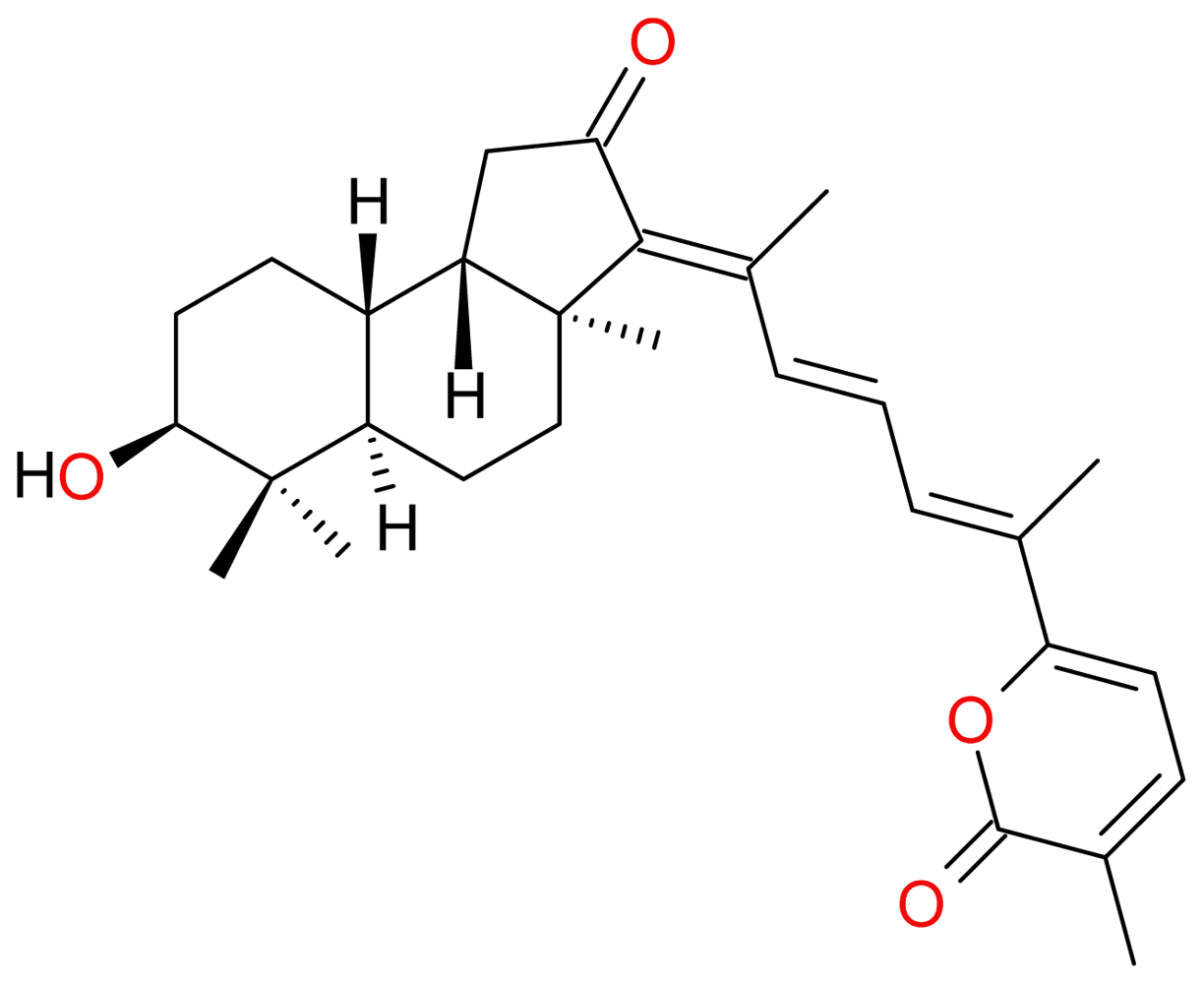
3.2.2. Stellettin A and B, and Geoditin A and B
3.2.3. Jaspolide B
3.2.4. Smenospongine
3.2.5. 13E,17E-Globostellatic Acid X Methyl Ester
3.3. Lipids
3.3.1. Petrosterol-3,6-dione and 5α,6α-epoxy-Petrosterol
3.3.2. Pachastrissamine
3.3.3. Rhizochalin
3.4. Macrolides
3.4.1. Salarin C
3.4.2. Spongistatin 1
3.4.3. Candidaspongiolide
3.4.4. Irciniastatin A
4. Discussion
5. Conclusions
References
- Kawabe, T. G2 checkpoint abrogators as anticancer drugs. Mol. Cancer Ther 2004, 3, 513–519. [Google Scholar]
- Maione, P; Rossi, A; Airoma, G; Ferrara, C; Castaldo, V; Gridelli, C. The role of targeted therapy in non-small cell lung cancer. Crit. Rev. Oncol. Hematol 2004, 51, 29–44. [Google Scholar]
- Rossi, D; Gaidano, G. Messengers of cell death: apoptotic signaling in health and disease. Haematologica 2003, 88, 212–218. [Google Scholar]
- Belarbi el, H; Contreras Gomez, A; Chisti, Y; Garcia Camacho, F; Molina Grima, E. Producing drugs from marine sponges. Biotechnol. Adv 2003, 21, 585–598. [Google Scholar]
- Wehrl, M; Steinert, M; Hentschel, U. Bacterial uptake by the marine sponge Aplysina aerophoba. Microb. Ecol 2007, 53, 355–365. [Google Scholar]
- Vacelet, J; Duport, E. Prey capture and digestion in the carnivorous sponge Asbestopluma hypogea (Porifera: Demospongiae). Zoomorphology 2004, 123, 179–190. [Google Scholar]
- Sokolover, N; Ilan, M. Assessing anti-predatory chemical defences among ten eastern Mediterranean sponges. J. Mar. Biol. Assoc. UK 2007, 87, 1785–1790. [Google Scholar]
- Peters, KJ; Amsler, CD; McClintock, JB; Baker, BJ. Potential chemical defenses of Antarctic sponges against sympatric microorganisms. Polar Biolol 2010, 33, 649–658. [Google Scholar]
- Chaves-Fonnegra, A; Castellanos, L; Zea, S; Duque, C; Rodriguez, J; Jimenez, C. Clionapyrrolidine A—a metabolite from the encrusting and excavating sponge Cliona tenuis that kills coral tissue upon contact. J. Chem. Ecol 2008, 34, 1565–1574. [Google Scholar]
- Blunt, JW; Copp, BR; Munro, MH; Northcote, PT; Prinsep, MR. Marine natural products. Nat. Prod. Rep 2011, 28, 196–268. [Google Scholar]
- Joseph, B; Sujatha, S. Pharmacologically Important Natural products from Marine Sponges. J. Nat. Prod 2011, 4, 5–12. [Google Scholar]
- Thomas, TR; Kavlekar, DP; LokaBharathi, PA. Marine drugs from sponge-microbe association—a review. Mar. Drugs 2010, 8, 1417–1468. [Google Scholar]
- Bergmann, W; Feeney, RJ. The isolation of a new thymine pentoside from sponges. J. Am. Chem. Soc 1950, 72, 2809–2810. [Google Scholar]
- Bergmann, W; Feeney, RJ. Contributions to the study of marine products. XXXII. The nucleosides of sponges. I. J. Org. Chem 1951, 16, 981–987. [Google Scholar]
- Proksch, P; Edrada, RA; Ebel, R. Drugs from the seas—current status and microbiological implications. Appl. Microbiol. Biotechnol 2002, 59, 125–134. [Google Scholar]
- Schwartsmann, G. Marine organisms and other novel natural sources of new cancer drugs. Ann. Oncol 2000, 11(Suppl 3), 235–243. [Google Scholar]
- Feldman, EJ; Lancet, JE; Kolitz, JE; Ritchie, EK; Roboz, GJ; List, AF; Allen, SL; Asatiani, E; Mayer, LD; Swenson, C; Louie, AC. First-In-Man Study of CPX-351: A Liposomal Carrier Containing Cytarabine and Daunorubicin in a Fixed 5:1 Molar Ratio for the Treatment of Relapsed and Refractory Acute Myeloid Leukemia. J. Clin. Oncol 2011, 29, 979–985. [Google Scholar]
- Agura, E; Cooper, B; Holmes, H; Vance, E; Berryman, RB; Maisel, C; Li, S; Saracino, G; Tadic-Ovcina, M; Fay, J. Report of a phase II study of clofarabine and cytarabine in de novo and relapsed and refractory AML patients and in selected elderly patients at high risk for anthracycline toxicity. Oncologist 2011, 16, 197–206. [Google Scholar]
- Walker, AR; Komrokji, RS; Ifthikharuddin, J; Messina, P; Mulford, D; Becker, M; Friedberg, J; Oliva, J; Phillips, G; Liesveld, JL; Abboud, C. Phase I study of cladribine, cytarabine (Ara-C), granulocyte colony stimulating factor (G-CSF) (CLAG Regimen) and simultaneous escalating doses of imatinib mesylate (Gleevec) in relapsed/refractory AML. Leuk. Res 2008, 32, 1830–1836. [Google Scholar]
- Sipkema, D; Osinga, R; Schatton, W; Mendola, D; Tramper, J; Wijffels, RH. Large-scale production of pharmaceuticals by marine sponges: sea, cell, or synthesis? Biotechnol. Bioeng 2005, 90, 201–222. [Google Scholar]
