Mycosporine-Like Amino Acids: Relevant Secondary Metabolites. Chemical and Ecological Aspects
Abstract
:1. Introduction
2. Molecular Structures and Properties
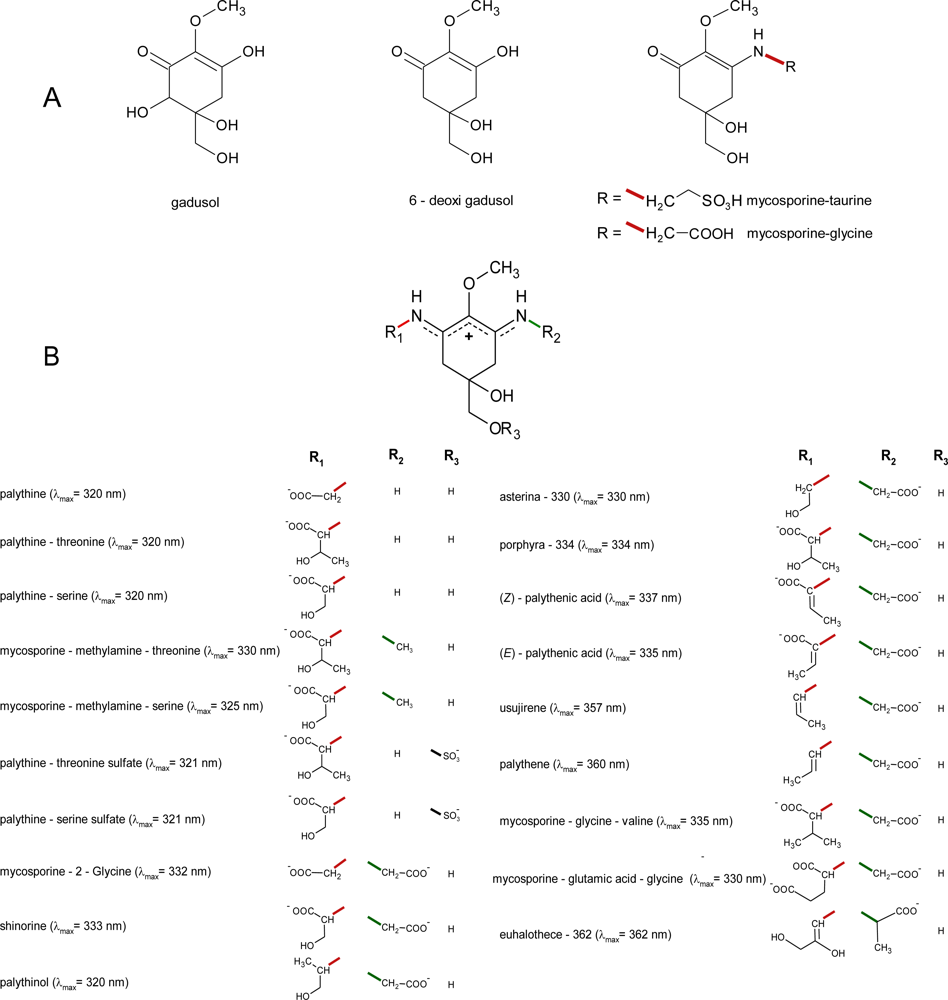
2.1. Structures
2.2. Properties of MAAs
2.2.1. Spectral Characteristics
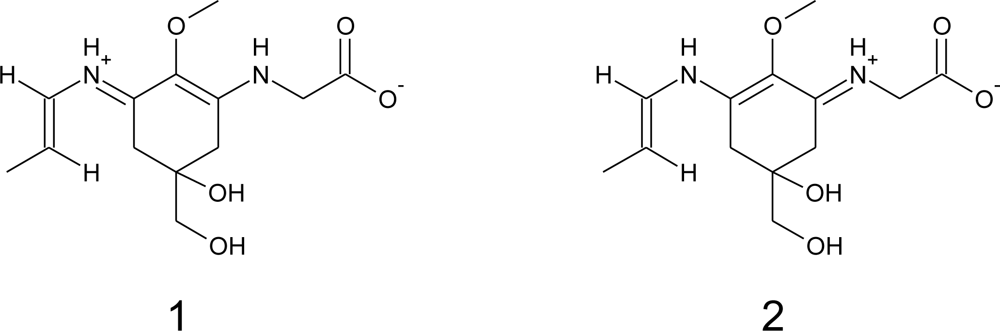
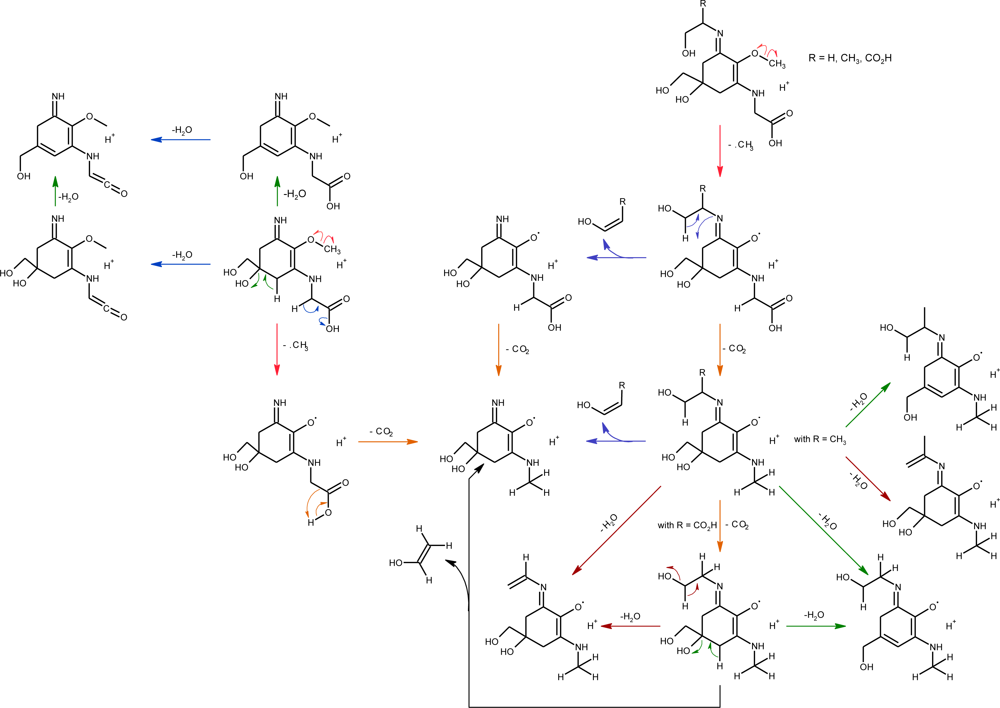
2.2.2. Photo-Induced Transformations
2.2.3. Chemical Reactivity
2.3. Methods for MAAs Isolation, Identification and Quantification
2.3.1. MAAs Isolation
2.3.2. HPLC-Approach for the Identification and Quantification of MAAs
2.3.2.1. HPLC-Separation
2.3.2.2. Identification
2.3.2.3. Quantification
2.4. Occurence of MAAs in Marine Organisms
2.5. MAAs in Producing Organisms
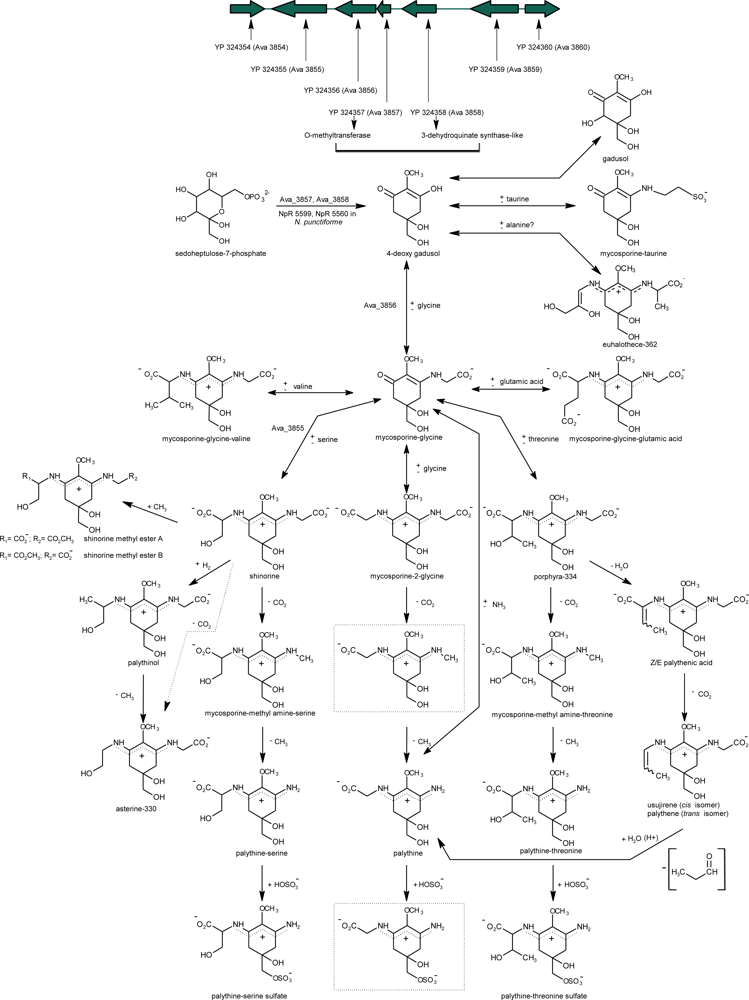
2.5.1. Biosynthesis of Primary MAAs
2.5.2. Biosynthesis Regulation
2.5.3. MAAs Distribution
2.5.3.1. Cyanobacteria
2.5.3.2. Microalgae
2.5.3.2.1. Dinoflagellates
2.5.3.2.2. Prymnesiophytes
2.5.3.2.3. Raphidophytes
2.5.3.2.4. Diatoms
2.5.3.2.5. Other Algae Groups
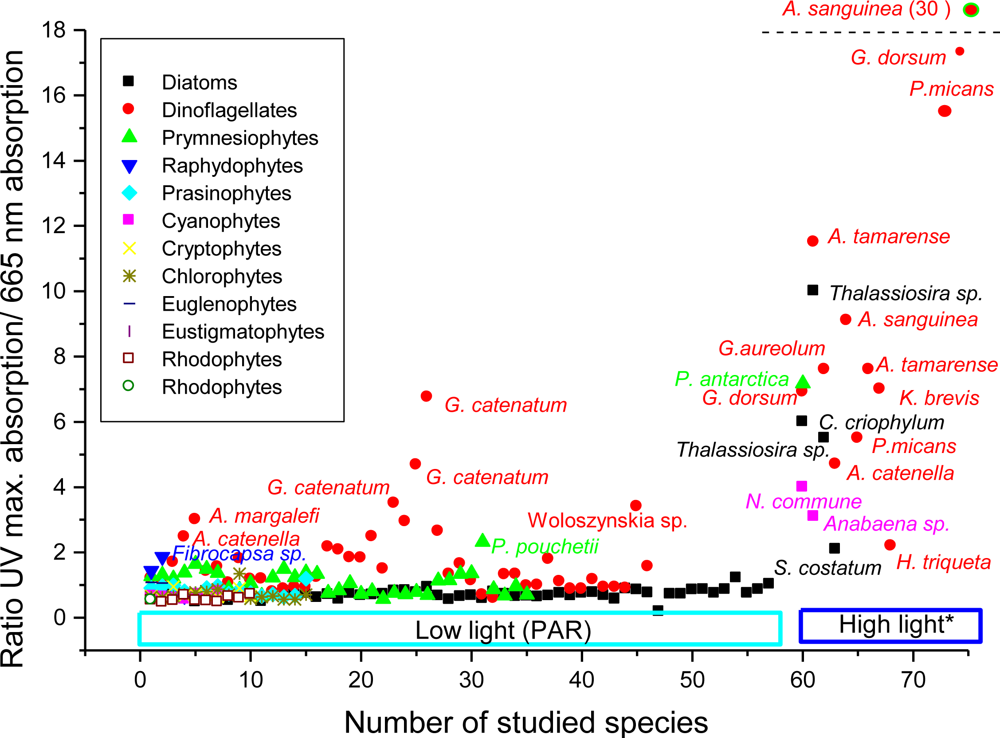
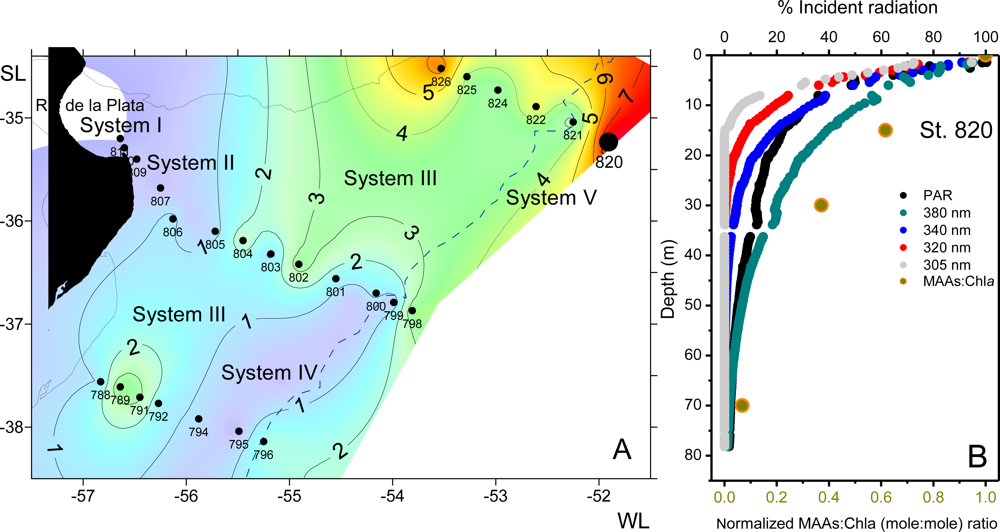
2.5.3.2.6. MAAs in Natural Phytoplankton Populations
2.5.3.3. Macroalgae
2.6. Biosynthesis and Interconversions of MAAs in Symbiotic Associations
2.7. Uptake, Biotransformation and Accumulation of Maas by Consumer Organisms
2.7.1. Selective Uptake
2.7.2. Interconversions of Dietary MAAs
3. Conclusions
Acknowledgments
- Samples Availability: Available from the authors.
