Chitin Deacetylases: Properties and Applications
Abstract
:1. Introduction
2. Occurrence of Chitin Deacetylases
2.1. Fungal Deacetylases
2.2. Insect Deacetylases
2.3. Deacetylases from Marine Bacteria
3. Properties of Chitin Deacetylases
3.1. Molecular Mass
3.2. Optimal pH and Temperature
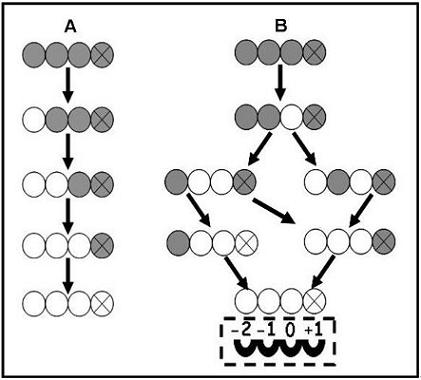
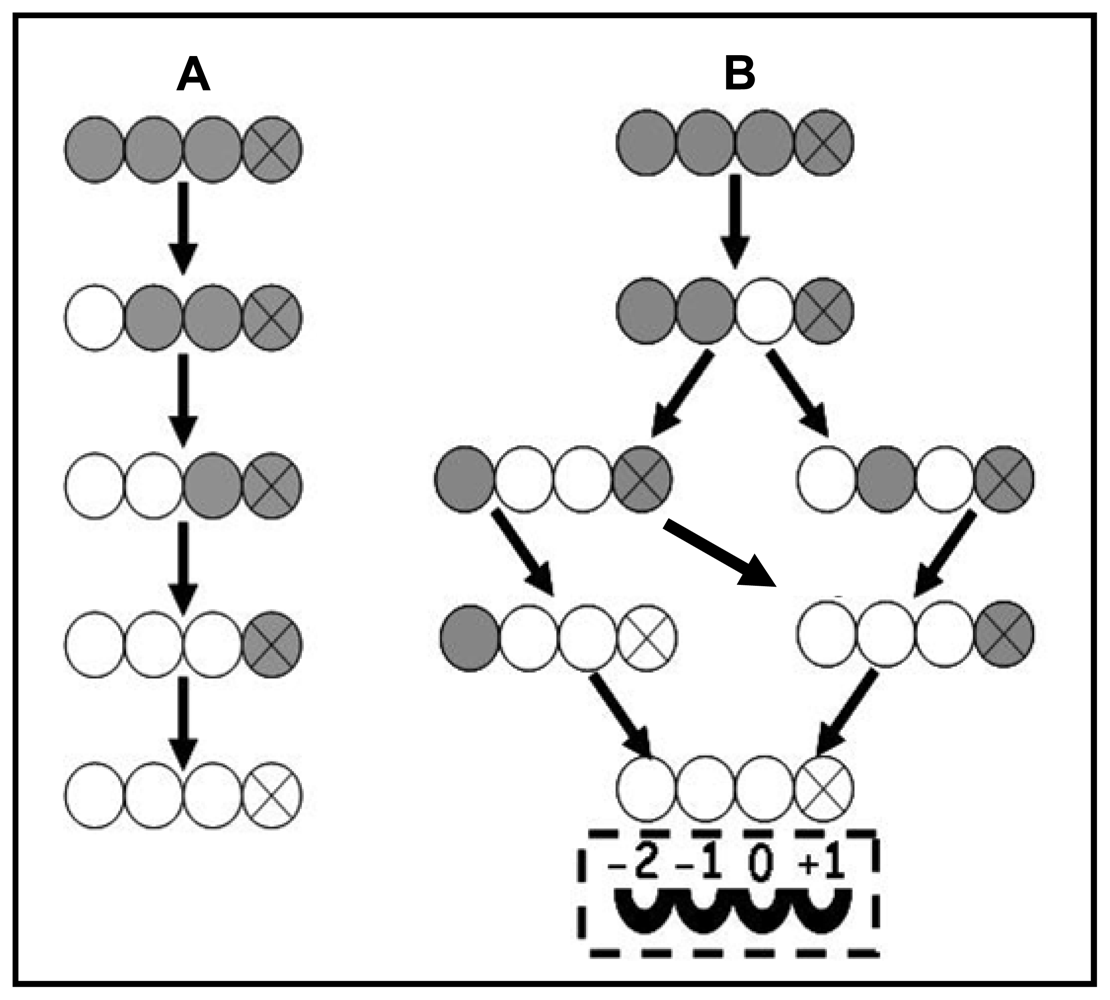
3.3. Enzymatic Deacetylation of Chitin
3.4. Substrate Specificity
3.5. Influence of Metal Ions on Chitin Deacetylase
3.6. Catalytic Mechanism
3.7. Gene and Structure
4. Biological Roles of Chitin Deacetylases
4.1. Biological Roles of Deacetylases from Marine Bacteria
4.2. Biological Roles of Fungal Deacetylases
4.3. Biological Roles of Insect Deacetylases
5. Applications of Chitin Deacetylases
5.1. Applications in the Marine Field
5.2. Preparations of Chitosan from Chitin
5.3. Applications in the Biochemical Area
Acknowledgements
References
- Coutinho, PM; Henrissat, B. Gilbert, HJ, Davies, G, Henrissat, B, Svensson, B, Eds.; Carbohydrate-active enzymes: An integrated database approach. In Recent Advances in Carbohydrate Bioengineering; The Royal Society of Chemistry: Cambridge, UK, 1999; pp. 3–12. [Google Scholar]
- John, M; Rohrig, H; Schmidt, J; Wieneke, U; Schell, J. Rhizobium NodB protein involved in nodulation signal synthesis is a chitooligosaccharide deacetylase. Proc Natl Acad Sci USA 1993, 90, 625–629. [Google Scholar]
- Gilmore, ME; Bandyopadhyay, D; Dean, AM; Linnstaedt, SD; Popham, DL. Production of muramic delta-lactam in Bacillus subtilis spore peptidoglycan. J Bacteriol 2004, 186, 80–89. [Google Scholar]
- Vollmer, W; Tomasz, A. The pgdA gene encodes for a peptidoglycan N-acetylglucosamine deacetylase in Streptococcus pneumoniae. J Biol Chem 2000, 275, 20496–20501. [Google Scholar]
- Araki, Y; Ito, E. A pathway of chitosan formation in Mucor rouxii. Eur J Biochem 1975, 55, 71–78. [Google Scholar]
- Davis, LL; Bartnicki-Garcia, S. Chitosan synthesis by the tandem action of chitin synthetase and chitin deacetylase from Mucor rouxii. Biochemistry 1984, 23, 1065–1073. [Google Scholar]
- Hunt, DE; Gevers, D; Vahora, NM; Polz, MF. Conservation of the chitin utilization pathway in the Vibrionaceae. Appl Environ Microbiol 2008, 74, 44–51. [Google Scholar]
- Gao, XD; Katsumoto, T; Onodera, K. Purification and characterization of chitin deacetylase from Absidia coerulea. J Biochem 1995, 117, 257–263. [Google Scholar]
- Gauthier, C; Clerisse, F; Dommes, J; Jaspar-Versali, MF. Characterization and cloning of chitin deacetylases from Rhizopus circinans. Protein Expr Purif 2008, 59, 127–137. [Google Scholar]
- Jeraj, N; Kunič, B; Lenasi, H; Breskvar, K. Purification and molecular characterization of chitin deacetylase from Rhizopus nigricans. Enzyme Microb Technol 2006, 39, 1294–1299. [Google Scholar]
- Kim, YJ; Zhao, Y; Oh, KT; Nguyen, VN; Park, RD. Enzymatic deacetylation of chitin by extracellular chitin deacetylase from a newly screened Mortierella sp. DY-52. J Microbiol Biotechnol 2008, 18, 759–766. [Google Scholar]
