Detection of Tetrodotoxin Shellfish Poisoning (TSP) Toxins and Causative Factors in Bivalve Molluscs from the UK
Abstract
:1. Introduction
2. Results
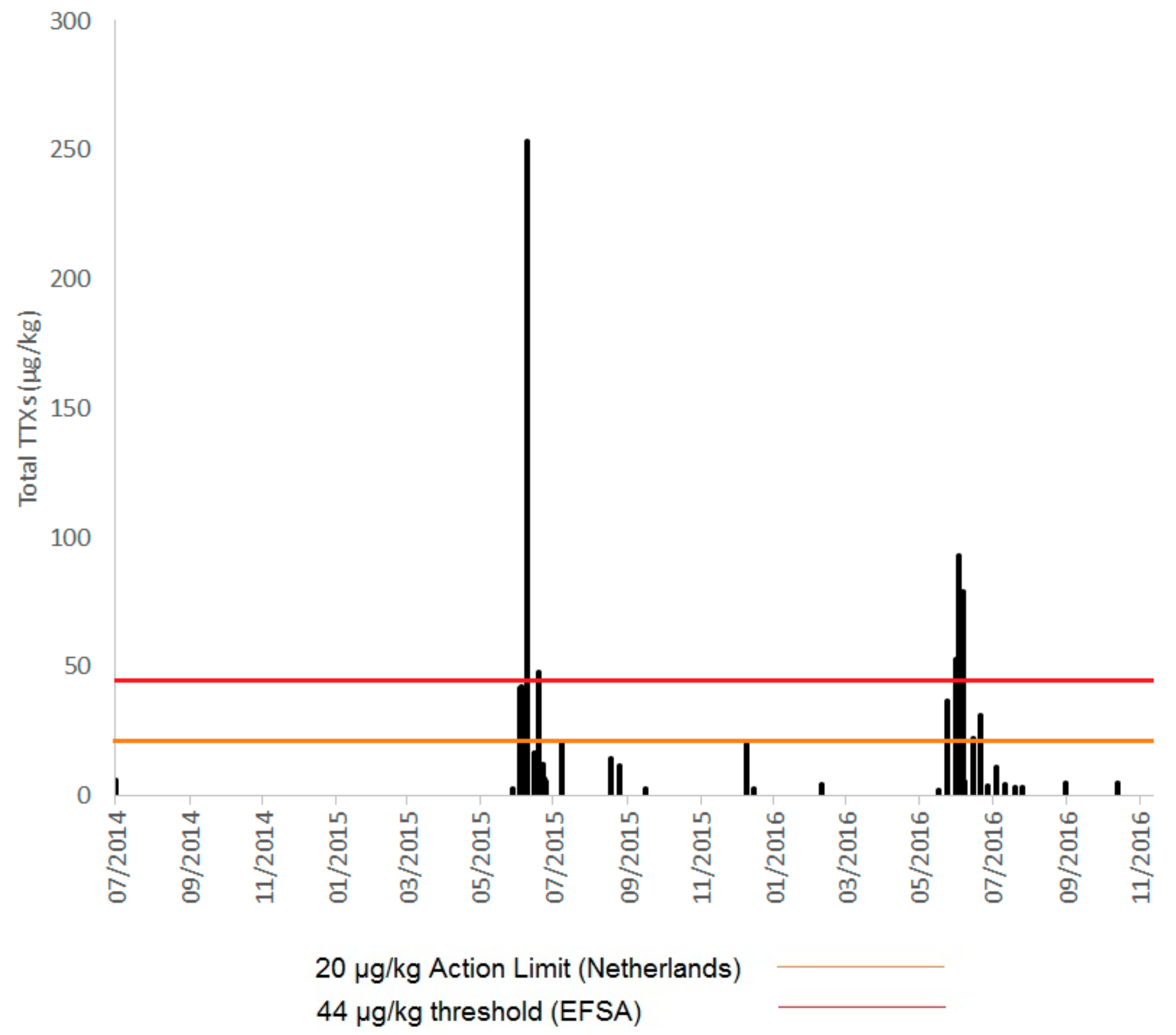
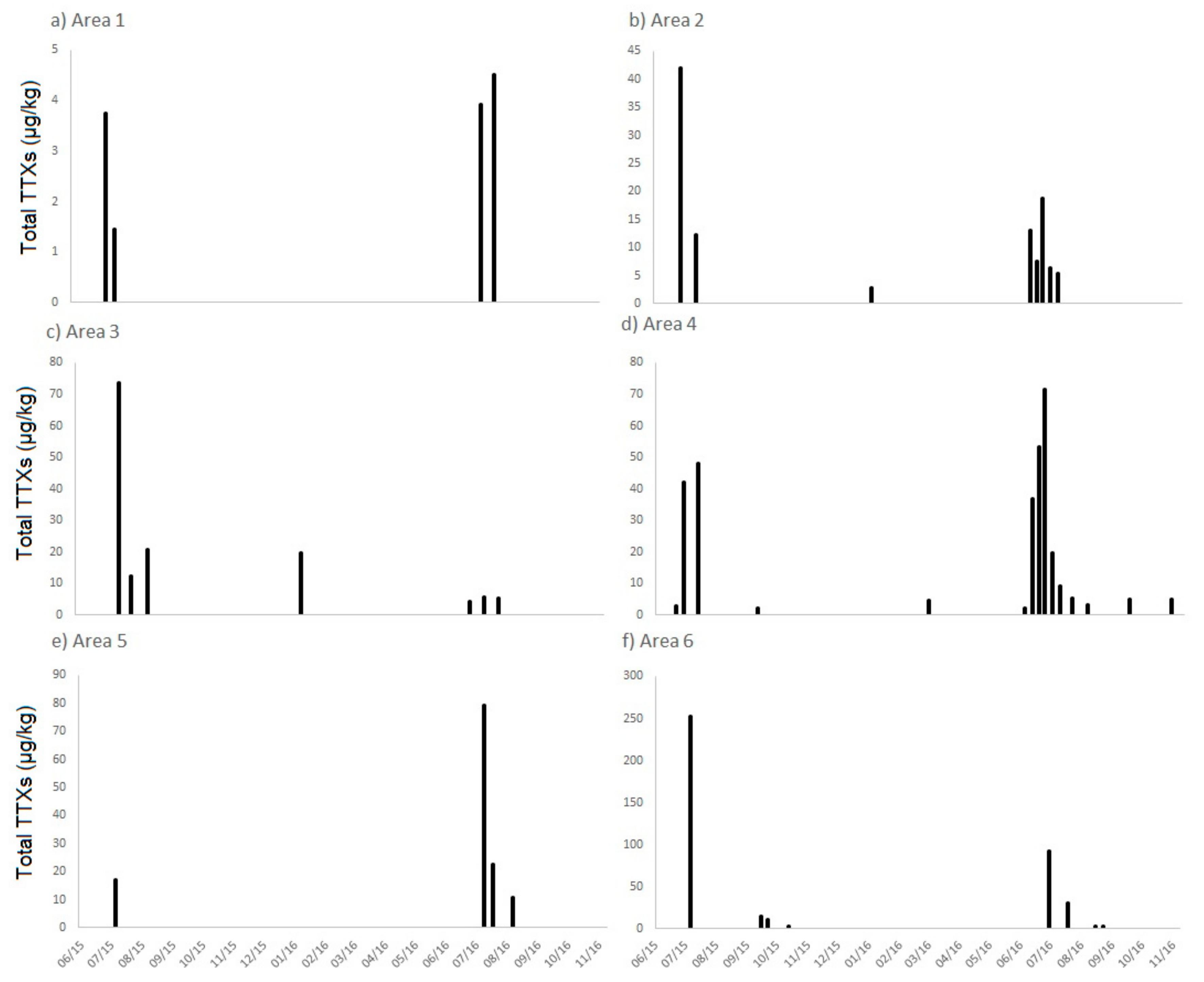
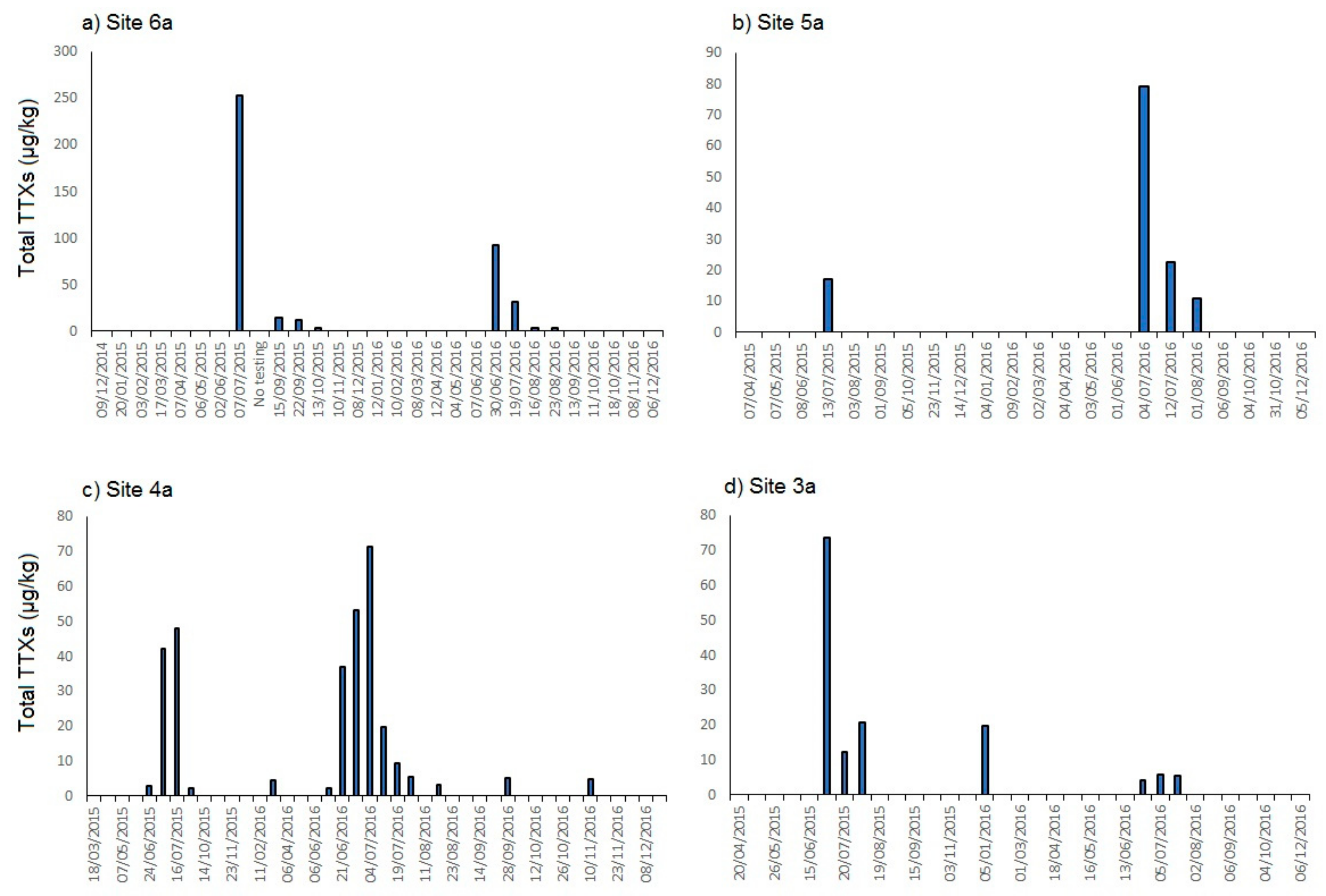