- Madajewicz, S; Zojwalla, NJ; Lucarelli, AG; Hentschel, P; Giardelli, J; Schuck, EL; Krivelevich, I; Wong, BY; Vogelzang, NJ; Sharma, S. A phase I trial of E7974 administered on days 1 and 15 of a 28-day cycle in patients with solid malignancies. J. Clin. Oncol 2007, 25, 2550. [Google Scholar]
- Yanagimoto, H; Shiomi, H; Satoi, S; Mine, T; Toyokawa, H; Yamamoto, T; Tani, T; Yamada, A; Kwon, AH; Komatsu, N; Itoh, K; Noguchi, M. A phase II study of personalized peptide vaccination combined with gemcitabine for non-resectable pancreatic cancer patients. Oncol. Rep 2010, 24, 795–801. [Google Scholar]
- Twelves, C; Cortes, J; Vahdat, LT; Wanders, J; Akerele, C; Kaufman, PA. Phase III trials of eribulin mesylate (E7389) in extensively pretreated patients with locally recurrent or metastatic breast cancer. Clin. Breast Cancer 2010, 10, 160–163. [Google Scholar]
- Newman, DJ; Cragg, GM. Marine natural products and related compounds in clinical and advanced preclinical trials. J. Nat. Prod 2004, 67, 1216–1238. [Google Scholar]
- Sayers, EW; Barrett, T; Benson, DA; Bolton, E; Bryant, SH; Canese, K; Chetvernin, V; Church, DM; DiCuccio, M; Federhen, S; et al. Database resources of the National Center for Biotechnology Information. Nucleic Acids Res 2011, 39, D38–D51. [Google Scholar]
- Shubina, LK; Makarieva, TN; Dyshlovoy, SA; Fedorov, SN; Dmitrenok, PS; Stonik, VA. Three new aaptamines from the marine sponge Aaptos sp. and their proapoptotic properties. Nat. Prod. Commun 2010, 5, 1881–1884. [Google Scholar]
- Kijjoa, A; Wattanadilok, R; Campos, N; Nascimento, MS; Pinto, M; Herz, W. Anticancer activity evaluation of kuanoniamines A and C isolated from the marine sponge Oceanapia sagittaria, collected from the Gulf of Thailand. Mar. Drugs 2007, 5, 6–22. [Google Scholar]
- Whibley, CE; Keyzers, RA; Soper, AG; Davies-Coleman, MT; Samaai, T; Hendricks, DT. Antiesophageal cancer activity from Southern African marine organisms. Ann. N. Y. Acad. Sci 2005, 1056, 405–412. [Google Scholar]
- Guzii, AG; Makarieva, TN; Denisenko, VA; Dmitrenok, PS; Kuzmich, AS; Dyshlovoy, SA; Krasokhin, VB; Stonik, VA. Monanchocidin: a new apoptosis-inducing polycyclic guanidine alkaloid from the marine sponge Monanchora pulchra. Org. Lett 2010, 12, 4292–4295. [Google Scholar]
- LaBarbera, DV; Modzelewska, K; Glazar, AI; Gray, PD; Kaur, M; Liu, T; Grossman, D; Harper, MK; Kuwada, SK; Moghal, N; Ireland, CM. The marine alkaloid naamidine A promotes caspase-dependent apoptosis in tumor cells. Anticancer Drugs 2009, 20, 425–436. [Google Scholar]
- Berry, E; Hardt, JL; Clardy, J; Lurain, JR; Kim, JJ. Induction of apoptosis in endometrial cancer cells by psammaplysene A involves FOXO1. Gynecol. Oncol 2009, 112, 331–336. [Google Scholar]
- Halim, H; Chunhacha, P; Suwanborirux, K; Chanvorachote, P. Anticancer and Antimetastatic Activities of Renieramycin M, a Marine Tetrahydroisoquinoline Alkaloid, in Human Non-small Cell Lung Cancer Cells. Anticancer Res 2011, 31, 193–201. [Google Scholar]
- Aoki, S; Cho, SH; Ono, M; Kuwano, T; Nakao, S; Kuwano, M; Nakagawa, S; Gao, JQ; Mayumi, T; Shibuya, M; Kobayashi, M. Bastadin 6, a spongean brominated tyrosine derivative, inhibits tumor angiogenesis by inducing selective apoptosis to endothelial cells. Anticancer Drugs 2006, 17, 269–278. [Google Scholar]
- Park, C; Kim, GY; Kim, WI; Hong, SH; Park, DI; Kim, ND; Bae, SJ; Jung, JH; Choi, YH. Induction of apoptosis by (Z)-stellettic acid C, an acetylenic acid from the sponge Stelletta sp., is associated with inhibition of telomerase activity in human leukemic U937 cells. Chemotherapy 2007, 53, 160–168. [Google Scholar]
- Tung, NH; Van Minh, C; Ha, TT; Van Kiem, P; Huong, HT; Dat, NT; Nhiem, NX; Tai, BH; Hyun, JH; Kang, HK; Kim, YH. C29 sterols with a cyclopropane ring at C-25 and 26 from the Vietnamese marine sponge Ianthella sp. and their anticancer properties. Bioorg. Med. Chem. Lett 2009, 19, 4584–4588. [Google Scholar]
- Umeyama, A; Matsuoka, N; Mine, R; Nakata, A; Arimoto, E; Matsui, M; Shoji, N; Arihara, S; Takei, M; Hashimoto, T. Polyacetylene diols with antiproliferative and driving Th1 polarization effects from the marine sponge Callyspongia sp. J. Nat. Med 2010, 64, 93–97. [Google Scholar]