- Tokuyasu, K; Ohnishi-Kameyama, M; Hayashi, K. Purification and characterization of extracellular chitin deacetylase from Colletotrichum lindemuthianum. Biosci Biotech Biochem 1996, 60, 1598–1603. [Google Scholar]
- Tsigos, I; Bouriotis, V. Purification and characterization of chitin deacetylase from Colletotrichum lindemuthianum. J Biol Chem 1995, 270, 26286–26291. [Google Scholar]
- Shrestha, B; Blondeau, K; Stevens, WF; Hegarat, FL. Expression of chitin deacetylase from Colletotrichum lindemuthianum in Pichia pastoris: Purification and characterization. Protein Expr Purif 2004, 38, 196–204. [Google Scholar]
- Alfonso, C; Nuero, OM; Santamaría, F; Reyes, F. Purification of a heat-stable chitin deacetylase from Aspergillus nidulans and its role in cell wall degradation. Curr Microbiol 1995, 30, 49–54. [Google Scholar]
- Nahar, P; Ghormade, V; Deshpande, MV. The extracellular constitutive production of chitin deacetylase in Metarhizium anisopliae: Possible edge to entomopathogenic fungi in the biological control of insect pests. J Invertebr Pathol 2004, 85, 80–88. [Google Scholar]
- Cai, J; Yang, J; Du, Y; Fan, L; Qiu, Y; Li, J; Kennedy, JF. Purification and characterization of chitin deacetylase from Scopulariopsis brevicaulis. Carbohydr Polym 2006, 65, 211–217. [Google Scholar]
- Martinou, A; Koutsioulis, D; Bouriotis, V. Expression, purification, and characterization of a cobalt-activated chitin deacetylase (Cda2p) from Saccharomyces cerevisiae. Protein Expr Purif 2002, 24, 111–116. [Google Scholar]
- Matsuo, Y; Tanaka, K; Matsuda, H; Kawamukai, M. cda1+, encoding chitin deacetylase is required for proper spore formation in Schizosaccharomyces pombe. FEBS Lett 2005, 579, 2737–2743. [Google Scholar]
- Yamada, M; Kurano, M; Inatomi, S; Taguchi, G; Okazaki, M; Shimosaka, M. Isolation and characterization of a gene coding for chitin deacetylase specifically expressed during fruiting body development in the basidiomycete Flammulina velutipes and its expression in the yeast Pichia pastoris. FEMS Microbiol Lett 2008, 298, 130–137. [Google Scholar]
- Baker, LG; Specht, CA; Donlin, MJ; Lodge, JK. Chitosan, the deacetylated form of chitin, is necessary for cell wall integrity in Cryptococcus neoformans. Eukaryotic Cell 2007, 6, 855–867. [Google Scholar]
- Blair, DE; Hekmat, O; Schuttelkopf, AW; Shrestha, B; Tokuyasu, K; Withers, SG; van Aalten, DMF. Structure and mechanism of chitin deacetylase from the fungal pathogen Colletotrichum lindemuthianum. Biochemistry 2006, 45, 9416–9426. [Google Scholar]
- Tokuyasu, K; Mitsutomi, M; Yamaguchi, I; Hayashi, K; Mori, Y. Recognition of chitooligosaccharides and their N-acetyl groups by putative subsites of chitin deacetylase from a Deuteromycete, Colletotrichum lindemuthianum. Biochemistry 2000, 39, 8837–8843. [Google Scholar]