References
- Farman, JC; Gardiner, BG; Shanklin, JD. Large losses of total ozone in Antarctica reveals seasonal ClOx/NOx interactions. Nature 1985, 315, 207–210. [Google Scholar]
- Madronich, S; Mackenzie, RL; Björn, LO; Caldwell, MM. Changes in biological active ultraviolet radiation reaching the Earth’s surface. J Photochem Photobiol B 1998, 46, 5–19. [Google Scholar]
- Vincent, WF; Neale, PJ. Mechanisms of UV damage to aquatic organisms. In The Effects of UV Radiation in the Marine Environment; De Mora, S, Demers, S, Vernet, M, Eds.; Cambridge Environmental Chemistry Series; University Press: Cambridge, UK, 2000; pp. 149–176. [Google Scholar]
- United Nations Environment Programme, Environmental Effects Assessment Panel. Environmental effects of ozone depletion and its interactions with climate change: Progress report, 2005. Photochem. Photobiol Sci 2006, 5, 13–24. [Google Scholar]
- Häder, D-P; Kumar, HD; Smith, RC; Worrest, RC. Effects of solar UV radiation on aquatic ecosystems and interactions with climate change. Photochem Photobiol Sci 2007, 6, 267–285. [Google Scholar]
- Lesser, MP; Farrel, JH. Exposure to solar radiation increases damage to both host tissues and algal symbionts of corals during thermal stress. Coral Reefs 2004, 23, 367–377. [Google Scholar]
- Ferrier-Pagés, C; Richard, C; Forcioli, D; Allemand, D; Pichon, M; Shick, JM. Effects of temperature and UV radiation increases on the photosynthetic efficiency in four scleractinian coral species. Biol Bull 2007, 213, 76–87. [Google Scholar]
- Baker, KS; Smith, RC; Green, AES. Middle ultraviolet radiation reaching the ocean surface. Photochem Photobiol 1980, 32, 367–374. [Google Scholar]
- DeMora, S; Demers, S; Vernet, M. The Effects of UV Radiation in the Marine Environment; Cambridge Environmental Chemistry Series; University Press: Cambridge, UK, 2000; p. 324. [Google Scholar]
- Carreto, JI; Carignan, MO; Daleo, G; De Marco, SG. Occurrence of mycosporine-like amino acids in the red-tide dinoflagellate Alexandrium excavatum: UV photoprotective compounds? J Plankton Res 1990, 12, 909–921. [Google Scholar]
- Carreto, JI; Carignan, MO; Montoya, NG. Comparative studies on mycosporine-like amino acids, paralytic shellfish toxins and pigment profiles of the toxic dinoflagellates Alexandrium tamarense, A. catenella and. A minutum Mar Ecol Prog Ser 2001, 223, 49–60. [Google Scholar]
- Ekelund, NGA. Influence of UV-B radiation on photosynthetic light response curves, absorption spectra and motility of four phytoplankton species. Physiol Plant 1994, 91, 696–702. [Google Scholar]
- Klisch, M; Häder, DP. Mycosporine-like amino acids in the marine dinoflagellate Gyrodinium dorsum: Induction by ultraviolet irradiation. J Photochem Photobiol B 2000, 55, 178–182. [Google Scholar]
- Banaszack, AT; Trench, RK. Ultraviolet sunscreens in dinoflagellates. Protist 2001, 152, 93–101. [Google Scholar]
- Cockell, CS; Hornet, G. The history of the UV radiation climate on earth-Theoretical and space-based observations. Photochem Photobiol 2001, 73, 447–451. [Google Scholar]
- Roy, S. Strategies for the minimisation of UV-induced damage. In The Effects of UV Radiation in the Marine Environment; de Mora, S, Demers, S, Vernet, M, Eds.; Cambridge University Press: Cambridge, UK, 2000; pp. 177–205. [Google Scholar]
- Bandaranayake, WM. Mycosporines. Are they nature’sunscreens. Nat Prod Rep 1998, 15, 159–172. [Google Scholar]
- Karentz, D. Chemical defences of marine organisms against solar radiation exposure: UV-absorbing mycosporine-like amino acids and scytonemin. In Marine Chemical Ecology; Mc-Clintock, JB, Baker, J, Eds.; CRC Press: Boca Raton, FL, USA, 2001; pp. 481–520. [Google Scholar]
- Shick, JM; Dunlap, WC. Mycosporine-like amino acids and related gadusols: Biosynthesis, accumulation, and UV-protective functions in aquatic organisms. Annu Rev Physiol 2002, 64, 223–262. [Google Scholar]
- Sinha, RP; Häder, D-P. UV-protectants in cyanobacteria. Plant Sci 2008, 174, 278–289. [Google Scholar]
- Ferroni, L; Klisch, M; Pancaldi, S; Häder, D. Complementary UV-absorption of mycosporine-like amino acids and scytonemin is responsible for UV-insensitivity of photosynthesis in Nostoc flagelliforme. Mar Drugs 2010, 8, 106–121. [Google Scholar]
- Rastogi, RP; Sinha, RP; Singh, SP; Häder, D-P. Photoprotective compounds from marine organisms. J Ind Microbiol Biotechnol 2010, 37, 537–558. [Google Scholar]
- Neale, PJ; Banaszak, AT; Jarriel, CR. Ultraviolet sunscreens in Gymnodinium sanguineum (Dinophyceae): Mycosporine-like amino acids protect against inhibition of photosynthesis. J Phycol 1998, 34, 928–938. [Google Scholar]
- Adams, NL; Shick, JM. Mycosporine-like amino acids provide protection against ultraviolet radiation in eggs of the green sea urchin Strongylocentrotus droebachiensis. Photochem Photobiol 1996, 64, 149–158. [Google Scholar]
- Adams, NL; Shick, JM. Mycosporine-like amino acids prevent UVB-induced abnormalities during early development of the green sea urchin Strongylocentrotus droebachiensis. Mar Biol 2001, 138, 267–280. [Google Scholar]
- Klisch, M; Sinha, RP; Richter, PE; Häder, D-P. Mycosporine-like amino acids (MAAs) protect against UV damage in Gyrodinium dorsum Kofoid. J Plant Physiol 2001, 158, 1449–1454. [Google Scholar]
- Dunlap, WC; Yamamoto, Y. Small-molecule antioxidants in marine organisms: Antioxidant activity of mycosporine-glycine. Comp Biochem Physiol 1995, 112, 105–114. [Google Scholar]
- Suhn, H; Lee, H; Jung, J. Mycosporine glycine protects biological systems against photodynamic damage by quenching singlet oxygen with a high efficiency. Photochem Photobiol 2003, 78, 109–113. [Google Scholar]
- Yoshiki, M; Tsuge, K; Tsuruta, Y; Yoshimura, T; Koganemaru, K; Sumi, T; Matsui, T; Matsumoto, K. Production of new antioxidant compound from mycosporine-like amino acid, porphyra-334 by heat treatment. Food Chem 2009, 113, 1127–1132. [Google Scholar]
- Conde, FR; Churio, MS; Previtali, CM. The photoprotector mechanism of mycosporine-like amino acids. Excited state properties and photostability of porphyra-334 in aqueous solution. J Photochem Photobiol B 2000, 56, 139–144. [Google Scholar]
- Conde, FR; Churio, MS; Previtali, CM. The deactivation pathways of the excited-states of the mycosporine-like amino acids shinorine and porphyra-334 in aqueous solution. Photochem Photobiol Sci 2004, 3, 960–967. [Google Scholar]
- Conde, FR; Churio, MS; Previtali, CM. Experimental study of excited-state properties and photostability of the mycosporine-like amino acid palythine in water solution. Photochem Photobiol Sci 2007, 6, 669–674. [Google Scholar]
- Yakovleva, IM; Bhagooli, R; Takemura, A; Hidaka, M. Differential susceptibility to oxidative stress of two scleractinian corals: Antioxidant functioning of mycosporine-glycine. Comp Biochem Physiol B 2004, 139, 721–730. [Google Scholar]
- Zhang, L; Li, L; Wu, Q. Protective effects of mycosporine-like amino acids of Synechocystis sp. PCC 6803 and their partial characterization. J Photochem Photobiol B 2007, 86, 240–245. [Google Scholar]
- Oyamada, C; Kaneniwa, M; Ebitani, K; Murata, M; Ishihara, K. Mycosporine-like amino-acids extracted from scallop (Patinopecten yessoensis) ovaries: UV protection and growth stimulation activities on human cells. Mar Biotechnol 2008, 10, 141–150. [Google Scholar]
- Nakayama, R; Tamura, Y; Kikuzaki, H; Nakatani, N. Antioxidant effect of the constituents of susabinori (Porphyra yezoensis). JAOCS 1999, 76, 649–653. [Google Scholar]
- de la Coba, F; Aguilera, J; Figueroa, FL; de Gálvez, MV; Herrera, E. Antioxidant activity of mycosporine-like amino acids isolated from three red macroalgae and one marine lichen. J Appl Phycol 2009, 21, 161–169. [Google Scholar]
- de la Coba, F; Aguilera, J; Figueroa, FL. Use of a mycosporin-type amino acid (shinorine) as an antioxidant. WIPO Patent Application WO/2007/026038 2007. [Google Scholar]
- Schmid, D; Schuerch, C; Zuelli, F. Hamless, natural cosmetic skin treatment composition, for protecting against UVA induced lipid oxidation and premature aging, containing mycosporine-like amino acids. Patent number EPI 1473028-AL 2004. [Google Scholar]
- Oren, A. Mycosporine-like amino acids as osmotic solutes in a community of halophilic cyanobacteria. Geomicrobiol J 1997, 14, 231–240. [Google Scholar]
- Oren, A; Gunde-Cimerman, N. Mycosporines and mycosporine-like amino acids: UV protectants or multipurpose secondary metabolites. FEMS Microbiol Lett 2007, 269, 1–10. [Google Scholar]
- Karsten, U. Effects of salinity and ultraviolet radiation on the concentration of mycosporine-like amino acids (MAAs) in various isolates of the benthic cyanobacterium Microcoleus chthonoplastes. Phycological Res 2002, 50, 129–134. [Google Scholar]
- Sinha, RP; Klisch, M; Gröniger, A; Häder, D-P. Ultraviolet-absorbing/screening substances in cyanobacteria, phytoplankton and macroalgae. J Photochem Photobiol B 1998, 47, 83–94. [Google Scholar]
- Gao, K; Wu, Y; Li, G; Wu, H; Villafane, VE; Helbling, EW. Solar UV radiation drives CO2 fixation in marine phytoplankton: A double-edged sword. Plant Physiol 2007, 144, 54–59. [Google Scholar]
