Pharmacological Analyses of Protein Kinases Regulating Egg Maturation in Marine Nemertean Worms: A Review and Comparison with Mammalian Eggs
Abstract
:1. Introduction
2. Results and Discussion
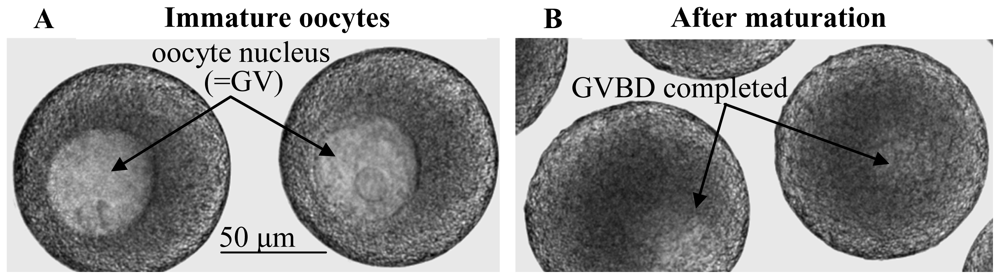
2.1. Elevations in intraoocytic cAMP trigger GVBD in nemerteans as opposed to generally inhibiting GVBD in mammals
2.2. ERK 1/2 MAPKs are activated before, but not required for, GVBD in nemerteans
2.3. Src-family kinase activation does not appear to be needed for GVBD in nemerteans
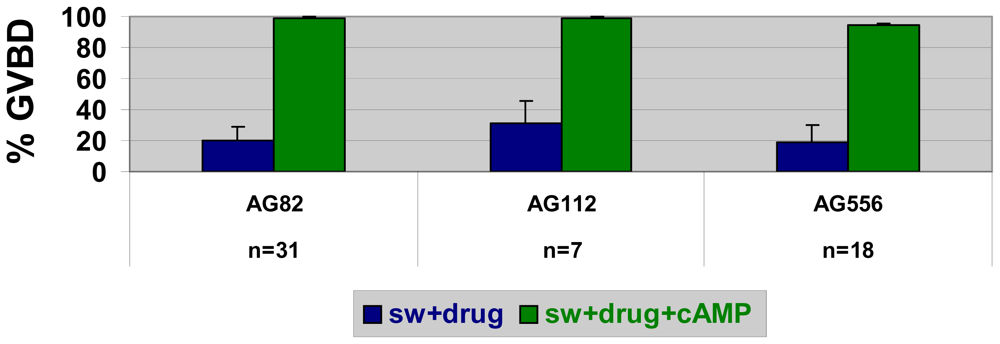
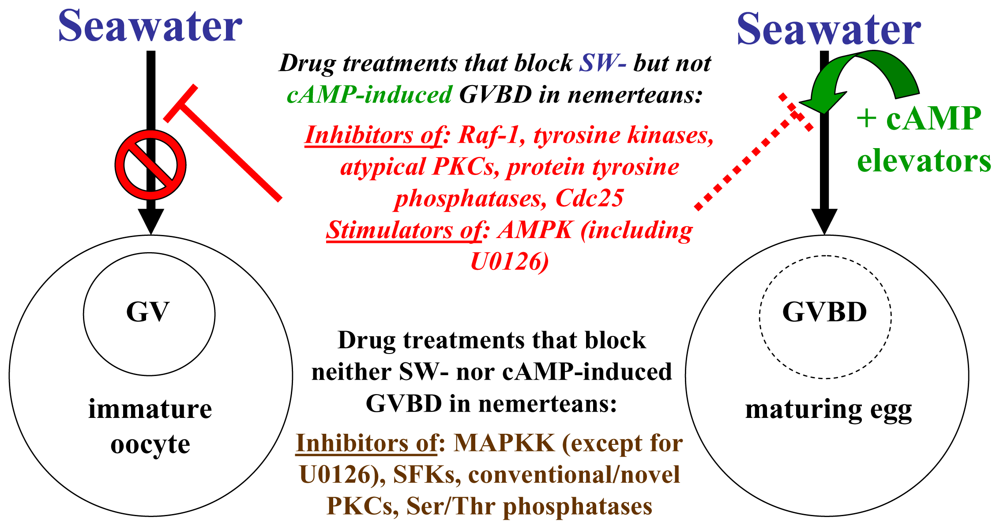
2.5. Stimulators of AMP-activated kinase block SW- but not cAMP-induced GVBD in nemerteans
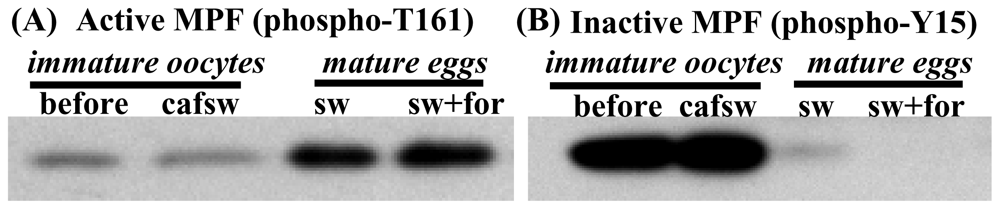
2.6. Egg maturation in nemerteans requires activation of pre-MPF following stimulation by either SW or cAMP elevators
3. Conclusions
4. Experimental
Acknowledgments
References
- Stricker, SA. Comparative biology of calcium signaling during fertilization and egg activation in animals. Dev. Biol 1999, 211, 157–176. [Google Scholar]
- Maller, JL. Maturation-promoting factor in the early days. Trends Biochem. Sci 1995, 20, 524–528. [Google Scholar]
- Nebreda, AR; Ferby, I. Regulation of the meiotic cell cycle in oocytes. Curr. Opin. Cell Biol 2000, 12, 666–675. [Google Scholar]
- Kishimoto, T. Cell-cycle control during meiotic maturation. Curr. Opin. Cell Biol 2003, 15, 654–663. [Google Scholar]
- Voronina, E; Wessel, GM. The regulation of oocyte maturation. Curr. Top. Dev. Biol 2003, 58, 53–110. [Google Scholar]
- Jones, KT. Turning it on and off: M-phase promoting factor during meiotic maturation and fertilization. Mol. Hum. Reprod 2004, 10, 1–5. [Google Scholar]
- Nagahama, Y; Yamashita, M. Regulation of oocyte maturation in fish. Dev. Growth Differ 2008, 50, S195–S219. [Google Scholar]
- Gibson, A. Nemerteans, 1st ed; Hutchinson & Co. Ltd: London, UK, 1972. [Google Scholar]
- Stricker, SA. Strathmann, MF, Ed.; Phylum Nemertea. In Reproduction and Development of Marine Invertebrates of the Northern Pacific Coast, 1st ed; University of Washington Press: Seattle, WA, USA, 1987; pp. 129–137. [Google Scholar]
- Stricker, SA; Smythe, TL; Miller, L; Norenburg, JL. Comparative biology of oogenesis in nemertean worms. Acta Zool 2001, 82, 213–230. [Google Scholar]
- Stricker, SA; Smythe, TL. Multiple triggers of oocyte maturation in nemertean worms: the roles of calcium and serotonin. J. Exp. Zool 2000, 287, 243–261. [Google Scholar]
- Mehlmann, LM. Stops and starts in mammalian oocytes: recent advances in understanding the regulation of meiotic arrest and oocyte maturation. Reproduction 2005, 130, 791–799. [Google Scholar]
- Tsafriri, A; Cao, X; Ashkenazi, H; Motola, S; Popliker, M; Pomerantz, SH. Resumption of oocyte meiosis in mammals: on models, meiosis activating sterols, steroids and EGF-like factors. Mol. Cell Endocrinol 2005, 234, 37–45. [Google Scholar]
