Advancement into the Arctic Region for Bioactive Sponge Secondary Metabolites
Abstract
:1. Introduction
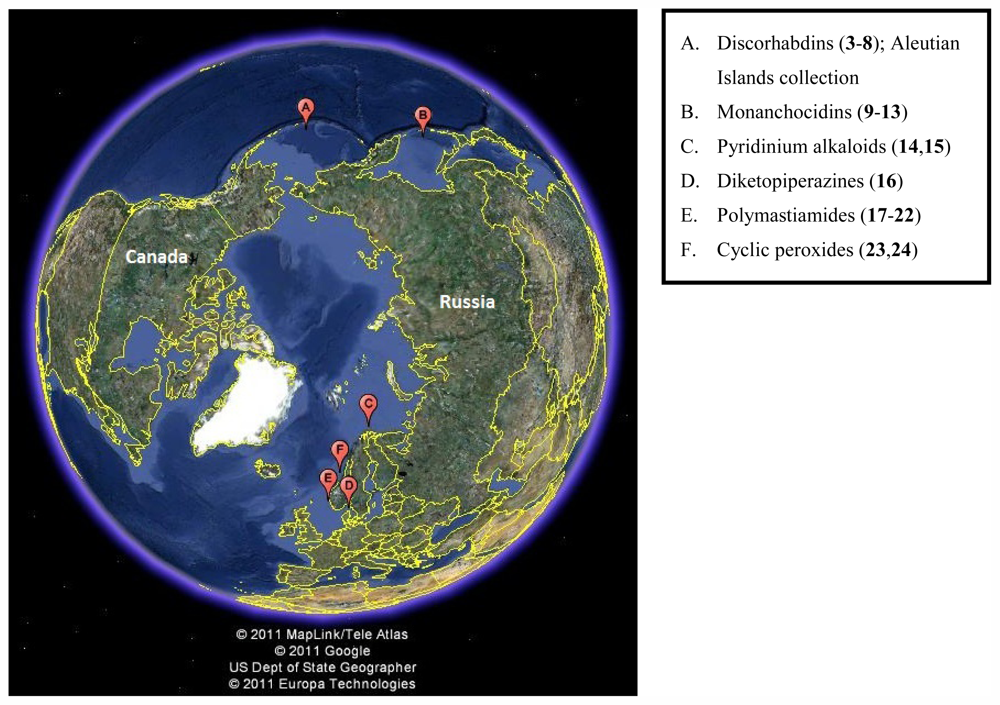
2. Secondary Metabolites from Polar Sponges
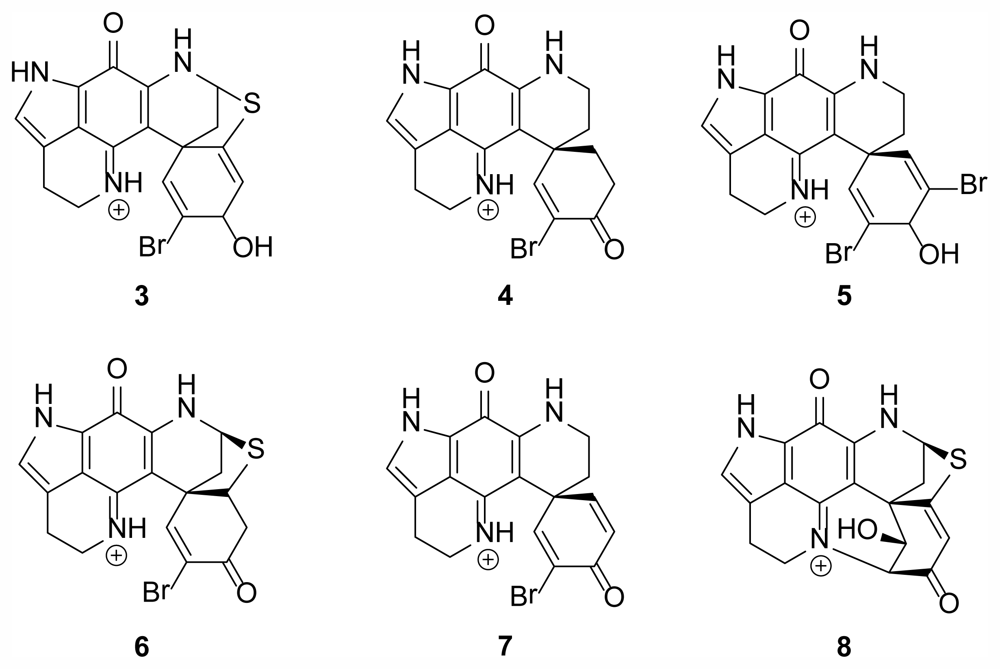
2.1. Discorhabdin Alkaloids
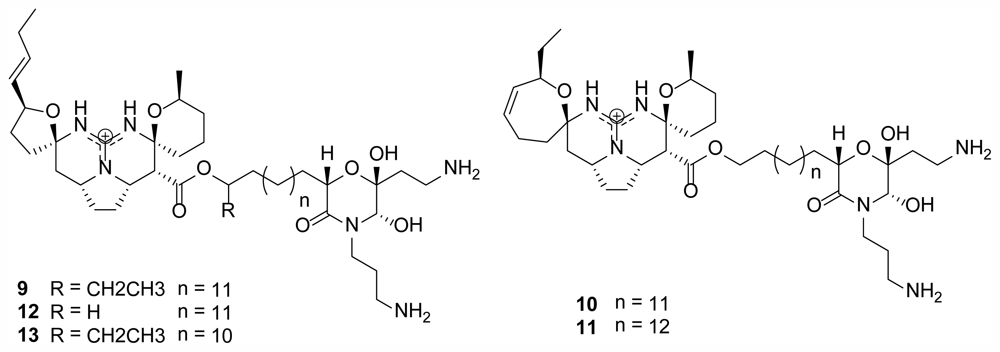
2.2. Monanchocidins
2.3. 3-Alkyl Pyridinium Alkaloids
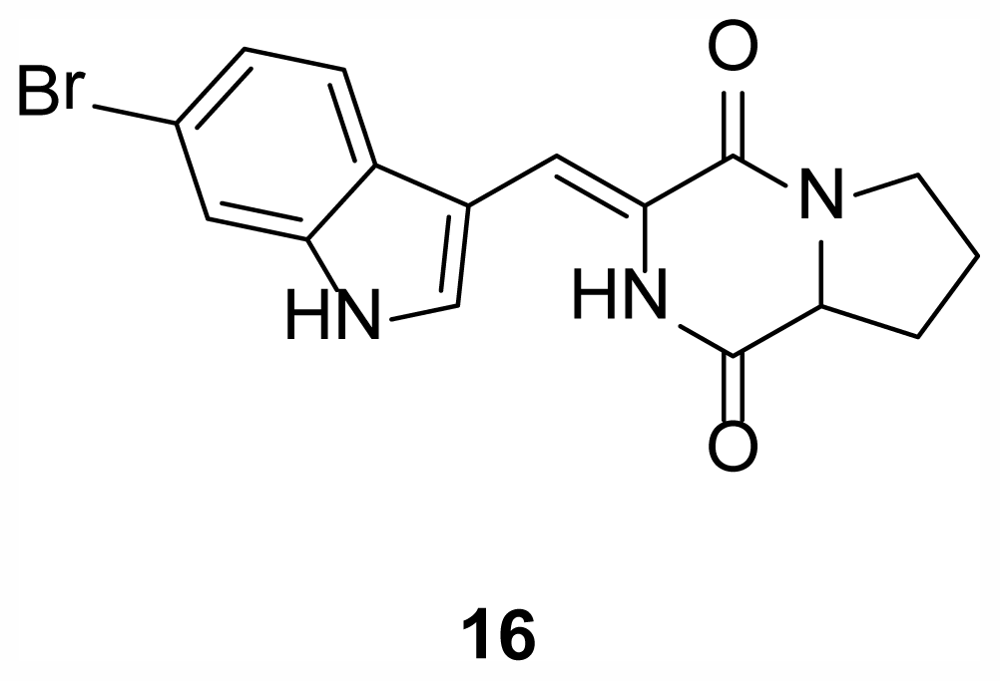
2.4. Diketopiperazines
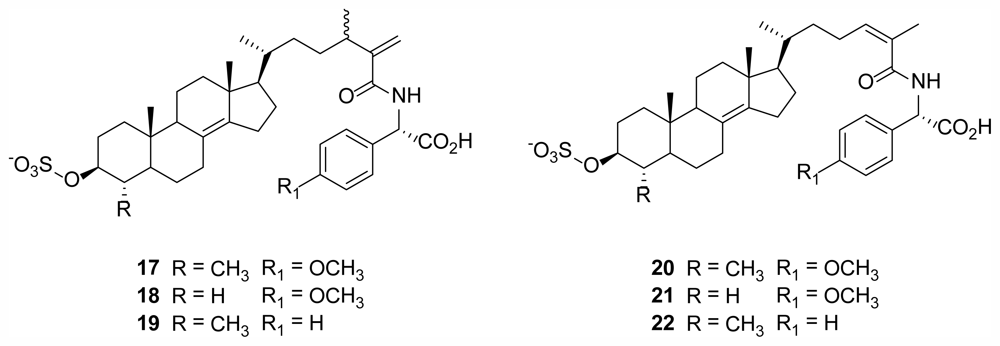
2.5. Polymastiamides
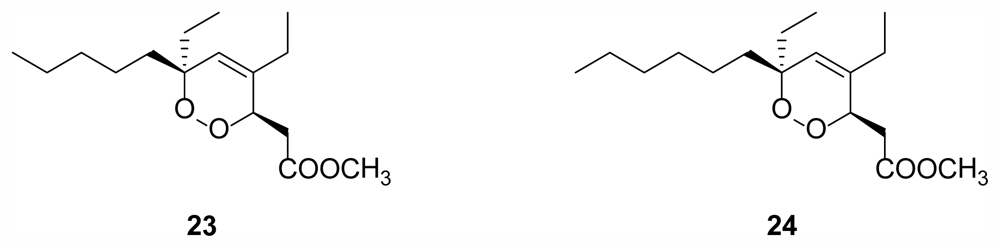
2.6. Cyclic Peroxides
3. Recent Collections Around the Aleutian Islands, Alaska
3.1. New Arctic Sponges
3.2. Isolation and Elucidation Efforts
3.3. Bioactivity
4. Conclusions
Supplementary Material
marinedrugs-09-02423-s001.pdfAcknowledgments
- Samples Availability: Available from the authors.
References
- Lebar, M.D.; Heimbegner, J.L.; Baker, B.J. Cold-water marine natural products. Nat. Prod. Rep 2007, 24, 774–797. [Google Scholar]
- Wilkins, S.P.; Blum, A.J.; Burkepile, D.E.; Rutland, T.J.; Wierzbicki, A.; Kelly, M.; Hamann, M.T. Isolation of an antifreeze peptide from the Antarctic sponge Homaxinella balfourensis. Cell Mol. Life Sci 2002, 59, 2210–2215. [Google Scholar]
- McClintock, J.B.; Amsler, C.D.; Baker, B.J.; van Soest, R.W.M. Ecology of antarctic marine sponges: An overview. Integr. Comp. Biol 2005, 45, 359–368. [Google Scholar]
- Na, M.; Ding, Y.; Wang, B.; Tekwani, B.L.; Schinazi, R.F.; Franzblau, S.; Kelly, M.; Stone, R.; Li, X.-C.; Ferreira, D.; et al. Anti-infective discorhabdins from deep-water Alaskan sponge of the genus Latrunculia. J. Nat. Prod 2010, 73, 383–387. [Google Scholar]
- Page, M.J.; Alcock, N.; Gordon, D.; Kelly, M.; Nelson, W.; Neill, K.; Watson, J. In Preliminary Assessment of the Biodiversity of Benthic Macrobiota of the Western Ross Sea, Antarctica; Final Research Report for the Ministry of Fisheries Research Project ZBD2000/02, Objectives 1–3 (Revised). National Institute of Water and Atmospheric Research Technical Report: Auckland, New Zealand, 2002. [Google Scholar]
