Verrucisidinol and Verrucosidinol Acetate, Two Pyrone-Type Polyketides Isolated from a Marine Derived Fungus, Penicillium aurantiogriseum
Abstract
:1. Introduction
2. Results and Discussion
3. Experimental Section
3.1. General Experimental Procedures
3.2. Fungal Material and Taxonomic Identification
3.3. Large Scale Fermentation
3.4. Extraction and Isolation
3.5. Antimicrobial Assay
3.6. Antifungal and Synergistic Antifungal Assay
4. Conclusions
Acknowledgements
References
- Blunt, JW; Copp, BR; Munro, MH; Northcote, PT; Prinsep, MR. Marine natural products. Nat Prod Rep 2010, 27, 165–237. [Google Scholar]
- Zhang, L. Integrated Approaches for Discovering Novel Drugs From Microbial Natural Products. In Natural Products; Zhang, L, Demain, AL, Eds.; Humana Press: Totowa, NJ, USA, 2005; pp. 33–55. [Google Scholar]
- Capon, RJ. Marine natural products chemistry: past, present and future. Aust J Chem 2010, 63, 851–854. [Google Scholar]
- Molinski, TF; Dalisay, DS; Lievens, SL; Saludes, JP. Drug development from marine natural products. Nat Rev Drug Discov 2009, 8, 69–85. [Google Scholar]
- Zhang, L; An, R; Wang, J; Sun, N; Zhang, S; Hu, J; Kuai, J. Exploring novel bioactive compounds from marine microbes. Curr Opin Microbiol 2005, 8, 276–281. [Google Scholar]
- Liu, X; Ashforth, E; Ren, B; Song, F; Dai, H; Liu, M; Wang, J; Xie, Q; Zhang, L. Bioprospecting microbial natural product libraries from the marine environment for drug discovery. J Antibiot 2010, 63, 415–422. [Google Scholar]
- Zhuo, Y; Zhang, W; Chen, D; Gao, H; Tao, J; Liu, M; Gou, Z; Zhou, X; Ye, B; Zhang, Q; Zhang, S; Zhang, L. Reverse biological engineering of hrdB to enhance the production of avermectins in an industrial strain of Streptomyces avermitilis. Proc Natl Acad Sci USA 2010, 107, 11250–11254. [Google Scholar]
- Song, F; Dai, H; Tong, Y; Ren, B; Chen, C; Sun, N; Liu, X; Bian, J; Liu, M; Gao, H; Liu, H; Chen, X; Zhang, L. Trichodermaketones A-D and 7-O-methylkoninginin D from the marine fungus Trichoderma koningii. J Nat Prod 2010, 73, 806–810. [Google Scholar]
- You, J; Dai, H; Chen, Z; Liu, G; He, Z; Song, F; Yang, X; Fu, H; Zhang, L; Chen, X. Trichoderone, a novel cytotoxic cyclopentenone and cholesta-7,22-diene-3β,5α,6β-triol with new activities from the marine-derived fungus Trichoderma sp. J Ind Microbiol Biot 2010, 37, 245–252. [Google Scholar]
- Bragulat, MR; Martínez, E; Castellá, G; Cabanes, FJ. Ochratoxin A and citrinin producing species of the genus Penicillium from feedstuffs. Int J Food Microbiol 2008, 126, 43–48. [Google Scholar]
- Wilson, BJ; Byerly, CS; Burka, LT. Neurologic disease of fungal origin in three herds of cattle. J Am Vet Med Assoc 1981, 179, 480–481. [Google Scholar]
- Hodge, RP; Harris, CM; Harris, TM. Verrucofortine, a major metabolite of Penicillium verrucosum var. cyclopium, the fungus that produces the mycotoxin verrucosidin. J Nat Prod 1988, 51, 66–73. [Google Scholar]
- Park, HR; Ryoo, IJ; Choo, SJ; Hwang, JH; Kim, JY; Cha, MR; Shin-Ya, K; Yoo, ID. Glucose-deprived HT-29 human colon carcinoma cells are sensitive to verrucosidin as a GRP78 down-regulator. Toxicology 2007, 229, 253–261. [Google Scholar]
- Yeulet, SE; Mantle, PG; Rudge, MS; Greig, JB. Nephrotoxicity of Penicillium aurantiogriseum, a possible factor in the aetiology of Balkan endemic nephropathy. Mycopathologia 1988, 102, 21–30. [Google Scholar]
- Mantle, PG; McHugh, KM; Adatia, R; Heaton, JM; Gray, T; Turner, DR. Penicillium aurantiogriseum-induced, persistent renal histopathological changes in rats; an experimental model for Balkan endemic nephropathy competitive with ochratoxin A. IARC Sci Publ 1991, 115, 119–127. [Google Scholar]