- Sun, QY; Miao, YL; Schatten, H. Towards a new understanding on the regulation of mammalian oocyte meiosis resumption. Cell Cycle 2009, 8, 2741–2747. [Google Scholar]
- Zhang, M; Ouyang, H; Xia, G. The signal pathway of gonadotropins-induced mammalian oocyte meiotic resumption. Mol. Hum. Reprod 2009, 15, 399–409. [Google Scholar]
- Stricker, SA; Smythe, TL. 5-HT causes an increase in cAMP that stimulates, rather than inhibits, oocyte maturation in marine nemertean worms. Development 2001, 128, 1415–1427. [Google Scholar]
- Cho, WK; Sterns, S; Biggers, JD. Inhibitory effect of dibutyryl cAMP on mouse oocyte maturation in vitro. J. Exp. Zool 1974, 187, 383–386. [Google Scholar]
- Eppig, JJ; Freter, RR; Ward-Bailey, PF; Schultz, RM. Inhibition of oocyte maturation in the mouse: participation of cAMP, steroid hormones and a putative maturation-inhibitory factor. Dev. Biol 1983, 100, 39–49. [Google Scholar]
- Dekel, N. Regulation of oocyte maturation. The role of cAMP. Ann. N.Y. Acad. Sci 1988, 541, 211–216. [Google Scholar]
- Downs, SM; Daniel, SAJ; Eppig, JJ. Induction of maturation in cumulus cell-enclosed mouse oocytes by follicle stimulating hormone and epidermal growth factor: evidence for a positive stimulus of somatic cell origin. J. Exp. Zool 1988, 245, 86–96. [Google Scholar]
- Tornell, J; Billig, H; Hillensjo, T. Regulation of oocyte maturation by changes in ovarian levels of cyclic nucleotides. Hum. Reprod 1991, 6, 411–422. [Google Scholar]
- Norris, RP; Ratzan, WJ; Freudzon, M; Mehlmann, LM; Krall, J; Movsesian, MA; Wang, H; Ke, H; Nidolaev, VO; Jaffe, LA. Cyclic GMP from the surrounding somatic cells regulates cyclic AMP and meiosis in the mouse oocyte. Development 2009, 136, 1869–1878. [Google Scholar]
- Downs, SM; Hunzicker-Dunn, M. Differential regulation of oocyte maturation and cumulus expansion in the mouse oocyte-cumulus cell complex by site-selective analogs of cyclic adenosine monophosphate. Dev. Biol 1995, 172, 72–85. [Google Scholar]
- Webb, RJ; Marshall, F; Swann, K; Carroll, J. Follicle-stimulating hormone induces a gap junction-dependent dynamic change in [cAMP] and protein kinase A in mammalian oocytes. Dev. Biol 2002, 246, 441–454. [Google Scholar]
- Sela-Abramovich, S; Edry, I; Galiani, D; Nevo, N; Dekel, N. Disruption of gap junctional communication within the ovarian follicle induces oocyte maturation. Endocrinology 2006, 147, 2280–2286. [Google Scholar]
- Norris, RP; Freudzon, M; Mehlmann, LM; Cowan, AE; Simon, AM; Paul, DL; Lampe, PD; Jaffe, LA. Luteinizing hormone causes MAP kinase-dependent phosphorylation and closure of connexin 43 gap junctions in mouse ovarian follicles: one of two paths to meiotic resumption. Development 2008, 135, 3229–3238. [Google Scholar]
- Sasseville, M; Gagnon, MC; Guillemette, C; Sullivan, R; Gilchrist, RB; Richard, FJ. Regulation of gap junctions in porcine cumulus-oocyte complexes: contributions of granulosa cell contact, gonadotropins, and lipid rafts. Mol. Endocrinol 2009, 23, 700–710. [Google Scholar]