- Sarà, M.; Balduzzi, A.; Barbieri, M.; Bavestrello, G.; Burlando, B. Biogeographic traits and checklist of Antarctic demosponges. Polar Biol 1992, 12, 559–585. [Google Scholar]
- Piepenburg, D. Recent research on Arctic benthos: Common notions need to be revised. Polar Biol 2005, 28, 733–755. [Google Scholar]
- Kelly, M.; Edwards, A.R.; Wilkinson, M.R.; Alvarez, B.; de Cook, S.C.; Bergquist, P.R.; Buckeridge, J.S.; Campbell, H.; Reiswig, H.M.; Valentine, C.; et al. Chapter 1. Phylum Porifera Sponges. In New Zealand Inventory of Biodiversity Volume 1. Kingdom Animalia: Radiata, Lophotrochozoa, and Deuterostomia; Gordon, D.P., Ed.; Canterbury University Press: Christchurch, New Zealand, 2009; pp. 23–46. [Google Scholar]
- McClintock, J.B.; Amsler, C.D.; Baker, B.J. Overview of the chemical ecology of benthic marine invertebrates along the Western Antarctic Peninsula. Integr. Comp. Biol 2010, 50, 967–980. [Google Scholar]
- Marine Literature Database, MarinLit, version vpc15.5; University of Canterbury: Christchurch, New Zealand, 2011.
- Perry, N.B.; Blunt, J.W.; McCombs, J.D.; Munro, M.H.G. Discorhabdin C, a highly cytotoxic pigment from a sponge of the genus Latrunculia. J. Org. Chem 1986, 51, 5476–5478. [Google Scholar]
- Blunt, J.W.; Munro, M.H.G.; Battershill, C.N.; Copp, B.R.; McCombs, J.D.; Perry, N.B.; Prinsep, M.R.; Thompson, A.M. From the Antarctic to the antipodes: 45 of marine chemistry. New. J. Chem 1990, 14, 761–775. [Google Scholar]
- Yang, A.; Baker, B.J.; Grimwade, J.; Leonard, A.; McClintock, J.B. Discorhabdin alkaloids from the Antarctic sponge Latrunculia apicalis. J. Nat. Prod 1995, 58, 1596–1599. [Google Scholar]
- McClintock, J.B.; Baker, B.J. A review of the chemical ecology of antarctic marine invertebrates. Am. Zool 1997, 37, 329–342. [Google Scholar]
