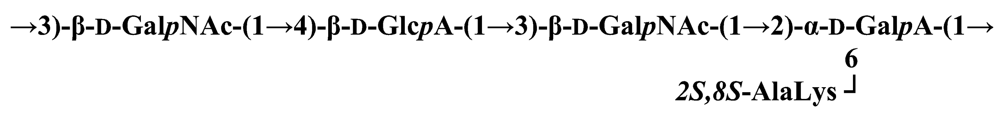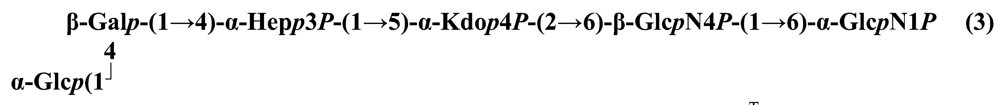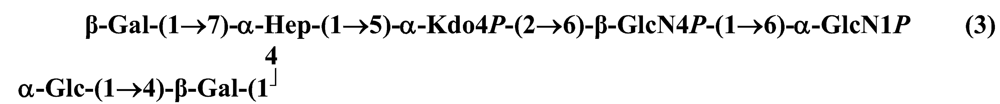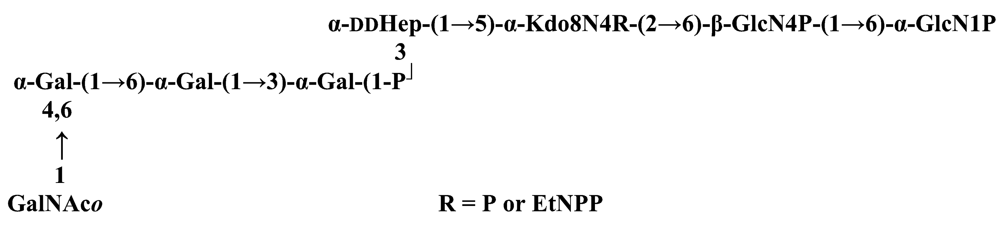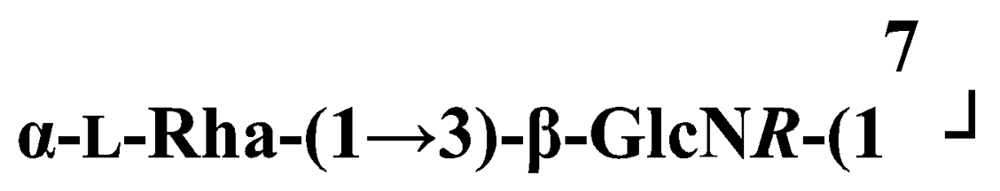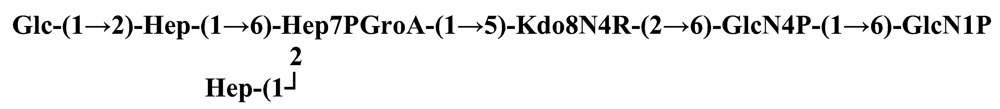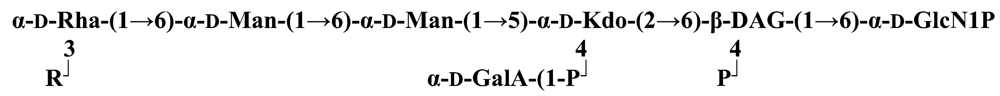Molecular Structure of Endotoxins from Gram-negative Marine Bacteria: An Update
Abstract
:1. Introduction
2. LPS molecular architecture and activity
3. Structure of O-specific polysaccharides from marine bacteria
→4)-α-l-GalpNAm3AcA-(1→3)-α-Sugp-(1→4)-β-d-GlcpNAc3NAcylA-(1→
Pseudoalteromonas rubra ATCC 29570T [29]
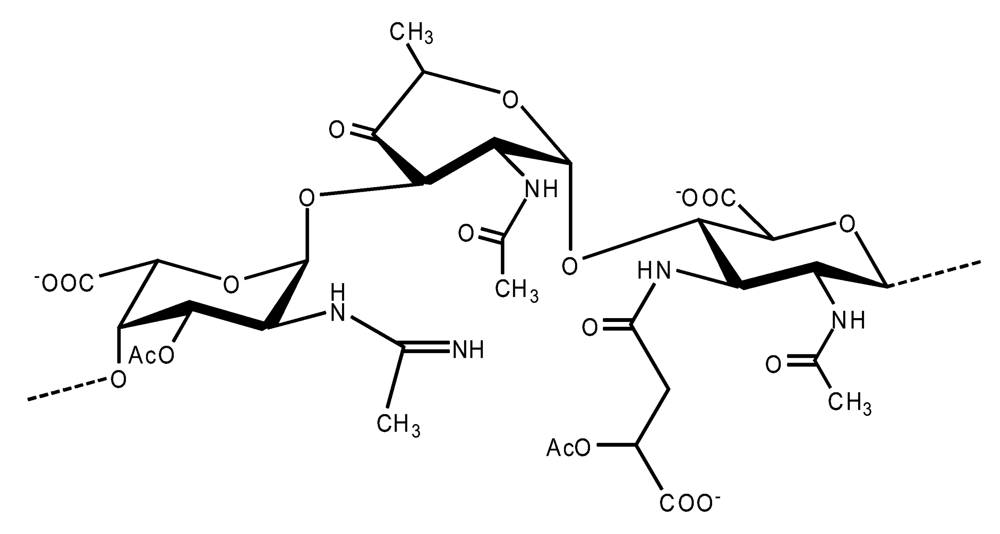
Pseudoalteromonas carrageenovora IAM 12662T [35]The acidic OPS from the type strain of P. aliena species, SW19T (=KMM 3562T=LMG 22059T), has been subjected to structural investigation [37]. It was found that the polymer contains two N-acetyl-d-glucosamine (GlcNAc) residues and one residue each of d-glucuronic acid (GlcA), 2-acetamido-2-deoxy-d-mannnuronic acid (ManNAcA) and 4-acetamido-4,6-dideoxy-d-glucose (N-acetyl-d-viosamine, d-Qui4NAc). Besides that, amino acid analysis revealed the presence of l-serine (l-Ser) as constituent of the PS. In this case, a 1H,13C band-selective constant-time heteronuclear multiple-bond connectivity experiment was used to determine amide linkages between serine and uronic acid residues, via 3JH,C correlations between Ser–Hα and C-6 of the uronic acids. As a result, the polysaccharide was shown to be a branched polymer made up of a pentasaccharide repeating unit containing two l-serine residues as amides of both uronic acid residues. This was the first time that two constituents as amides of uronic acids with l-serine have been found as part of a polysaccharide, namely N-(2-acetamido-2-deoxy-d-mannuronoyl)-l-serine and N-(d-glucuronoyl)-l-serine. Furthermore, this polymer contains another sugar, 4-acetamido-4,6-dideoxy-d-glucose, identified earlier in antigenic polysaccharides from Escherichia coli O:7 [38], Shigella dysenteriae type 7 [39], Francisella tularensis strain 15 [40], Vibrio anguillarum V-123 [41] and Hafnia alvei 1205 [42].Back to TopTopPseudoalteromonas aliena KMM 3562T [37]The antigenic polysaccharide from Pseudoalteromonas atlantica strain IAM 14165 [43] is an example of a polysaccharide containing, within its oligosaccharide repeating unit, a higher monosaccharide with a nine carbon atom skeleton. On the basis of structural characterization data, a pentasaccharide repeating unit was found, containing along with d-Gal, d-GlcNAc, d-QuiNAc and d-GalA, 5,7-diacetamido-3,5,7,9-tetradeoxy-l-glycero-l-manno-non-2-ulosonic acid, one of the stereoisomers of pseudaminic acid [43]. This sugar has been identified earlier as the component of the antigenic PSs from Shigella boydii serogroup 7 [44], some serogroups of Pseudomonas aeruginosa [45,46], Vibrio cholerae serogroup 2 [47] and Escherichia coli O136 [48]. The identified structure is presented below:Pseudoalteromonas atlantica IAM 14165 [43]The genus Shewanella comprises more than 20 validly described species, including both free-living and symbiotic forms, and members of this family have been isolated from various marine sources, including water, sediments, fish, algae and marine animals. These bacteria are responsible of the spoilage of the protein-rich foods and two strains, S. putrefaciens and S. algae, are known as opportunistic pathogens of humans and marine animals and recognised as the causative agents of soft tissues bacteraemia and sepsis. Members of this genus have also been studied for their involvement in a variety of anaerobic processes including the dissimilar reduction of manganese and iron oxides, uranium, thiosulphate and elemental sulphur among others. Because of their metabolic versatility and wide distribution in a variety of aquatic habitats, Shewanella and related microrganisms play a crucial role in the cycling of organic carbon and other nutrients [2]. Several O-polysaccharides from Shewanella have been isolated and characterised. Also in this case, as for Pseudoalteromonas, acidic polysaccharides have been identified, often containing peculiar monosaccharides or acidic non carbohydrate substituents, i.e. malic acid in the O-polysaccharide from S. algae BrY [30] or the peculiar monosaccharide Shewanellose, a novel C-branched sugar [(2-acetamido-2,6-dideoxy-4-C-(3’carboxamide-2’,2’-dihydroxypropyl)-d-galactose, She] first found in the O-polysaccharide from S. putrefaciens A6, together with a derivative of the 8-epilegionamminic acid [49]. In recent years, the structures of two new O-polysaccharides isolated from Shewanella LPS have been added to the vast literature describing such kind of molecules (for a review, see Nazarenko et al. [9]).The recently described species S. fidelis was isolated from sediment samples from the South China Sea [50]. The O-specific polysaccharide from the type strain KMM 3582T has a tetrasaccharide repeating unit consisting of two d-GalNAc, one d-GlcA and one d-GalA residues. The latter is amidated with Nε-[(S)-1-carboxyethyl]-Nα-(d-galacturonoyl)-l-lysine (alaninolysine, AlaLys) [51]. The occurrence of amides of alaninolysine with GalA and GlcA has been reported previously only for the O-specific polysaccharides from a number of members of Enterobacteriaceae family: Providencia rustigianii O14 [52], Providencia alcalifaciens O23 [53], Proteus myxofaciens [54] and Proteus mirabilis O13 [55]. However, this is the first time that this amide compound was found in the O-antigenic polysaccharide of a bacterium from a different taxon. The presence of such characteristic functional groups might be an indication of a common ancestor for bacteria of these genera as members of the γ-subclass of Proteobacteria [56].Shewanella fidelis KMM 3582T [51]An O-specific polysaccharide was also isolated from the LPS of the type strain of the new species S. japonica, which has a linear structure and contains an amino-deoxysugars, N-acetyl-l-fucosamine, 4-acetamido-4,6-dideoxy-d-galactose (4-aminofucose, tomosamine, Fuc4NAc) and N-acetyl-d-bacillosamine (QuiNAc4NAc), together with d-GalA [57]. Bacillosamine has previously been found in the capsular polysaccharide from another marine bacteria, Pseudoalteromonas sp. KMM 155 [58] and in the O-antigens, among the others, of Pseudomonas aeruginosa [59], Pseudomonas aurantiaca IMB 31 [60], Pseudomonas reactans [61], Vibrio cholerae O3 [62], O5 [63], and O8 [64], Pseudoalteromonas haloplanktis KMM 223 [65], ATCC 14393 [66], KMM 634 [67,68]. Fuc4NAc has been found before in the cyclic enterobacterial common antigen from Yersinia pestis [69] and in the repeating unit of the O-chain polysaccharide of Pseudomonas fluorescens biovar A strain IMV 1152 [70] and of Pseudomonas sp. OX1 [71].→3)-α-d-Fucp4NAc-(1→4)-α-d-GalpA-(1→3)-α-l-FucpNAc-(1→3)-β-d-QuipNAc4NAc-(1→Shewanella japonica KMM 3299T [57]In order to present a complete picture of the enormous structural variability that may be encountered in marine bacteria LPSs, we also report here the structure of the OPS from the fish pathogen Flexibacter maritimus [72]. This microrganism belongs to the Cytophaga-Flavobacterium-Bacteroides group. These are a large, heterogeneous group of filamentous, gliding, Gram-negative bacteria infecting a wide variety of fish species and usually forming filamentous biofilms [73], primarily on the tissues associated with the oral cavity. F. maritimus has been associated with a disease (Flexibacteriosis) in a number of fish species [74]. The OPS from this bacterium has been characterized by means of extensive spectroscopical analyses performed on the polysaccharide and on its methanolysis products, revealing the occurrence of a higher monosaccharide, 5-acetamido-7-[(S)-3-hydroxybutyramido]-8-amino-3,5,7,8,9-pentadeoxynonulopyranosonic acid, together with 2-acet-amido-3-O-acetyl-4-[(S)-2-hydroxyglutar-5-ylamido]-2,4,6-trideoxy-β-glucose (QuiNAc3Ac4NAcyl). The repeating unit includes a linkage via the (S)-2-hydroxyglutaric acid residue, that was reported for the first time in a bacterial polysaccharide. The repeating unit is represented in Figure 2:A similar feature, namely the occurrence of O-glycosylated amide linked (R)-malic acid, was reported to be a component of the OPS from another fish pathogen, Flavobacterium psychrophilum, belonging to the same phylum of F. maritimus [75]. The structure found in that case is reported below, and again shows the occurrence of both amino-sugars and non glycidic components.→4)-α-l-FucpNAc-(1→3)-α-d-Quip2NAc4NR-(1→2)-α-L-Rhap-(1→R = (3S,5S)-CH3CH(OH)CH2CH(OH)CH2COFlavobacterium psychrophilum [75]Finally, the OPS from another member of the family of Flavobacteriaceae, Cellulophaga fucicola, was identified, after mild acid hydrolysis with acetate buffer, as constituted by a trisaccharide repeating unit containing a nonulosonic acid residue, pseudaminic acid (5,7-diacetamido-3,5,7,9-tetradeoxy-l-glycero-l-manno-non-2-ulosonic acid, Psep) [76] . The structure found is the following:→4)-β-d-Galp-(1→4)-β-d-Glcp-(1→4)-β-Psep-(2→Cellulophaga fucicola [76]4. Structure of core oligosaccharides from marine bacteria