- Xin, Z; Fang, Y; Du, L; Zhu, T; Duan, L; Chen, J; Gu, Q; Zhu, W. Aurantiomides A-C, quinazoline alkaloids from the sponge-derived fungus Penicillium aurantiogriseum SP0-19. J Nat Prod 2007, 70, 853–855. [Google Scholar]
- Igarashi, Y; Sekine, A; Fukazawa, H; Uehara, Y; Yamaguchi, K; Okuda, T; Furumai, T; Oki, T. Anicequol, a novel inhibitor for anchorage-independent growth of tumor cells from Penicillium aurantiogriseum Dierckx TP-F0213. J Antibiot 2002, 55, 371–376. [Google Scholar]
- Manabu, N. Terrestric acid as a phytotoxic metabolite from Pyricularia oryzae Cavara. Agric Biol Chem 1988. [Google Scholar]
- Ganguli, M; Burka, LT; Harris, TM. Structure studies of the mycotoxin verrucosidin. J Org Chem 1984, 49, 3762–3766. [Google Scholar]
- Williams, DR; White, FH. Total synthesis of (±)-citreoviridin. J Org Chem 1987, 52, 5067–5079. [Google Scholar]
- Whang, K; Cooke, RJ; Okay, G; Cha, JK. Total synthesis of (+)-verrucosidin. J Am Chem Soc 1990, 112, 8985–8987. [Google Scholar]
- Hatakeyama, S; Sakurai, K; Numata, H; Ochi, N; Takano, S. A novel chiral route to substituted tetrahydrofurans. Total synthesis of (±)-verrucosidin and formal synthesis of (−)-citreoviridin. J Am Chem Soc 1988, 110, 5201–5203. [Google Scholar]
- Hanaki, N; Link, JT; MacMillan, DWC; Overman, LE; Trankle, WG; Wurster, JA. Stereoselection in the prins-pinacol synthesis of 2,2-disubstituted 4-acyltetrahydrofurans. Enantioselective synthesis of (−)-citreoviral. Org Lett 1999, 2, 223–226. [Google Scholar]
- Burka, LT; Ganguli, M; Wilson, BJ. Verrucosidin, a tremorgen from Penicillium verrucosum var cyclopium. J Chem Soc Chem Commun 1983, 544–545. [Google Scholar]
- Nunez, F; Rodriguez, MM; Bermudez, ME; Cordoba, JJ; Asensio, MA. Composition and toxigenic potential of the mould population on dry-cured Iberian ham. Int J Food Microbiol 1996. [Google Scholar]
- Lunda, F; Frisvada, JC. Chemotaxonomy of Penicillium aurantiogriseum and related species. Mycol Res 1994, 98, 481–492. [Google Scholar]
- Jacobsen, JP; Reffstrup, T; Cox, RE; Holker, JSE; Boll, PM. Revision of the naturally occurring acyl tetronic acid: dihydrocarolic acid, terrestric acid and carlic acid. Tetrahedron Lett 1978, 19, 1081–1084. [Google Scholar]
- Birkinshaw, JH; Raistrick, H. Studies in the biochemistry of micro-organisms. LII. Isolation, properties and constitution of terrestric acid (ethylcarolic acid), a metabolic product of Penicillium terrestre Jensen. Biochem J 1936. [Google Scholar]
- Thompson, JD; Higgins, DG; Gibson, TJ. CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 1994, 22, 4673–4680. [Google Scholar]
- Saitou, N; Nei, M. The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 1987, 4, 406–425. [Google Scholar]
- Tamura, K; Dudley, J; Nei, M; Kumar, S. MEGA4: Molecular Evolutionary Genetics Analysis (MEGA) software version 4.0. Mol Biol Evol 2007, 24, 1596–1599. [Google Scholar]
- Felsenstein, J. Confidence limits on phylogenies: an approach using the bootstrap. Evolution 1985, 39, 783–791. [Google Scholar]
- Zhang, L; Yan, K; Zhang, Y; Huang, R; Bian, J; Zheng, C; Sun, H; Chen, Z; Sun, N; An, R; Song, Y; Min, F; Zhao, W; Zhuo, Y; You, J; Song, Y; Yu, Z; Liu, Z; Yang, K; Gao, H; Dai, H; Zhang, X; Wang, J; Fu, C; Pei, G; Liu, J; Zhang, S; Goodfellow, M; Jiang, Y; Kuai, J; Zhou, G; Chen, X. High-throughput synergy screening identifies microbial metabolites as combination agents for the treatment of fungal infections. Proc Natl Acad Sci USA 2007, 104, 4606–4611. [Google Scholar]
- National Committee for Clinical Laboratory Standards, 2002; Reference Method for Broth Dilution Antifungal Susceptibility Testing of Yeasts: Approved Standard, Document M27-A2; National Committee for Clinical Laboratory Standards: Wayne, PA, USA, 2002.

