Characterization of the Exopolysaccharide Produced by Salipiger mucosus A3T, a Halophilic Species Belonging to the Alphaproteobacteria, Isolated on the Spanish Mediterranean Seaboard
Abstract
:1. Introduction
2. Results
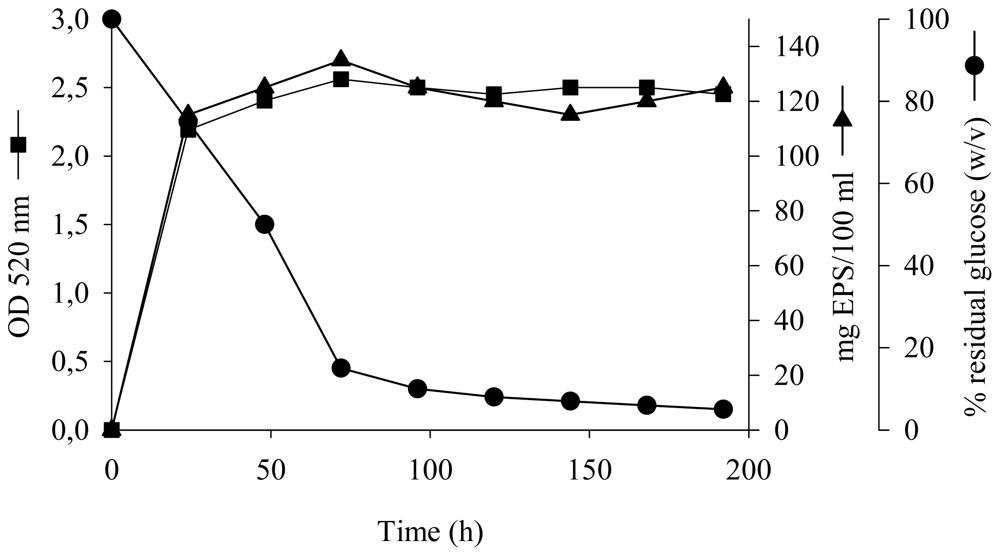
2.1. Kinetics of Growth and EPS Production
2.2. Influence of Culture Parameters on Growth and EPS Synthesis
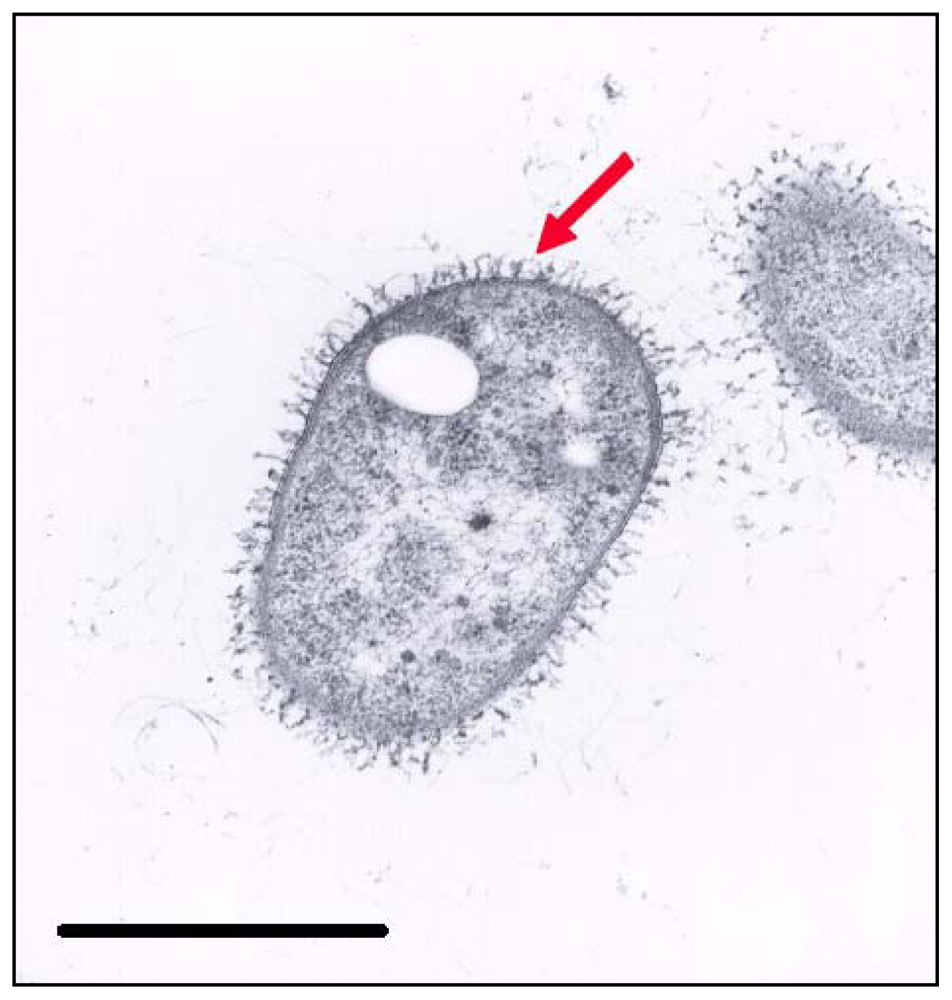