- Arpin, N; Bouillant, ML. Light and Mycosporines. In The Fungal Spore, Morphogenetic Controls; Turian, G, Hohl, HR, Eds.; Academic Press: London, UK, 1981; pp. 159–173. [Google Scholar]
- Bandaranayake, WM; Bourne, DJ; Sim, RG. Chemical composition during maturing and spawning of the sponge Dysidea herbacea (Porifera: Demospongiae). Comp Biochem Physiol 1997, 118B, 851–859. [Google Scholar]
- Bandaranayake, WM; des Rocher, A. Role of secondary metabolites and pigments in the epidermal tissues, ripe ovaries, viscera, gut contents and diet of the sea cucumber. Holothuria atra Mar Biol 1999, 133, 163–169. [Google Scholar]
- Gates, RD; Hoegh-Guldberg, O; McFall-Ngais, MJ; Bil, KY; Muscatine, L. Free amino acids exhibit anthozoan “host factor” activity: They induce the release of photosynthate from symbiotic dinoflagellates in vitro. Proc Natl Acad Sci USA 1995, 92, 7430–7434. [Google Scholar]
- Jiang, H; Gao, K; Helbling, EW. UV-absorbing compounds in Porphyra haitanensis (Rhodophyta) with special references to effects of desiccation. J Appl Phycol 2008, 20, 387–395. [Google Scholar]
- Korbee Peinado, N; Abdala Díaz, RT; Figueroa, FL; Helbling, EW. Ammonium and UV radiation stimulate the accumulation of mycosporine-like amino acids in Porphyra columbina (Rhodophyta) from Patagonia, Argentina. J Phycol 2004, 40, 248–259. [Google Scholar]
- Kicklighter, C; Kamio, M; Gemann, M; Derby, C. Pyrimidines and mycosporine-like amino acids function as alarm cues in the defensive secretions of the sea hare Aplysia californica. Chem Senses 2007, 32, A30. [Google Scholar]
- Derby, CD. Escape by inking and secreting: Marine molluscs avoid predators through a rich array of chemicals mechanisms. Biol Bull 2007, 213, 274–289. [Google Scholar]
- Sivalingam, PM; Ikawa, T; Yokohama, Y; Nisizawa, K. Distribution of a 334 UV-absorbing-substance in algae, with special regard of its possible physiological roles. Bot Mar 1974, 17, 23–29. [Google Scholar]
- Tsujino, I; Saito, T. Studies on the compounds specific for each group of marine algae. I. Presence of characteristic ultraviolet absorbing material in Rhodophyceae. Bull Fac Fish Hokkaido Univ 1961, 7, 49–57. [Google Scholar]
- Bon, WF; Ruttenburg, G; Dohrn, A; Batnik, H. Comparative physicochemical investigations on the lens proteins of fishes. Exp Eye Res 1968, 7, 603–610. [Google Scholar]
- Shibata, K. Pigments and a UV-absorbing substance in corals and a blue-green alga living in the Great Barrier Reef. Plant Cell Physiol 1969, 10, 325–335. [Google Scholar]
- Yoshida, T; Sivalingam, PM. UV-absorbing substance in red alga, Porphyra yezoensis Ueda. J Tokyo Univ Fish 1969, 2, 427–434. [Google Scholar]
- Maragos, JE. A study of the ecology of Hawaiian reef corals. Ph.D. Thesis; University of Hawaii: Honolulu, HI, USA, 1972. [Google Scholar]
- Siebeck, O. Photoreactivation and depth-dependent UV tolerance in reef coral in the Great Barrier Reef/Australia. Naturwissenschaften 1981, 68, 426–428. [Google Scholar]
- Jokiel, PL; York, RH, Jr. Solar ultraviolet photobiology of the reef coral Pocillopora damicornis and symbiotic zooxanthellae. Bull Mar Sci 1982, 32, 301–315. [Google Scholar]
- Dunlap, WC; Chalker, BE. Identification and quantification of near-UV absorbing compound (S-320) in a hermatypic scleractinian. Coral Reefs 1986, 5, 1–5. [Google Scholar]
- Haxo, FT. The wavelength dependence of photosynthesis and the role of accessory pigments. In Comparative Biochemistry of Photoreactive Systems; Allen, MB, Ed.; Academic Press: New York, NY, USA, 1960; pp. 339–360. [Google Scholar]
- Yentsch, CS; Yentsch, CM. The attenuation of light by marine phytoplankton with specific reference to the absorption of near-UV radiation. In The Role of Solar Ultraviolet Radiation in Marine Ecosystems; NATO Conference Series 4; Plenum Publishing Corporation: New York, NY, USA, 1982; pp. 691–700. [Google Scholar]
- Balch, WM; Haxo, FT. Spectral properties of Noctiluca miliaris Suriray, a heterotrophic dinoflagellate. J Plankton Res 1984, 6, 515–525. [Google Scholar]
- Carreto, JI; de Marco, S; Lutz, VA. UV-absorbing pigments in the dinoflagellates Alexandrium excavatum and Prorocentrum micans: Effects of light intensity. In Red Tides: Biology, Environmental Science and Toxicology; Okaichi, T, Anderson, DM, Nemoto, T, Eds.; Elsevier: New York, NY, USA, 1989; pp. 37–40. [Google Scholar]
- Vernet, M; Neori, A; Haxo, FT. Spectral properties and photosynthetic action in red-tide populations of Prorocentrum micans and. Gonyaulax polyedra Mar Biol 1989, 103, 365–371. [Google Scholar]
- Ito, S; Hirata, Y. Isolation and structure of a mycosporine from the zoanthidian Palythoa tuberculosa. Tetrahedron Lett 1977, 28, 2429–2430. [Google Scholar]
- Favre-Bonvin, J; Arpin, N; Brevard, C. Structure de la mycosporine (P-31O). Can J Chem 1976, 54, 1105–1113. [Google Scholar]
- Gröniger, A; Sinha, RP; Klisch, M; Häder, D-P. Photoprotective compounds in cyanobacteria, phytoplankton and macroalgae—a database. J Photochem Photobiol B 2000, 58, 115–122. [Google Scholar]
- Sinha, RP; Singh, SP; Häder, D-P. Database on mycosporines and mycosporine-like amino acids (MAAs) in fungi, cyanobacteria, macroalgae, phytoplankton and animals. J Photochem Photobiol B 2007, 89, 29–35. [Google Scholar]
- Rozema, J; Björn, LO; Bornman, JF; Gaberščik, A; Häder, D-P; Trošt, T; Germ, M; Klisch, M; Gröniger, A; Sinha, RP; Lebert, M; He, Y-Y; Buffoni-Hall, R; de Bakker, NVJ; van de Staaij, J; Meijkamp, BB. The role of UV-B radiation in aquatic and terrestrial ecosystems-an experimental and functional analysis of the evolution of UV-absorbing compounds. J Photochem Photobiol B 2002, 66, 2–12. [Google Scholar]
- Starcevic, A; Akthar, S; Dunlap, WC; Shick, JM; Hranueli, D; Cullum, J; Long, PF. Enzymes of the shikimic acid pathway encoded in the genome of a basal metazoan, Nematostella vectensis, have microbial origins. Proc Natl Acad Sci USA 2008, 105, 2533–2537. [Google Scholar]
- Singh, SP; Klisch, M; Sinha, RP; Häder, D-P. Genome mining of mycosporine-like amino acid (MAA) synthesizing and non-synthesizing cyanobacteria: A bioinformatics study. Genomics 2010, 95, 120–128. [Google Scholar]
- Karentz, D; Mc Euen, FS; Land, MV; Dunlap, WC. Survey of mycosporine-like amino acid compounds in Antarctic marine organisms: Potential protection from ultraviolet exposure. Mar Biol 1991, 108, 157–166. [Google Scholar]
- Carreto, JI; Carignan, MO; Montoya, NG. A high-resolution reverse-phase liquid chromatography method for the analysis of mycosporine-like amino acids (MAAs) in marine organisms. Mar Biol 2005, 146, 237–252. [Google Scholar]
- Volkmann, M; Whitehead, K; Rütters, H; Rullkötter, J; Gorbushina, AA. Mycosporine-glutamicol-glucoside: A natural UV-absorbing secondary metabolite of rock-inhabiting microcolonial fungi. Rapid Commun Mass Spectrom 2003, 17, 897–902. [Google Scholar]
- Volkmann, M; Gorbushina, AA; Kedar, L; Oren, A. Structure of euhalothece-362, a novel red-shifted mycosporine-like amino acid, from a halophilic cyanobacterium (Euhalothece sp). Microbiol Lett 2006, 258, 50–54. [Google Scholar]
- Volkmann, M; Gorbushina, AA. A broadly applicable method for extraction and characterization of mycosporines and mycosporine-like amino acids of terrestrial, marine and freshwater origin. Microbiol Lett 2006, 255, 286–295. [Google Scholar]
- Carignan, MO; Cardozo, KHM; Oliveira-Silva, D; Colepicolo, P; Carreto, JI. Palythine-treonine, a major novel mycosporine-like amino acid (MAA) isolated from the hermatypic coral Pocillopora capitata. J Photochem Photobiol B 2009, 94, 191–200. [Google Scholar]
- Korbee, N; Figueroa, FL; Aguilera, J. Acumulación de aminoácidos tipo micosporina (MAAs): Biosíntesis, fotocontrol y funciones ecofisiológicas. Rev Chil Hist Nat 2006, 79, 119–132. [Google Scholar]
- Favre-Bonvin, J; Arpin, N; Brevard, C. Structure de la mycosporine (P-31O). Can J Chem 1976, 54, 1105–1113. [Google Scholar]
- Favre-Bonvin, J; Favre-Bonvin, J; Bernillon, J; Salin, N; Arpin, N. Biosynthesis of mycosporines: Mycosporine glutaminol in Trichothecium roseum. Phytochemistry 1987, 26, 2509–2514. [Google Scholar]
- Gorbushina, AA; Dornieden, T; Niesse, A; Schulte, A; Hedges, JI. Black fungal colonies as units of survival: Hyphal mycosporines synthesized by rock-dwelling microcolonial fungi. Can J Bot 2003, 81, 131–138. [Google Scholar]
- Teai, T; Raharivelomanana, P; Bianchini, JP; Faura, R; Martín, PMV; Cambon, A. Structure de deux nouvelles iminomycosporines isolées de Pocillopora eydouxy. Tetrahedron Lett 1997, 38, 5799–5800. [Google Scholar]
- Stochaj, WR; Dunlap, WC; Shick, JM. Two new UV-absorbing mycosporine-like amino acids from the sea anemone Anthopleura elegantissima and the effects of zooxanthellae and spectral irradiance on chemical composition and content. Mar Biol 1994, 118, 149–156. [Google Scholar]
- Shick, JM; Dunlap, WC; Pearse, JS; Pearse, AV. Mycosporine-like amino acid content in four species of sea anemones in the genus Anthopleura reflects phylogenetic but not environmental or symbiotic relationships. Biol Bull 2002, 203, 315–330. [Google Scholar]