The structure elucidation of the core region of bacterial LPS is attained after complete deacylation of the LOS. This is normally achieved by alkaline treatment with hydrazine followed by hydrolysis with aqueous KOH, that allows the recovering of the oligosaccharide backbone comprehensive of the lipid A glucosamine disaccharide. This treatment is responsible for the loss of base-labile groups, that are conventionally characterised through a complementary approach that cleaves the linkage between Kdo and lipid A under extremely mild condition (1% acetic acid). In this way, water soluble oligosaccharides can be analysed in aqueous medium. Alternatively, it is possible to simply O-deacylate the LOS and carry out spectroscopic analyses in denaturing solvent system (SDS-d12 in D2O). This latter approach allows one to preserve either acid- and base-labile groups, that can be in this way identified. The chemical and spectroscopical analyses are completed and integrated by means MALDI MS, that allows the identification of the exact molecular weight of these structure, and at the same time allows to determine the occurrence of different glycoforms, varying in the glycosylation and/or phosphorylation pattern.Exploiting this approach, the core oligosaccharides (OSs) from two new core oligosaccharides from Pseudoalteromonas have been recently described, from P. carrageenovora IAM 12662T [35] and P. issachenkonii KMM 3549T [77]. The OS from P. carrageenovora, whose OPS has been described in the previous section, has been elucidated by means of compositional analysis, matrix-assisted laser desorption/ionization mass spectrometry and complete 1H-, 13C- and 31P-NMR spectroscopy. It is composed by a mixture of three glycoforms, differing for the length of the sugar chain and the phosphorylation pattern [35], and is characterized by a strong accumulation of phosphate groups in the lipid A-core portion, creating a region with a high charge density, as depicted below.α-Hepp3P-(1→5)-α-Kdop4P-(2→6)-β-GlcpN4P-(1→6)-α-GlcpN1P (1)β-Galp-(1→4)-α-Hepp3P-(1→5)-α-Kdop4P-(2→6)-β-GlcpN4P-(1→6)-α-GlcpN1P (2)Pseudoalteromonas carrageenovora IAM 12662T [35]A closely related chemical architecture has been found also in the core region of the LPS from P. issachenkonii KMM 3549T [77], isolated from the thallus of the brown alga Fucus evanescens, collected in the Kraternaya Bight of the Kurile Islands in the Pacific Ocean [78]. This halophilic microrganism has bacteriolytic, proteolytic and haemolytic activity and degrades algal polysaccharides, producing a number of glycosyl hydrolases (fucoidanases, laminaranases, alginases, agarases, pullulanases, β-glucosidases, β-galactosidases, β-N-acetylglucosaminidases and β-xylosidases). The core oligosaccharide structure has been depicted after alkaline de-acylation, and on the basis of the MALDI MS and NMR data, it has been described as composed by a mixture of three glycoforms differing in the glycosylation and phosphorylation profile.β-Gal-(1→7)-α-Hep3R-(1→5)-α-Kdo4P-(2→6)-β-GlcN4P-(1→6)-α-GlcN1P (1)a R = Pb R = Ha R = Pb R = HPseudoalteromonas issachenkonii KMM 3549T [77]Interestingly, glycoform 3 was present only in its tri-phosphorylated form, where the three phosphate residues on lipid A and Kdo were plainly assigned, thus, with no phosphate residue at O-3 of the heptose unit. It should be noted, that in two core glycoforms of the LOS from P. issachenkonii (2 and 3), a 4,7-di-substituted heptose is present and this is, at our knowledge, the first time that in a core region a heptose with such a substitution pattern is found. It is noteworthy that the only one more core structure of LPS from Pseudoalteromonas has been reported to date, P. haloplanktis TAC 125 [79]. As in the case of P. carrageenovora and P.issachenkonii, it possesses the carbohydrate skeletonβ-Gal-(1→4)-α-Hep3P-(1→5)-α-Kdo4P-(2→6)-β-GlcN4P-(1→6)-β-GlcN1P,that, up to now, can be considered a unique structural feature of the Pseudoalteromonas genus.From the structural investigation on the core oligosaccharides of LPSs from Shewanella, interesting information have emerged. For instance, the oligosaccharide from S. oneidensis MR-1 constitutes the first case of identification of a monosaccharide other than Kdo, namely 8-amino-3,8-dideoxy-d-manno-octulosonic acid (Kdo8N) [10], as junction of the OS with lipid A. This is not the only odd feature of such OS, since a residue of 2-acetamido-2-deoxy-d-galactose in an open-chain form (GalNAco), linked as cyclic acetal to O-4 and O-6 of d-galactopyranose has also been detected by means of NMR spectroscopy [10]. An open-chain acetal linkage was previously found only in the core part of some Proteus LPSs [80]. In addition, the structure contains a phosphodiester linkage between the α-d-galactopyranose and d-glycero-d-manno-heptose (dd-Hep) residues:Shewanella oneidensis MR-1 [10]The occurrence of Kdo8N replacing Kdo has been also detected in the oligosaccharides from S. algae BrY [11] and from the three strains KMM 3601, KMM 3605 and KMM 3772 of S. pacifica [12], which revealed identical core structures, and constitute, together with the OS structure from S. putrefaciens CN32 [81], the only known oligosaccharide structures for this genus. Although this latter does not include in its architecture the Kdo8N, it is still possible to suggest that this sugar residue may be considered as a taxonomic marker for the genus. The core structure from S. algae BrY is a blend of diverse glycoforms, but all variants are only mere truncations of the following maximal structure.R=Ac or HShewanella algae BrY [11]The combined chemical, MS and NMR approach has led to the complete elucidation of the novel oligosaccharide structure from S. pacifica KMM 3601, KMM 3605 and KMM 3772 [12]. As in the two examples presented above, in S. pacifica LOS the Kdo residue is replaced by Kdo8N. Moreover, as in all Shewanella LPSs, this Kdo8N residue bears an unusual heptose residue (d,d-Hep) at the C-5 position, and this carries a non-carbohydrate substituents linked by a phosphodiester linkage, as reported for S. oneidensis MR-1 LPS.The replacement of Kdo by a derivative in the inner core of LPS is rather rare and, to the best of our knowledge, the only other occurrence is the presence of the 3-hydroxy-derivative of Kdo, d-glycero-d-talo-octulosonic acid (Ko), which non-stoichiometrically replaces Kdo in Acinetobacter haemolyticus LPS [82], whilst Kdo itself is only present as a branching residue of the oligosaccharide. A feature of the LPS from S. pacifica is the presence of glyceric acid, which is attached via phosphodiester linkage to the d,d-Hep, contributing to the increasing of the total negative charge of the inner-core region. Although this is a key molecule of the primary metabolism of Gram-negative bacteria, it has never been detected in the core of LPS molecules.R = P or PPEtNShewanella pacifica KMM 3772 [12]The revision of the genus Alteromonas, first established by Baumann and co-workers [83], resulted in its partition into two genera: Pseudoalteromonas and Alteromonas [84]. The newly defined Alteromonas genus comprises few validly described species, namely A. macleodii, A. marina, A. stellipolaris, A. litorea and A. addita. The structure investigation of the LPS structures from bacteria belonging to this genus has only recently begun, and, up to now, only two structures have been given, from A. macleodii ATCC 27126T [85] and from A. addita KMM 3600T [14]. Interestingly, in both cases the bacteria have been found to produce only a R-LPS, provided with an extremely short oligosaccharide chain with a high negative charge density. The structure of the glycidic portion of the LPS therefore coincides, in such cases, with the description of the OS. Within the core oligosaccharide from A. macleodii ATCC 27126T, the negative charge is inferred, among the others, by the occurrence of Kdo residue that is present as β-configured residue. This feature is rather rare in polysaccharides in general, being sometimes detected in polysaccharide capsules [86], and is very rare in LPSs. In the OPS from Serratia marcescens, β-Kdo is present as a single residue at the end of the chain, functioning as a monosaccharide cap in the regulation of the length of the molecule [87].