2.3. Electron Microscopy
2.4. Chemical Composition
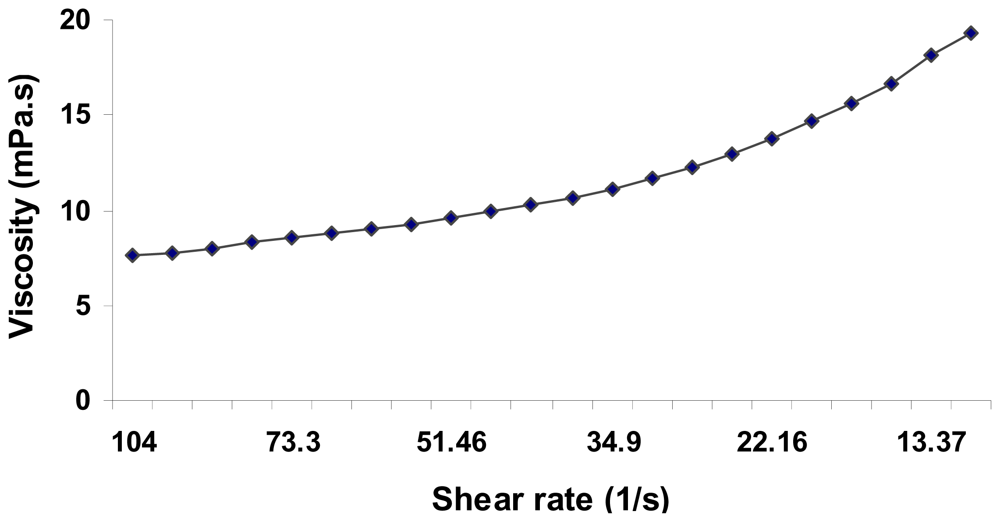
2.5. Physical Properties
3. Discussion
4. Experimental
4.1. Bacterial Strain
4.2. Optimization and Analysis of the Kinetics of EPS Production
4.3. Electron Microscopy
4.4. Chemical Analysis
4.5. Physical Properties
5. Conclusions
Acknowledgements
- Samples Availability: Available from the authors.