- Chen, J; Chi, MM; Moley, KH; Downs, SM. cAMP pulsing of denuded mouse oocytes increases meiotic resumption via activation of AMP-activated protein kinase. Reproduction 2009, 138, 759–770. [Google Scholar]
- Freeman, G; Ridgway, EB. The role of cAMP in oocyte maturation and the role of the germinal vesicle contents in mediating maturation and subsequent developmental events in hydrozoans. Roux’s Arch. Dev. Biol 1988, 197, 197–211. [Google Scholar]
- Yamashita, M. Involvement of cAMP in initiating maturation of the brittle-star Amphipholis kochii oocytes: induction of oocyte maturation by inhibitors of cyclic nucleotide phosphodiesterase and activators of adenylate cyclase. Dev. Biol 1988, 125, 109–114. [Google Scholar]
- Yi, JH; Lefièvre, L; Gagnon, C; Anctil, M; Dubé, F. Increase of cAMP upon release from prophase arrest in surf clam oocytes. J. Cell. Sci 2002, 115, 311–320. [Google Scholar]
- Takeda, N; Kyozuka, K; Deguchi, R. Increase in intracellular cAMP is a prerequisite signal for initiation of physiological oocyte meiotic maturation in the hydrozoan Cytaeis uchidae. Dev. Biol 2006, 298, 248–258. [Google Scholar]
- Lambert, CC. Signaling pathways in ascidian oocyte maturation: the role of cyclic AMP and follicle cells in germinal vesicle breakdown. Dev. Growth Differ 2008, 50, 181–188. [Google Scholar]
- Webb, RJ; Tinworth, L; Thomas, GM; Zaccolo, M; Carroll, J. Developmentally acquired PKA localisation in mouse oocytes and embryos. Dev. Biol 2008, 317, 36–45. [Google Scholar]
- Stricker, SA; Smythe, TL. Differing mechanisms of cAMP- versus seawater-induced oocyte maturation in marine nemertean worms. II. The roles of tyrosine kinases and phosphatases. Mol. Reprod. Dev 2006, 73, 1564–1577. [Google Scholar]
- Stricker, SA. Roles of protein kinase C isotypes during seawater- versus cAMP-induced oocyte maturation in a marine worm. Mol. Reprod. Dev 2009, 76, 693–707. [Google Scholar]
- Wilkinson, MG; Millar, JB. Control of the eukaryotic cell cycle by MAP kinase signaling pathways. FASEB J 2000, 14, 2147–2157. [Google Scholar]
- Chang, L; Karin, M. Mammalian MAP kinase signalling cascades. Nature 2001, 410, 37–40. [Google Scholar]
- Raman, M; Chen, W; Cobb, MH. Differential regulation and properties of MAPKs. Oncogene 2007, 26, 3100–3112. [Google Scholar]
- Sturgill, TW. MAP kinase: it’s been longer than fifteen minutes. Biochem. Biophys. Res. Commun 2008, 371, 1–4. [Google Scholar]
- Stricker, SA; Smythe, TL. Endoplasmic reticulum reorganizations and Ca2+ signaling in maturing and fertilized oocytes of marine protostome worms: The roles of MAPKs and MPF. Development 2003, 130, 2867–2879. [Google Scholar]
- Smythe, TL; Stricker, SA. Germinal vesicle breakdown is not fully dependent on MAPK activation in maturing oocytes of marine nemertean worms. Mol. Reprod. Dev 2005, 70, 91–102. [Google Scholar]