β-Kdo-(2→7)-α-Hep-(1→5)-α-KdoP-(2→6)-β-GlcN4P-(1→6)-α-GlcN1PAlteromonas macleodii ATCC 27126T [85]The newest species added to the genus, A. addita, was first isolated from sea water samples collected at various depth in the Pacific Ocean region of Chazma Bay (Sea of Japan), during a study on free-living microbial colonies in radionuclides contaminated environments [88]. The core oligosaccharide of the LOS from this strain was characterized by means of 1H-, 13C- and 31P-NMR spectroscopy and MALDI MS, after complete de-acylation and selective mild O-deacylation [14]. This latter approach allowed the detection of the base labile substitution of a Glc residue linked via phosphodiester bond to a heptose moiety:α-Glc-(1-P-3)-□α-Hep-(1→5)-α-Kdo4P-(2→6)-β-GlcN4P-(1→6)-α-GlcN1PAlteromonas addita KMM 3600T [14]The same feature of a phosphodiester bond connecting two monosaccharides units was previously reported in the core oligosaccharide of another marine bacterium, Arenibacter certesii KMM 3941T [13]. The recently described genus Arenibacter was established to accommodate Gram-negative, strictly aerobic, heterotrophic, dark-orange pigmented, non-motile marine bacteria belonging to the Cytophaga–Flavobacterium–Bacteroides phylum [89]. Also in this case, a combined approach based on spectroscopical and MS analyses was performed on the product of either complete and selective O-deacylation. The structural analysis showed the replacement of the β-GlcN (GlcN II) moiety of the lipid A backbone with a unit of 2,3-diamino-2,3-dideoxy-glucose (DAG). DAG has been previously identified as component of the lipid A backbone in other LPSs [90,91]. The DAG-GlcpN β-(1→6)-linked disaccharide was found in lipid A from Campylobacter jejuni and Rhodospirillum salinarum [90], whereas the DAG-DAG β-(1→6)-linked disaccharide is present in the lipid A from the LPS of Aquifex pyrophilus [91], Bordetella pertussis, and Legionella pneumophila, and, as a minor component, in lipid A from C. jejuni [90]. The presence of the DAG moiety in the lipid A backbone of A. certesii contributes to assure the bacterium a major resistance to environment, given the superior resistance of the amide linkages to the hydrolysis, which can be seen as an adaptation of the bacterium to external hostile environment.R = H or α-d-RhaArenibacter certesii KMM 3941T [13]The occurrence of the GalA moiety contributes also in this case to the increase of the total charge density of the molecule. It is deemed that these negatively charged groups allow the establishment of ionic bridges between LPS molecules due to electrostatic interactions with environmentally available bivalent cations (Ca2+, Mg2+), and that these contribute to the rigidity and stability of the Gram-negative cell wall [92]. Thus, the high number of negative charges in these short oligosaccharides from Pseudoalteromonas, Shewanella, Alteromonas and Arenibacter could be important for maintaining the integrity of the outer membrane exposed to a peculiar external surrounding.5. Structure of lipids A from marine bacteria
In order to perform the structure elucidation of lipid A, the glycolipid moiety has to be released from the LPS or LOS. This is generally achieved through the same mild treatment used for the isolation of OPSs, which exploits the susceptibility to acid conditions of the glycosidic linkage of Kdo to β-GlcN. The lipid A is then characterized by means of selective chemical analyses, NMR spectroscopy and, mostly, mass spectrometry, that allows the depiction of the intrinsic heterogeneity of this portion. Selective deacylation with NH4OH, which cleaves the acyloxy-acylester groups but leaves unaffected the acyloxy-acylamide substituents before MS analysis provides useful insight to the description of the acylation pattern of lipid A [93,94]. The information about the lipid A structure may also derive from the MALDI MS analysis on the intact LOS fraction. In fact, due to the energy associated to the laser source, a phenomenon of β-elimination [95] may be observed, that yields either oligosaccharide ion (termed B-type ions, according to the Domon and Costello nomenclature [96]) and lipid A ions.The structural study of marine bacteria lipid A is still at an early stage compared to that of oligo- and poly-saccharides. Nevertheless, in the past few years, a number of structures has been described. In order to give a synoptic description of lipid A, the fatty acid substitution profile referring to the main form of the studied species is reported in Table 1. Unless specifically stated, all the molecules share the conventional glucosamine disaccharide backbone and show phosphorylation at the positions C1 and C4’, as exemplified in Figure 3, where the lipid A from E. coli is shown.The lipid A portion of the LPS is the most conservative region of the molecule, and exhibits a low intra-genus variability. Therefore, only slight differences are encountered, among the genus, in the lipids A from the bacteria herein presented. For instance, three lipid As have been described from the LPSs of bacteria belonging to the genus Pseudoalteromonas, namely from P. haloplanktis ATCC 14393T [97] and TAC 125 [98] and from P. issachenkonii KMM 3549T [77].A structure of the lipid A from P. haloplanktis ATCC 14393T has been proposed on the basis of chemical analyses and NMR spectroscopy, pointing out to two possible penta-acyl distributions, with a variable localization of the secondary amide linked fatty acid, namely a dodecanoic acid moiety (12:0), on GlcN I or GlcN II. Moreover, primary fatty acid substitution at C-3 and C-3’ is non-stoichiometrically given by (R)-3-hydroxy-dodecanoic acid [12:0(3-OH)], (R)-3-hydroxy-undecanoic acid [11:0(3-OH)], by their iso-branched isomers [iso-12:0(3-OH) and iso-11:0(3-OH), respectively], or (R)-3-hydroxy-decanoic acid [10:0(3-OH)] [97].In the case of the strain TAC 125 [98], the structure depiction has been achieved through the use of Electrospray Ionization (ESI) tandem mass spectrometry (MS/MS). Also in this case, the most abundant species is the penta-acyl lipid A with 12:0(3-OH) linked both as ester and amide to 2’,3’ (distal glucosamine) and 2,3 positions (proximal glucosamine) of the sugar backbone. Secondary substitution was realized at the OH group of the 12:0(3-OH) at position 3’ by a 12:0. In addition to the penta-acyl component, a minor tetra-acyl form, lacking the acyl residue at position 3, was also detected. The slight differences among the lipid A fractions from these two strains of the same species have been attributed to a difference in the culture condition employed, since it is known that growth conditions can modify the molecular architecture of lipid A.The lipid A structure from P. issachenkonii KMM 3549T has been described directly from the MS data obtained for the intact LOS sample [77]. As the opposite of P. haloplanktis, in P. issachenkonii the tetra-acyl lipid A is the most abundant species, carrying three C12:0(3-OH) and a C12:0 residues. Minor penta-acyl [bearing an additional C10:0(3-OH)] and tri-acyl [lacking a 12:0(3-OH)] forms were also present. The identical composition and acylation profile was found in the lipid A from the LOS of P. carrageenovora IAM 12662T (unpublished data). Also in these two cases, the prevalence of the less acylated species compared to P. haloplanktis may be originated by the different environmental conditions from which this microrganism was first isolated.Together with the structure elucidation of the core oligosaccharide, the molecular characterization of the lipids A from Alteromonas macleodii ATCC 27126T [85] and Alteromonas addita KMM 3600T [14] has been performed. MALDI MS analyses of the intact and NH4OH-treated lipid A from A. macleodii ATCC 27126T allowed the description of a blend in which the tetra-acyl species constituted the major form, together with minor amounts of tri- and penta-acyl species. In particular, the penta-acyl lipid A carried two C12:0(3-OH) residues in an ester linkage and two C12:0(3-OH) in amide linkages as primary fatty acids and one C12:0 residue as a secondary fatty acid at C2’. One or two ester-linked 12:0(3-OH) lack in the tetra- and tri-acyl forms [85].Using the same approach, the lipid A from A. addita KMM 3600T has been determined [14], and it presented a predominance of the penta-acyl species, with the same [3+2] distribution of the fatty acids on the glucosamine disaccharide. In this case, the major species exhibited two units of (R)-3-hydroxy-tetradecanoic acid [14:0(3-OH)] as amide substituents. This residue was eventually replaced, on the GlcN I, by a (R)-3-hydroxy-tridecanoic acid [13:0(3-OH)]. Secondary acylation at GlcN II was realized by a 14:0 or a 12:0 residue, whereas the ester linked fatty acid at C3 position was a 10:0(3-OH).The acylation pattern for the Lipid A from Shewanella pacifica KMM 3772 has been completely described exploiting the MS data associated to the lipid A fraction and to either the intact and O-deacylated LOS [12]. In both cases, ions produced by the in source β-elimination of lipid A were in fact visible. The main lipid A species from S. pacifica KMM 3772 is a hexa-acyl form in which both amide- and ester-linked primary fatty acids are (R)-3-hydroxy-tridecanoic acid [13:0(3-OH)]. The primary substituents at the GlcN II both bear secondary tridecanoic acid (13:0) residues. Such residues are missing in the minor penta- and tetra-acyl species.The structure of the lipid A from the LPS of another γ-proteobacterium, Marinomonas vaga (formerly Alteromonas vaga) ATCC 27119T, has been described [98]. This microrganism was isolated from the waters off the coast of the Hawaiian archipelago and has a psychrophilic