- Salma, Y; Lafont, E; Therville, N; Carpentier, S; Bonnafe, MJ; Levade, T; Genisson, Y; Andrieu-Abadie, N. The natural marine anhydrophytosphingosine, Jaspine B, induces apoptosis in melanoma cells by interfering with ceramide metabolism. Biochem. Pharmacol 2009, 78, 477–485. [Google Scholar]
- Fedorov, SN; Makarieva, TN; Guzii, AG; Shubina, LK; Kwak, JY; Stonik, VA. Marine two-headed sphingolipid-like compound rhizochalin inhibits EGF-induced transformation of JB6 P+ Cl41 cells. Lipids 2009, 44, 777–785. [Google Scholar]
- Trisciuoglio, D; Uranchimeg, B; Cardellina, JH; Meragelman, TL; Matsunaga, S; Fusetani, N; Del Bufalo, D; Shoemaker, RH; Melillo, G. Induction of apoptosis in human cancer cells by candidaspongiolide, a novel sponge polyketide. J. Natl. Cancer Inst 2008, 100, 1233–1246. [Google Scholar]
- Chinen, T; Nagumo, Y; Watanabe, T; Imaizumi, T; Shibuya, M; Kataoka, T; Kanoh, N; Iwabuchi, Y; Usui, T. Irciniastatin A induces JNK activation that is involved in caspase-8-dependent apoptosis via the mitochondrial pathway. Toxicol. Lett 2010, 199, 341–346. [Google Scholar]
- Konishi, H; Kikuchi, S; Ochiai, T; Ikoma, H; Kubota, T; Ichikawa, D; Fujiwara, H; Okamoto, K; Sakakura, C; Sonoyama, T; Kokuba, Y; Sasaki, H; Matsui, T; Otsuji, E. Latrunculin a has a strong anticancer effect in a peritoneal dissemination model of human gastric cancer in mice. Anticancer Res 2009, 29, 2091–2097. [Google Scholar]
- Ben-Califa, N; Bishara, A; Kashman, Y; Neumann, D. Salarin C, a member of the salarin superfamily of marine compounds, is a potent inducer of apoptosis. Invest New Drugs 2010. [Google Scholar] [CrossRef]
- Schyschka, L; Rudy, A; Jeremias, I; Barth, N; Pettit, GR; Vollmar, AM. Spongistatin 1: a new chemosensitizing marine compound that degrades XIAP. Leukemia 2008, 22, 1737–1745. [Google Scholar]
- Aoki, S; Sanagawa, M; Watanabe, Y; Setiawan, A; Arai, M; Kobayashi, M. Novel isomarabarican triterpenes, exhibiting selective anti-proliferative activity against vascular endothelial cells, from marine sponge Rhabdastrella globostellata. Bioorg. Med. Chem 2007, 15, 4818–4828. [Google Scholar]
- Dai, J; Fishback, JA; Zhou, YD; Nagle, DG. Sodwanone and yardenone triterpenes from a South African species of the marine sponge Axinella inhibit hypoxia-inducible factor-1 (HIF-1) activation in both breast and prostate tumor cells. J. Nat. Prod 2006, 69, 1715–1720. [Google Scholar]
- Liu, WK; Ho, JC; Che, CT. Apoptotic activity of isomalabaricane triterpenes on human promyelocytic leukemia HL60 cells. Cancer Lett 2005, 230, 102–110. [Google Scholar]
- Cheung, FW; Li, C; Che, CT; Liu, BP; Wang, L; Liu, WK. Geoditin A induces oxidative stress and apoptosis on human colon HT29 cells. Mar. Drugs 2010, 8, 80–90. [Google Scholar]
- Schumacher, M; Cerella, C; Eifes, S; Chateauvieux, S; Morceau, F; Jaspars, M; Dicato, M; Diederich, M. Heteronemin, a spongean sesterterpene, inhibits TNF alpha-induced NF-kappa B activation through proteasome inhibition and induces apoptotic cell death. Biochem. Pharmacol 2010, 79, 610–622. [Google Scholar]
- Lu, PH; Chueh, SC; Kung, FL; Pan, SL; Shen, YC; Guh, JH. Ilimaquinone, a marine sponge metabolite, displays anticancer activity via GADD153-mediated pathway. Eur. J. Pharmacol 2007, 556, 45–54. [Google Scholar]
- Wei, SY; Li, M; Tang, SA; Sun, W; Xu, B; Cui, JR; Lin, WH. Induction of apoptosis accompanying with G(1) phase arrest and microtubule disassembly in human hepatoma cells by jaspolide B, a new isomalabaricane-type triterpene. Cancer Lett 2008, 262, 114–122. [Google Scholar]
- Guo, JF; Zhou, JM; Feng, GK; Liu, ZC; Xiao, DJ; Deng, SZ; Deng, R; Zhu, XF. Apoptosis of human leukemia HL-60 cells induced by rhabdastrellic acid-A and its mechanisms. Ai Zheng 2007, 26, 809–814. (in Chinese). [Google Scholar]
- Shi, Z; Jain, S; Kim, IW; Peng, XX; Abraham, I; Youssef, DT; Fu, LW; El Sayed, K; Ambudkar, SV; Chen, ZS. Sipholenol A, a marine-derived sipholane triterpene, potently reverses P-glycoprotein (ABCB1)-mediated multidrug resistance in cancer cells. Cancer Sci 2007, 98, 1373–1380. [Google Scholar]
- Kong, D; Aoki, S; Sowa, Y; Sakai, T; Kobayashi, M. Smenospongine, a sesquiterpene aminoquinone from a marine sponge, induces G1 arrest or apoptosis in different leukemia cells. Mar. Drugs 2008, 6, 480–488. [Google Scholar]
- Peter, ME; Krammer, PH. The CD95(APO-1/Fas) DISC and beyond. Cell Death Differ 2003, 10, 26–35. [Google Scholar]