- Arbeloa, EM; Carignan, MO; Acuna, FH; Churio, MS; Carreto, JI. Mycosporine-like amino acid content in the sea anemones Aulactinia marplatensis, Oulactis muscosa and Anthothoe chilensis. Comp Biochem Physiol B 2010, 156, 216–221. [Google Scholar]
- Wu, JJ; Chalker, BE; Rideout, JA. Two new UV-absorbing compounds from Stylophora pistillata: Sulfate esters of mycosporine-like amino acids. Tetrahedron Lett 1997, 38, 2525–2526. [Google Scholar]
- Zhang, L; Li, L; Wu, Q. Protective effects of mycosporine-like amino acids of Synechocystis sp. PCC 6803 and their partial characterization. J Photochem Photobiol B 2007, 86, 240–245. [Google Scholar]
- Klisch, M; Richter, P; Puchta, R; Häder, D-P; Bauer, W. The Stereostructure of Porphyra-334: An Experimental and Calculational NMR Investigation. Evidence for an Efficient “Proton Sponge”. Helvetica Chim Acta 2007, 90, 488–511. [Google Scholar]
- Takano, S; Uemura, D; Hirata, Y. Isolation and structure of a 334 nm UV absorbing substance, porphyra-334 from the red algae Porphyra tenera Kjellman. Chem Lett 1979, 26, 419–420. [Google Scholar]
- Kobayashi, J; Nakamura, H; Hirata, Y. Isolation and structure of a UV-absorbing substance 337 from the ascidian Halocynthia roretzi. Tetrahedron Lett 1981, 22, 3001–3002. [Google Scholar]
- White, JD; Cammack, JH; Sakuma, K; Rewcastle, GW; Widener, RK. Transformations of quinic acid. Asymmetric synthesis and absolute configuration of mycosporine I and mycosporine-gly. J Org Chem 1995, 60, 3600–3611. [Google Scholar]
- Torres, A; Enk, CD; Hochberg, M; Srebnik, M. Porphyra-334, a potential natural source for UVA protective sunscreens. Photochem Photobiol Sci 2006, 5, 432–435. [Google Scholar]
- Furusaki, A; Matsumoto, T; Tsujino, I; Sekikawa, I. The crystal and molecular structure of palythine, trihydrate. Bull Chem Soc Jpn 1980, 53, 319–323. [Google Scholar]
- Uemura, D; Katayama, C; Wada, A; Hirata, Y. Crystal and molecular structure of palythene possessing a novel 360 nm chromophore. Chem Lett 1980, 9, 755–756. [Google Scholar]
- Nakamura, H; Kobayashi, J; Hirata, Y. Isolation and structure of a 330 nm UV-absorbing substance, asterina-330 from the starfish Asterina pectinifera. Chem Lett 1981, 28, 1413–1414. [Google Scholar]
- Cardozo, KHM; Carvalho, VM; Pinto, E. Fragmentation of mycosporine-like amino acids by hydrogen/deuterium exchange and electrospray ionisation tandem mass spectrometry. Rapid Comm Mass Spectrom 2006, 20, 253–258. [Google Scholar]
- Whitehead, K; Karentz, D; Hedges, JI. Mycosporine-like amino acids (MAAs) in phytoplankton, a herbivorous pteropod (Limacina helicina), and its pteropod predator (Clione antarctica) in Mc Murdo Bay, Antarctica. Mar Biol 2001, 139, 1013–1019. [Google Scholar]
- Kedar, L; Kashman, Y; Oren, A. Mycosporine-2-glycine is the major mycosporine-like amino acid in a unicellular cyanobacterium (Euhalothece sp.) isolated from a gypsum crust in a hypersaline saltern pond. FEMS Microbiol Lett 2002, 208, 233–237. [Google Scholar]
- Bandaranayake, WM; Bemis, JE; Bourne, DJ. Ultraviolet absorbing pigments from the marine sponge Dysidea herbacea: Isolation and structure of a new mycosporine. Comp Biochem Physiol 1996, 115C, 281–286. [Google Scholar]
- Balskus, EP; Walsh, CT. The genetic and molecular basis for sunscreen biosynthesis in cyanobacteria. Science 2010, 329, 1653–1656. [Google Scholar]
- Wu, JJ; Rideout, JA; Chalker, BE. Isolation and structure of a novel mycosporine-like amino acid from the reef-building corals Pocillopora damicornis and Stylophora pistillata. Tetrahedron Lett 1995, 36, 5255–5256. [Google Scholar]
- Takano, S; Uemura, D; Hirata, Y. Isolation and structure of two new amino acids, palythinol and palythene, from the zoanthid Palythoa tuberculosa. Tetrahedron Lett 1978, 49, 4909–4912. [Google Scholar]
- Tsujino, I; Yabe, K; Sekikawa, I; Hamanaka, N. Isolation and structure of a mycosporine from the red alga Chondrus yendoi. Tetrahedron Lett 1978, 16, 1401–1402. [Google Scholar]
- Cardozo, KH; Vessecchi, VM; Carvalho, VM; Pinto, E; Gates, PJ; Colepicolo, P; Galembeck, SE; Lopes, NP. A theoretical and MAAs spectrometry study of the fragmentation of mycosporine-like amino acids. Int J Mass Spectrom 2008, 273, 11–19. [Google Scholar]
- Tsujino, I; Yabe, K; Sekikawa, I. Isolation and structure of an new amino acid, shinorine from the red alga Chondrus yendoi Yamada et Mikami. Bot Mar 1980, 23, 65–68. [Google Scholar]
- Sekikawa, I; Kubota, C; Hiraoki, T; Tsujino, I. Isolation and structure of a 357 nm UV absorbing substance, usujirene, from the red alga Palmaria palmata (L.) O. Kuntze. Jpn J Phycol 1986, 34, 185–188. [Google Scholar]
- Callone, AI; Carignan, MO; Montoya, NG; Carreto, JI. Biotransformation of mycosporine-like amino acids (MAAs) in the toxic dinoflagellate Alexandrium tamarense. J Photochem Photobiol B 2006, 84, 204–212. [Google Scholar]
- Laurion, I; Roy, S. Growth and photoprotection in three dinoflagellates (including two strains of Alexandrium tamarense) and one diatom exposed to four weeks of natural and enhanced UVB radiation. J Phycol 2009, 45, 16–33. [Google Scholar]
- Llewellyn, CA; Airs, RL. Distribution and abundance of MAAs in 33 species of microalgae across 13 Classes. Mar Drugs 2010, 8, 1273–1291. [Google Scholar]
- Jeffrey, SW; MacTavish, HS; Dunlap, WC; Vesk, M; Groenewould, K. Occurrence of UVA and UVB-absorbing compounds in 152 species (206 strains) of marine microalgae. Mar Ecol Prog Ser 1999, 189, 35–51. [Google Scholar]
- Subramanian, A; Carpenter, EJ; Karentz, D; Falkowski, PG. Bio-optical properties of the marine diazotrophic cyanobacteria Trichodesmium spp. I. Absorption and photosynthetic action spectra. Limnol Oceanogr 1999, 44, 608–617. [Google Scholar]
- Liu, Z; Häder, D-P; Sommaruga, R. Occurrence of mycosporine-like amino acids (MAAs) in the bloom-forming cyanobacterium Microcystis aeruginosa. J Plankton Res 2004, 26, 963–966. [Google Scholar]
- Laurion, I; Blouin, F; Roy, S. The quantitative filter technique for measuring phytoplankton absorption: Interference by MAAs in the UV waveband. Limnol Oceanogr Methods 2003, 1, 1–9. [Google Scholar]
- Laurion, I; Blouin, F; Roy, S. Packaging of mycosporine-like amino acids in dinoflagellates. Mar Ecol Prog Ser 2004, 279, 297–303. [Google Scholar]
- Böhm, GA; Pfleiderer, W; Böger, P; Scherer, S. Structure of a novel olygosaccharidemycosporine-amino acid ultraviolet A/B sunscreen pigment from the terrestrial cyanobacterium Nostoc comune. J Biol Chem 1995, 270, 9–17. [Google Scholar]
- Dunlap, WC; Williams, DM; Chalker, BE; Banaszak, AT. Biochemical photoadaptations in vision: UV-absorbing pigments in fish eye tissues. Comp Biochem Physiol 1989, 93B, 601–607. [Google Scholar]
- Shashar, N; Hárosi, FI; Banaszak, AT; Hanlon, RT. UV Radiation blocking compounds in the eye of the cuttlefish Sepia officinalis. Biol Bull 1998, 195, 187–188. [Google Scholar]
- Ingalls, AE; Kenia Whitehead, K; Bridouxa, MC. Tinted windows: The presence of the UV absorbing compounds called mycosporine-like amino acids embedded in the frustules of marine diatoms. Geochim Cosmochim Acta 2010, 74, 104–115. [Google Scholar]
- Daniel, S; Cornelia, S; Fred, Z. UV-A sunscreen from red algae for protection against premature skin aging. Cosmet Toilet Manuf Worldw 2000, 115, 139–143. [Google Scholar]
- Bjornland, T. UV-vis spectroscopy of carotenoids. Monographs on oceanographic methodology. In Phytoplankton Pigments in Oceanography: Guidelines to Modern Methods; Jeffrey, SW, Mantoura, RFC, Wright, SW, Eds.; UNESCO: Paris, France, 1997; pp. 578–594. [Google Scholar]
- Conde, FR; Carignan, MO; Churio, MS; Carreto, JI. In vitro cis-trans photoisomerization of palythene and usujirene. Implications on the in vivo transformation of mycosporine-like amino acids. Photochem Photobiol 2003, 77, 146–150. [Google Scholar]
- Zhang, Z; Tashiro, Y; Matsukawa, S; Ogawa, H. Influence of pH and temperature on the ultraviolet-absorbing properties of porphyra-334. Fish Sci 2005, 71, 1382–1384. [Google Scholar]
- Gröniger, A; Häder, D-P. Stability of mycosporine-like amino acids. Recent Res Devel Photochem Photobiol 2000, 4, 247–252. [Google Scholar]
- Shick, JM; Dunlap, WC; Buettner, R. Ultraviolet (UV) protection in marine organisms II. Biosynthesis, accumulation, and sunscreening function of mycosporine-like amino acids. In Free Radicals in Chemistry, Biology and Medicine; Yoshikawa, T, Toyokuni, S, Yamamoto, Y, Naito, Y, Eds.; OICA International: London, UK, 2000; pp. 215–228. [Google Scholar]
- Sinha, RP; Klisch, M; Gröniger, A; Häder, D-P. Mycosporine-like amino acids in the marine red alga Gracilaria cornea—effects of UV and heat. Environ Exp Bot 2000, 43, 33–43. [Google Scholar]
- Arbeloa, EM; Uez, MJ; Bertolotti, SG; Churio, MS. Antioxidant activity of gadusol and occurrence in fish roes from Argentine Sea. Food Chem 2010, 119, 586–591. [Google Scholar]
- Gao, K; Yu, H; Brown, MT. Solar PAR and UV radiation affects the physiology and morphology of the cyanobacterium Anabaena sp. PCC 7120. J Photochem Photobiol B 2007, 89, 117–124. [Google Scholar]