and moderately halophilic character. Its lipid A shows stoichiometric lack of the phosphate ester group at C4’. Moreover, also in this case, the main form is represented by a penta-acyl species, with a [3+2] distribution of fatty acids, and acylation is principally performed by the short chained 10:0(3-OH). It has been reported for the first time in this occasion that the 3-hydroxy moiety is also present as the secondary substituent at the amide linked fatty acid of GlcN II [98].The lipid A from Arenibacter certesii KMM 3941T, from the Cytophaga-Flavobacterium-Bacteroides phylum, is characterised by the prevalence of a penta-acyl form (Silipo et al., unpublished results). As discussed above in the core oligosaccharide section, this glycolipid possesses a glycidic backbone constituted by a DAG-GlcpN β-(1→6) linked disaccharide. Phosphorylation occurs, also in this case, at the conventional C1 and C4’ positions. Singularly, substitution is performed, in the most abundant penta-acyl species, only by fatty acids with an odd number of carbon atoms, namely four units of (R)-3-hydroxy-pentadecanoic acid [15:0(3-OH)], and one secondary pentadecanoic acid (15:0). From the MS analyses on the partially degraded lipid A, it has been possible to locate the 15:0 residue in ester linkage on one of the two primary amide substituents on the DAG unit, although it is not yet well understood whether it is the one at position C2’ or C3’.To the same phylum of A. certesii KMM 3941T belongs the bacterium Chryseobacterium scophtalmum CIP 104199T, whose LOS possessed an extremely unusual lipid A that was isolated and characterized [100]. The peculiarity of this molecule consists in the occurrence of a monosaccharidic lipid A moiety, in which a single glucosamine unit, phosphorylated at C1, carries one (R)-3-hydroxy-15-methylhexadecanoic [iso-17:0(3-OH)] and one (R)-3-hydroxy-13-methyltetradecanoic [iso-15:0(3-OH)] residues, as primary amide and ester substituents, respectively. This structure closely resembles the so-called lipid X, the biosynthetic precursor of lipid A in E. coli. This event probably relates with the absence, in C. scophtalmum membranes, of phosphatidylglycerol (PG), one of the three main phospholipids of Gram-negative bacteria. It is been in fact observed that the accumulation of lipid X in cells of mutant bacteria correlates with the deficiency of PG [101–103]. The absence of this phospholipid has been also detected in other bacteria of the same genus, i.e. C. indoltheticum and C. defluvii sp. [104]. Apparently, there is a certain relationship between lipid A synthesis and the presence of PG in bacterial cells: when PG is absent, lipid As with incomplete monosaccharide structure are synthesized. However, some substantial differences between the E. coli mutant strain and C. scophtalmum should be mentioned. Primarily, despite of the absence of PG and lipid A (in its classical structural variant), C. scophtalmum retains its viability, while the mutant E. coli cells grow only at nonpermissive temperatures (42°C) and, already after 3 h, a cessation of their growth is observed [101]. In addition, lipid A from C. scophtalmum can be isolated only after acidic hydrolysis of bacterial cells, indicating the existence of a strong, probably chemical, bond with other components of the outer membrane.Besides the lipid A structures presented above, preliminary studies have been performed on the chemical characterization of the fatty acid compositions (but not their distribution) of the glycolipid moieties from other Pseudoalteromonas species [105]. In all the cases examined, the fatty acid composition resembles that from the microrganisms here described, with a strong prevalence of shorter chained fatty acids compared to the wide-spread enterobacteria.6. Concluding remarks
Marine bacteria are habitual colonizers of all watery environments, and, either as free living colonies or symbiotic forms, they are present in sea habitats worldwide. It is widely accepted that the molecular architecture of the LPSs from Gram-negative marine bacteria strictly relates to the possibility of thriving in such environments. These molecules represent the outermost barrier of the microbial cell towards the surrounding environment, being therefore involved in the cellular resistance against the stressors typifying marine environments. Moreover, LPS endotoxins play a fundamental role in a number of essential processes, like the adhesion to the host tissues as well as to sediments, and the recognition either in the event of symbiotic interaction or animal and human pathogenicity. In this latter case, the most conservative portions of the LPSs structure act as PAMPs, being recognised by specialised receptors of the host immune system, thus eliciting the immune response. In the present paper, the structures of the LPSs from a number of marine Gram-negative bacteria is reviewed. This work provides an overlook on the growing body of information about oligo- and polysaccharides from marine Gram-negative bacteria, and also evaluates, for the first time, a collection of structures of lipids A. Many features encountered during our investigation can be ascribed to the environmental demand imposed by stress factors like elevated pressures, high salinity or low temperatures, to which they might represent an adaptive response. For instance, the majority of the O-specific polysaccharides structures here reported is endowed with anionic groups, variously supplied by acidic (often uncommon) monosaccharides, amino-acid or non-glycidic appendages. This is a general tendency observed in the smooth marine strains, and was already identified in the OPSs that were earlier characterised. A comparison between the structures presented suggests the idea that bacteria acquainted with marine environments provide the outer membrane with molecules with uncommon negative charge density, adapted during evolution in order to provide optimal protection of the cell. In the case of rough strains, producing only lipooligosaccharides (e.g. A. addita and A. macleodii), the enhancement of the cation binding capacity of the membrane may have been achieved through the constitutive shortening of the oligosaccharide chains and the addition of anionic decorations of both glycidic and non-glycidic nature (i.e. phosphate groups). Such structural features increase the capability of binding naturally available cations (Ca+2 and Mg+2), through which the molecules overcome the electrostatic repulsion, otherwise arising among them. In this way, charged glycolipids can organize themselves in a rigid net of cross-linkages, providing higher resistance to physical stressors. This effect is in our opinion closely related to barophily, thus to mechanical pressures acting on the cell. Halophily may reflect in the enhancement of water retention in the immediate surrounding of the cell, as a reaction to the osmotic pressure generating between the cytoplasm and the external sea environment. The salt content in the different cell compartments may be modulated through the creation of a microenvironment around the cell with an increased water attraction capability due to charged groups, confirming the trend already outlined for glycolipids within the cytoplasmic membrane [106].The present work also reports for the first time a collection of lipid A structures from marine bacteria. These are the first few examples for which the complete acylation pattern has been described, and some interesting conclusions can be made. For instance, a preference for shorter acyl chains [10:0(3-OH); 12:0; 12:0(3-OH); 13:0(3-OH)] with respect to the lipids A from Enterobacteriaceae is evident. The shortening of lipid A fatty acids has been observed in mesophilic bacteria as a short term response to cold stress [20,107] and is in agreement with the fact that marine bacteria presented in this paper express a certain psychrophilic character or at least do not live at the “canonical” 37° C. Evolutionary adaptation in obliged psycrophiles may have led to similar stratagems in membrane glycolipids, bringing to the preference for structures more suitable to counterbalance the freezing effects of cold on long chain fatty acids. Shorter lipids have less Van der Waals interactions between the chains, preserving the physiological fluidity of the Outer Membrane. Moreover, the majority of the lipids A characterized from marine bacteria produces mainly penta- and tetra-acyl species, i.e. species with less fatty acids than endotoxically active molecules. Some species, like M. vaga, also possess a single phosphate group on the disaccharide backbone, another feature that is related to a lowering in toxicity. These two features make marine lipid As potential endotoxin antagonists candidates [108–110]. The rather low innate immune system elicitation showed by Marinomonas vaga [99], Alteromonas macleodii and A. addita (unpublished results) lipids A fits well with the above suggestion. Further investigations are needed to confirm this theory.