References
- Sutherland, IW. Biotechnology of Microbial Exopolysaccharides; Cambridge University: New York, NY, USA, 1990. [Google Scholar]
- Matou, S; Colliec-Jouault, S; Galy-Fauroux, I; Ratiskol, J; Sinquin, C; Guezennec, J; Fischer, AM; Helley, D. Effect of an oversulfated exopolysaccharide on angiogenesis induced by fibroblast growth factor-2 or vascular endothelial growth factor in vitro. Biochem. Pharmacol 2005, 69, 751–759. [Google Scholar]
- Arena, A; Gugliandolo, C; Stassi, G; Pavone, B; Iannello, D; Bisignano, G; Maugeri, TL. An exopolysaccharide produced by Geobacillus thermodenitrificans strain B3-72: antiviral activity on immunocompetent cells. Immunol. Lett 2009, 123, 132–137. [Google Scholar]
- Sutherland, IW. Vandamme, EJ, De Baets, S, Steinbüchel, A, Eds.; Polysaccharides from Prokaryotes. In Biopolymers: Polysaccharides I; Wiley-VCH: Weinheim, Germany, 2002; pp. 1–19. [Google Scholar]
- MacCormick, CA; Harris, JE; Jay, AJ; Ridout, EJ; Colquhoun, IJ; Morris, VJ. Isolation and characterization of new extracellular polysaccharide from an Acetobacter species. J. Appl. Bacteriol 1996, 81, 419–424. [Google Scholar]
- Quesada, E; Valderrama, MJ; Béjar, V; Ventosa, A; Gutiérrez, MC; Ruiz-Berraquero, F; Ramos-Cormenzana, A. Volcaniella eurihalina gen. nov., sp. nov., a moderately halophilic non motile gram-negative rod. Int. J. Syst. Evol. Microbiol 1990, 40, 261–267. [Google Scholar]
- Bouchotroch, S; Quesada, E; del Moral, A; Llamas, I; Béjar, V. Halomonas maura sp. nov., a novel moderately halophilic, exopolysaccharide-producing bacteria. Int. J. Syst. Evol. Microbiol 2001, 51, 1625–1632. [Google Scholar]
- Martínez-Cánovas, MJ; Quesada, E; Llamas, I; Béjar, V. Halomonas ventosae a new moderately halophilic, denitrifying, exopolysaccharide-producing bacterium. Int. J. Syst. Evol. Microbiol 2004, 54, 733–734. [Google Scholar]
- Martínez-Cánovas, MJ; Béjar, V; Martínez-Checa, F; Quesada, E. Halomonas anticariensis sp. nov., from Fuente de Piedra, a saline-wetland wild-fowl reserve in Malaga, southern Spain. Int. J. Syst. Evol. Microbiol 2004, 54, 1329–1332. [Google Scholar]
- Martínez-Checa, F; Béjar, V; Llamas, I; del Moral, A; Quesada, E. Alteromonas hispanica sp. nov., a polyunsaturated-fatty-acid-producing, halophilic bacterium isolated from Fuente de Piedra, south-east Spain. Int. J. Syst. Evol. Microbiol 2005, 55, 2385–2390. [Google Scholar]
- Martínez-Cánovas, MJ; Béjar, V; Martínez-Checa, F; Páez, R; Quesada, E. Idiomarina fontislapidosi sp. nov. and Idiomarina ramblicola sp. nov., isolated from inland hypersaline habitats in Spain. Int. J. Syst. Evol. Microbiol 2004, 54, 1793–1797. [Google Scholar]
- Quesada, E; Béjar, V; Ferrer, MR; Calvo, C; Llamas, I; Martínez-Checa, F; Arias, S; Ruíz-, García C; Páez, R; Martínez-Cánovas, MJ; del Moral, A. Ventosa, A, Ed.; Moderately halophilic exopolysaccharide-producing bacteria. In Halophilic Microorganisms; Springer: Berlin, Germany, 2004; pp. 295–314. [Google Scholar]
- Mata, JA; Béjar, V; Llamas, I; Arias, S; Bressollier, P; Tallon, R; Urdaci, MC; Quesada, E. Exopolysaccharides produced by the recently described bacteria Halomonas ventosae and Halomonas anticariensis. Res. Microbiol 2006, 157, 827–835. [Google Scholar]
- Mata, JA; Béjar, V; Bressollier, P; Tallon, R; Urdaci, MC; Quesada, E; Llamas, I. Characterization of exopolysaccharides produced by thres moderately halophilic bacteria belonging to the family Alteromonadaceae. J. Appl. Microbiol 2008, 105, 521–528. [Google Scholar]