- Lin, FL; Hsu, JL; Chou, CH; Wu, WJ; Chang, CI; Liu, HJ. Activation of p38 MAPK by damnacanthal mediates apoptosis in SKHep 1 cells through the DR5/TRAIL and TNFR1/TNF-alpha and p53 pathways. Eur. J. Pharmacol 2011, 650, 120–129. [Google Scholar]
- Singh, A; Ni, J; Aggarwal, BB. Death domain receptors and their role in cell demise. J. Interferon Cytokine Res 1998, 18, 439–450. [Google Scholar]
- Merino, D; Lalaoui, N; Morizot, A; Schneider, P; Solary, E; Micheau, O. Differential inhibition of TRAIL-mediated DR5-DISC formation by decoy receptors 1 and 2. Mol. Cell. Biol 2006, 26, 7046–7055. [Google Scholar]
- Bodmer, JL; Holler, N; Reynard, S; Vinciguerra, P; Schneider, P; Juo, P; Blenis, J; Tschopp, J. TRAIL receptor-2 signals apoptosis through FADD and caspase-8. Nat. Cell Biol 2000, 2, 241–243. [Google Scholar]
- Scaffidi, C; Fulda, S; Srinivasan, A; Friesen, C; Li, F; Tomaselli, KJ; Debatin, KM; Krammer, PH; Peter, ME. Two CD95 (APO-1/Fas) signaling pathways. EMBO J 1998, 17, 1675–1687. [Google Scholar]
- Belizario, JE; Alves, J; Occhiucci, JM; Garay-Malpartida, M; Sesso, A. A mechanistic view of mitochondrial death decision pores. Braz. J. Med. Biol. Res 2007, 40, 1011–1024. [Google Scholar]
- Luo, X; Budihardjo, I; Zou, H; Slaughter, C; Wang, X. Bid, a Bcl2 interacting protein, mediates cytochrome c release from mitochondria in response to activation of cell surface death receptors. Cell 1998, 94, 481–490. [Google Scholar]
- Kluck, RM; Bossy-Wetzel, E; Green, DR; Newmeyer, DD. The release of cytochrome c from mitochondria: a primary site for Bcl-2 regulation of apoptosis. Science 1997, 275, 1132–1136. [Google Scholar]
- Susin, SA; Lorenzo, HK; Zamzami, N; Marzo, I; Snow, BE; Brothers, GM; Mangion, J; Jacotot, E; Costantini, P; Loeffler, M; et al. Molecular characterization of mitochondrial apoptosis-inducing factor. Nature 1999, 397, 441–446. [Google Scholar]
- Du, C; Fang, M; Li, Y; Li, L; Wang, X. Smac, a mitochondrial protein that promotes cytochrome c-dependent caspase activation by eliminating IAP inhibition. Cell 2000, 102, 33–42. [Google Scholar]
- Li, LY; Luo, X; Wang, X. Endonuclease G is an apoptotic DNase when released from mitochondria. Nature 2001, 412, 95–99. [Google Scholar]
- Suzuki, Y; Imai, Y; Nakayama, H; Takahashi, K; Takio, K; Takahashi, R. A serine protease, HtrA2, is released from the mitochondria and interacts with XIAP, inducing cell death. Mol. Cell 2001, 8, 613–621. [Google Scholar]
- Acehan, D; Jiang, X; Morgan, DG; Heuser, JE; Wang, X; Akey, CW. Three-dimensional structure of the apoptosome: implications for assembly, procaspase-9 binding, and activation. Mol Cell 2002, 9, 423–432. [Google Scholar]
- Slee, EA; Harte, MT; Kluck, RM; Wolf, BB; Casiano, CA; Newmeyer, DD; Wang, HG; Reed, JC; Nicholson, DW; Alnemri, ES; Green, DR; Martin, SJ. Ordering the cytochrome c-initiated caspase cascade: hierarchical activation of caspases-2, -3, -6, -7, -8, and -10 in a caspase-9-dependent manner. J. Cell Biol 1999, 144, 281–292. [Google Scholar]
- Sakahira, H; Enari, M; Nagata, S. Cleavage of CAD inhibitor in CAD activation and DNA degradation during apoptosis. Nature 1998, 391, 96–99. [Google Scholar]
- Boulares, AH; Yakovlev, AG; Ivanova, V; Stoica, BA; Wang, G; Iyer, S; Smulson, M. Role of poly(ADP-ribose) polymerase (PARP) cleavage in apoptosis. Caspase 3-resistant PARP mutant increases rates of apoptosis in transfected cells. J. Biol. Chem 1999, 274, 22932–22940. [Google Scholar]
- Hirata, H; Takahashi, A; Kobayashi, S; Yonehara, S; Sawai, H; Okazaki, T; Yamamoto, K; Sasada, M. Caspases are activated in a branched protease cascade and control distinct downstream processes in Fas-induced apoptosis. J. Exp. Med 1998, 187, 587–600. [Google Scholar]
- Kothakota, S; Azuma, T; Reinhard, C; Klippel, A; Tang, J; Chu, K; McGarry, TJ; Kirschner, MW; Koths, K; Kwiatkowski, DJ; Williams, LT. Caspase-3-generated fragment of gelsolin: effector of morphological change in apoptosis. Science 1997, 278, 294–298. [Google Scholar]
- Takahashi, A; Alnemri, ES; Lazebnik, YA; Fernandes-Alnemri, T; Litwack, G; Moir, RD; Goldman, RD; Poirier, GG; Kaufmann, SH; Earnshaw, WC. Cleavage of lamin A by Mch2 alpha but not CPP32: multiple interleukin 1 beta-converting enzyme-related proteases with distinct substrate recognition properties are active in apoptosis. Proc. Natl. Acad. Sci. USA 1996, 93, 8395–8400. [Google Scholar]