References and Notes
- Head, IM; Jones, DM; Röling, WFM. Marine microorganisms make a meal of oil. Nat Rev Microbiol 2006, 4, 173–182. [Google Scholar]
- Hau, HH; Gralnick, JA. Ecology and biotechnology of the genus Shewanella. Ann Rev Microbiol 2007, 61, 237–258. [Google Scholar]
- Rothschild, LJ; Mancinelli, RL. Life in extreme environments. Nature 2001, 409, 1092–1100. [Google Scholar]
- Alexander, C; Rietschel, ET. Bacterial lipopolysaccharides and innate immunity. J Endotoxin Res 2001, 7, 167–202. [Google Scholar]
- Dinarello, CA. Cytokines as mediators in the pathogenesis of septic shock. Curr Top Microbiol Immunol 1996, 216, 133–65. [Google Scholar]
- Caroff, M; Karibian, D. Structure of bacterial lipopolysaccharides. Carbohydr Res 2003, 338, 2431–2447. [Google Scholar]
- Holst, O. Brade, H, Morrison, DC, Opal, S, Vogel, S, Eds.; Endotoxin in Health and Disease; Marcel Dekker Inc: New York NY, 1999; pp. 115–154. [Google Scholar]
- Rietschel, ET; Kirikae, T; Shade, UF; Ulmer, AJ; Holst, O; Brade, H; Schmidt, G; Mamat, U; Grimmecke, HD; Kusumoto, S; Zahringer, U. The chemical structure of bacterial endotoxin in relation to bioactivity. Immunobiology 1993, 187, 169–190. [Google Scholar]
- Nazarenko, EL; Komandrova, NA; Gorshkova, RP; Tomshich, SV; Zubrov, VA; Kilcoyne, M; Savage, AV. Structures of polysaccharides and oligosaccharides of some Gram-negative marine Proteobacteria. Carbohydr Res 2003, 338, 2449–2457. [Google Scholar]
- The structure of the rough-type lipopolysaccharide from Shewanella oneidensis MR-1, containing 8-amino-8-deoxy-Kdo and an open-chain form of 2-acetamido-2-deoxy-d-galactose. Carbohydr Res 2003, 338, 1991–1997.
- Vinogradov, E; Korenevsky, A; Beveridge, TJ. The structure of the core region of the lipopolysaccharide from Shewanella algae BrY, containing 8-amino-3,8-dideoxy-d-manno-oct-2-ulosonic acid. Carbohydr Res 2004, 339, 737–740. [Google Scholar]
- Silipo, A; Leone, S; Molinaro, A; Sturiale, L; Garozzo, D; Nazarenko, EL; Gorshkova, RP; Ivanova, EP; Lanzetta, R; Parrilli, M. Complete structural elucidation of a novel lipooligosaccharide from the Outer Membrane of the marine bacterium Shewanella pacifica. Eur J Org Chem 2005, 2281–2291. [Google Scholar]
- Silipo, A; Molinaro, A; Nazarenko, EL; Sturiale, L; Garozzo, D; Gorshkova, RP; Nedashkovskaya, OI; Lanzetta, R; Parrilli, M. Structural characterization of the carbohydrate backbone of the lipooligosaccharide of the marine bacterium Arenibacter certesii strain KMM 3941T. Carbohydr Res 2005, 340, 2540–2549. [Google Scholar]
- Leone, S; Molinaro, A; Sturiale, L; Garozzo, D; Nazarenko, EL; Gorshkova, RP; Ivanova, EP; Shevchenko, LS; Lanzetta, R; Parrilli, M. The Outer Membrane of the Marine Gram-Negative Bacterium Alteromonas addita is Composed of a Very Short-Chain Lipopolysaccharide with a High Negative Charge Density. Eur J Org Chem 2007, 1113–1122. [Google Scholar]
- Zähringer, U; Lindner, B; Rietschel, ET. Molecular structure of Lipid A, the endotoxic center of bacterial lipopolysaccharides. Adv Carbohydr Chem Biochem 1994, 50, 211–276. [Google Scholar]
- Zähringer, U; Lindner, B; Rietschel, ET. Brade, H, Morrison, DC, Opal, S, Vogel, S, Eds.; Endotoxin in Health and Disease; Marcel Dekker Inc: New York NY, 1999; pp. 93–114. [Google Scholar]
- Wilkinson, SG. Bacterial lipopolysaccharides - themes and variations. Prog Lipid Res 1996, 35, 283–343. [Google Scholar]
- Akira, S; Uematsu, S; Takeuchi, O. Pathogen recognition and innate immunity. Cell 2006, 124, 783–801. [Google Scholar]
- Seydel, U; Oikava, M; Fukase, K; Kusumoto, S; Brandeburg, K. Intrinsic conformation of lipid A is responsible for agonistic and antagonistic activity. Eur J Biochem 2000, 267, 3032–3039. [Google Scholar]
- Carty, SM; Sreekumar, KR; Raetz, CR. Effect of cold shock on lipid A biosynthesis in Escherichia coli. Induction at 12C of an acyltransferase specific for palmitoleoyl-acyl carrier protein. J Biol Chem 1999, 274, 9677–9685. [Google Scholar]
- Ramos, JL; Gallegos, MT; Marques, S; Ramos-Gonzalez, MI; Espinosa-Urgel, M; Segua, A. Responses of Gram-negative bacteria to certain environmental stressors. Curr Opin Microbiol 2001, 4, 166–171. [Google Scholar]
- Vorobeva, EV; Krasikova, IN; Solov’eva, TF. Influence of lipopolysaccharides and lipids A from some marine bacteria on spontaneous and Escherichia coli LPS-induced TNF-α release from peripheral human blood cells. Biochemistry (Moscow) 2006, 71, 759–766. [Google Scholar]
- Coats, SR; Do, CT; Karimi-Naser, LM; Braham, PH; Darveau, RP. Antagonistic lipopolysaccharides block E. coli lipopolysaccharide function at human TLR4 via interaction with the human MD-2 lipopolysaccharide binding site. Cell Microbiol 2007, 9, 1191–202. [Google Scholar]
- Galanos, C; Lüderitz, O; Westphal, O. A new method for the extraction of R lipopolysaccharides. Eur J Biochem 1969, 9, 245–249. [Google Scholar]
- Westphal, O; Jann, K. Bacterial lipopolysaccharides. Extraction with phenol / water and further application of the procedure. Meth Carbohydr Chem 1965, 5, 83–91. [Google Scholar]
- http://www.ncbi.nlm.nih.gov/Taxonomy/Browser/wwwtax.cgi?id=22
- Jensen, PR; Fenical, W. Strategies for the discovery of secondary metabolites from marine bacteria: ecological perspectives. Annu Rev Microbiol 1994, 48, 559–584. [Google Scholar]
- Holmström, C; Kjelleberg, S. Marine Pseudoalteromonas species are associated with higher organisms and produce biologically active extracellular agents. FEMS Microbiol Ecology 1999, 30, 285–293. [Google Scholar]
- Kilcoyne, M; Shashkov, AS; Knirel, YA; Gorshkova, RP; Nazarenko, EL; Ivanova, EP; Gorshkova, NM; Senchenkova, SN; Savage, AV. The structure of the O-polysaccharide of the Pseudoalteromonas rubra ATCC 29570T lipopolysaccharide containing a keto sugar. Carbohydr Res 2005, 340, 2369–75. [Google Scholar]
- Vinogradov, E; Korenevsky, A; Beveridge, TJ. The structure of the O-specific polysaccharide chain of the Shewanella algae BrY lipopolysaccharide. Carbohydr Res 2003, 338, 385–388. [Google Scholar]
- Forsberg, LS; Noel, KD; Box, J; Carlson, RW. Genetic locus and structural characterization of the biochemical defect in the O-antigenic polysaccharide of the symbiotically deficient Rhizobium etli mutant, CE166. Replacement of N-acetylquinovosamine with its hexosyl-4-ulose precursor. J Biol Chem 2003, 278, 51347–59. [Google Scholar]
- Jansson, PE; Lindberg, B; Lindquist, U. Structural studies of the capsular polysaccharide from Streptococcus pneumoniae type 5. Carbohydr Res 1985, 140, 101–10. [Google Scholar]
- Sadovskaya, I; Brisson, JR; Khieu, NH; Mutharia, LM; Altman, E. Structural characterization of the lipopolysaccharide O-antigen and capsular polysaccharide of Vibrio ordalii serotype O:2. Eur J Biochem 1998, 253, 319–27. [Google Scholar]
- MacLean, LL; Perry, MB; Crump, EM; Kay, WW. Structural characterization of the lipopolysaccharide O-polysaccharide antigen produced by Flavobacterium columnare ATCC 43622T. Eur J Biochem 2003, 270, 3440–3446. [Google Scholar]
- Silipo, A; Molinaro, A; Nazareno, EL; Gorshkova, RP; Ivanova, EP; Lanzetta, R; Parrilli, M. The O-chain structure from the LPS of marine halophilic bacterium Pseudoalteromonas carrageenovora-type strain IAM 12662T. Carbohydr Res 2005, 340, 2693–2697. [Google Scholar]