- Stricker, SA; Smythe, TL. Differing mechanisms of cAMP- versus seawater-induced oocyte maturation in marine nemertean worms I. The roles of serine/threonine kinases and phosphatases. Mol. Reprod. Dev 2006, 73, 1578–1590. [Google Scholar]
- Stricker, SA. Interactions between mitogen-activated protein kinase and protein kinase C signaling during oocyte maturation and fertilization in a marine worm. Mol. Reprod. Dev 2009, 76, 708–721. [Google Scholar]
- Fan, HY; Sun, QY. Involvement of mitogen-activated protein kinase cascade during oocyte maturation and fertilization in mammals. Biol. Reprod 2004, 70, 535–547. [Google Scholar]
- Liang, CG; Su, YQ; Fan, HY; Schatten, H; Sun, QY. Mechanisms regulating oocyte meiotic resumption: roles of mitogen-activated protein kinase. Mol. Endocrin 2007, 21, 2037–2055. [Google Scholar]
- Fan, HY; Liu, Z; Shimada, M; Sterneck, E; Johnson, PF; Hedrick, SM; Richards, JS. MAPK3/1 (ERK1/2) in ovarian granulosa cells are essential for female fertility. Science 2009, 324, 938–941. [Google Scholar]
- Yamashita, Y; Hishinuma, M; Shimada, M. Activation of PKA, p38 MAPK and ERK1/2 by gonadotropins in cumulus cells is critical for induction of EGF-like factor and TACE/ADAM17 gene expression during in vitro maturation of porcine COCs. J. Ovarian Res 2009, 2, 20:1–20:9. [Google Scholar]
- Spivack, JG; Maller, JL. Phosphorylation and protein synthetic events in Xenopus laevis oocytes microinjected with pp60v-src. Mol. Cell. Biol 1985, 5, 3629–3633. [Google Scholar]
- Stricker, SA; Carroll, DJ; Tsui, WL. Roles of Src family kinase signaling during fertilization and the first cell cycle in the marine protostome worm Cerebratulus. Int. J. Dev. Biol 2010, 54, 787–793. [Google Scholar]
- McGinnis, LK; Kinsey, WH; Albertini, DF. Functions of Fyn kinase in the completion of meiosis in mouse oocytes. Dev. Biol 2009, 327, 280–287. [Google Scholar]
- Zheng, KG; Meng, XQ; Yang, Y; Yu, YS; Liu, DC; Li, YL. Requirements of Src family kinase during meiotic maturation in mouse oocyte. Mol. Reprod. Dev 2007, 74, 125–130. [Google Scholar]
- Sette, C; Paronetto, MP; Barchi, M; Bevilacqua, A; Geremia, R; Rossi, P. Tr-kit-induced resumption of the cell cycle in mouse eggs requires activation of a Src-like kinase. EMBO J 2002, 21, 5386–5395. [Google Scholar]
- Talmor-Cohen, A; Tomashov-Matar, R; Eliyahu, E; Shapiro, R; Shalgi, R. Are Src family kinases involved in cell cycle resumption in rat eggs? Reproduction 2004, 127, 455–463. [Google Scholar]
- McGinnis, LK; Albertini, DF; Kinsey, WH. Localized activation of Src-family protein kinases in the mouse egg. Dev. Biol 2007, 306, 241–254. [Google Scholar]
- Reut, TM; Mattan, L; Dafna, T; Ruth, KK; Ruth, S. The role of Src family kinases in egg activation. Dev. Biol 2007, 312, 77–89. [Google Scholar]
- Mellor, H; Parker, PJ. The extended protein kinase C superfamily. Biochem. J 1998, 332, 281–292. [Google Scholar]