- Hekmat, O; Tokuyasu, K; Withers, SG. Subsite structure of the endo-type chitin deacetylase from a Deuteromycete, Colletotrichum lindemuthianum: An investigation using steady-state kinetic analysis and MS. Biochem J 2003, 374, 369–380. [Google Scholar]
- Kauss, H; Jeblick, W; Young, DH. Chitin deacetylase from the plant pathogen Colletotrichum lindemuthianum. Plant Sci 1983, 28, 231–236. [Google Scholar]
- Guo, W; Li, G; Pang, Y; Wang, P. A novel chitin-binding protein identified from the peritrophic membrane of the cabbage looper, Trichoplusia ni. Insect Biochem Mol Biol 2005, 35, 1224–1234. [Google Scholar]
- Dixit, R; Arakane, Y; Specht, CA; Richard, C; Kramer, KJ; Beeman, RW; Muthukrishnan, S. Domain organization and phylogenetic analysis of proteins from the chitin deacetylase gene family of Tribolium castaneum and three other species of insects. Insect Biochem Mol Biol 2008, 38, 440–451. [Google Scholar]
- Ruiz-Herrera, J; Martinez-Espinoza, AD. Jollès, P, Muzzarelli, RAA, Eds.; Chitin biosynthesis and structural organization in vivo. In Chitin and chitinases; Birkhauser Verlag: Basel, Switzerland, 1999; pp. 39–53. [Google Scholar]
- Brosson, D; Kuhn, L; Prensier, G; Vivarès, CP; Texier, C. The putative chitin deacetylase of Encephalitozoon cuniculi: A surface protein implicated in microsporidian spore-wall formation. FEMS Microbiol Lett 2005, 247, 81–90. [Google Scholar]
- Deising, H; Siegrist, J. Chitin deacetylase activity of the rust Uromyces viciae-fabae is controlled by fungal morphogenesis. FEMS Microbiol Lett 1995, 127, 207–212. [Google Scholar]
- Christodoulidou, A; Briza, P; Ellinger, A; Bouriotis, V. Yeast ascospore wall assembly requires two chitin deacetylase isozymes. FEBS Lett 1999, 460, 275–279. [Google Scholar]
- Meens, J; Schreiber, C; Deshpande, MV. Muzzarelli, RAA, Ed.; Screening of marine fungi for new chitin deacetylase enzymes. In Chitin Enzymology; Atec: Grottammare, Italy, 2001; pp. 533–540. [Google Scholar]
- Luschnig, S; Bätz, T; Armbruster, K; Krasnow, MA. serpentine and vermiform encode matrix proteins with chitin binding and deacetylation domains that limit tracheal tube length in Drosophila. Curr Biol 2006, 16, 186–194. [Google Scholar]
- Wang, S; Jayaram, SA; Hemphälä, J; Senti, KA; Tsarouhas, V; Jin, H; Samakovlis, C. Septate-junction-dependent luminal deposition of chitin deacetylases restricts tube elongation in the Drosophila Trachea. Curr Biol 2006, 16, 180–185. [Google Scholar]
- Campbell, PM; Cao, AT; Hines, ER; East, PD; Gordon, KHJ. Proteomic analysis of the peritrophic matrix from the gut of the caterpillar, Helicoverpa armigera. Insect Biochem Mol Biol 2008, 38, 950–958. [Google Scholar]
- Toprak, U; Baldwin, D; Erlandson, M; Gillott, C; Hou, X; Coutu, C; Hegedus, DD. A chitin deacetylase and putative insect intestinal lipases are components of the Mamestra configurata (Lepidoptera: Noctuidae) peritrophic matrix. Insect Mol Biol 2008, 17, 573–585. [Google Scholar]