- Ford, J.; Capon, R.J. Discorhabdin R: A new antibacterial pyrroloiminoquinone from two Latrunculiid marine sponges, Latrunculia sp. and Negombata sp. J. Nat. Prod 2000, 63, 1527–1528. [Google Scholar]
- Urban, S.; Hickford, S.J.H.; Blunt, J.W.; Munro, M.H.G. Bioactive marine alkaloids. Curr. Org. Chem 2000, 4, 793–795. [Google Scholar]
- Samaai, T.; Gibbons, M.J.; Kelly, M.; Davies-Coleman, M. South African Latrunculiidae (Porifera: Demospongiae: Poecilosclerida): Descriptions of new species of Latrunculia du Bocage, Strongylodesma Lévi, and Tsitsikamma Samaai & Kelly. Zootaxa 2003, 371, 1–26. [Google Scholar]
- Samaai, T.; Govender, V.; Kelly, M. Cyclacanthia n. g. (Demospongiae: Poecilosclerida: Latrunculiidae incertae sedis), a new genus of marine sponges from South African waters, and description of two new species. Zootaxa 2004, 725, 1–18. [Google Scholar]
- Samaai, T.; Gibbons, M.J.; Kelly, M. Revision of the genus Latrunculia du Bocage, 1869 (Porifera: Demospongiae: Latrunculiidae) with descriptions of new species from New Caledonia and the Northeastern Pacific. Zootaxa 2006, 1127, 1–71. [Google Scholar]
- Guzii, A.G.; Makarieva, T.N.; Denisenko, V.A.; Dmitrenok, P.S.; Kuzmich, A.S.; Dyshlovoy, S.A.; Krasokhin, V.B.; Stonik, V.A. Monanchocidin: A new apoptosis-inducing polycyclic guanidine alkaloid from the marine sponge Monanchora pulchra. Org. Lett 2010, 12, 4292–4295. [Google Scholar]
- Makarieva, T.N.; Tabakmaher, K.M.; Guzii, A.G.; Denisenko, V.A.; Dmitrenok, P.S.; Shubina, L.K.; Kuzmich, A.S.; Lee, H-S.; Stonik, V.A.; Monanchocidins, B–E. Polycyclic guanidine alkaloids with potent antileukemic activities from the sponge Monanchora pulchra. J. Nat. Prod 2011, 74, 1952–1958. [Google Scholar]
- Timm, C.; Mordhorst, T.; Kock, M. Synthesis of 3-alkyl pyridinium alkaloids from the arctic sponge Haliclona viscosa. Mar. Drugs 2010, 8, 483–497. [Google Scholar]