- Inoue, Y; Sakurai, T; Tokitomo, Y; Saito, J; Misonou, T. Measurements of fluorescence quantum yield of ultraviolet-absorbing substance extracted from red alga: Porphyra yezoensis and its phototermal spectroscopy. Opt Rev 2002, 9, 75–80. [Google Scholar]
- Bernillon, J; Parussini, E; Letoublon, R; Favre-Bonvin, J; Arpin, N. Flavin-mediated photolysis of mycosporines. Phytochemistry 1990, 29, 81–84. [Google Scholar]
- Whitehead, K; Hedges, JI. Photodegradation and photosensitization of mycosporine-like aminoacids. J Photochem Photobiol B Biol 2005, 80, 115–121. [Google Scholar]
- Bosca, F; Lhiaubet-Vallet, V; Cuquerella, MC; Castell, JV; Miranda, MA. The triplet energy of thymine in DNA. J Am Chem Soc 2006, 128, 6318–6319. [Google Scholar]
- Misonou, T; Saitoh, J; Oshiba, S; Tokimoto, Y; Maegawa, M; Inoue, Y; Hori, H; Sakurai, T. UV-absorbing substance in the red alga Porphyra yezoensis (Bangiales, Rhodophyta) block thymine photodimer production. Mar Biotechnol 2003, 5, 194–200. [Google Scholar]
- Hirata, Y; Uemura, D; Ueda, K; Takano, S. Several compounds from Palythoa tuberculosa (Coelenterata). Pure Appl Chem 1979, 51, 1875–1883. [Google Scholar]
- Ogasawara, M; Matsunaga, T; Suzuki, H. Differential effects of antioxidants on the in vitro invasion, growth and lung metastasis of murine colon cancer cells. Biol Pharm Bull 2007, 30, 200–204. [Google Scholar]
- Tartarotti, B; Sommaruga, R. The effect of different methanol concentrations and temperatures on the extraction of mycosporine-like amino acids (MAAs) in algae and zooplankton. Arch Hydrobiol 2002, 154, 691–703. [Google Scholar]
- Dunlap, WC; Shick, JM; Yamamoto, Y. UV protection in marine organisms. I. Sunscreens, oxidative stress and antioxidants. In Free Radicals in Chemistry, Biology and Medicine; Yoshikawa, S, Toyokuni, S, Yamamoto, Y, Naito, Y, Eds.; OICA International: London, UK, 2000; pp. 200–214. [Google Scholar]
- Yuan, YV; Walsh, NA. Antioxidant and antiproliferative activities of extracts from a variety of edible seaweeds. Food Chem Toxicol 2006, 44, 1144–1150. [Google Scholar]
- Gleason, DF; Wellington, GM. Variation in UVB sensitivity of planula larvae of the coral Agaricia agaricites along a depth gradient. Mar Biol 1995, 123, 693–703. [Google Scholar]
- Chioccara, F; Della Gala, A; de Rosa, M; Novellino, E; Prota, G. Mycosporine amino acids and related compounds from the eggs of fish. Bull Soc Chim Belg 1980, 89, 1101–1106. [Google Scholar]
- Teai, T; Drollet, JH; Bianchini, JP; Cambon, A; Martín, PMV. Occurrence of ultraviolet radiation-absorbing mycosporine-like amino acids in coral mucus and whole corals of French Polynesia. Mar Freshw Res 1998, 49, 127–132. [Google Scholar]
- Carreto, JI; Roy, S; Whitehead, K; Llewellyn, C; Carignan, MO. UV-absorbing “pigments”: Mycosporine-like Amino Acids. In Phytoplankton Pigments in Oceanography; Roy, S, Egeland, ES, Llewellyn, C, Johnsen, G, Wright, S, Eds.; Cambridge University Press: Cambridge, UK, 2011; in press. [Google Scholar]
- Tsujino, I; Yabe, K; Sakurai, M. Presence of the near 358 nm UV-absorbing substances in red algae. Bull Fac Fish Hokkaido Univ 1979, 30, 100–108. [Google Scholar]
- Karsten, U; Escoubeyrou, K; Charles, F. The effect of re-dissolution solvents and HPLC columns on the analysis of mycosporine-like amino acids in the eulittoral macroalgae Prasiola crispa and Porphyra umbilicalis. Helgol Mar Res 2009, 63, 231–238. [Google Scholar]
- Nakamura, H; Kobayashi, J; Hirata, Y. Separation of mycosporine-like amino acids in marine organisms using reversed-phase high-performance liquid chromatography. J Chromatogr 1982, 250, 113–118. [Google Scholar]
- Shick, JM; Romaine-Lioud, S; Ferrier-Pagès, C; Gattuso, JP. Ultraviolet-B radiation stimulates shikimate pathway-dependent accumulation of mycosporine-like amino acids in the coral Stylophora pistillata despite decreases in its population of symbiotic dinoflagellates. Limnol Oceanogr 1999, 44, 1667–1682. [Google Scholar]
- Whitehead, K; Hedges, JI. Analysis of mycosporine-like amino acids in plankton by liquid chromatography electrospray ionization mass spectrometry. Mar Chem 2002, 80, 27–39. [Google Scholar]
- Helbling, EW; Chalker, BE; Dunlap, WC; Holm-Hansen, O; Villafane, VE. Photoacclimation of antartic marine diatoms to solar ultraviolet radiation. J Exp Mar Biol Ecol 1996, 204, 85–101. [Google Scholar]
- Apffel, A; Fisher, S; Goldberg, G; Goodley, PC; Kuhlmann, FE. Enhanced sensitivity for peptide mapping with electrospray liquid chromatography-mass spectrometry in the presence of signal suppression due to trifluoroacetic acid-containing mobile phases. J Chromatogr A 1995, 712, 177–190. [Google Scholar]
- Mirza, UA; Chait, BT. Effects of anions on the positive ion electrospray ionization mass spectra of peptides and proteins. Anal Chem 1994, 66, 2898–2904. [Google Scholar]
- Whitehead, K; Hedges, JI. Electrospray ionization tandem mass spectrometric and electron impact mass spectrometric characterization of mycosporine-like amino acids. Rapid Commun Mass Spectrom 2003, 17, 2133–2138. [Google Scholar]
- Cardozo, KH. Estudos de compostos fotoprotetores da radiaçao ultravioleta em algas: Aminoácidos tipo micosporinas (MAAs). Ph.D. Thesis; Universidade de Sao Paulo, Instituto de Química: Brasil, 2007. [Google Scholar]
- Cardozo, KH; Vessecchi, VM; Galembeck, SE; Guaratini, T; Gates, PJ; Pinto, E; Lopes, NP; Colepicolo, PA. Fragmentation study of di-acidic mycosporine-like amino acids in electrospray and nanospray mass spectrometry. J Braz Chem Soc 2009, 20, 1625–1631. [Google Scholar]
- Sommaruga, R; Whitehead, K; Shick, JM; Lobban, CS. Mycosporine-like amino acids in the zooxanthella-cilliate symbiosis Maristentor dinoferus. Protist 2006, 157, 185–191. [Google Scholar]
- Souverain, S; Rudaz, S; Veuthey, J-L. Matrix Effect in LC-ESI-MS and LC-APCI-LC-MS with off-line and on-line extraction procedures. J Chromatogr A 2004, 1058, 61–66. [Google Scholar]
- Martens-Lobenhoffer, J; Bode-Böger, SM. Chromatographic-mass spectrometric methods for the quantification of l-arginine and its methylated metabolites in biological fluids. J Chromatogr B 2007, 851, 30–41. [Google Scholar]
- Matuszewski, BK; Constanzer, ML; Chavez-Eng, CM. Strategies for the assessment of matrix effect in quantitative bioanalytical methods based on HPLC-MS/MS. Anal Chem 2003, 75, 3019. [Google Scholar]
- Garcia-Pichel, F; Castenholz, RW. Occurence of UV-absorbing, mycosporine-like compounds among cyanobacterial isolates and an estimate of their screening capacity. Appl Environ Microbiol 1993, 59, 163–169. [Google Scholar]
- Karsten, U; Sawall, T; Hanelt, D; Bishop, K; Flores-Moya, A; Figueroa, FL; Wiencke, C. Contents of UV absorbing mycosporine-like amino acids in macroalgae from polar to warm-temperate regions. Bot Mar 1998, 41, 443–453. [Google Scholar]
- Michalek-Wagner, K. Seasonal and sex specific variation in levels of photo-protecting mycosporine-like amino acids (MAAs) in softcorals. Mar Biol 2001, 139, 651–660. [Google Scholar]
- Shick, JM; Lesser, MP; Dunlap, WC; Stochaj, WR; Chalker, BE; Wu Won, J. Depth-dependent responses to solar ultraviolet radiation and oxidative stress in the zooxanthellate coral Acropora microphthalma. Mar Biol 1995, 122, 41–51. [Google Scholar]
- Shick, JM. The continuity and intensity of ultraviolet irradiation affect the kinetics of biosynthesis, accumulation, and conversion of mycosporine-like amino acids (MAAs) in the coral Stylophora pistillata. LimnolOceanogr 2004, 49, 442–458. [Google Scholar]
- Banaszak, AT; Santos, MG; LaJeunesse, TC; Lesser, MP. The distribution of mycosporine-like amino acids (MAAs) and the phylogenetic identity of symbiotic dinoflagellates in cnidarian hosts from the Mexican Caribbean. J Exp Mar Biol Ecol 2006, 337, 131–146. [Google Scholar]
- Banaszak, AT; Trench, RK. Effects of ultraviolet (UV) radiation on microalgal-invertebrate symbiosis. II. The synthesis of mycosporine-like amino acids in response to exposure to UV in Anthopleura elegantissima and Cassiopeia xamachana. J Exp Mar Biol Ecol 1995, 194, 233–250. [Google Scholar]
- Grant, PT; Middleton, C; Plack, PA; Thompson, RH. The isolation of four aminocyclohexenimines (mycosporines) and a structurally related derivative of cyclohexane-1:3-dione (gadusol) from the brine shrimp. Artemia Comp Biochem Physiol B 1985, 80, 755–759. [Google Scholar]
- Carroll, AK; Shick, JM. Dietary accumulation of mycosporine-like amino acids (MAAs) by the green sea urchin (Strongylocentrus droebachiensis). Mar Biol 1996, 124, 561–569. [Google Scholar]
- Shick, JM; Dunlap, WC; Chalker, BE; Banaszak, AT; Rosenzweig, TK. Survey of ultraviolet radiation-absorbing mycosporine-like amino acids in organs of coral reef holothuroids. Mar Ecol Prog Ser 1992, 90, 139–148. [Google Scholar]
- Ishikura, M; Kato, C; Maruyama, T. UV-absorbing substances in zooxanthellate and azooxanthellate clams. Mar Biol 1997, 128, 649–655. [Google Scholar]
- Dionisio-Sese, ML; Ishikura, M; Maruyama, T; Miyachi, S. UV-absorbing substances in the tunic of a colonial ascidian protect its symbiont, Prochloron sp., from damage by UV-B radiation. Mar Biol 1997, 128, 455–461. [Google Scholar]