- Ferguson, MJL; Gooday, GW. Muzzarelli, RAA, Ed.; Environmental recycling of chitin. In Chitin Enzymology; Atec: Grottammare, Italy, 1996; pp. 393–396. [Google Scholar]
- Kafetzopoulos, D; Martinou, A; Bouriotis, V. Bioconversion of chitin to chitosan: Purification and characterization of chitin deacetylase from Mucor rouxii. Proc Natl Acad Sci USA 1993, 90, 2564–2568. [Google Scholar]
- Trudel, J; Asselin, A. Detection of chitin deacetylase activity after polyacrylamide gel electrophoresis. Anal Biochem 1990, 189, 249–253. [Google Scholar]
- Win, NN; Stevens, WF. Shrimp chitin as substrate for fungal chitin deacetylase. Appl Microbiol Biotechnol 2001, 57, 334–341. [Google Scholar]
- Caufrier, F; Martinou, A; Dupont, C; Bouriotis, V. Carbohydrate esterase family 4 enzymes: Substrate specificity. Carbohydr Res 2003, 338, 687–692. [Google Scholar]
- Blair, DE; Schuttelkopf, AW; Macrae, JI; van Aalten, DM. Structure and metal-dependent mechanism of peptidoglycan deacetylase, a streptococcal virulence factor. Proc Natl Acad Sci USA 2005, 102, 15429–10975. [Google Scholar]
- Taylor, EJ; Gloster, TM; Turkenburg, JP; Vincent, F; Brzozowski, AM; Dupont, C; Shareck, F; Centeno, MS; Prates, JA; Puchart, M; et al. Structure and activity of two metal-ion dependent acetyl xylan esterases involved in plant cell wall degradation reveals a close similarity to peptidoglycan deacetylase. J Biol Chem 2006, 281, 10968–10975. [Google Scholar]
- Martinou, A; Bouriotis, V; Stokke, BT; Vårum, KM. Mode of action of chitin deacetylase from Mucor rouxii on partially N-acetylated chitosans. Carbohydr Res 1998, 311, 71–78. [Google Scholar]
- Tsigos, I; Zydowicz, N; Martinou, A; Domard, A; Bouriotis, V. Mode of action of chitin deacetylase from Mucor rouxii on N-acetylchitooligosaccharides. Eur J Biochem 1999, 261, 698–705. [Google Scholar]
- Tokuyasu, K; Ono, H; Ohnishi-Kameyama, M; Hayashi, K; Mori, Y. Deacetylation of chitin oligosaccharides of dp 2–4 by chitin deacetylase from Colletotrichum lindemuthianum. Carbohydr Res 1997, 303, 353–358. [Google Scholar]
- Tokuyasu, K; Ohnishi-kameyama, M; Hayashi, K; Mori, Y. Cloning and expression of chitin deacetylase gene from a Deuteromycete, Colletotrichum lindemuthianum. J Biosci Bioeng 1999, 87, 418–423. [Google Scholar]
- Kafetzopoulos, D; Thireos, G; Vournakis, J; Bouriotis, V. The primary structure of fungal chitin deacetylase reveals the function for two bacterial gene products. Proc Natl Acad Sci USA 1993, 90, 8005–8008. [Google Scholar]
- Mishra, C; Semino, CE; Mccreath, KJ; Vega, HDL; Jones, BJ; Specht, CA; Robbins, PW. Cloning and expression of two chitin deacetylase genes of Saccharomyces cerevisiae. Yeast 1997, 13, 327–336. [Google Scholar]
- Tsigos, I; Martinou, A; Kafetzopoulos, D; Bouriotis, V. Chitin deacetylases: New, versatile tools in biotechnology. Trends Biotechnol 2000, 18, 305–312. [Google Scholar]
- Arakane, Y; Dixit, R; Begum, K; Park, Y; Specht, CA; Merzendorfer, H; Kramer, KJ; Muthukrishnan, S; Beeman, RW. Analysis of functions of the chitin deacetylase gene family in Tribolium castaneum. Insect Biochem Mol Biol 2009, 39, 355–365. [Google Scholar]