- Muldoon, J; Perepelov, AV; Shashkov, AS; Gorshkova, RP; Nazarenko, EL; Zubkov, VA; Ivanova, EP; Knirel, YA; Savage, AV. Structure of a colitose-containing O-specific polysaccharide of the marine bacterium Pseudoalteromonas tetraodonis IAM 14160(T). Carbohydr Res 2001, 333, 41–46. [Google Scholar]
- Nordmark, EL; Perepelov, AV; Shashkov, AS; Nazarenko, EL; Gorshkova, RP; Ivanova, EP; Widmalm, G. Structure of an acidic polysaccharide from the marine bacterium Pseudoalteromonas aliena type strain KMM 3562T containing two residues of l-serine in the repeating unit. Carbohydr Res 2005, 340, 1483–1487. [Google Scholar]
- L'vov, VL; Shashkov, AS; Dmitriev, BA; Kochetkov, NK; Jann, B; Jann, K. Structural studies of the O-specific side chain of the lipopolysaccharide from Escherichia coli O:7. Carbohydr Res 1984, 126, 249–259. [Google Scholar]
- Knirel, YA; Dashunin, VV; Shashkov, AS; Kochetkov, NK; Dmitriev, BA; Hoffman, IL. Somatic antigens of Shigella: Structure of the O-specific polysaccharide chain of the Shigella dysenteriae type 7 lipopolysaccharide. Carbohydr Res 1988, 179, 51–60. [Google Scholar]
- Vinogradov, EV; Shashkov, AS; Knirel, YA; Kochetkov, NK; Tochtamysheva, NV; Averin, SF; Goncharova, OV; Khlebnikov, VS. Structure of the O-antigen of Francisella tularensis strain 15. Carbohydr Res 1991, 219, 289–297. [Google Scholar]
- Eguchi, H; Kaya, S; Araki, Y; Kojima, N; Yokota, SI. Structure of the O-polysaccharide chain of the lipopolysaccharide of Vibrio anguillarum V-123. Carbohydr Res 1992, 231, 159–169. [Google Scholar]
- Katzenellenbogen, E; Romanowska, E; Kocharova, NA; Knirel, YA; Shashkov, AS; Kochetkov, NK. The structure of glycerol teichoic acid-like O-specific polysaccharide of Hafnia alvei 1205. Carbohydr Res 1992, 231, 249–260. [Google Scholar]
- Perepelov, AV; Shashkov, AS; Torgov, VI; Nazarenko, EL; Gorshkova, RP; Ivanova, EP; Gorshkova, NM; Widmalm, G. Structure of an acidic polysaccharide from the agar-decomposing marine bacterium Pseudoalteromonas atlantica strain IAM 14165 containing 5,7-diacetamido-3,5,7,9-tetradeoxy-l-glycero-l-manno-non-2- ulosonic acid. Carbohydr Res 2005, 340, 69–74. [Google Scholar]
- L’vov, VL; Shashkov, AS; Dmitriev, BA. Bacterial antigenic polysaccharides. The structure of the repeating unit of the specific polysaccharide from Shigella boydii type 7. Russ J Bioorg Chem 1987, 13, 223–233. [Google Scholar]
- Knirel, YA; Kocharova, NA; Shashkov, AS; Dmitriev, BA; Kochetkov, NK; Stanislavsky, ES; Mashilova, GM. Somatic antigens of Pseudomonas aeruginosa. The structure of O-specific polysaccharide chains of the lipopolysaccharides from P. aeruginosa O5 (Lanyi) and immunotype 6 (Fisher). Eur J Biochem 1987, 163, 639–652. [Google Scholar]
- Knirel, YA; Vinogradov, EV; Shashkov, AS; Kochetkov, NK; L'vov, VL; Dmitriev, BA. Identification of 5-acetamido-3,5,7,9-tetradeoxy-7-[(R)-3- hydroxybutyramido]-l-glycero-l-manno-nonulosonic acid as a component of bacterial polysaccharides. Carbohydr Res 1985, 141, C1–C3. [Google Scholar]
- Kenne, L; Lindberg, B; Schweda, E; Gustavsson, B; Holme, T. Structural studies of the O-antigen from Vibrio cholerae O:2. Carbohydr Res 1988, 180, 285–294. [Google Scholar]
- Staaf, M; Weintraub, A; Widmalm, G. Structure determination of the O-antigenic polysaccharide from the enteroinvasive Escherichia coli O136. Eur J Biochem 1999, 263, 656–661. [Google Scholar]
- Shashkov, AS; Torgov, VI; Nazarenko, EL; Zubkov, VA; Gorshkova, NM; Gorshkova, RP; Widmalm, G. Structure of the phenol-soluble polysaccharide from Shewanella putrefaciens strain A6. Carbohydr Res 2002, 337, 1119–1127. [Google Scholar]
- Ivanova, EP; Sawabe, T; Hayashi, K; Gorshkova, NM; Zhukova, NV; Nedashkovskaya, OI; Mikhailov, VV; Nicolau, DV; Christen, R. Shewanella fidelis sp. nov., isolated from sediments and sea water. Int J Syst Evol Microbiol 2003, 53, 577–582. [Google Scholar]
- Kilcoyne, M; Perepelov, AV; Shashkov, AS; Nazarenko, EL; Ivanova, EP; Gorshkova, NM; Gorshkova, RP; Savage, AV. Structure of an acidic O-specific polysaccharide from marine bacterium Shewanella fidelis KMM 3582T containing N∊-[(S)-1-carboxyethyl]-Nα-(d-galacturonoyl)-l-lysine. Carbohydr Res 2004, 339, 1655–1661. [Google Scholar]
- Kocharova, NA; Zatonsky, GV; Torzewska, A; Macieja, Z; Bystrova, OV; Shashkov, AS; Knirel, YA; Rozalski, A. Structure of the O-specific polysaccharide of Providencia rustigisnii O14 containing N∊-[(S)-1-carboxyethyl]-Nα-(d-galacturonoyl)-l- lysine. Carbohydr Res 2003, 338, 1009–1016. [Google Scholar]
- Kocharova, NA; Vinogradov, EV; Borisova, SA; Shashkov, AS; Knirel, YA. Identification of N∑-[(R)-1-carboxyethyl]-l-lysine in, and the complete structure of, the repeating unit of the O-specific polysaccharide of Providencia alcalifaciens O23. Carbohydr Res 1998, 309, 131–133. [Google Scholar]
- Sidorczyk, Z; Kondakova, AN; Zych, K; Senchenkova, SN; Shashkov, AS; Drzewiecka, D; Knirel, YA. Structure of the O-polysaccharide from Proteus myxofaciens. Eur J Biochem 2003, 270, 3182–3188. [Google Scholar]
- Perepelov, AV; Senchenkova, SN; Cedzynski, M; Ziolkowski, A; Vinogradov, EV; Kaca, W; Shashkov, AS; Knirel, YA. Isolation using triflic acid solvolysis and identification of N∊- [(R)-1-carboxyethyl]-Nα-(d-galacturonoyl)-l-lysine as a component of the O-specific polysaccharide of Proteus mirabilis O13. Carbohydr Res 2000, 328, 441–444. [Google Scholar]
- Penner, JL. Ballows, A, Truper, HG, Dworkin, M, Harder, W, Schleifer, KH, Eds.; The Genera Proteus, Providencia and Morganella; Springer-Verlag: Berlin, 1992; pp. 2849–2863. [Google Scholar]
- Kilcoyne, M; Shashkov, AS; Perepelov, AV; Nazarenko, EL; Gorshkova, RP; Ivanova, EP; Widmalm, G; Savage, AV. Structure of the O-specific polysaccharide from Shewanella japonica type strain KMM 3299T containing the rare amino sugar Fuc4NAc. Carbohydr Res 2005, 340, 1557–1561. [Google Scholar]
- Zubkov, VA; Nazarenko, EL; Gorshkova, RP; Ivanova, EP; Shashkov, AS; Knirel, YA; Paramonov, NA; Ovodov, YS. Structure of the capsular polysaccharide from Alteromonas sp. CMM 155. Carbohydr Res 1995, 275, 1147–1154. [Google Scholar]
- Knirel, YA; Vinogradov, EV; Shashkov, AS; Wilkinson, SG; Tahara, Y; Dmitriev, BA; Kochetkov, NK; Stanislavsky, ES; Mashilova, GM. Somatic antigens of Pseudomonas aeruginosa. The structure of O-specific polysaccharide chains of the lipopolysaccharides from P. aeruginosa O1 (Lanyi), O3 (Habs) and O14 (Wokatch), and the serologically related strain NCTC 8505. Eur J Biochem 1986, 155, 659–669. [Google Scholar]
- Knirel, YA; Zdorovenko, GM; Veremeychenko, SN; Lipkind, GM; Shashkov, AS; Zakharova, IY; Kochetkov, NK. Antigenic polysaccharides of bacteria. 31 . The structure of the O-specific polysaccharide chain of Pseudomonas aurantiaca IMB 31 lipopolysaccharide. Russ J Bioorgan Chem 1988, 14, 352–358. [Google Scholar]
- Molinaro, A; Evidente, A; Sante Iacobellis, N; Lanzetta, R; Lo Cantore, P; Mancino, A; Parrilli, M. O-specific chain structure from the lipopolysaccharide fraction of Pseudomonas reactans: a pathogen of the cultivated mushrooms. Carbohydr Res 2002, 337, 467–471. [Google Scholar]