- Maruyama, T; Hirose, E; Ishikura, M. Ultraviolet-light-absorbing tunic cells in didemnid ascidians hosting a symbiotic photo-oxygenic prokaryote Prochloron. Biol Bull 2003, 204, 109–113. [Google Scholar]
- Mason, DS; Schafer, F; Shick, JM; Dunlap, WC. Ultraviolet radiation-absorbing mycosporine-like amino acids (MAAs) are acquired from their diet by medaka fish (Oryzias latipes) but not by SKH-1 hairless mice. Comp Biochem Physiol A 1998, 120, 587–598. [Google Scholar]
- Eckes, M; Ulrike, E; Siebeck, UE; Dove, S; Grutter, AS. Ultraviolet sunscreens in reef fish mucus. Mar Ecol Progr Ser 2008, 353, 203–211. [Google Scholar]
- Newman, SJ; Dunlap, WC; Nicol, S; Ritz, D. Antarctic krill (Euphausia superba) acquire a UV-absorbing mycosporine-like amino acid from dietary algae. J Exper Mar Biol Ecol 2000, 255, 93–110. [Google Scholar]
- Portwich, A; Garcia-Pichel, F. Biosynthetic pathway of mycosporines (mycosporine-like amino acids) in the cyanobacterium Chlorogloeopsis sp. strain PCC 6912. Phycologia 2003, 42, 384–392. [Google Scholar]
- Singh, SP. Study on mycosporine-like amino acids (MAAs) in cyanobacteria: A biochemical, bioinformatics and molecular biology approach. Ph.D. Thesis; University of Erlangen-Nürnberg: Erlangen, Germany, 2009. [Google Scholar]
- Taira, H; Aoki, S; Yamanoha, Y; Taguchi, S. Daily variation in cellular content of UV-absorbing compounds mycosporine-like amino acids in the marine dinoflagellate Scrippsiella sweeneyae. J Photochem Photobiol B 2004, 75, 145–155. [Google Scholar]
- Carreto, JI; Lutz, VA; De Marco, SG; Carignan, MO. Fluence and wavelength dependence of mycosporine-like amino acid synthesis in the dinoflagellate Alexandrium excavatum. In Toxic Marine Phytoplankton; Graneli, E, Edler, L, Anderson, DM, Eds.; Elsevier: New York, NY, USA, 1990; pp. 275–279. [Google Scholar]
- Carreto, JI; Carignan, MO; Montoya, NG. Short-term effects of ultraviolet radiation on the dinoflagellate Alexandrium catenella. Pigment bleaching and MAAs synthesis inhibition. In Aquaculture, Environment and Marine Phytoplankton; Arzul, G, Ed.; IFREMER: Brest, France, 2002; pp. 173–190. [Google Scholar]
- Klisch, M; Häder, D-P. Wavelength dependence of mycosporine-like amino acid synthesis in Gyrodinium dorsum. J Photochem Photobiol B 2002, 66, 60–66. [Google Scholar]
- Riegger, L; Robinson, D. Photoinduction of UV-absorbing compounds in Antarctic diatoms and Phaeocystis antartica. Mar Ecol Prog Ser 1997, 160, 13–25. [Google Scholar]
- Hannach, G; Sigleo, AC. Photoinduction of UV-absorbing compounds in six species of marine phytoplankton. Mar Ecol Prog Ser 1998, 174, 207–222. [Google Scholar]
- Hernando, M; Carreto, JI; Carignan, MO; Ferreyra, GA; Gross, C. Effects of solar radiation on growth and mycosporine-like amino acids content in Thalassiosira sp., an Antarctic diatom. Polar Biol 2002, 25, 12–20. [Google Scholar]
- Singh, SP; Klisch, M; Sinha, RP; Häder, D-P. Effects of abiotic stressors on synthesis of the mycosporine-like amino acid shinorine in the cyanobacterium Anabaena variabilis PCC 7937. Photochem. Photobiol 2008, 84, 1500–1505. [Google Scholar]
- Castenholz, RW; Garcia-Pichel, F. Cyanobacterial responses to UV-radiation. In The Ecology of Cyanobacteria: Their Diversity in Time and Space; Whitton, BA, Potts, M, Eds.; Kluwer: Dordrecht, The Netherlands, 2000; pp. 591–611. [Google Scholar]
- Lesser, MP. Depth-dependent photoacclimatization to solar ultraviolet radiation in the Caribbean coral Montastraea faveolata. Mar Ecol Prog Ser 2000, 192, 137–151. [Google Scholar]
- Hoyer, K; Karsten, U; Wiencke, C. Induction of sunscreen compounds in Antarctic macroalgae by different radiation conditions. Mar Biol 2002, 141, 619–627. [Google Scholar]
- Zudaire, L; Roy, S. Photoprotection and long-term acclimation to UV radiation in the marine diatom Thalassiosira weissflogii. J Photochem Photobiol B 2001, 62, 26–34. [Google Scholar]
- Carignan, MO; Montoya, NG; Carreto, JI. Long-term effects of ultraviolet radiation on the composition of pigment and mycosporine-like amino acids (MAAs). In Aquaculture, Environment and Marine Phytoplankton; Arzul, G, Ed.; IFREMER: Brest, France, 2002; pp. 191–207. [Google Scholar]
- Franklin, L; Kräbs, G; Kuhlenkamp, R. Blue light and UV-A radiation control the synthesis of mycosporine-like amino acids in Chondrus crispus (Florideophyceae). J Phycol 2001, 37, 257–270. [Google Scholar]
- Kräbs, G; Bischof, K; Hanelt, D; Tüg, H; Wienke, C. Wavelength-dependence induction of UV-absorbing mycosporine-like amino acids in the red alga Chondrus crispus under natural solar radiation. J Exp Mar Biol 2002, 268, 69–82. [Google Scholar]
- Kräbs, G; Watanabe, M; Wiencke, CA. Monochromatic action spectrum for the photoinduction of the UV-absorbing mycosporine-like amino acid shinorine in the red alga Chondrus crispus. Photochem Photobiol 2004, 79, 515–519. [Google Scholar]
- Portwich, A; Garcia-Pichel, F. A novel prokaryotic UV-B photoreceptor in the cyanobacterium Chlorogloeopsis PCC 6912. Photochem Photobiol 2000, 71, 493–498. [Google Scholar]
- Portwich, A; Garcia-Pichel, F. Ultraviolet and osmotic stress induce and regulate the synthesis of mycosporines in the cyanobacterium Chlorogloeopsis PCC 6912. Arch. Microbiol 1999, 172, 187–192. [Google Scholar]
- Vernet, M; Whitehead, K. Release of ultraviolet-absorbing compounds by the red-tide dinoflagellate Lingulodinium polyedra. Mar Biol 1996, 127, 35–44. [Google Scholar]
- Litchman, E; Neale, PJ; Banaszak, AT. Increased sensitivity to ultraviolet radiation in nitrogen-limited dinoflagellates: Photoprotection and repair. Limnol Oceanogr 2002, 47, 86–94. [Google Scholar]
- Frame, ER. Mycosporine-like amino acids (MAAs) in bloom forming phytoplankton: The influence of nitrogen, ultraviolet radiation and species composition. PhD Thesis; University of California: San Diego, CA, USA, 2004. [Google Scholar]
- Klisch, M; Häder, D-P. Mycosporine-like amino acids and marine toxins—The common and the different. Mar Drugs 2008, 6, 147–163. [Google Scholar]
- Montoya, NG; Fulco, VK; Carignan, MO; Carreto, JI. Toxin variability in cultured and natural populations of Alexandrium tamarense from southern South America—Evidences of diversity and environmental regulation. Toxicon 2010, 56, 1408–1418. [Google Scholar]
- Etheridge, SM. Paralytic shellfish poisoning: Sea food safety and human health perspectives. Toxicon 2010, 56, 108–122. [Google Scholar]
- Singh, SP; Klisch, M; Sinha, RP; Häder, D-P. Sulfur deficiency changes mycosporine-like amino acid (MAA) composition of Anabaena variabilis PCC 7937: A possible role of sulfur in MAA bioconversion. Photochem Photobiol 2010, 86, 862–870. [Google Scholar]
- Karsten, U; Bischof, K; Hanelt, D; Tüg, H; Wienke, C. The effect of ultraviolet radiation on photosynthesis and ultraviolet-absorbing substances in the endemic macroalgae Devaleraea ramentacea (Rhodophyta). Physiol Plant 1999, 105, 58–66. [Google Scholar]
- Karsten, U; Sawall, T; Hanelt, D; Bischof, K; Figueroa, FL; Flores-Moya, A; Wiencke, C. An inventory of UV-absorbing mycosporine like amino acids in macroalgae from polar to warm temperate regions. Bot Mar 1998, 41, 443–453. [Google Scholar]
- Franklin, LA; Yakovleva, I; Karsten, U; Lüning, K. Synthesis of mycosporine-like amino acids in Chondrus crispus (Florideophyceae) and the consequences for sensitivity to ultraviolet B radiation. J Phycol 1999, 35, 682–693. [Google Scholar]
- He, YY; Klisch, M; Häder, D-P. Adaptation of cyanobacteria to UV-B stress correlated with oxidative stress and oxidative damage. Photochem Photobiol 2002, 76, 188–196. [Google Scholar]
- Wu, X; Flatt, PM; Oliver Schlörke, O; Zeeck, A; Dairi, T; Mahmud, T. A comparative analysis of the sugar phosphate cyclase superfamily involved in primary and secondary metabolism. ChemBioChem 2007, 8, 239–248. [Google Scholar]
- Sinha, RP; Ambasht, NK; Sinha, JP; Klisch, M; Häder, D-P. UV-B-induced synthesis of mycosporine-like amino acids in three strains of Nodularia (cyanobacteria). J Photochem Photobiol B 2003, 71, 51–58. [Google Scholar]
- Wulff, A; Mohlin, M; Sundback, K. Intraspecific variation in the response of the cyanobacterium Nodularia spumigena to moderate UV-B radiation. Harmful Algae 2007, 6, 388–399. [Google Scholar]
- Lesser, MP; Stochaj, WR; Tapley, DW; Shick, JM. Bleaching in coral reef anthozoans: Effects of irradiance, ultraviolet radiation and temperature on the activities of protective enzymes against active oxygen. Coral Reefs 1990, 8, 225–232. [Google Scholar]
- Karsten, U; García Pichel, F. Carotenoids and mycosporine-like amino acids compounds in members of the genus Microcoleus (Cyanobacteria): A chemosystematic study. System Appl Microbiol 1996, 19, 285–294. [Google Scholar]
- Sommaruga, R; Garcia-Pichel, F. UV-absorbing mycosporine-like compounds in planktonic and benthic organisms from a high-mountain lake. Arch Hydrobiol 1999, 144, 255–269. [Google Scholar]
- Partensky, F; Hess, WR; Vaulot, D. Prochlorococcus, a marine photosynthetic prokaryote of global significance. Microbiol Mol Biol Rev 1999, 63, 106–127. [Google Scholar]
- Carignan, MO; Carreto, JI. Instituto Nacional de Investigación y Desarrollo Pesquero, Mar del Plata, Argentina. Unpublished work, 2008.