- Volk, C.A.; Kock, M. Viscosaline: New 3-alkyl pyridinium alkaloid from the Artic sponge Haliclona viscosa. Org. Biomol. Chem 2004, 2, 1827–1830. [Google Scholar]
- Lidgren, G.; Bohlin, L.; Bergman, J. Studies of Swedish marine organisms VII. A novel biologically active indole alkaloid from the sponge Geodia baretti. Tetrahedron Lett 1986, 27, 3283–3284. [Google Scholar]
- Kong, F.; Andersen, R.J. Polymastiamide A, a novel steroid/amino acid conjugate isolated from the Norwegian marine sponge Polymastia boletiformis (Lamarck, 1815). J. Org. Chem 1993, 58, 6924–6927. [Google Scholar]
- Kong, F.; Andersen, R.J. Polymastiamides B–F, novel steroid/amino acid conjugates isolated from the Norwegian marine sponge Polymastia boletiformis. J. Nat. Prod 1996, 59, 379–385. [Google Scholar]
- Holzwarth, M.; Trendel, J.; Albrecht, P.; Maier, A.; Michaelis, W. Cyclic peroxides derived from marine sponge Plakortis simplex. J. Nat. Prod 2005, 68, 759–761. [Google Scholar]
- Lehnert, H.; Stone, R.; Heimler, W. New species of deep-sea demosponges (Porifera) from the Aleutian Islands (Alaska, USA). Zootaxa 2006, 1250, 1–35. [Google Scholar]
- Austin, W.C. Porifera. In An Annotated Check List of Marine Invertebrates in the Cold Temperate Northeast Pacific; Brinkhurst, R.O., Ed.; Khoystan Marine Laboratory: Cowichan Bay, Canada, 1985; Volume 1, pp. 21–42. [Google Scholar]
- Austin, W.C.; Ott, B. Phylum Porifera. In Marine Invertebrates of the Pacific Northwest; Kozloff, E.N., Ed.; University of Washington Press: Washington, D.C., USA, 1996; pp. 6–31. [Google Scholar]
- De Laubenfels, M.W. Sponges of the Alaskan Arctic; Smithsonian Institution: Washington, D.C., USA, 1953; Volume 121, pp. 1–22. [Google Scholar]
- Fristedt, K. Sponges from the Atlantic and Arctic Oceans and the Behring Sea. Vega-Expeditionens Vetenskap. In Iakttagelser (Nordenskiöld); 1887; Volume 4, pp. 401–471, plates 22–31. [Google Scholar]