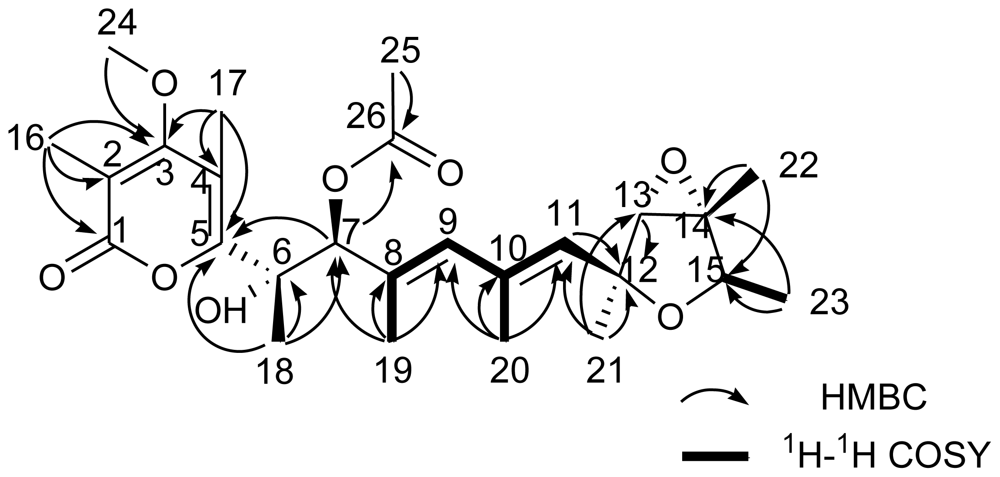
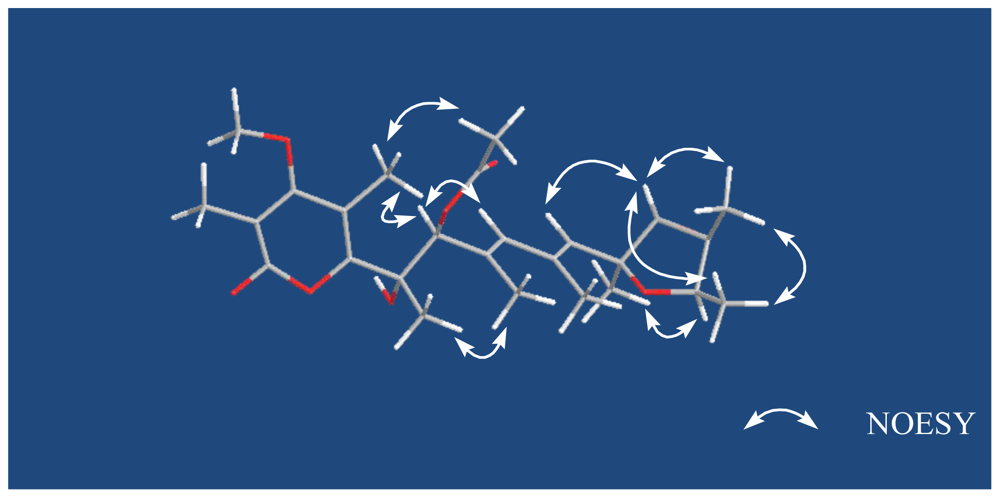

| verrucosidinol (1) | verrucosidinol acetate (2) | |||||||
|---|---|---|---|---|---|---|---|---|
| Position | δC | δHa (J in Hz) | HMBCa | NOESY | δC | δHa (J in Hz) | HMBCa | NOESY |
| 1 | 165.0, qC | 164.3, qC | ||||||
| 2 | 110.4, qC | 110.9, qC | ||||||
| 3 | 169.2, qC | 168.5, qC | ||||||
| 4 | 111.8, qC | 111.5, qC | ||||||
| 5 | 159.7, qC | 157.5, qC | ||||||
| 6 | 78.8, qC | 78.2, qC | ||||||
| 7 | 79.8, CH | 4.61, s | 5, 6, 8, 9, 18, 19 | 9, 17 | 82.1, CH | 5.43, s | 6, 8, 9, 19, 26 | 9, 17 |
| 8 | 133.9, qC | 130.4, qC | ||||||
| 9 | 134.3, CH | 5.87, s | 7, 11, 19, 20 | 7 | 135.4, CH | 5.85, s | 7, 11, 19, 20 | 7 |
| 10 | 134.4, qC | 134.3, qC | ||||||
| 11 | 133.0, CH | 5.43, s | 9, 12, 13, 20, 21, | 13, 23 | 133.2, CH | 5.41, s | 9, 12, 13, 20 | 13, 23 |
| 12 | 80.1, qC | 80.0, qC | ||||||
| 13 | 67.5, CH | 3.43, s | 12 | 11, 22 | 67.4, CH | 3.41, s | 12 | 11, 22 |
| 14 | 67.4, qC | 67.4, qC | ||||||
| 15 | 76.7, CH | 4.12, q (7.0) | 12, 13 | 21 | 76.7, CH | 4.11, q (7.0) | 12, 13 | 21 |
| 16 | 10.2, CH3 | 2.01, s | 1, 2, 3 | 10.2, CH3 | 2.02, s | 1, 2, 3 | ||
| 17 | 9.9, CH3 | 2.21, s | 3, 4, 5 | 7 | 10.1, CH3 | 2.20, s | 3, 4, 5 | 7, 25 |
| 18 | 23.4, CH3 | 1.40, s | 5, 6, 7 | 19 | 23.8, CH3 | 1.55, s | 5, 6, 7 | 19 |
| 19 | 14.8, CH3 | 1.82, s | 7, 8, 9, | 18 | 15.4, CH3 | 1.80, s | 7, 8, 9 | 18 |
| 20 | 18.6, CH3 | 1.89, s | 9,10,11 | 18.4, CH3 | 1.87, s | 9, 10, 11 | ||
| 21 | 21.9, CH3 | 1.41, s | 11, 12, 13 | 15 | 21.8, CH3 | 1.39, s | 11, 12, 13 | 15 |