- Chowdhury, TA; Jansson, PE; Lindberg, B; Gustavsson, B; Holme, T. Structural studies of the Vibrio cholerae O:3 O-antigen polysaccharide. Carbohydr Res 1991, 215, 303–314. [Google Scholar]
- Hermansson, K; Jansson, PE; Holme, T; Gustavsson, B. Structural studies of the Vibrio cholerae O:5 O-antigen polysaccharide. Carbohydr Res 1993, 348, 199–211. [Google Scholar]
- Kocharova, NA; Perepelov, AV; Zatonsky, GV; Shashkov, AS; Knirel, YA; Jansson, PE; Weintraub, A. Structural studies of the O-specific polysaccharide of Vibrio cholerae O:8 using solvolysis with triflic acid. Carbohydr Res 2001, 330, 83–92. [Google Scholar]
- Hanniffy, OM; Shashkov, AS; Senchenkova, SN; Tomshich, SV; Komandrova, NA; Romanenko, LA; Knirel, YA; Savage, AV. Structure of a highly acidic O-specific polysaccharide of lipopolysaccharide of Pseudoalteromonas haloplanktis KMM 223 containing l-iduronic acid and d-QuiNHb4NHb. Carbohydr Res 1998, 307, 291–298. [Google Scholar]
- Hanniffy, OM; Shashkov, AS; Senchenkova, SN; Tomshich, SV; Komandrova, NA; Romanenko, LA; Knirel, YA; Savage, AV. Structure of an acidic O-specific polysaccharide of Pseudoalteromonas haloplanktis type strain ATCC 14393 containing 2-acetamido-2-deoxy-d- and -l-galacturonic acids and 3-(N-acetyl-d-alanyl)amino-3,6-dideoxy-d-glucose. Carbohydr Res 1999, 321, 132–138. [Google Scholar]
- Komandrova, NA; Tomshich, SV; Shevchenko, LS; Perepelov, AV; Senchenkova, SN; Shashkov, AS; Knirel, YA. Structure of an acidic O-specific polysaccharide from marine bacterium Pseudoalteromonas sp. KMM 634. Biochemistry (Moscow) 2000, 65, 1253–1261. [Google Scholar]
- Perepelov, AV; Senchenkova, SN; Shashkov, AS; Komandrova, NA; Tomshich, SV; Shevchenko, LS; Knirel, YA; Kochetkov, NK. First application of triflic acid for selective cleavage of glycosidic linkages in structural studies of a bacterial polysaccharide from Pseudoalteromonas sp. KMM 634. J Chem Soc Perk Trans I 2000, 363–366. [Google Scholar]
- Vinogradov, EV; Knirel, YA; Thomas-Oates, JE; Shashkov, AS; L'vov, VL. The structure of the cyclic enterobacterial common antigen (ECA) from Yersinia pestis. Carbohydr Res 1994, 258, 223–232. [Google Scholar]
- Knirel, YA; Paramonov, NA; Shashkov, AS; Kochetkov, NK; Zdorovenko, GM; Veremeychenko, SN; Zakharova, IY. Somatic antigens of pseudomonads: structure of the O-specific polysaccharide of Pseudomonas fluorescens biovar A strain IMV 1152. Carbohydr Res 1993, 243, 205–210. [Google Scholar]
- Leone, S; Izzo, V; Lanzetta, R; Molinaro, A; Parrilli, M; Di Donato, A. The structure of the O-polysaccharide from Pseudomonas stutzeri OX1 containing two different 4-acylamido-4,6-dideoxy-residues, tomosamine and perosamine. Carbohydr Res 2005, 340, 651–656. [Google Scholar]
- Vinogradov, E; MacLean, LL; Crump, EM; Perry, MB; Kay, WW. Structure of the polysaccharide chain of the lipopolysaccharide from Flexibacter maritimus. Eur J Biochem 2003, 270, 1810–1815. [Google Scholar]
- Bernadet, JF; Segers, P; Vancanneyt, M; Berthe, F; Kersters, K; Vandamme, P. Cutting the Gordian knot: emended classification and description of the genus Flavobacterium, emended description of the family Flavobacteriaceae, and proposal of Flavobacterium hydatis nom. nov, (Basonym, Cytophaga aquatalis Strohl and Tait (1978). Int J Syst Bacteriol 1996, 46, 128–148. [Google Scholar]
- Handlinger, J; Soltani, M; Percival, S. The pathology of Flexibacter maritimus in aquaculture species in Tasmania, Australia. J Fish Dis 1997, 20, 159–169. [Google Scholar]
- MacLean, LL; Vinogradov, E; Crump, EM; Perry, MB; Kay, WW. The structure of the lipopolysaccharide O-antigen produced by Flavobacterium psychrophilum. Eur J Biochem 2001, 268, 1–8. [Google Scholar]
- Perepelov, AV; Shashkov, AS; Tomshich, SV; Komandrova, NA; Nedashkovskaya, OI. A pseudoaminic acid-containing O-specific polysaccharide from a marine bacterium Cellulophaga fucicola. Carbohydr Res 2007, 342, 1378–1381. [Google Scholar]
- Silipo, A; Leone, S; Lanzetta, R; Parrilli, M; Sturiale, L; Garozzo, D; Nazarenko, EL; Gorshkova, RP; Ivanova, EP; Gorshkova, NM; Molinaro, A. The complete structure of the lipooligosaccharide from the halophilic bacterium Pseudoalteromonas issachenkonii KMM 3549T. Carbohydr Res 2004, 339, 1985–1993. [Google Scholar]
- Ivanova, EP; Sawabe, T; Alexeeva, YA; Lysenko, AM; Gorshkova, NM; Hayashi, K; Zhukova, NV; Christen, R; Mikhailov, VV. Pseudoalteromonas issachenkonii sp. nov., a bacterium that degrades the thallus of the brown alga Fucus evanescens. Int J Syst Evol Microbiol 2002, 52, 229–232. [Google Scholar]
- Corsaro, MM; Lanzetta, R; Parrilli, E; Parrilli, M; Tutino, ML. Structural investigation on the lipooligosaccharide fraction of psychrophylic Pseudoalteromonas haloplanktis TAC 125 bacterium. Eur J Biochem 2001, 268, 5092–5097. [Google Scholar]
- Vinogradov, EV; Bock, K. A new type of glycosidic linkage: an open-chain acetal-linked N-acetylgalactosamine in the core part of the lipopolysaccharides from Proteus microorganisms. Angew Chem Int Ed 1999, 38, 671–674. [Google Scholar]
- Vinogradov, E; Korenevsky, A; Beveridge, TJ. The structure of the carbohydrate backbone of the LPS from Shewanella putrefaciens CN32. Carbohydr Res 2002, 337, 1285–1289. [Google Scholar]
- Vinogradov, E; Bock, K; Petersen, BO; Holst, O; Brade, H. The structure of the carbohydrate backbone of the lipopolysaccharide from Acinetobacter strain ATCC 17905. Eur J Biochem 1997, 243, 122–127. [Google Scholar]
- Baumann, L; Baumann, P; Mandel, M; Allen, RD. Taxonomy of aerobic marine eubacteria. J Bacteriol 1972, 110, 402–429. [Google Scholar]
- Gauthier, G; Gauthier, M; Christen, R. Phylogenetic analysis of the genera Alteromonas, Shewanella, and Moritella using genes coding for small-subunit rRNA sequences and division of the genus Alteromonas into two genera, Alteromonas (emended) and Pseudoalteromonas gen. nov., and proposal of twelve new species combinations. Int J Syst Bacteriol 1995, 45, 755–761. [Google Scholar]
- Liparoti, V; Molinaro, A; Sturiale, L; Garozzo, D; Nazarenko, EL; Gorshkova, RP; Ivanova, EP; Shevcenko, LS; Lanzetta, R; Parrilli, M. Structural Analysis of the Deep Rough Lipopolysaccharide from Gram Negative Bacterium Alteromonas macleodii ATCC 27126T: The First Finding of β-Kdo in the Inner Core of Lipopolysaccharides. Eur J Org Chem 2006, 4710–4716. [Google Scholar]
- Fraysse, N; Lindner, B; Kaczynski, Z; Sharypova, L; Holst, O; Niehaus, K; Poinsot, V. Sinorhizobium meliloti strain 1021 produces a low-molecular-mass capsular polysaccharide that is a homopolymer of 3-deoxy-d-manno-oct-2-ulosonic acid harboring a phospholipid anchor. Glycobiology 2005, 15, 101–110. [Google Scholar]
- Vinogradov, E; Petersen, BO; Duus, JO; Radziejewska-Lebrecht, J. The structure of the polysaccharide part of the LPS from Serratia marcescens serotype O19, including linkage region to the core and the residue at the non-reducing end. Carbohydr Res 2003, 338, 2757–2761. [Google Scholar]
- Ivanova, EP; Bowman, JP; Lysenko, AM; Zhukova, NV; Gorshkova, NM; Sergeev, AF; Mikhailov, VV. Alteromonas addita sp. nov. Int J Syst Evol Microbiol 2005, 55, 1065–1068. [Google Scholar]