- Burton, M. Some sponges from the Okhotsk Sea and the Sea of Japan. Explor Mers l’URSS 1935, 22, 61–79. [Google Scholar]
- Koltun, V.M. New genera and species of sponges (Spongia, Cornacuspongida) from the Okhotsk and Bering Seas. Trudÿ Zool. Instituta Akad. Nauk SSSR 1955, 18, 13–18. plate 1 (in Russian). [Google Scholar]
- Koltun, V.M. Sponge Fauna of the Northwestern Pacific from the Shallows to the Hadal Depths. In Fauna of the Kurile-Kamchatka Trench and Its Environment; Bogorov, V.G., Ed.; Institute of Oceanology of the Academy of Sciences of the U.S.S.R: Moscow, Russia, 1970; pp. 165–221. [Google Scholar]
- Lambe, L.M. On some sponges from the Pacific Coast of Canada and Behring Sea. Proc. Trans. R. Soc. Can 1892, 10, 67–78, plates III–VI. [Google Scholar]
- Lambe, L.M. Sponges from the Pacific coast of Canada. Proc. Trans. R. Soc. Can 1893, 11, 25–43, plates II–IV. [Google Scholar]
- Lambe, L.M. Sponges from the western coast of North America. Trans. R. Soc. Can 1894, 12, 113–138, plates II–IV. [Google Scholar]
- Lee, W.L.; Elvin, D.W.; Reiswig, H.M. The Sponges of California. In A Guide and Key to the Marine Sponges of California; Monterey Bay Sanctuary Foundation: Monterey, CA, USA, 2007; Volume i-x, pp. 1–265. [Google Scholar]
- Vacelet, J.; Kelly, M.; Schlacher-Hoernlinger, M. Two new species of Chondrocladia (Demospongiae: Cladorhizidae) with a new spicule type from the deep south Pacific, and a discussion of the genus Meliiderma. Zootaxa 2009, 2073, 57–68. [Google Scholar]
- Wratten, S.J.; Wolfe, M.S.; Andersen, R.J.; Faulkner, D.J. Antibiotic metabolites from a marine pseudomonad. Antimicrob. Agents Chemother 1977, 11, 411–414. [Google Scholar]
- Andersen, R.J.; Wolfe, M.S.; Faulkner, D.J. Autotoxic antibiotic production by a marine Chromobacterium. Mar. Biol 1974, 27, 281–285. [Google Scholar]