- Blair, DE; Van Aalten, DM. Structures of Bacillus subtilis PdaA, a family 4 carbohydrate esterase, and a complex with N-acetyl-glucosamine. FEBS Lett 2004, 570, 13–19. [Google Scholar]
- Pruzzo, C; Vezzulli, L; Colwell, RR. Global impact of Vibrio cholerae interactions with chitin. Environ Microbiol 2008, 10, 1400–1410. [Google Scholar]
- Meibom, KL; Li, XBB; Nielsen, AT; Wu, CY; Roseman, S; Schoolnik, GK. The Vibrio cholerae chitin utilization program. Proc Nat Acad Sci USA 2004, 101, 2524–2529. [Google Scholar]
- Li, XB; Wang, LX; Wang, XS; Roseman, S. The chitin catabolic cascade in the marine bacterium Vibrio cholerae: Characterization of a unique chitin oligosaccharide deacetylase. Glycobiology 2007, 17, 1377–1387. [Google Scholar]
- Jung, BO; Roseman, S; Park, JK. The central concept for chitin catabolic cascade in marine bacterium, Vibrios. Macromol Res 2008, 16, 1–5. [Google Scholar]
- Kadokura, K; Rokutani, A; Yamamoto, M; Ikegami, T; Sugita, H; Itoi, S; Hakamata, W; Oku, T; Nishio, T. Purification and characterization of Vibrio parahaemolyticus extracellular chitinase and chitin oligosaccharide deacetylase involved in the producation of heterodisaccharide from chitin. Appl Microbiol Biotechnol 2007, 75, 357–365. [Google Scholar]
- Hirano, T; Kadokura, K; Ikegami, T; Shigeta, Y; Kumaki, Y; Hakamata, W; Oku, T; Nishio, T. Heterodisaccharide 4-O-(N-acetyl-beta-d-glucosaminyl)-d-glucosamine is a specific inducer of chitinolytic enzyme production in Vibrios harboring chitin oligosaccharide deacetylase genes. Glycobiology 2009, 19, 1046–1053. [Google Scholar]
- Lee, BC; Kim, MS; Choi, SH; Kim, KY; Kim, TS. In vitro and in vivo antimicrobial activity of water-soluble chitosan oligo saccharides against Vibrio vulnificus. Int J Mol Med 2009, 24, 327–333. [Google Scholar]
- Chaiyakosa, S; Charernjiratragul, W; Umsakul, K; Vuddhakul, V. Comparing the efficiency of chitosan with chlorine for reducing Vibrio parahaemolyticus in shrimp. Food Control 2007, 18, 1031–1035. [Google Scholar]
- Coluccio, A; Neiman, AM. Interspore bridges: A new feature of the Saccharomyces cerevisiae spore wall. Microbiol 2004, 150, 3189–3196. [Google Scholar]
- Gueddari, NEE; Rauchhaus, U; Moerschbacher, BM; Deising, HB. Developmentally regulated conversion of surface-exposed chitin to chitosan in cell walls of plant pathogenic fungi. New Phytol 2002, 156, 103–112. [Google Scholar]
- van den Burg, HA; Harrison, SJ; Joosten, MHAJ; Vervoort, J; de Wit, PJGM. Cladosporium fulvum Avr4 protects fungal cell walls against hydrolysis by plant chitinases accumulating during infection. Am Phytopathol Soc 2006, 19, 1420–1430. [Google Scholar]
- Aye, KN; Karuppuswamy, R; Ahamed, T; Stevens, WF. Peripheral enzymatic deacetylation of chitin and reprecipitated chitin particles. Bioresour Technol 2006, 97, 577–582. [Google Scholar]