- Carreto, JI; Montoya, NG; Akselman, R; Carignan, MO; Silva, RI; Cucchi Colleoni, DA. Algal pigment patterns and phytoplankton assemblages in different water masses of the Río de la Plata maritime front. Cont Shelf Res 2008, 28, 1589–1607. [Google Scholar]
- Sinha, RP; Kumari, S; Rastogi, RP. Impacts of ultraviolet-B radiation on cyanobacteria photoprotection and repair. J Sci Res 2008, 52, 125–142. [Google Scholar]
- Bhattachaarya, D; Yoon, HS; Hackett, JD. Photosynthetic eukaryotes unite: Endosymbiosis connects the dots. Bioessays 2004, 26, 50–60. [Google Scholar]
- Waller, RF; Slamovits, CH; Keeling, PJ. Lateral gene transfer of a multigene region from cyanobacteria to dinoflagellates resulting in a novel plastid-targeted fusion protein. Mol Biol Evol 2006, 23, 1437–1443. [Google Scholar]
- Banaszak, AT; Lesser, MP. Survey of mycosporine-like amino acids in macrophytes of Kaneohe Bay. In Ultraviolet Radiation and Coral Reefs Hawaii Institute of Marine Biology Technical Report 41; Gulko, D, Jokiel, PL, Eds.; University of Hawaii: Honolulu, HI, USA, 1995; pp. 171–179. [Google Scholar]
- Lesser, MP. Elevated temperatures and ultraviolet radiation cause oxidative stress and inhibit photosynthesis in symbiotic dinoflagellates. Limnol Oceanogr 1996, 41, 271–283. [Google Scholar]
- Lesser, MP. Acclimation of phytoplankton to UV-B radiation: Oxidative stress and photoinhibition of photosynthesis are not preventing by UV-absorbing compounds in the dinoflagellate Prorocentrum micans. Mar Ecol Prog Ser 1996, 132, 287–297. [Google Scholar]
- Banaszak, AT; LaJeunesse, TC; Trench, RK. The synthesis of mycosporine-like amino acids (MAAs) by cultured, symbiotic dinoflagellates. J Exp Mar Biol Ecol 2000, 249, 219–233. [Google Scholar]
- Cembella, AD. Ecophysiology and metabolism of paralytic shellfish toxins in marine microalgae. In Physiological Ecology of Harmful Algal Blooms; Anderson, DM, Cembella, AD, Hallegraeff, GM, Eds.; Springer-Verlag: New York, USA, 1998; pp. 381–403. [Google Scholar]
- Wang, D-Z; Zhang, S-G; Gu, H-F; Chana, LL; Hua-Sheng Hong, H-S. Paralytic shellfish toxin profiles and toxin variability of the genus Alexandrium (Dinophyceae) isolated from the Southeast China Sea. Toxicon 2006, 48, 138–151. [Google Scholar]
- Moustafa, JE; Loram, J; Hackett, D; Anderson, DM; Plumley, GF; Bhattacharya, D. Origin of saxitoxin biosynthetic genes in cyanobacteria. PLoS ONE 2009, 4, 57–58. [Google Scholar]
- Doucette, GJ. Assessment of the interaction of prokaryotic cells with harmful algal species. In Harmful Marine Algae Blooms Technique & Documentation; Lassus, P, Arzul, G, Erard, E, Gentien, P, Marcaillou, C, Eds.; Lavoisier Ltd: Paris, France, 1995; pp. 385–400. [Google Scholar]
- Arai, T; Nishijima, M; Adachi, K; Sano, H. Isolation and Structure of a UV Absorbing Substance from the Marine Bacterium Micrococcus sp. AK-334; Marine Biotechnology Institute: Tokyo, Japan, 1992; pp. 88–94. [Google Scholar]
- Singh, SP; Kumari, S; Rastogi, RP; Singh, KL; Sinha, RP. Mycosporine-like amino acids (MAAs): Chemical structure, biosynthesis and significance as UV-absorbing/screening compounds. Indian J Exp Biol 2008, 46, 7–17. [Google Scholar]
- Carreto, JI; Carignan, MO. Instituto Nacional de Investigación y Desarrollo Pesquero, Mar del Plata, Argentina. Unpublished work, 2011.
- Marchant, HJ; Davidson, AT; Kelly, GJ. UV-B protecting compounds in the marine alga Phaeocystis pouchetii from Antarctica. Mar Biol 1991, 109, 391–395. [Google Scholar]
- Moisan, TA; Mitchell, BG. UV absorption by mycosporine-like amino acids in Phaeocystis antarctica induced by photosynthetically available radiation. Mar Biol 2001, 138, 217–227. [Google Scholar]
- Buma, AGJ; van Oijen, T; van de Poll, W; Veldhuis, MJW; Gieskes, WWC. The sensitivity of Emiliania huxleyi (Prymnesiophyceae) to ultraviolet-B radiation. J Phycol 2000, 36, 296–303. [Google Scholar]
- Marshall, JA; Newman, S. Differences in photoprotective pigment production between Japanese and Australian strains of Chattonella marina (Raphidophyceae). J Exp Mar Biol Ecol 2002, 272, 13–27. [Google Scholar]
- Xiong, F; Komenda, J; Kopecky, J; Nedbal, L. Strategies of ultraviolet-B protection in microscopic algae. Physiol Plant 1997, 100, 378–388. [Google Scholar]
- Sonntag, B; Summerer, M; Sommaruga, R. Sources of mycosporine-like amino acids in planktonic Chlorella-bearing ciliates (Ciliophora). Freshw Biol 2007, 52, 1476–1485. [Google Scholar]
- Karsten, U; Friedl, T; Schumann, R; Hoyer, K; Lembcke, S. Mycosporine-like amino acids and phylogenies in green algae: Prasiola and its relatives from the Trebouxiophyceae (Chlorophyta). J Phycol 2005, 41, 557–566. [Google Scholar]
- Karsten, U; Lembcke, S; Schumann, R. The effect of ultraviolet radiation on photosynthetic performance, growth and sunscreen compound in aeroterrestrial biofilm algae isolated from building facades. Planta 2007, 225, 991–1000. [Google Scholar]
- Vernet, M; Brody, EA; Holm-Hansen, O; Mitchell, BG. The response of Antarctic phytoplankton to ultraviolet radiation: Ansorption, photosynthesis, and taxonomic composition. Antarct Res Ser 1994, 62, 143–158. [Google Scholar]
- Ferreyra, GA; Schloss, I; Demers, S; Neale, PJ. Phytoplankton responses to natural ultraviolet irradiance during early spring in the Weddell-Scotia Confluence: An experimental approach. Antarc J Unit States 1994, 29, 268–270. [Google Scholar]
- Whitehead, K; Vernet, M. Influence of mycosporine-like amino acids (MAAs) on UV absorption by particulate and dissolved organic matter in La Jolla Bay. Limnol Oceanogr 2000, 45, 1788–1796. [Google Scholar]
- Negri, RM; Carreto, JI; Benavides, H; Lutz, VA; Akselman, R. An unusual bloom of Gyrodinium cf. aureolum in the Argentine Sea. Community structure and conditioning factors. J Plankton Res 1992, 14, 261–269. [Google Scholar]
- Llewellyn, CA; Harbour, DS. A temporal study of mycosporine-like amino acids in surface water phytoplankton from the English Channel and correlation with solar irradiation. J Mar Biol Ass UK 2003, 83, 1–9. [Google Scholar]
- Riemer, U; Lamare, BM; Peake, BM. Temporal concentrations of sunscreen compounds (mycosporine-like amino acids) in phytoplankton and in the New Zealand krill, Nyctiphanes australis G.O. Sars. J Plankton Res 2007, 29, 1077–1086. [Google Scholar]
- Morrison, JR; Nelson, NB. Seasonal study of phytoplankton UV absorption at the Bermuda Atlantic Time-series Study (BATS) site. Limnol Oceanogr 2004, 49, 215–224. [Google Scholar]
- Steinberg, DK; Nelson, NB; Carlson, CA; Prusak, AC. Production of chromophoric dissolved organic matter (CDOM) in the open ocean by zooplankton and the colonial cyanobacterium Trichodesmium spp. Mar Ecol Prog Ser 2004, 267, 45–56. [Google Scholar]
- Dupouy, C; Neveux, J; Subramaniam, A; Mulholland, M; Campbell, M; Montoya, J; Carpenter, E; Capone, DG. Satellite captures Trichodesmium bloom in the Southwestern Tropical Pacific. EOS Trans Am Geophys Union 2000, 1, 13–16. [Google Scholar]
- Westberry, TK; Siegel, DA; Subramaniam, A. An improved bio-optical model for the remote sensing of Trichodesmium spp. blooms. J Geophys Res 2005, 110, C06012. [Google Scholar]
- Dupouy, C; Neveux, J; Dirbergc, G; Röttgersd, R; Tenórioe, MMB; Ouillonc, S. Bio-optical properties of the marine cyanobacteria Trichodesmium spp. J Appl Remote Sens 2008, 2, 1–17. [Google Scholar]
- Kahru, M; Mitchell, BG. Spectral reflectance and absorption of a massive red tide off southern California. J Geophys Res 1998, 103, 601–609. [Google Scholar]
- Mitchell, BG; Kahru, M. Bio-Optical Algorithms for ADEOS-GLI. J Remote Sens Soc Jpn 2009, 29, 80–85. [Google Scholar]
- Hoyer, K; Karsten, U; Wiencke, C. Photoprotective substances in Antactic macroalgae and their variations with respect to depth distributions, different tissues and developmental stages. Mar Ecol Prog Ser 2001, 211, 117–129. [Google Scholar]
- Aguilera, J; Bischof, K; Karsten, U; Hanelt, D. Seasonal variation in ecophysiological patterns in macroalgae from an Arctic fjord. II. Pigment accumulation and biochemical defence systems against high light stress. Mar Biol 2002, 140, 1087–1095. [Google Scholar]
- Yuan, YV; Westcott, ND; Huc, Ch; Kitts, DD. Mycosporine-like amino acid composition of the edible red alga, Palmaria palmata (dulse) harvested from the west and east coasts of Grand Manan Island, New Brunswick. Food Chem 2009, 112, 321–328. [Google Scholar]
- Cardozo, KHM; Marques, LG; Carvalho, VM; Carignan, MO; Pinto, E; Marinho-Soriano, E; Colepicolo, P. Analyses of photoprotective compounds in red algae from the Brazilian coast. Braz J Pharmacogn 2011, in press. [Google Scholar]
- Helbling, EW; Barbieri, ES; Sinha, RJ; Villafane, VE; Häder, D-P. Dynamics of potentially protective compounds in Rhodophyta species from Patagonia (Argentina) exposed to solar radiation. J Photochem Photobiol B 2004, 75, 63–71. [Google Scholar]
- Karsten, U; Sawall, T; Wiencke, C. A survey of the distribution of UV-absorbing substances in tropical macroalgae. Phycol Res 1998, 46, 271–279. [Google Scholar]
- Gröeniger, A; Häder, D-P. Induction of the synthesis of an UV-absorbing substance in the green alga Prasiola stipitata. J Photochem Photobiol B 2002, 66, 54–59. [Google Scholar]
- LaJeunesse, TC; Thornhill, DJ; Cox, EF; Stanton, FG; Fitt, WK; Schimdt, GW. High diversity and host specificity observed among symbiotic dinoflagellates in reef coral communities from Hawaii. Coral Reefs 2004, 23, 596–603. [Google Scholar]
- Shashar, N; Banaszak, AT; Lesser, MP; Amrami, D. Coral endolithic algae: Life in a protected environment. Pac Sci 1997, 51, 167–173. [Google Scholar]
- Yakovleva, IM; Hidaka, M. Diel fluctuations of mycosporine-like amino acids (MAAs) in shallow water scleractinian corals. Mar Biol 2004, 145, 863–873. [Google Scholar]
- Torres-Pérez, JL. Responses of two species of caribbean shallow-water branching corals to changes in ultraviolet radiation. Ph.D. Thesis; University of Puerto Rico: San Juan, Puerto Rico, 2005. [Google Scholar]
- Gleason, DF. Differential effects of ultraviolet radiation on green and brown morphs of the Caribbean coral Porites astreoides. Limnol Oceanogr 1993, 38, 1452–1463. [Google Scholar]