- Ha, J.-H.; Lee, D.-U.; Lee, J.-T.; Kim, J.-S.; Yong, C.-S.; Kim, J.-A.; Ha, J.-S.; Huh, K. 4-Hydroxybenzaldehyde from Gastrodia elata B1. Is active in the antioxidation of GABAergic neuromodulation of the rat brain. J. Ethnopharmacol 2000, 73, 329–333. [Google Scholar]
- Blunt, J.W.; Copp, B.R.; Hu, W.-P.; Munro, M.H.G. Marine natural products. Nat. Prod. Rep 2009, 26, 170–244. [Google Scholar]




| Monanchora pulchra | Monanchora n. sp. 1 (yellow fan) | Monanchora alaskensis | Monanchora laminachela | |
|---|---|---|---|---|
| shape | ramose fan | fan | short flabby | subglobular |
| color in life | red | yellow | brown | yellow |
| styles (interior) (μm) | 1100 | 480–510 | 262 | 840–1170 |
| styles (dermal) (μm) | 176–478 | 200–250 | 144 | 350–395 |
| microscleres 1 (μm) | 19 | 30–35 | 91 | 22–25 |
| microscleres 2 (μm) | 13 | 20 | 32 | 19–23 |
© 2011 by the authors; licensee MDPI, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Abbas, S.; Kelly, M.; Bowling, J.; Sims, J.; Waters, A.; Hamann, M. Advancement into the Arctic Region for Bioactive Sponge Secondary Metabolites. Mar. Drugs 2011, 9, 2423-2437. https://doi.org/10.3390/md9112423
Abbas S, Kelly M, Bowling J, Sims J, Waters A, Hamann M. Advancement into the Arctic Region for Bioactive Sponge Secondary Metabolites. Marine Drugs. 2011; 9(11):2423-2437. https://doi.org/10.3390/md9112423
Chicago/Turabian StyleAbbas, Samuel, Michelle Kelly, John Bowling, James Sims, Amanda Waters, and Mark Hamann. 2011. "Advancement into the Arctic Region for Bioactive Sponge Secondary Metabolites" Marine Drugs 9, no. 11: 2423-2437. https://doi.org/10.3390/md9112423
APA StyleAbbas, S., Kelly, M., Bowling, J., Sims, J., Waters, A., & Hamann, M. (2011). Advancement into the Arctic Region for Bioactive Sponge Secondary Metabolites. Marine Drugs, 9(11), 2423-2437. https://doi.org/10.3390/md9112423