- Jaworska, MM; Bryjak, J; Liesiene, J. A search of an optimal carrier for immobilization of chitin deacetylase. Cellulose 2009, 16, 261–270. [Google Scholar]
- Cao, R; Xue, CH; Liu, Q. Changes in microbial flora of Pacific oysters (Crassostrea gigas) during refrigerated storage and its shelf-life extension by chitosan. Int J Food Microbiol 2009, 131, 272–276. [Google Scholar]
- Powell, A; Rowley, AF. The effect of dietary chitin supplementation on the survival and immune reactivity of the shore crab, Carcinus maenas. Compar Biochem Physiol 2007, A-147, 122–128. [Google Scholar]
- Das, S; Dellen, KV; Bulik, D; Magnelli, P; Cui, J; Head, J; Robbins, PW; Samuelson, J. The cyst wall of Entamoeba invadens contains chitosan (deacetylated chitin). Mol Biochem Parasitol 2006, 148, 86–92. [Google Scholar]
- Howard, MB; Ekborg, NA; Weiner, RM; Hutcheson, SW. Detection and characterization of chitinases and other chitin-modifying enzymes. J Ind Microbiol Biotechnol 2003, 30, 627–635. [Google Scholar]
- Muzzarelli, RAA; RTarsi, R; Filippini, O; Giovanetti, E; Biagini, G; Varaldo, PE. Antimicrobial properties of N-carboxybutyl chitosan. Antimicrob Agents Chemother 1990, 34, 2019–2023. [Google Scholar]
- Vezzulli, L; Pezzati, E; Repetto, B; Stauder, M; Giusto, G; Pruzzo, C. General role for surface membrane proteins in attachment to chitin particles and copepods of environmental and clinical vibrios. Lett Appl Microbiol 2008, 46, 119–125. [Google Scholar]
- Sugita, H; Ito, Y. Identification of intestinal bacteria from Japanese flounder (Paralichthys olivaceus) and their ability to digest chitin. Lett Appl Microbiol 2006, 43, 336–342. [Google Scholar]
- Itoi, S; Okamura, T; Koyama, Y; Sugita, H. Chitinolytic bacteria in the intestinal tract of Japanese coastal fishes. Can J Microbiol 2006, 52, 1158–1163. [Google Scholar]
- Weinhold, MX; Sauvageau, JCM; Keddig, N; Matzke, M; Tartsch, B; Grunwald, I; Kubel, C; Jastorff, B; Thoming, J. Strategy to improve the characterization of chitosan for sustainable biomedical applications: SAR guided multi-dimensional analysis. Green Chem 2009, 11, 498–509. [Google Scholar]
- Yamada, M; Kurano, M; Inatomi, S; Taguchi, G; Okazaki, M; Shimosaka, M. Isolation and characterization of a gene coding for chitin deacetylase specifically expressed during fruiting body development in the basidiomycete Flammulina velutipes and its expression in the yeast Pichia pastoris. FEMS Microbiol Lett 2008, 289, 130–137. [Google Scholar]
- Martinou, A; Tsigos, I; Bouriotis, V. Muzzarelli, RAA, Peter, MG, Eds.; Preparation of chitosan by enzymatic deacetylation. In Chitin Handbook; Atec: Grottammare, Italy, 1997; pp. 501–506. [Google Scholar]
- Martinou, A; Kafetsopoulos, D; Bouriotis, V. Chitin deacetylation by enzymatic means: Monitoring of deacetylation process. Carbohydr Res 1995, 273, 235–242. [Google Scholar]
- Beaney, PD; Gan, Q; Magee, TRA; Flealy, M; Lizardi-Mendoza, J. Modification of chitin properties for enzymatic deacetylation. J Chem Technol Biotechnol 2007, 82, 165–173. [Google Scholar]