- Jokiel, PL; Lesser, MP; Ondrusek, ME. UV-absorbing compounds in the coral Pocillopora damicornis: Interactive effects of UV radiation, photosynthetically active radiation, and water flow. Limnol Oceanogr 1997, 42, 1468–1473. [Google Scholar]
- Corredor, JE; Bruckner, AW; Muszynski, FZ; Armstrong, RA; R García, R; Morell, JM. UV-absorbing compounds in three species of Caribbean zooxanthellate corals: Depth distribution and spectral response. Bull Mar Sci 2000, 67, 821–830. [Google Scholar]
- LaJeunesse, TC; Loh, WKW; van Woesik, R; Hoegh-Guldberg, O; Schmidt, GW; Fitt, WK. Low symbiont diversity in southern Great Barrier Reef corals, relative to those of the Caribbean. Limnol Oceanogr 2003, 48, 2046–2054. [Google Scholar]
- Dunlap, WC; Shick, JM. Ultraviolet Radiation-absorbing mycosporine-like aminoacids in coral reef organism: A biochemical and environmental perspective. J Phycol 1998, 34, 418–430. [Google Scholar]
- Yakovleva, IM; Baird, AH. Ontogenetic change in the abundance of mycosporine-like amino acids in non-zooxanthellate coral larvae. Coral Reefs 2005, 24, 443–452. [Google Scholar]
- Zeevi, BY; Kashman, Y; Benayahu, Y. Mycosporine-like amino acids in azooxanthellate and zooxanthellate early development stages of the soft coral Heteroxenia fuscescens. J Exp Mar Biol Ecol 2008, 355, 12–17. [Google Scholar]
- Geller, JB; Fitzgerald, LJ; King, CE. Fission in sea anemones: Integrative studies of life cycle evolution. Integr Comp Biol 2005, 45, 615–622. [Google Scholar]
- Daly, M; Chaudhuri, A; Gusmão, LC; Rodriguez, E. Phylogenetic relationships among sea anemones (Cnidaria: Anthozoa: Actiniaria). Mol Phylogenet Evol 2008, 48, 292–301. [Google Scholar]
- Wang, J-T; Chou, Y-J; Chou, J-H; Chen, CA; Chen, W-M. Tenacibaculum aiptasiae sp. nov., isolated from a sea anemone Aiptasia pulchella. Int J Syst Evol Microbiol 2008, 58, 761–766. [Google Scholar]
- Swanson, R; Hoegh-Guldberg, O. Amino acid synthesis in the symbiotic sea anemone Aiptasia pulchella. Mar Biol 1998, 131, 83–93. [Google Scholar]
- Huxtable, RJ. Physiological actions of taurine. Physiol Rev 1992, 72, 101–163. [Google Scholar]
- Lobban, CS; Schefter, M; Simpson, AGB; Pochon, X; Pawlowski, J; Foissner, W. Maristentor dinoferus n. gen., n. sp., a giant heterotrich ciliate (Spirotrichea: Heterotrichia) with zooxanthellae, from coral reefs on Guam, Mariana Islands. Mar Biol 2002, 140, 411–423. [Google Scholar]
- Tartarotti, B; Baffico, G; Temporetti, P; Zagarese, HE. Mycosporine-like amino acids in planktonic organisms living under different UV exposure conditions in Patagonian lakes. J Plankton Res 2004, 26, 753–762. [Google Scholar]
- Hirose, E; Otsuka, K; Ishikura, M; Maruyama, T. Ultraviolet absorption in ascidian tunic and ascidian-Prochloron symbiosis. J Mar Biol Assoc UK 2004, 84, 789–794. [Google Scholar]
- Hirose, E; Hirabayashi, S; Hori1, K; Kasai, F; Watanabe, MM. UV protection in the photosymbiotic ascidian Didemnum molle inhabiting different depths. Zool Sci 2006, 23, 57–63. [Google Scholar]
- Hirabayashi, S; Kasai, F; Watanabe, MM; Hirose, E. Contents of ultraviolet-absorbing substances in two color morphs of the photosymbiotic ascidian Didemnum molle. Hydrobiologia 2006, 571, 419–422. [Google Scholar]
- Steindler, L; Huchon, D; Avni, A; Ilan, M. 16S rRNA Phylogeny of Sponge-Associated Cyanobacteria. Appl Environ Microbiol 2005, 71, 4127–4131. [Google Scholar]
- McClintock, JB; Karentz, D. Mycosporine-like amino acids in 38 species of subtidal marine organisms from Mc Murdo Sound, Antarctica. Antartic Sci 1997, 9, 392–398. [Google Scholar]
- Büdel, B; Karsten, U; García-Pichel, F. Ultraviolet-absorbing scytonemin and mycosporine-like amino acid derivarives in exposed, rock-inhabiting cyanobacterial lichens. Oecología 1997, 112, 165–172. [Google Scholar]
- Banaszak, AT; Lesser, MP; Kuffner, IB; Ondrusek, M. Relationships between ultraviolet (UV) radiation and mycosporine-like amino acids (MAAs) in marine organisms. Bull Mar Sci 1998, 63, 617–628. [Google Scholar]
- Hylander, S; Boeing, WJ; Granéli, W; Karlsson, J; von Einem, J; Gutseit, K; Hanssona, L-A. Complementary UV-protective compounds in zooplankton. Limnol Oceanogr 2009, 54, 1883–1893. [Google Scholar]
- García, PE; Diéguez;, MC; Ferraro, MA; Zagarese, HE; Pérez, AP. Mycosporine-like amino acids in freshwater copepods: Potential sources and some factors that affect their bioaccumulation. Photochem Photobiol 2010, 86, 353–359. [Google Scholar]
- Przeslawski, R; Benkendorff, K; Davis, AR. A quantitative survey of mycosporine-like amino acids (MAAs) in intertidal egg masses from temperate rocky shores. J Chem Ecol 2006, 31, 2417–2438. [Google Scholar]
- Grynbaum, MD; Hentschel, P; Putzbach, K; Rehbein, J; Krucker, M; Nicholson, G; Albert, K. Unambiguous detection of astaxanthin and astaxanthin fatty acid esters in krill (Euphausia superba Dana). J Sep Sci 2005, 28, 1685–1693. [Google Scholar]
- Zamzow, JP. Effects of diet, ultraviolet exposure, and gender on the ultraviolet absorbance of fish mucus and ocular structures. Mar Biol 2004, 144, 1057–1064. [Google Scholar]
- Zamzow, JP. Ultraviolet-absorbing compounds in the mucus of shallow-dwelling tropical reef fishes correlate with environmental water clarity. Mar Ecol Prog Ser 2007, 343, 263–271. [Google Scholar]
- Zamzow, JP; Losey, GS. Ultraviolet radiation absorbance by coral reef fish mucus: Photo-protection and visual communication. Environ Biol Fish 2002, 63, 41–47. [Google Scholar]
- Drollet, JH; Teai, T; Faucon, M; Martin, PMV. Field study of compensatory changes in UV-absorbing compounds in the mucus of the solitary coral Fungia repanda (Scleractinia: Fungiidae) in relation to solar UV radiation, sea-water temperature, and other coincident physico-chemical parameters. Mar Freshw Res 1997, 48, 329–333. [Google Scholar]


| MAAs | Chemical Assay | m.p. Determination | Elemental Analysis | UV-Vis Spectroscopy | Optical Activity | IR Spectroscopy | MS Spectroscopy | NMR Spectroscopy | X Ray Diffraction | References |
|---|---|---|---|---|---|---|---|---|---|---|
| asterina-330 | • Methylation • H/D Interchange | No | No | Yes ɛ determined | No | No | EI-MS (high resolution) ESI-MS (high resolution) ESI-MSn | 1H-NMR | No | [17,97–99] |
| dehydroxylusujirene | No | No | No | Yes | No | No | ESI-MS/MS | No | No | [34] |
| euhalothece 362 | • Amino acid composition after acid hydrolysis | No | No | Yes | No | No | ESI-MSn (high resolution) | No | No | [77] |
| mycosporine-2-glycine | • Amino acid composition after acid hydrolysis | No | No | Yes | No | No | ESI-MSn (high resolution) | 1H-NMR 13C-NMR | No | [77,85,100] |
| mycosporine-glutamic acid-glycine | • Amino acid composition after alcaline hydrolysis | Yes | No | Yes ɛ determined | No | No | SI-MS | 1H-NMR 13C-NMR | No | [101] |
| mycosporine-glycine | • Amino acid composition after neutral or alkaline hydrolysis • Methylation | Yes after derivatization | No | Yes ɛ determined | Yes after derivatization | No | ESI-MS (high resolution) | 1H-NMR 13C-NMR | No | [67,101,102] |
| mycosporine-glycine-valine | • Amino acid composition after alkaline hydrolysis | No | No | No | No | No | No | No | No | [74] |
| mycosporine-methylamine-serine | No | No | No | Yes ɛ determined | No | No | ESI-MSn | 1H-NMR | No | [84] |
| mycosporine-methylamine-threonine | • Amino acid composition after hydrolysis (not detailed) | No | No | Yes ɛ determined | No | No | ESI-MS (high resolution) | 1H-NMR 13C-NMR | No | [103] |
| mycosporine-taurine | • Amino acid composition after hydrolysis (not detailed) | No | No | Yes | No | No | ESI-MS | No | No | [85] |
| palythene | • MAA residue after acid hydrolysis. • Catalytic hydrogenation | Yes | Yes | Yes ɛ determined | Yes | Yes | ESI-MSn | 1H-NMR 13C-NMR | Yes | [96,99,104] |
| palythine | • Amino acid composition after alcaline hydrolysis • Interchange H/D | Yes | Yes | Yes ɛ determined | Yes | Yes | ESI-MS (high resolution) ESI-MSn | 1H-NMR 13C-NMR | Yes | [95,98,99, 104,105] |
| palythine-serine | No | No | No | Yes. ɛ determined | No | No | ESI-MSn | 1H-NMR | No | [84] |
| palythine-serine sulfate | No | No | No | No | No | No | ESI-MS (high resolution) | 1H-NMR 13C-NMR | No | [88] |
| palythine-threonine sulfate | No | No | No | No | No | No | ESI-MS (high resolution) | 1H-NMR 13C-NMR | No | [88] |
| palythine-threonine | • Amino acid composition after alkaline hydrolysis • Interchange H/D | No | No | Yes | No | No | ESI-MS (high resolution) ESI-MSn | No | No | [79] |
| palythinol | • Amino acid composition after alkaline hydrolysis • Methylation • Interchange H/D | Yes | Yes | Yes. ɛ determined | Yes | Yes | ESI-FTICR-MSn (high resolution) ESI-MSn | 1H-NMR 13C-NMR | No | [98,99,104,106] |
| porphyra-334 | • Amino acid composition after alkaline hydrolysis • Methylation. | No | Yes | Yes. ɛ determined | Yes | Yes | ESI-MSn | 1H-NMR 13C-NMR | No | [90,91,99] |
| shinorine | • Amino acid composition after acid hydrolysis • Interchange H/D | Yes | Yes | Yes. ɛ determined | Yes | Yes | ESI-MS (high resolution) FD-MS ESI-MSn | 1H-NMR 13C-NMR | No | [98,99,107] |
| usujirene | • Amino acid composition after alkaline hydrolysis • MAA residue after acid hydrolysis • Catalytic hydrogenation | Yes | No | Yes | Yes | Yes | FD-MS (high resolution) FAB-MS (high resolution) | 1H-NMR 13C-NMR | No | [36,108] |
| Z-palythenic acid | • Amino acid composition after hydrolysis (not detailed) | Yes | No | Yes ɛ determined | Yes | No | FD-MS ESI-MSn | Yes | No | [92,99] |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Carreto, J.I.; Carignan, M.O. Mycosporine-Like Amino Acids: Relevant Secondary Metabolites. Chemical and Ecological Aspects. Mar. Drugs 2011, 9, 387-446. https://doi.org/10.3390/md9030387
Carreto JI, Carignan MO. Mycosporine-Like Amino Acids: Relevant Secondary Metabolites. Chemical and Ecological Aspects. Marine Drugs. 2011; 9(3):387-446. https://doi.org/10.3390/md9030387
Chicago/Turabian StyleCarreto, Jose I., and Mario O. Carignan. 2011. "Mycosporine-Like Amino Acids: Relevant Secondary Metabolites. Chemical and Ecological Aspects" Marine Drugs 9, no. 3: 387-446. https://doi.org/10.3390/md9030387