- Kumar, MV; Shirley, R; Ma, Y; Lewis, RW. Role of genomics-based strategies in overcoming chemotherapeutic resistance. Curr. Pharm. Biotechnol 2004, 5, 471–480. [Google Scholar]
- Joza, N; Susin, SA; Daugas, E; Stanford, WL; Cho, SK; Li, CY; Sasaki, T; Elia, AJ; Cheng, HY; Ravagnan, L; et al. Essential role of the mitochondrial apoptosis-inducing factor in programmed cell death. Nature 2001, 410, 549–554. [Google Scholar]
- Wang, X; Wang, J; Gengyo-Ando, K; Gu, L; Sun, CL; Yang, C; Shi, Y; Kobayashi, T; Shi, Y; Mitani, S; Xie, XS; Xue, D. C. elegans mitochondrial factor WAH-1 promotes phosphatidylserine externalization in apoptotic cells through phospholipid scramblase SCRM-1. Nat. Cell Biol 2007, 9, 541–549. [Google Scholar]
- Studzinski, GP. Overview of apoptosis. In Apoptosis: A Practical Approach; Oxford University Press: New York, NY, USA, 1999; p. 8. [Google Scholar]
- Indran, IR; Tufo, G; Pervaiz, S; Brenner, C. Recent advances in apoptosis, mitochondria and drug resistance in cancer cells. Biochim. Biophys. Acta 2011, 1807, 735–745. [Google Scholar]
- Chen, L; Gilkes, DM; Pan, Y; Lane, WS; Chen, J. ATM and Chk2-dependent phosphorylation of MDMX contribute to p53 activation after DNA damage. EMBO J 2005, 24, 3411–3422. [Google Scholar]
- Vogelstein, B; Lane, D; Levine, AJ. Surfing the p53 network. Nature 2000, 408, 307–310. [Google Scholar]
- Cheng, EH; Kirsch, DG; Clem, RJ; Ravi, R; Kastan, MB; Bedi, A; Ueno, K; Hardwick, JM. Conversion of Bcl-2 to a Bax-like death effector by caspases. Science 1997, 278, 1966–1968. [Google Scholar]
- Blunt, JW; Copp, BR; Hu, WP; Munro, MH; Northcote, PT; Prinsep, MR. Marine natural products. Nat. Prod. Rep 2008, 25, 35–94. [Google Scholar]
- Frincke, JM; Faulkner, DJ. Antimicrobial metabolites of the sponge Reniera sp. J. Am. Chem. Soc 1981, 104, 265–269. [Google Scholar]
- Davidson, BS. Renieramycin G, a new alkaloid from the sponge Xestospongia caycedoi. Tetrahedron Lett 1992, 33, 3721–3724. [Google Scholar]
- Suwanborirux, K; Amnuoypol, S; Plubrukarn, A; Pummangura, S; Kubo, A; Tanaka, C; Saito, N. Chemistry of renieramycins. Part 3. Isolation and structure of stabilized renieramycin type derivatives possessing antitumor activity from Thai sponge Xestospongia species, pretreated with potassium cyanide. J. Nat. Prod 2003, 66, 1441–1446. [Google Scholar]
- Pettit, GR; Knight, JC; Collins, JC; Herald, DL; Pettit, RK; Boyd, MR; Young, VG. Antineoplastic agents 430. Isolation and structure of cribrostatins 3, 4, and 5 from the republic of maldives cribrochalina species. J. Nat. Prod 2000, 63, 793–798. [Google Scholar]
- Oku, N; Matsunaga, S; van Soest, RW; Fusetani, N. Renieramycin J, a highly cytotoxic tetrahydroisoquinoline alkaloid, from a marine sponge Neopetrosia sp. J. Nat. Prod 2003, 66, 1136–1139. [Google Scholar]
- Lane, JW; Estevez, A; Mortara, K; Callan, O; Spencer, JR; Williams, RM. Antitumor activity of tetrahydroisoquinoline analogues 3-epi-jorumycin and 3-epi-renieramycin G. Bioorg. Med. Chem. Lett 2006, 16, 3180–3183. [Google Scholar]
- Scott, JD; Williams, RM. Chemistry and biology of the tetrahydroisoquinoline antitumor antibiotics. Chem. Rev 2002, 102, 1669–1730. [Google Scholar]
- Copp, BR; Fairchild, CR; Cornell, L; Casazza, AM; Robinson, S; Ireland, CM. Naamidine A is an antagonist of the epidermal growth factor receptor and an in vivo active antitumor agent. J. Med. Chem 1998, 41, 3909–3911. [Google Scholar]
- Schroeder, FC; Kau, TR; Silver, PA; Clardy, J. The psammaplysenes, specific inhibitors of FOXO1a nuclear export. J. Nat. Prod 2005, 68, 574–576. [Google Scholar]
- McDonald, LA; Eldredge, GS; Barrows, LR; Ireland, CM. Inhibition of topoisomerase II catalytic activity by pyridoacridine alkaloids from a Cystodytes sp. ascidian: a mechanism for the apparent intercalator-induced inhibition of topoisomerase II. J. Med. Chem 1994, 37, 3819–3827. [Google Scholar]
- Li, DD; Guo, JF; Huang, JJ; Wang, LL; Deng, R; Liu, JN; Feng, GK; Xiao, DJ; Deng, SZ; Zhang, XS; Zhu, XF. Rhabdastrellic acid-A induced autophagy-associated cell death through blocking Akt pathway in human cancer cells. PLoS One 2010, 5, e12176. [Google Scholar]
- Li, J; Xu, B; Cui, J; Deng, Z; de Voogd, NJ; Proksch, P; Lin, W. Globostelletins A–I, cytotoxic isomalabaricane derivatives from the marine sponge Rhabdastrella globostellata. Bioorg. Med. Chem 2010, 18, 4639–4647. [Google Scholar]