- Webb, LJ; Hirst, SJ; Giembycz, MA. Protein kinase C isoenzymes: a review of their structure, regulation, and role in regulating airways smooth muscle tone and mitogenesis. Br. J. Pharmcol 2000, 130, 1433–1452. [Google Scholar]
- Kalive, M; Faust, JJ; Koeneman, BA; Capco, DG. Involvement of the PKC family in regulation of early development. Mol. Reprod. Dev 2010, 77, 95–104. [Google Scholar]
- Downs, SM; Cottom, J; Hunzicker-Dunn, M. Protein kinase C and meiotic regulation in isolated mouse oocytes. Mol. Reprod. Dev 2001, 58, 101–115. [Google Scholar]
- Avazeri, N; Courtot, AM; Lefevre, B. Regulation of spontaneous meiosis resumption in mouse oocytes by various conventional PKC isozymes depends on cellular compartmentalization. J. Cell Sci 2004, 117, 4969–4978. [Google Scholar]
- Bornslaeger, EA; Pouymirou, WT; Mattei, P; Schultz, RM. Effects of protein kinase C activators on germinal vesicle breakdown and polar body emission of mouse oocytes. Exp. Cell Res 1986, 165, 507–517. [Google Scholar]
- Lefevre, B; Pesty, AR; Koziak, K; Testart, J. Protein kinase C modulators influence meiosis kinetics but not fertilizability of mouse oocytes. J. Exp. Zool 1992, 264, 206–213. [Google Scholar]
- Luria, A; Tennenbaum, T; Sun, QY; Rubinstein, S; Breitbart, H. Differential localization of conventional protein kinase C isoforms during mouse oocyte development. Biol. Reprod 2000, 62, 1564–1570. [Google Scholar]
- Lu, Q; Smith, GD; Chen, DY; Yang, Z; Han, ZM; Schatten, H; Sun, QY. Phosphorylation of mitogen-activated protein kinase is regulated by protein kinase C, cyclic 3′5′-adenosine monophosphate, and protein phosphatase modulators during meiosis resumption in rat oocytes. Biol. Reprod 2001, 64, 1444–1450. [Google Scholar]
- Denys, A; Avazeri, N; Lefevre, B. The PKC pathway and in particular its beta 1 isoform is clearly involved in meiotic arrest maintenance but poorly in FSH-induced meiosis resumption of the mouse cumulus cell enclosed oocyte. Mol. Reprod. Dev 2007, 74, 1575–1580. [Google Scholar]
- Gallicano, GI; McGaughey, RW; Capco, DG. Activation of protein kinase C after fertilization is required for remodeling the mouse egg into zygote. Mol. Reprod. Dev 1997, 46, 587–601. [Google Scholar]
- Quan, HM; Fan, HY; Meng, XQ; Chen, DY; Schatten, H; Yang, PM; Sun, QY. Effects of PKC activation of the meiotic maturation, fertilization and early development of mouse oocytes. Zygote 2003, 11, 329–337. [Google Scholar]
- Hallet, G; Tunwell, R; Parkinson, SJ; Carroll, J. Conventional PKCs regulate the temporal pattern of Ca2+ oscillations at fertilization in mouse eggs. J. Cell Biol 2004, 164, 1033–1044. [Google Scholar]
- Ducibella, T; LeFevre, L. Study of protein kinase C antagonists on cortical granule exocytosis and cell cycle resumption in fertilized mouse eggs. Mol. Reprod. Dev 1997, 46, 216–226. [Google Scholar]