- Arias, S; del Moral, A; Ferrer, MR; Tallon, R; Quesada, E; Béjar, V. Mauran, an exopolysaccharide produced by the halophilic bacterium Halomonas maura, with a novel composition and interesting properties for biotechnology. Extremophiles 2003, 7, 319–326. [Google Scholar]
- Bouchotroch, S; Quesada, E; Izquierdo, I; Rodríguez, M; Béjar, V. Bacterial exopolysaccharides produced by newly discovered bacteria belonging to the genus Halomonas, isolated from hypersaline habitats in Morocco. J. Ind. Microbiol. Biotechnol 2000, 24, 374–378. [Google Scholar]
- Béjar, V; Calvo, C; Moliz, J; Díaz-Martínez, F; Quesada, E. Effect of growth conditions on the rheological properties and chemical composition of Volcaniella eurihalina exopolisaccharide. Appl. Biochem. Biotechnol 1996, 59, 77–86. [Google Scholar]
- Béjar, B; Llamas, I; Calvo, C; Quesada, E. Characterization of exopolysaccharides produced by 19 halophilic strains of the species Halomonas eurihalina. J. Biotechnol 1998, 61, 135–141. [Google Scholar]
- Calvo, C; Ferrer, MR; Martínez-Checa, F; Béjar, V; Quesada, E. Some rheological properties of the extracellular polysaccharide produced by Volcaniella eurihalina F2-7. Appl. Biochem. Biotechnol 1995, 55, 45–54. [Google Scholar]
- Calvo, C; Martínez-Checa, F; Mota, A; Béjar, V; Quesada, E. Effect of cations, pH and sulphate content on the viscosity and emulsifying activity of the Halomonas eurihalina exopolysaccharide. J. Ind. Microbiol. Biotechnol 1998, 20, 205–209. [Google Scholar]
- Calvo, C; Martinez-Checa, F; Toledo, FL; Porcel, J; Quesada, E. Characteristics of bioemulsifiers synthesised in crude oil media by Halomonas eurihalina and their effectiveness in the isolation of bacteria able to grow in the presence of hydrocarbons. Appl. Microbiol. Biotechnol 2002, 60, 347–351. [Google Scholar]
- Martínez-Checa, F; Calvo, C; Caba, MA; Ferrer, MR; Béjar, V; Quesada, E. Efecto de las condiciones nutricionales sobre la viscosidad y la capacidad emulgente del biopolímero V2-7 de Volcaniella eurihalina. Microbiología SEM 1996, 12, 55–60. [Google Scholar]
- Quesada, E; Béjar, V; Calvo, C. Exopolysaccharide production by Volcaniella eurihalina. Experientia 1993, 49, 1037–1041. [Google Scholar]
- Martínez-Cánovas, MJ; Quesada, E; Martínez-Checa, F; del Moral, A; Béjar, V. Salipiger mucosus gen. nov., sp. nov., a moderately halophilic, exopolysaccharide-producing bacterium isolated from hypersaline soil, belonging to the α-Proteobacteria. Int. J. Syst. Evol. Microbiol 2004, 54, 1735–1740. [Google Scholar]
- Cheirslip, B; Shimizu, H; Shioya, S. Modelling and optimization of environmental conditions for kefiran production by Lactobacillus kefiranofaciens. Appl. Microbiol. Biotechnol 2001, 57, 639–643. [Google Scholar]
- Gorret, AU; Maubois, N; Engasser, JL; Ghoul, JM. Study of the effects of temperature, pH and yeast extract on growth and exopolysaccharide production by Propionibacterium acidipropionici on milk microfiltrate using a response surface methodology. J. Appl. Microbiol 2001, 90, 788–796. [Google Scholar]
- Pham, PL; Dupont, I; Roy, D; Lapointe, G; Cerning, J. Production of exopolysaccharide by Lactobacillus rhamnosus and analysis of its enzymatic degradation during prolonged fermentation. Appl. Environ. Microbiol 2000, 66, 2302–2310. [Google Scholar]