- Zhang, WH; Che, CT. Isomalabaricane-type nortriterpenoids and other constituents of the marine sponge Geodia japonica. J. Nat. Prod 2001, 64, 1489–1492. [Google Scholar]
- Tang, S; Pei, Y; Fu, H; Deng, Z; Li, J; Proksch, P; Lin, W. Jaspolides A–F, six new isomalabricane-type terpenoids from the sponge Jaspis sp. Chem. Pharm. Bull. (Tokyo) 2006, 54, 4–8. [Google Scholar]
- Kondracki, ML; Guyot, M. Smenospongine: a cytotoxic and antimicrobial aminoquinone isolated from smenospongia. Tetrahedron Lett 1987, 27, 5815–5818. [Google Scholar]
- Aoki, S; Kong, D; Matsui, K; Kobayashi, M. Smenospongine, a spongean sesquiterpene aminoquinone, induces erythroid differentiation in K562 cells. Anticancer Drugs 2004, 15, 363–369. [Google Scholar]
- Kong, D; Yamori, T; Kobayashi, M; Duan, H. Antiproliferative and antiangiogenic activities of smenospongine, a marine sponge sesquiterpene aminoquinone. Mar. Drugs 2011, 9, 154–161. [Google Scholar]
- Kuroda, I; Musman, M; Ohtani, II; Ichiba, T; Tanaka, J; Gravalos, DG; Higa, T. Pachastrissamine, a cytotoxic anhydrophytosphingosine from a marine sponge, Pachastrissa sp. J. Nat. Prod 2002, 65, 1505–1506. [Google Scholar]
- Makarieva, TN; Denisenko, VA; Stonik, VA; Milgrom, YM; Rashkes, YW. Rhizochalin, a novel secondary metabolite of mixed biosynthesis from the sponge Rhizochalina incrustata. Tetrahedron Lett 1989, 30, 6581–6584. [Google Scholar]
- Jin, JO; Shastina, V; Park, JI; Han, JY; Makarieva, T; Fedorov, S; Rasskazov, V; Stonik, V; Kwak, JY. Differential induction of apoptosis of leukemic cells by rhizochalin, two headed sphingolipids from sponge and its derivatives. Biol. Pharm. Bull 2009, 32, 955–962. [Google Scholar]
- Pettit, GR; Cichacz, ZA; Gao, F; Herald, CL; Boyd, MR; Schmidt, JM; Hooper, JNA. Isolation and Structure of Spongistatin-1. J. Org. Chem 1993, 58, 1302–1304. [Google Scholar]
- Bai, R; Cichacz, ZA; Herald, CL; Pettit, GR; Hamel, E. Spongistatin 1, a highly cytotoxic, sponge-derived, marine natural product that inhibits mitosis, microtubule assembly, and the binding of vinblastine to tubulin. Mol. Pharmacol 1993, 44, 757–766. [Google Scholar]
- Schneiders, UM; Schyschka, L; Rudy, A; Vollmar, AM. BH3-only proteins Mcl-1 and Bim as well as endonuclease G are targeted in spongistatin 1-induced apoptosis in breast cancer cells. Mol. Cancer Ther 2009, 8, 2914–2925. [Google Scholar]
- Rothmeier, AS; Schneiders, UM; Wiedmann, RM; Ischenko, I; Bruns, CJ; Rudy, A; Zahler, S; Vollmar, AM. The marine compound spongistatin 1 targets pancreatic tumor progression and metastasis. Int. J. Cancer 2010, 127, 1096–1105. [Google Scholar]
- Meragelman, TL; Willis, RH; Woldemichael, GM; Heaton, A; Murphy, PT; Snader, KM; Newman, DJ; van Soest, R; Boyd, MR; Cardellina, JH, II; McKee, TC. Candidaspongiolides, distinctive analogues of tedanolide from sponges of the genus Candidaspongia. J. Nat. Prod 2007, 70, 1133–1138. [Google Scholar]
- Pettit, GR; Xu, JP; Chapuis, JC; Pettit, RK; Tackett, LP; Doubek, DL; Hooper, JN; Schmidt, JM. Antineoplastic agents. 520. Isolation and structure of irciniastatins A and B from the Indo-Pacific marine sponge Ircinia ramosa. J. Med. Chem 2004, 47, 1149–1152. [Google Scholar]
- Cichewicz, RH; Valeriote, FA; Crews, P. Psymberin, a potent sponge-derived cytotoxin from Psammocinia distantly related to the pederin family. Org. Lett 2004, 6, 1951–1954. [Google Scholar]
- Blunt, JW; Copp, BR; Hu, WP; Munro, MH; Northcote, PT; Prinsep, MR. Marine natural products. Nat. Prod. Rep 2007, 24, 31–86. [Google Scholar]
- Blunt, JW; Copp, BR; Munro, MH; Northcote, PT; Prinsep, MR. Marine natural products. Nat. Prod. Rep 2004, 21, 1–49. [Google Scholar]
- Blunt, JW; Copp, BR; Munro, MH; Northcote, PT; Prinsep, MR. Marine natural products. Nat. Prod. Rep 2005, 22, 15–61. [Google Scholar]
- Blunt, JW; Copp, BR; Munro, MH; Northcote, PT; Prinsep, MR. Marine natural products. Nat. Prod. Rep 2006, 23, 26–78. [Google Scholar]
- Blunt, JW; Copp, BR; Hu, WP; Munro, MH; Northcote, PT; Prinsep, MR. Marine natural products. Nat. Prod. Rep 2009, 26, 170–244. [Google Scholar]
- Blunt, JW; Copp, BR; Munro, MH; Northcote, PT; Prinsep, MR. Marine natural products. Nat. Prod. Rep 2010, 27, 165–237. [Google Scholar]