- Carling, D. AMP-activated protein kinase: balancing the scales. Biochimie 2005, 87, 87–91. [Google Scholar]
- Alessi, DR; Sakamoto, K; Bayascas, JR. LKB1-dependent signaling pathways. Ann. Rev. Biochem 2006, 75, 137–163. [Google Scholar]
- Hardie, DG. AMP-activated/SNF1 protein kinases: conserved guardians of cellular energy. Nat. Rev. Cell Mol. Biol 2007, 8, 774–785. [Google Scholar]
- Stricker, SA; Swiderek, L; Nguyen, T. Stimulators of AMP-activated kinase inhibit seawater-but not cAMP-induced oocyte maturation in a marine worm: implications for interactions between cAMP and AMPK signaling. Mol. Reprod. Dev 2010, 77, 497–510. [Google Scholar]
- Downs, SM; Hudson, ER; Hardie, DG. A potential role for AMP-activated protein kinase in meiotic induction in mouse oocytes. Dev. Biol 2002, 245, 200–212. [Google Scholar]
- Chen, J; Hudson, E; Chi, MM; Chang, AS; Moley, KH; Hardie, DG; Downs, SM. AMPK regulation of mouse oocyte meiotic resumption in vitro. Dev. Biol 2006, 291, 227–238. [Google Scholar]
- Chen, J; Downs, SM. AMP-activated protein kinase is involved in hormone-induced mouse oocyte meiotic maturation in vitro. Dev. Biol 2008, 313, 47–57. [Google Scholar]
- Bilodeau-Goeseels, S; Sasseville, M; Guillemette, C; Richard, FJ. Effects of adenosine monophosphate-activated kinase activators on bovine oocyte nuclear maturation in vitro. Mol. Reprod. Dev 2007, 74, 1021–1034. [Google Scholar]
- Mayes, MA; Laforest, MF; Guillemette, C; Gilchrist, RB; Richard, FJ. Adenosine 5′-monophosphate kinase-activated protein kinase (PRKA) activators delay meiotic resumption in porcine oocytes. Biol. Reprod 2007, 76, 589–597. [Google Scholar]
- Tosca, L; Uzbekova, S; Chabrolle, C; Dupont, J. Possible role of 5′ AMP-activated protein kinase in the metformin-mediated arrest of bovine oocytes at the germinal vesicle stage during in vitro maturation. Biol. Reprod 2007, 77, 452–465. [Google Scholar]
- Bilodeau-Goeseels, S. Review: The regulation of meiotic maturation in bovine oocytes. Can. J. Anim. Sci 2008, 88, 343–349. [Google Scholar]
- Ferby, I; Blazquez, M; Palmer, A; Eritja, R; Nebreda, A. A novel p34cdc2 binding and activating protein that is necessary and sufficient to trigger G2/M preogression in Xenopus oocytes. Genes Dev 1999, 13, 2177–2189. [Google Scholar]
- Kume, S; Endo, T; Nishimura, Y; Kano, K; Naito, K. Porcine SPDYA2 (RINGOA2) stimulates CDC2 activity and accelerates meiotic maturation of porcine oocytes. Biol. Reprod 2007, 76, 440–447. [Google Scholar]
- Yamashita, M; Mita, K; Yoshida, N; Kondo, T. Molecular mechanisms of the initiation of oocyte maturation: General and species specific aspects. Prog. Cell Cycle Res 2000, 4, 115–129. [Google Scholar]
- Lincoln, AJ; Wickramasinghe, D; Stein, P; Schultz, RM; Palko, ME; De Miguel, MP; Tessarollo, L; Donovan, PJ. Cdc25b phosphatase is required for resumption of meiosis during oocyte maturation. Nat. Gen 2002, 30, 446–449. [Google Scholar]