- Yim, JH; Son, E; Pyo, S; Lee, HK. Novel sulphated polysaccharide derived from red-tide microalga Gyrodinium impudicum strain KG03 with immunostimulating activity in vivo. Mar. Biotechnol 2005, 7, 331–338. [Google Scholar]
- Kitazawa, H; Ishii, Y; Uemura, J; Kawai, Y; Saito, T; Kaneko, T; Noda, K; Itoh, T. Augmentation of macrophage functions by an extracellular phosphopolysaccharide from Lactobacillus delbrueckii ssp. bulgaricus. Food Microbiol 2000, 17, 109–118. [Google Scholar]
- Nishimura-Uemura, J; Kitazawa, H; Kawai, Y; Itoh, T; Oda, M; Saito, T. Functional alteration of murine macrophages stimulated with extracellular polysaccharides from Lactobacillus delbrueckii ssp. bulgaricus OLL1073R-1. Food Microbiol 2003, 20, 267–273. [Google Scholar]
- Ebina, T; Ogata, N; Murata, K. Antitumor effect of Lactobacillus bulgaricus 878R. Biotherapy 1995, 9, 65–70. [Google Scholar]
- Vanhooren, PT; Vandamme, EJ. l-fucose: occurrence, physiological role, chemical, enzymatic and microbial synthesis. J. Chem. Technol. Biotechnol 1999, 74, 479–497. [Google Scholar]
- Desai, JD; Banat, IM. Microbial production of surfactants and their commercial potential. Microbiol. Mol. Biol. Rev 1997, 61, 47–64. [Google Scholar]
- Navon-Venezia, S; Banin, E; Ron, EZ; Rosenberg, E. The bioemulsifier alasan: role of protein in maintaining structure and activity. Appl. Microbiol. Biotechnol 1998, 49, 382–384. [Google Scholar]
- Rosenberg, E; Zuckerberg, A; Rubinovitz, C; Gutnick, DL. Emulsifier of Arthrobacter RAG-1: isolation and emulsifying properties. Appl. Environ. Microbiol 1979, 37, 402–408. [Google Scholar]
- Ashtaputre, AA; Shah, AK. Emulsifying property of a viscous exopolysaccharide from Sphingomonas paucimobilis. World J. Microbiol. Biotechnol 1995, 11, 219–222. [Google Scholar]
- Sutherland, IW. Structure-function relationships in microbial exopolysaccharides. Biotechnol. Adv 1994, 12, 393–448. [Google Scholar]
- Geddie, JL; Sutherland, IW. Uptake of metals by bacterial polysaccharides. J. Appl. Bacteriol 1993, 74, 467–472. [Google Scholar]
- Valls, M; de Lorenzo, V. Exploiting the genetic and biochemical capacities of bacteria for the remediation of heavy metal pollution. FEMS Microbiol. Rev 2002, 26, 327–338. [Google Scholar]
- Martínez-Cánovas, MJ; Quesada, E; Martínez-Checa, F; Béjar, V. A taxonomic study to establish the relationship between exopolysaccharide-producing bacterial strains living in diverse hypersaline habitats. Curr. Microbiol 2004, 48, 348–353. [Google Scholar]
- Moraine, RA; Rogovin, P. Kinetics of polysaccharide B-1459 fermentation. Biotechnol. Bioeng 1996, 8, 511–524. [Google Scholar]
- Rodríguez-Valera, F; Ruíz-Berraquero, F; Ramos-Comenzana, A. Characteristics of the heterotrophic bacterial populations in hypersaline environments of different salt concentrations. Microbiol. Ecol 1981, 7, 235–243. [Google Scholar]
- Quesada, E; del Moral, A; Béjar, V. Comparative methods for isolation of Volcaniella eurihalina exopolysaccharide. Biotechnol. Tech 1994, 8, 701–706. [Google Scholar]
- Bergmeyer, HV; Bent, E. Method of Enzymatic Analysis; Academic: New York, NY, USA, 1965; pp. 123–130. [Google Scholar]
- Dubois, M; Gilles, KA; Hamilton, JK; Rebers, PA; Smith, F. Colorimetric method for determination of sugars and related substances. Anal. Chem 1956, 28, 350–356. [Google Scholar]