- Bao, B; Sun, Q; Yao, X; Hong, J; Lee, CO; Cho, HY; Jung, JH. Bisindole alkaloids of the topsentin and hamacanthin classes from a marine sponge Spongosorites sp. J. Nat. Prod 2007, 70, 2–8. [Google Scholar]
- Luo, X; Li, F; Hong, J; Lee, CO; Sim, CJ; Im, KS; Jung, JH. Cytotoxic oxylipins from a marine sponge Topsentia sp. J. Nat. Prod 2006, 69, 567–571. [Google Scholar]
- Mansoor, TA; Lee, YM; Hong, J; Lee, CO; Im, KS; Jung, JH. 5,6:8,9-diepoxy and other cytotoxic sterols from the marine sponge Homaxinella sp. J. Nat. Prod 2006, 69, 131–134. [Google Scholar]
- Zheng, L; Yan, X; Han, X; Chen, H; Lin, W; Lee, FS; Wang, X. Identification of norharman as the cytotoxic compound produced by the sponge (Hymeniacidon perleve)-associated marine bacterium Pseudoalteromonas piscicida and its apoptotic effect on cancer cells. Biotechnol. Appl. Biochem 2006, 44, 135–142. [Google Scholar]
- Folmer, F; Jaspars, M; Dicato, M; Diederich, M. Marine cytotoxins: callers for the various dances of death. Gastroenterol. Hepatol. Bed Bench 2009, 2, S34–S50. [Google Scholar]
- Keyzers, RA; Daoust, J; Davies-Coleman, MT; Van Soest, R; Balgi, A; Donohue, E; Roberge, M; Andersen, RJ. Autophagy-modulating aminosteroids isolated from the sponge Cliona celata. Org. Lett 2008, 10, 2959–2962. [Google Scholar]
- Vicencio, JM; Ortiz, C; Criollo, A; Jones, AW; Kepp, O; Galluzzi, L; Joza, N; Vitale, I; Morselli, E; Tailler, M; Castedo, M; Maiuri, MC; Molgo, J; Szabadkai, G; Lavandero, S; Kroemer, G. The inositol 1,4,5-trisphosphate receptor regulates autophagy through its interaction with Beclin 1. Cell Death Differ 2009, 16, 1006–1017. [Google Scholar]
- Andrews, PA; Howell, SB. Cellular pharmacology of cisplatin: perspectives on mechanisms of acquired resistance. Cancer Cells 1990, 2, 35–43. [Google Scholar]
- Huang, GS; Lopez-Barcons, L; Freeze, BS; Smith, AB, III; Goldberg, GL; Horwitz, SB; McDaid, HM. Potentiation of taxol efficacy and by discodermolide in ovarian carcinoma xenograft-bearing mice. Clin. Cancer Res 2006, 12, 298–304. [Google Scholar]
- Hadaschik, BA; Adomat, H; Fazli, L; Fradet, Y; Andersen, RJ; Gleave, ME; So, AI. Intravesical chemotherapy of high-grade bladder cancer with HTI-286, a synthetic analogue of the marine sponge product hemiasterlin. Clin. Cancer Res 2008, 14, 1510–1518. [Google Scholar]
- Vahdat, LT; Pruitt, B; Fabian, CJ; Rivera, RR; Smith, DA; Tan-Chiu, E; Wright, J; Tan, AR; Dacosta, NA; Chuang, E; et al. Phase II study of eribulin mesylate, a halichondrin B analog, in patients with metastatic breast cancer previously treated with an anthracycline and a taxane. J. Clin. Oncol 2009, 27, 2954–2961. [Google Scholar]
- Wu, YC; Zhu, J. Asymmetric total syntheses of (−)-renieramycin M and G and (−)-jorumycin using aziridine as a lynchpin. Org. Lett 2009, 11, 5558–5561. [Google Scholar]
- Aberle, NS; Lessene, G; Watson, KG. A concise total synthesis of naamidine A. Org. Lett 2006, 8, 419–421. [Google Scholar]
- Georgiades, SN; Clardy, J. Total synthesis of psammaplysenes A and B, naturally occurring inhibitors of FOXO1a nuclear export. Org. Lett 2005, 7, 4091–4094. [Google Scholar]
- Kotoku, N; Tamada, N; Hayashi, A; Kobayashi, M. Synthesis of BC-ring model of globostellatic acid X methyl ester, an anti-angiogenic substance from marine sponge. Bioorg. Med. Chem. Lett 2008, 18, 3532–3535. [Google Scholar]
- Passiniemi, M; Koskinen, AM. Asymmetric synthesis of Pachastrissamine (Jaspine B) and its diastereomers viaeta(3)-allylpalladium intermediates. Org. Biomol. Chem 2011, 9, 1774–1783. [Google Scholar]
- Bhaket, P; Morris, K; Stauffer, CS; Datta, A. Total synthesis of cytotoxic anhydrophytosphingosine pachastrissamine (jaspine B). Org. Lett 2005, 7, 875–876. [Google Scholar]
- Crimmins, MT; Katz, JD; Washburn, DG; Allwein, SP; McAtee, LF. Asymmetric total synthesis of spongistatins 1 and 2. J. Am. Chem. Soc 2002, 124, 5661–5663. [Google Scholar]
- Smith, AB, III; Zhu, W; Shirakami, S; Sfouggatakis, C; Doughty, VA; Bennett, CS; Sakamoto, Y. Total synthesis of (+)-spongistatin 1. An effective second-generation construction of an advanced EF Wittig salt, fragment union, and final elaboration. Org. Lett 2003, 5, 761–764. [Google Scholar]
- Huang, X; Shao, N; Palani, A; Aslanian, R; Buevich, A. The total synthesis of psymberin. Org. Lett 2007, 9, 2597–2600. [Google Scholar]