- Ivanova, EP; Nedashkovskaya, OI; Chun, J; Lysenko, AM; Frolova, GM; Svetashev, VI; Vysotskii, MV; Mikhailov, VV; Huq, A; Colwell, RR. Arenibacter gen. nov., new genus of the family Flavobacteriaceae and description of a new species, Arenibacter latericius sp. nov. Int J Syst Evol Microbiol 2001, 51, 1987–1995. [Google Scholar]
- Zähringer, U; Lindner, B; Rietschel, ET. Molecular structure of lipid A, the endotoxic center of bacterial lipopolysaccharides. Adv Carbohydr Chem Biochem 1994, 50, 211–276. [Google Scholar]
- Plotz, BM; Lindner, B; Stetter, KO; Holst, O. Characterization of a novel lipid A containing d-galacturonic acid that replaces phosphate residues. The structure of the lipid a of the lipopolysaccharide from the hyperthermophilic bacterium Aquifex pyrophilus. J Biol Chem 2000, 275, 11222–11228. [Google Scholar]
- Alexander, C. and Rietschel ET. Bacterial lipopolysaccharides and innate immunity. J Endotox Res 2001, 7, 167–202. [Google Scholar]
- Silipo, A; Lanzetta, R; Amoresano, A; Parrilli, M; Molinaro, A. Ammonium hydroxide hydrolysis: a valuable support in the MALDI-TOF mass spectrometry analysis of Lipid A fatty acid distribution. J Lipid Res 2002, 43, 2188–2195. [Google Scholar]
- Sforza, S; Silipo, A; Molinaro, A; Marchelli, R; Parrilli, M; Lanzetta, R. Determination of fatty acid positions in native lipid A by positive and negative electrospray ionization mass spectrometry. J Mass Spec 2004, 39, 378–383. [Google Scholar]
- Gibson, BW; Engstrom, JJ; Hines, CMW; Falick, AM. Characterization of bacterial lipooligosaccharides by delayed extraction matrix-assisted laser desorption ionization time-of-flight mass spectrometry. J Am Soc Mass Spectrom 1997, 8, 645–658. [Google Scholar]
- Domon, B; Costello, CE. Structure elucidation of glycosphingolipids and gangliosides using high-performance tandem mass spectrometry. Glycoconj J 1988, 5, 397–409. [Google Scholar]
- Krasikova, IN; Kapustina, NV; Isakov, VV; Gorshkova, NM; Solov’eva, TF. Elucidation of structure of lipid A from the marine Gram-negative bacterium Pseudoalteromonas haloplanktis ATCC 14393T. Russ J Bioorgan Chem 2003, 30, 367–373. [Google Scholar]
- Corsaro, MM; Piaz, FD; Lanzetta, R; Parrilli, M. Lipid A structure of Pseudoalteromonas haloplanktis TAC 125: use of electrospray ionization tandem mass spectrometry for the determination of fatty acid distribution. J Mass Spectrom 2002, 37, 481–488. [Google Scholar]
- Krasikova, IN; Kapustina, NV; Isakov, VV; Dmitrenok, AS; Dmitrenok, PS; Gorshkova, NM; Solov'eva, TF. Detailed structure of lipid A isolated from lipopolysaccharide from the marine proteobacterium Marinomonas vaga ATCC 27119T. Eur J Biochem 2004, 271, 2895–904. [Google Scholar]
- Vorobeva, EV; Krasikova, IN; Dmitrenok, AS; Isakov, VV; Nedashkovskaya, OI; Solov’eva, TF. An Unusual Lipid A from a Marine Bacterium Chryseobacterium scophtalmum CIP 104199T. Russ J Bioorg Chem 2006, 32, 485–491. [Google Scholar]
- Nishijima, M; Raetz, CR. Membrane lipid biogenesis in Escherichia coli: identification of genetic loci for phosphatidylglycerophosphate synthetase and construction of mutants lacking phosphatidylglycerol. J Biol Chem 1979, 254, 7837–7844. [Google Scholar]
- Nishijima, M; Bulawa, CE; Raetz, CR. Two interacting mutations causing temperature-sensitive phosphatidylglycerol synthesis in Escherichia coli membranes. J Bacteriol 1981, 145, 113–121. [Google Scholar]
- Nishijima, M; Raetz, CR. Characterization of two membrane-associated glycolipids from an Escherichia coli mutant deficient in phosphatidylglycerol. J Biol Chem 1981, 256, 10690–10696. [Google Scholar]
- Vorob’eva, EV; Krasikova, IN; Dmitrenok, AS; Dmitrenok, PS; Drozdov, AL; Reunov, AV; Lapshina, LA; Solov’eva, TF. Some Peculiarities of the Outer Membrane Composition of Marine Gram-Negative Bacterium Chryseobacterium indoltheticum CIP 103168T. Biochemistry (Moscow) Suppl Ser A: Membr Cell Biol 2007, 1, 114–121. [Google Scholar]
- Krasikova, IN; Kapustina, NV; Svetashev, VI; Gorshkova, RP; Tomshich, SV; Nazarenko, EL; Komandrova, NA; Ivanova, EP; Gorshkova, NM; Romanenko, LA; Mikhailov, VV; Solov'eva, TF. Chemical characterization of lipid A from some marine proteobacteria. Biochemistry (Moscow) 2001, 66, 1047–1054. [Google Scholar]
- Hart, DJ; Vreeland, RH. Changes in the Hydrophilic-hydrophobic cell surface character of Halomonas elongata in response to NaCl. J Bacteriol 1988, 37, 345–350. [Google Scholar]
- Chintalapati, S; Kiran, MD; Shivaji, S. Role of membrane lipid fatty acids in cold adaptation. Cell Mol Biol 2004, 50, 631–642. [Google Scholar]
- Baker, PJ; Hraba, T; Taylor, CE; Stashak, PW; Fauntleroy, MB; Takayama, K; Sievert, TR; Hronowski, XP; Cotter, RJ; Perez-perez, G. Molecular structures that influence the immunomodulatory properties of the lipid A and inner-core region oligosaccharides of bacterial lipopolysaccharides. Infect Immun 1994, 62, 2257–2269. [Google Scholar]
- Jarvis, BW; Lichenstein, H; Qureshi, N. Diphosphoryl lipid A from Rhodobacter sphaeroides inhibits complexes that form in vitro between lipopolysaccharide (LPS)-binding protein, soluble CD14, and spectrally pure LPS. Infect Immun 1997, 65, 3011–3016. [Google Scholar]
- Kawata, T; Bristol, JR; Rossignol, DP; Rose, JR; Kobayashi, S; Yokohama, H; Ishibashi, A; Christ, WJ; Katayama, K; Yamatsu, I; Kishi, Y. E5531, a synthetic non-toxic lipid A derivative blocks the immunobiological activities of lipopolysaccharide. Br J Pharmacol 1999, 127, 853–862. [Google Scholar]
Table 1. Acylation pattern of the main lipid A species from Gram-negative marine bacteria. Bacterium Nature and linkage of acyl substituents bound to GlcN IIa GlcN I Ref. 3’ 2’ 3 2 P. haloplanktis ATCC 14393T 10:0(3-OH)b 12:0(3-O-12:0)c 10:0(3-OH)b 12:0(3-OH)c 97 P. haloplanktis TAC 125 12:0(3-OH) 12:0(3-O-12:0) 12:0(3-OH) 12:0(3-OH) 98 P. issachenkonii KMM 3549T 10:0(3-OH)d 12:0(3-O-12:0) 12:0(3-OH) 77 P. carrageenovora IAM 12662T 10:0(3-OH)d 12:0(3-O-12:0) 12:0(3-OH) - A. macleodii ATCC 27126T 12:0(3-OH)d 12:0(3-O-12:0)c 12:0(3-OH) 85 A. addita KMM 3600T 12:0(3-OH) 14:0(3-O-12:0) 10:0(3-OH) 14:0(3-OH) 14 S. pacifica KMM 3772 13:0(3-O-13:0) 13:0(3-O-13:0) 13:0(3-OH) 13:0(3-OH) 12 Marinomonas vaga ATCC 27119T 10:0[3-O-10:0(3-OH)] 10:0(3-OH) 10:0(3-O-10:0) 99 A. certesii KMM 3941T 15:0(3-OH)c 15:0(3-O-15:0)c 15:0(3-OH) 15:0(3-OH) - C. scophtalmum CIP 104199T iso-15:0(3-OH) iso-17:0(3-OH) 100 aDAG in Arenibacter certesii KMM 3941Tbalternative substitution with 11:0(3-OH); 12:0(3-OH); iso-11:0(3-OH) or iso-12:0(3-OH).cinterchangeabledalternative substitution at position C3Share and Cite
MDPI and ACS StyleLeone, S.; Silipo, A.; Nazarenko, E.L.; Lanzetta, R.; Parrilli, M.; Molinaro, A. Molecular Structure of Endotoxins from Gram-negative Marine Bacteria: An Update. Mar. Drugs 2007, 5, 85-112. https://doi.org/10.3390/md503085
AMA Style
Leone S, Silipo A, Nazarenko EL, Lanzetta R, Parrilli M, Molinaro A. Molecular Structure of Endotoxins from Gram-negative Marine Bacteria: An Update. Marine Drugs. 2007; 5(3):85-112. https://doi.org/10.3390/md503085
Chicago/Turabian Style
Leone, Serena, Alba Silipo, Evgeny L. Nazarenko, Rosa Lanzetta, Michelangelo Parrilli, and Antonio Molinaro. 2007. "Molecular Structure of Endotoxins from Gram-negative Marine Bacteria: An Update" Marine Drugs 5, no. 3: 85-112. https://doi.org/10.3390/md503085
Article Metrics