| 22 | 13.8, CH3 | 1.47, s | 13, 14, 15 | 13, 23 | 13.8, CH3 | 1.46, s | 13, 14, 15 | 13, 23 |
| 23 | 18.8, CH3 | 1.18, d (7.0) | 14, 15 | 11, 22 | 18.8, CH3 | 1.16, d (7.0) | 14, 15 | 11, 22 |
| 24 | 60.3, CH3 | 3.79, s | 3 | 60.3, CH3 | 3.78, s | 3 | ||
| 25 | 21.0, CH3 | 2.06, s | 26 | 17 | ||||
| 26 | 169.4, qC | |||||||
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yu, K.; Ren, B.; Wei, J.; Chen, C.; Sun, J.; Song, F.; Dai, H.; Zhang, L. Verrucisidinol and Verrucosidinol Acetate, Two Pyrone-Type Polyketides Isolated from a Marine Derived Fungus, Penicillium aurantiogriseum. Mar. Drugs 2010, 8, 2744-2754. https://doi.org/10.3390/md8112744
Yu K, Ren B, Wei J, Chen C, Sun J, Song F, Dai H, Zhang L. Verrucisidinol and Verrucosidinol Acetate, Two Pyrone-Type Polyketides Isolated from a Marine Derived Fungus, Penicillium aurantiogriseum. Marine Drugs. 2010; 8(11):2744-2754. https://doi.org/10.3390/md8112744
Chicago/Turabian StyleYu, Ke, Biao Ren, Junli Wei, Caixia Chen, Jinsheng Sun, Fuhang Song, Huanqin Dai, and Lixin Zhang. 2010. "Verrucisidinol and Verrucosidinol Acetate, Two Pyrone-Type Polyketides Isolated from a Marine Derived Fungus, Penicillium aurantiogriseum" Marine Drugs 8, no. 11: 2744-2754. https://doi.org/10.3390/md8112744
APA StyleYu, K., Ren, B., Wei, J., Chen, C., Sun, J., Song, F., Dai, H., & Zhang, L. (2010). Verrucisidinol and Verrucosidinol Acetate, Two Pyrone-Type Polyketides Isolated from a Marine Derived Fungus, Penicillium aurantiogriseum. Marine Drugs, 8(11), 2744-2754. https://doi.org/10.3390/md8112744