- Bradford, MM. A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle protein-dye binding. Anal. Biochem 1976, 72, 248–254. [Google Scholar]
- McComb, EA; McCready, RM. Determination of acetyl in pectin and in acetylated carbohydrate polymers. Anal. Chem 1957, 29, 819–821. [Google Scholar]
- Sloneker, JH; Orentas, DG. Quantitative determination of pyruvic acid. Nature 1962, 194, 478. [Google Scholar]
- Johnson, AR. Improved method of hexosamine determination. Anal. Biochem 1971, 44, 628–635. [Google Scholar]
- Chaplin, MF. A rapid and sensitive method for the analysis of carbohydrate components in glycoproteins using gas-liquid chromatography. Anal. Biochem 1982, 123, 336–341. [Google Scholar]
- Cooper, D; Goldenberg, G. Surface active agents from two Bacillus species. Appl. Environ. Microbiol 1987, 54, 224–229. [Google Scholar]
| S. mucosus A3T | Triton X-100 | Tween 80 | Xanthan gum | |
|---|---|---|---|---|
| Sunflower oil | 70 ± 1.41 | 62.5 ± 1.82 | 62 ± 1.67 | 88.95 ± 1.60 |
| Mineral oil | 71 ± 1.60 | 67.5 ± 1.41 | 70 ± 1.15 | 90.3 ± 1.43 |
| Olive oil | 60.3 ± 1.52 | 60 ± 1.52 | 62.5 ± 1.64 | 100 ± 1.94 |
| Tetradecane | 75 ± 2.08 | 62.5 ± 1.82 | 62.5 ± 1.10 | 90.3 ± 0.98 |
| Octane | 70 ± 1.51 | 60 ± 1.52 | 60 ± 0.70 | 93.3 ± 2.18 |
| Kerosene | 70 ± 0.51 | 62.1 ± 2.12 | 60 ± 1.40 | 89 ± 1.02 |
| Isopropyl myristate | 67.5 ± 0.70 | 67.5 ± 0.70 | 67.5 ± 1.02 | 100 ± 1.34 |
| Petrol | 20 ± 3.01 | 70 ± 1.52 | 32.5 ± 2.60 | 92.5 ± 2.45 |
| Diesel | 37.5 ± 0.70 | 62.5 ± 2.12 | 65 ± 1.70 | 87.5 ± 3.01 |
| Crude oil | 95 ± 0.57 | 60 ± 0.70 | 60 ± 0.60 | 89.75 ± 1.32 |
| Xylene | 47.5 ± 0.70 | 12.5 ± 1.52 | 12.5 ± 1.30 | 86.5 ± 1.78 |
| Toluene | 52.6 ± 3.53 | 12.5 ± 0.70 | 60 ± 1.80 | 100 ± 1.75 |
| Vaseline oil | 52.6 ± 2.50 | 65 ± 0.57 | 60 ± 2.55 | 100 ± 1.67 |
| Hexane | 50 ± 2.51 | 50 ± 1.52 | 42 ± 0.66 | 100 ± 2.25 |
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Llamas, I.; Mata, J.A.; Tallon, R.; Bressollier, P.; Urdaci, M.C.; Quesada, E.; Béjar, V. Characterization of the Exopolysaccharide Produced by Salipiger mucosus A3T, a Halophilic Species Belonging to the Alphaproteobacteria, Isolated on the Spanish Mediterranean Seaboard. Mar. Drugs 2010, 8, 2240-2251. https://doi.org/10.3390/md8082240
Llamas I, Mata JA, Tallon R, Bressollier P, Urdaci MC, Quesada E, Béjar V. Characterization of the Exopolysaccharide Produced by Salipiger mucosus A3T, a Halophilic Species Belonging to the Alphaproteobacteria, Isolated on the Spanish Mediterranean Seaboard. Marine Drugs. 2010; 8(8):2240-2251. https://doi.org/10.3390/md8082240
Chicago/Turabian StyleLlamas, Inmaculada, Juan Antonio Mata, Richard Tallon, Philippe Bressollier, María C. Urdaci, Emilia Quesada, and Victoria Béjar. 2010. "Characterization of the Exopolysaccharide Produced by Salipiger mucosus A3T, a Halophilic Species Belonging to the Alphaproteobacteria, Isolated on the Spanish Mediterranean Seaboard" Marine Drugs 8, no. 8: 2240-2251. https://doi.org/10.3390/md8082240
APA StyleLlamas, I., Mata, J. A., Tallon, R., Bressollier, P., Urdaci, M. C., Quesada, E., & Béjar, V. (2010). Characterization of the Exopolysaccharide Produced by Salipiger mucosus A3T, a Halophilic Species Belonging to the Alphaproteobacteria, Isolated on the Spanish Mediterranean Seaboard. Marine Drugs, 8(8), 2240-2251. https://doi.org/10.3390/md8082240