- Li, ZL; Guo, JS. Optimizing producing conditions of chitin deacetylase. Proceedings of the Fiber Society 2009 Spring Conference; Donghua Univ. Press: Shanghai, China, 2009; pp. 437–439. [Google Scholar]
- Kadokura, K; Sakamoto, Y; Saito, K; Ikegami, T; Hirano, T; Hakamata, W; Oku, T; Nishio, T. Production of a recombinant chitin oligosaccharide deacetylase from Vibrio parahaemolyticus in the culture medium of Escherichia coli cells. Biotechnol Lett 2007, 29, 1209–1215. [Google Scholar]
- Martinou, A; Tsigos, I; Bouriotis, V. Muzzarelli, RAA, Peter, MG, Eds.; Enzymatic deacetylation of chitooligosaccharides. In Chitin Handbook; Atec: Grottammare, Italy, 1997; pp. 191–194. [Google Scholar]
| Fungi | Phylum | Optimal pH/Temp. (°C) | pI | Molecular Weight (kDa) | Carbohydrate contents (%) | Refs |
|---|---|---|---|---|---|---|
| Mucor rouxii | Mucoromycotina | 4.5, 50 | 3.0 | 75–80 | 30 | [7] |
| Absidia coerulea | Mucoromycotina | 5.0, 50 | NA | 75 | NA | [8] |
| Rhizpus circinans | Mucoromycotina | 5.5–6.0, 37 | NA | 75 | NA | [9] |
| Rhizopus nigricans | Mucoromycotina | NA | NA | 100 | 53 | [10] |
| Mortierella sp. DY-52# | Mucoromycotina | 5.5, 60 | NA | 50, 59 | NA | [11] |
| Colletotrichum lindemuthianum (ATCC 56676)# | Ascomycotina | 12, 60 | 3.7 | 32–33 | NA | [12] |
| Colletotrichum lindemuthianum (DSM 63144)# | Ascomycotina | 8.5, 50 | 3.0 | 150 | 67 | [13] |
| Colletotrichum lindemuthianum (UPS 9) #,* | Ascomycotina | 8.0, 60 | NA | 25 | 0 | [14] |
| Aspergillus nidulans # | Ascomycotina | 7.0, 50 | 2.8 | 27 | 28 | [15] |
| Metarhizium anisopliae # | Ascomycotina | 8.5, NA | 3.6 | 70, 37, 26 | NA | [16] |
| Scopulariopsis brevicaulis | Ascomycotina | 7.5, 55 | NA | 55 | NA | [17] |
| Saccharomyces cerevisiae Cda2p | Ascomycotina | 8.0, 50 | NA | 43 | 18 | [18] |
| Schizosaccharomyces pombe | Ascomycotina | NA | NA | NA | NA | [19] |
| Flammulina velutipes | Basidiomycotina | 7.0, 60 | NA | 31 | 0 | [20] |
| Cryptococcus neoformans | Basidiomycotina | NA | NA | NA | NA | [21] |
| Applications | Refs. |
|---|---|
| Preparations of chitosan from chitin | [11,40,64,65] |
| Protection of fishes and crustaceans via inhibition of Vibrio deacetylases | [66,67] |
| Biological control of some pest insects | [16] |
| Applications as target for biological control of fungal human/plant pathogens | [12,21,29,68] |
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhao, Y.; Park, R.-D.; Muzzarelli, R.A.A. Chitin Deacetylases: Properties and Applications. Mar. Drugs 2010, 8, 24-46. https://doi.org/10.3390/md8010024
Zhao Y, Park R-D, Muzzarelli RAA. Chitin Deacetylases: Properties and Applications. Marine Drugs. 2010; 8(1):24-46. https://doi.org/10.3390/md8010024
Chicago/Turabian StyleZhao, Yong, Ro-Dong Park, and Riccardo A.A. Muzzarelli. 2010. "Chitin Deacetylases: Properties and Applications" Marine Drugs 8, no. 1: 24-46. https://doi.org/10.3390/md8010024