- Oh, JS; Han, JS; Conti, M. Wee 1B, Myt1, and Cdc25 function in distinct compartment of the mouse oocyte to control meiotic resumption. J. Cell Biol 2010, 188, 199–207. [Google Scholar]
- Alexandre, H; van Cauwenberge, A; Tsukitani, Y; Mulnard, J. Pleiotropic effect of okadaic acid on maturing mouse oocytes. Development 1991, 112, 971–980. [Google Scholar]
- Lu, Q; Smith, GD; Chen, DY; Yang, Z; Han, ZM; Schatten, H; Sun, QY. Phosphorylation of mitogen-activated protein kinase is regulated by protein kinase C, cyclic 3′5′-adenosine monophosphate, and protein phosphatase modulators during meiosis resumption in rat oocytes. Biol. Reprod 2001, 64, 1444–1450. [Google Scholar]
- Stricker, SA; Silva, R; Smythe, TL. Calcium and endoplasmic reticulum dynamics during oocyte maturation and fertilization in the marine worm Cerebratulus lacteus. Dev. Biol 1998, 203, 305–322. [Google Scholar]
- Schroeder, TE; Stricker, SA. Morphological changes during maturation of starfish oocytes: Surface ultrastructure and cortical actin. Dev. Biol 1983, 98, 373–384. [Google Scholar]
- Stricker, SA; Schatten, G. Nuclear envelope disassembly and nuclear lamina depolymerization during germinal vesicle breakdown in starfish. Dev. Biol 1989, 135, 87–98. [Google Scholar]
- Davies, SP; Reddy, H; Caivano, M; Cohen, P. Specificity and mechanism of action of some commonly used protein kinase inhibitors. Biochem. J 2000, 351, 95–105. [Google Scholar]

| Signal regulating egg maturation: | Positive (+) or negative (−) effect of signal on GVBD | |
|---|---|---|
| in nemerteans [refs] (notes): | in mammals [refs] (notes): | |
| Intraoocytic cAMP elevation | + [16,35,36,43] | − [17–27] |
| ERK 1/2 MAPK activation | + [41–44] (a) | + [14,45–48] (a,b) |
| Src family kinase activation | − [50] | − [51] or + [52] |
| Protein kinase C activation | + [36,44] [c] | − [60–66] (d) |
| Intraoocytic AMP kinase activation | − [74] | + [75–77] (e) |
| Increased Cdc2 kinase activity via conversion of pre-MPF into MPF | + [43] | + [4,14] |
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Stricker, S.A.; Escalona, J.R.; Abernathy, S.; Marquardt, A. Pharmacological Analyses of Protein Kinases Regulating Egg Maturation in Marine Nemertean Worms: A Review and Comparison with Mammalian Eggs. Mar. Drugs 2010, 8, 2417-2434. https://doi.org/10.3390/md8082417
Stricker SA, Escalona JR, Abernathy S, Marquardt A. Pharmacological Analyses of Protein Kinases Regulating Egg Maturation in Marine Nemertean Worms: A Review and Comparison with Mammalian Eggs. Marine Drugs. 2010; 8(8):2417-2434. https://doi.org/10.3390/md8082417
Chicago/Turabian StyleStricker, Stephen A., Jose R. Escalona, Samuel Abernathy, and Alicia Marquardt. 2010. "Pharmacological Analyses of Protein Kinases Regulating Egg Maturation in Marine Nemertean Worms: A Review and Comparison with Mammalian Eggs" Marine Drugs 8, no. 8: 2417-2434. https://doi.org/10.3390/md8082417