- Jiang, X; Garcia-Fortanet, J; De Brabander, JK. Synthesis and complete stereochemical assignment of psymberin/irciniastatin A. J. Am. Chem. Soc 2005, 127, 11254–11255. [Google Scholar]
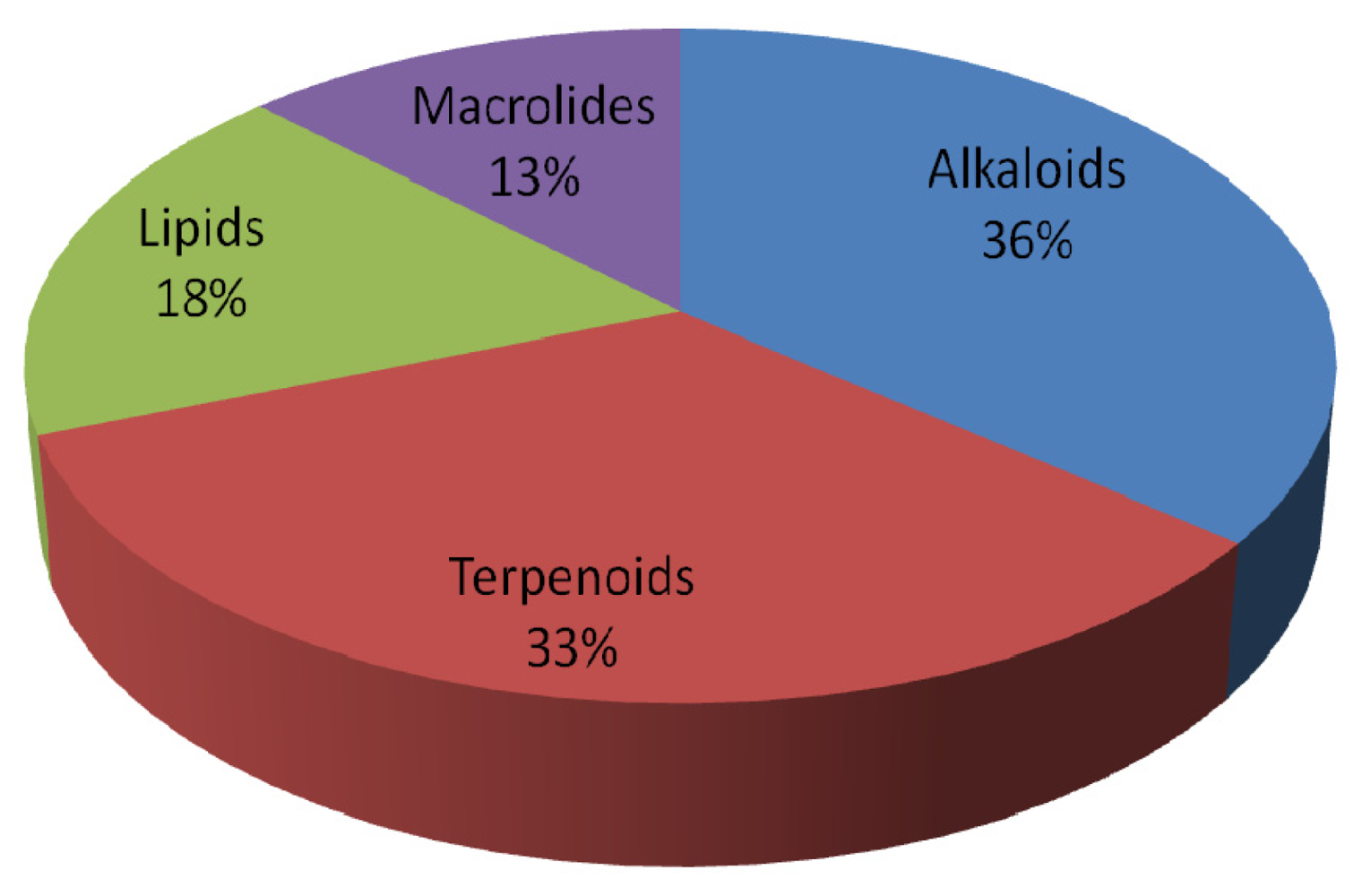



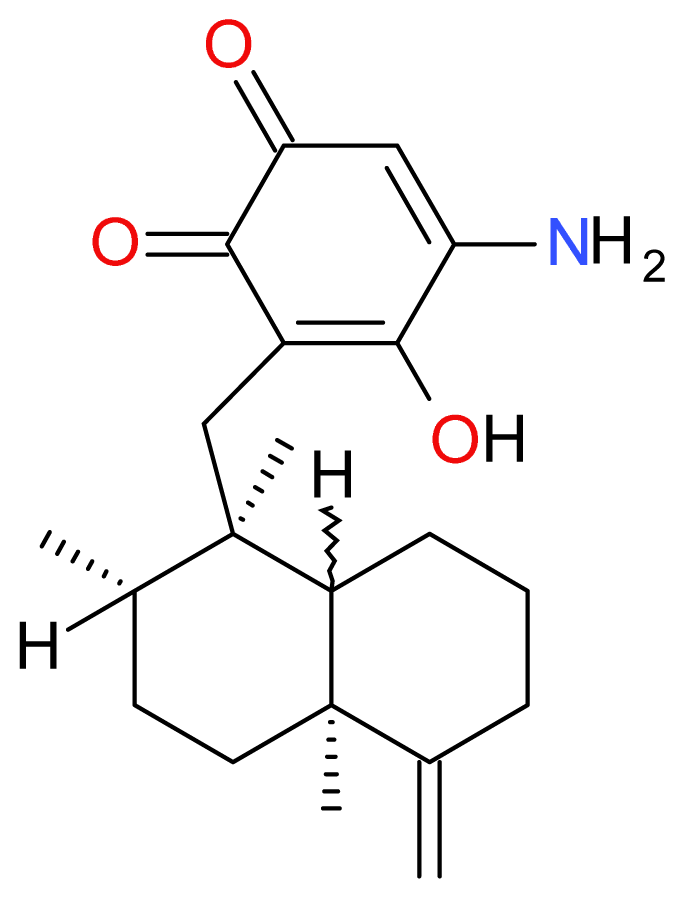


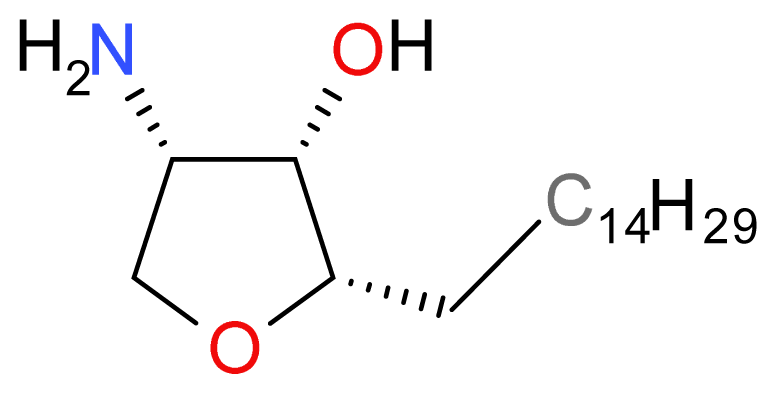
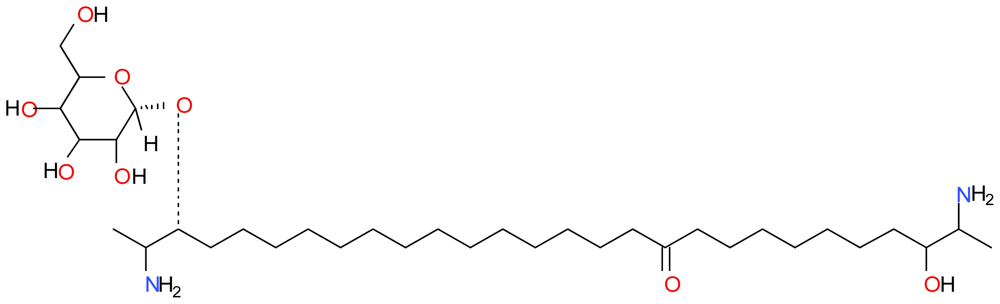


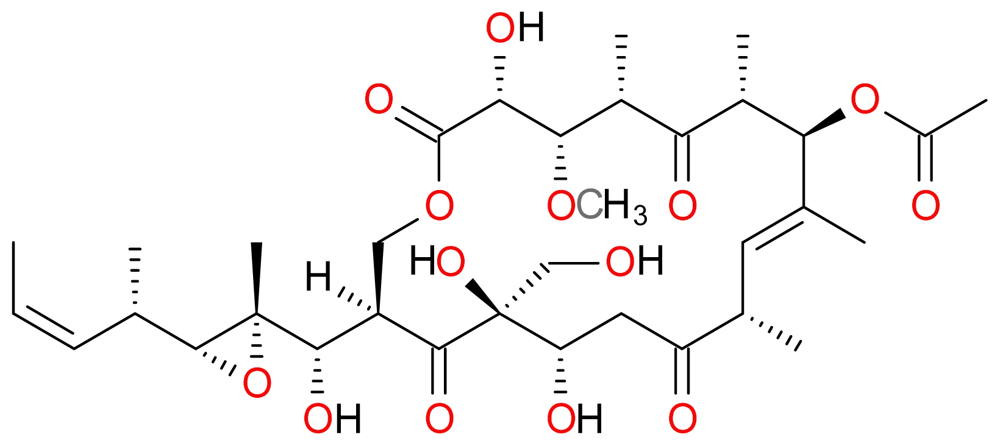

| Compound | Type | Sponge | Cell line | Reference |
|---|---|---|---|---|
| (1) 2,3-dihydro-2,3-dioxoaaptamine | alkaloid | Aaptos sp. | THP-1 | [26] (abstract) |
| (2) 3-(methylamino)demethyl (oxy)aaptamine | alkaloid | Aaptos sp. | THP-1 | [26] (abstract) |
| (3) 6-(N-morpholinyl)-4,5-dihydro-5-oxo-demethyl (oxy)aaptamine | alkaloid | Aaptos sp. | THP-1 | [26] (abstract) |
| (4) kuanoniamine A | alkaloid | Oceanapia sagittaria | MCF-7 | [27] |
| (5) kuanoniamine C | alkaloid | Oceanapia sagittaria | MCF-7 | [27] |
| (6) Makaluvic acid C | alkaloid | Strongylodesma aliwaliensis | WHC01 WHC06 KYSE30 | [28] |
| (7) Monanchocidin | alkaloid | Monanchora pulchra | THP-1 HeLa | [29] |
| (8) N-1-β-d-ribofuranosyl damirone C | alkaloid | Strongylodesma aliwaliensis | WHC01 WHC06 KYSE30 | [28] |
| (9) N-1-β-d-ribofuranosyl makalu-vamine I | alkaloid | Strongylodesma aliwaliensis | WHC01 WHC06 KYSE30 | [28] |
| (10) N-1-β-d-ribofuranosyl makaluvic acid C | alkaloid | Strongylodesma aliwaliensis | WHC01 WHC06 KYSE30 | [28] |
| (11) Naamidine A | alkaloid | Leucetta chagosensis | A-431 | [30] |
| (12) Psammaplysene A | alkaloid | Psammaplysilla | Ishikawa ECC1 | [31] |
| (13) Renieramycin M | alkaloid | Xestospongia sp. | NSCLC | [32] |
| (14) Bastadin 6 | alkaloid | lanthella sp. | HUVEC | [33] (abstract) |
| (15) (Z)-stellettic acid C | lipid | Stelletta sp. | U937 | [34] (abstract) |
| (16) 5-alpha,6-alpha-epoxypetrosterol | lipid | Lanthella sp. | HL-60 | [35] |
| (17) Callyspongidiol | lipid | Callyspongia sp. | HL-60 | [36] (abstract) |
| (18) Jaspine B | lipid | Jaspis sp. | SK-Mel28 | [37] |
| (19) Petrosterol | lipid | Lanthella sp. | HL-60 | [35] |
| (20) Petrosterol-3,6-dione | lipid | Lanthella sp. | HL-60 | [35] |
| (21) Rhizochalin | lipid | Rhizochalina incrustata | THP-1 HeLa SNU-C4 | [38] |
| (22) Candidaspongiolide | macrolide | Candidaspongia sp. | U251 HCT116 | [39] |
| (23) Irciniastatin A | macrolide | Ircinia ramose | Jurkat T | [40] |
| (24) Latrunculins A and B | macrolide | Negombata magnifica | MKN45 NUGC-4 | [41] (abstract) |
| (25) Salarin C | macrolide | Fascaplysinopsis sp. | K562 | [42] |
| (26) Spongistatin 1 | macrolide | Spongia | Jurkat T | [43] |
| (27) 13E,17E-globostellatic acid X methyl ester | terpenoid | Rhabdastrella globostellata | HUVEC | [44] |
| (28) 3-epi-sodwanone K 3-acetate | terpenoid | Axinella sp. | T47D | [45] |
| (29) Geoditin A | terpenoid | Geodia japonica | HL-60 HT29 | [46] [47] |
| (30) Geoditin B | terpenoid | Geodia japonica | HL-60 | [46] |
| (31) Heteronemin | terpenoid | Hyrtios sp. | K562 Jurkat T | [48] |
| (32) Ilimaquinone | terpenoid | Hippospongia metachromia | PC3 | [49] |
| (33) Jaspolide B | terpenoid | Jaspis sp. | Bel-7402 HepG2 | [50] |
| (34) Rhabdastrellic acid-A | terpenoid | Rhabdastrella globostellata | HL-60 | [51] |
| (35) Sipholenol A | terpenoid | Callyspongia siphonella | KB-3-1 KB-C2 KB-V1 | [52] |
| (36) Smenospongine | terpenoid | Dactylospongia elegans | K56 | [53] |
| (37) Sodwanone V | terpenoid | Axinella sp. | MDA-MB-231 | [45] |
| (38) Stellettin A | terpenoid | Geodia japonica | HL-60 | [46] |
| (39) Stellettin B | terpenoid | Geodia japonica | HL-60 | [46] |
© 2011 by the authors; licensee MDPI, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Essack, M.; Bajic, V.B.; Archer, J.A.C. Recently Confirmed Apoptosis-Inducing Lead Compounds Isolated from Marine Sponge of Potential Relevance in Cancer Treatment. Mar. Drugs 2011, 9, 1580-1606. https://doi.org/10.3390/md9091580
Essack M, Bajic VB, Archer JAC. Recently Confirmed Apoptosis-Inducing Lead Compounds Isolated from Marine Sponge of Potential Relevance in Cancer Treatment. Marine Drugs. 2011; 9(9):1580-1606. https://doi.org/10.3390/md9091580
Chicago/Turabian StyleEssack, Magbubah, Vladimir B. Bajic, and John A.C. Archer. 2011. "Recently Confirmed Apoptosis-Inducing Lead Compounds Isolated from Marine Sponge of Potential Relevance in Cancer Treatment" Marine Drugs 9, no. 9: 1580-1606. https://doi.org/10.3390/md9091580