2.1. TTXs in UK Shellfish
2.1.1. England
2.1.2. Scotland
2.1.3. Wales
2.1.4. Northern Ireland
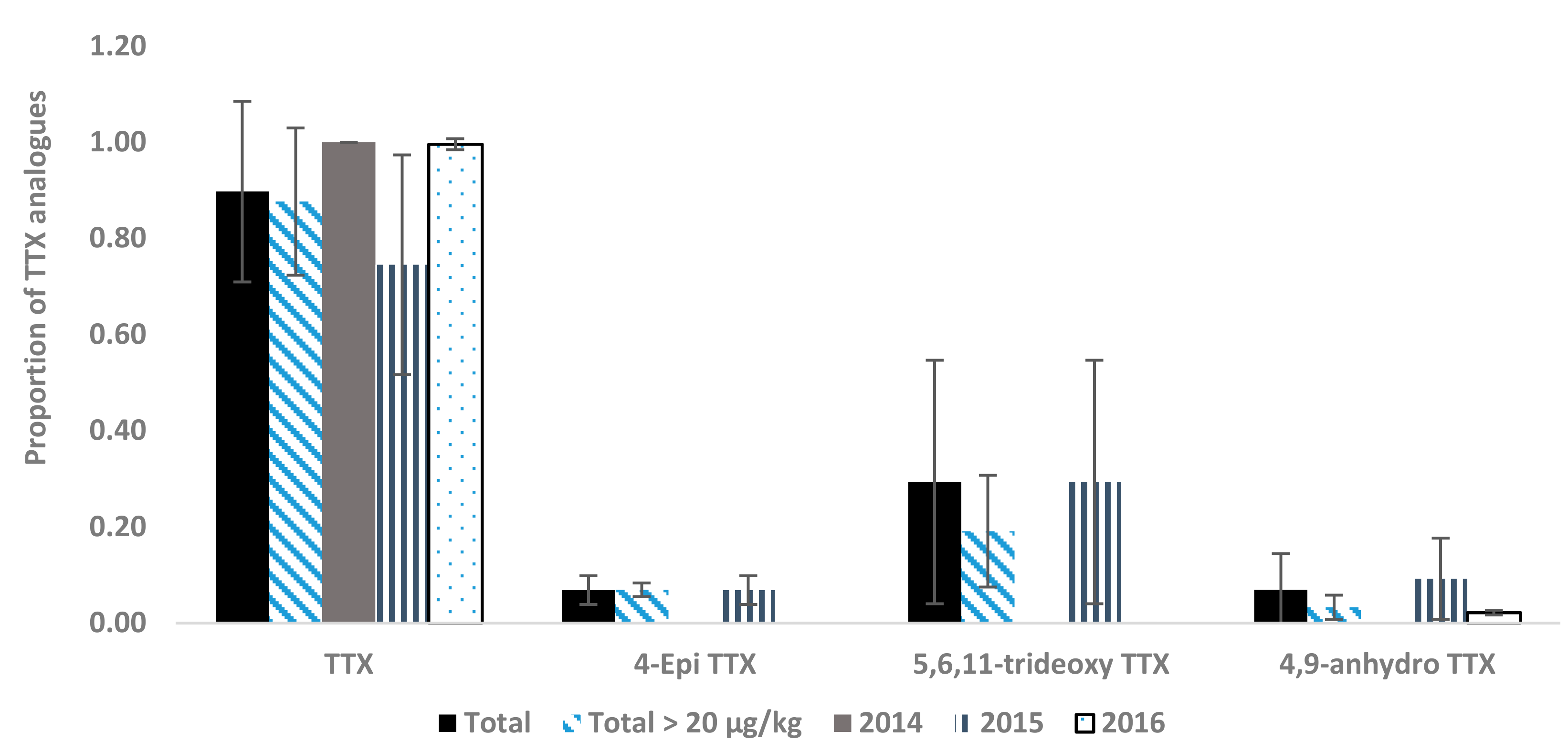
2.2. Toxin Profiles
2.3. Links to Environmental Conditions
2.3.1. Water Temperatures
2.3.2. Water Depth and Salinity
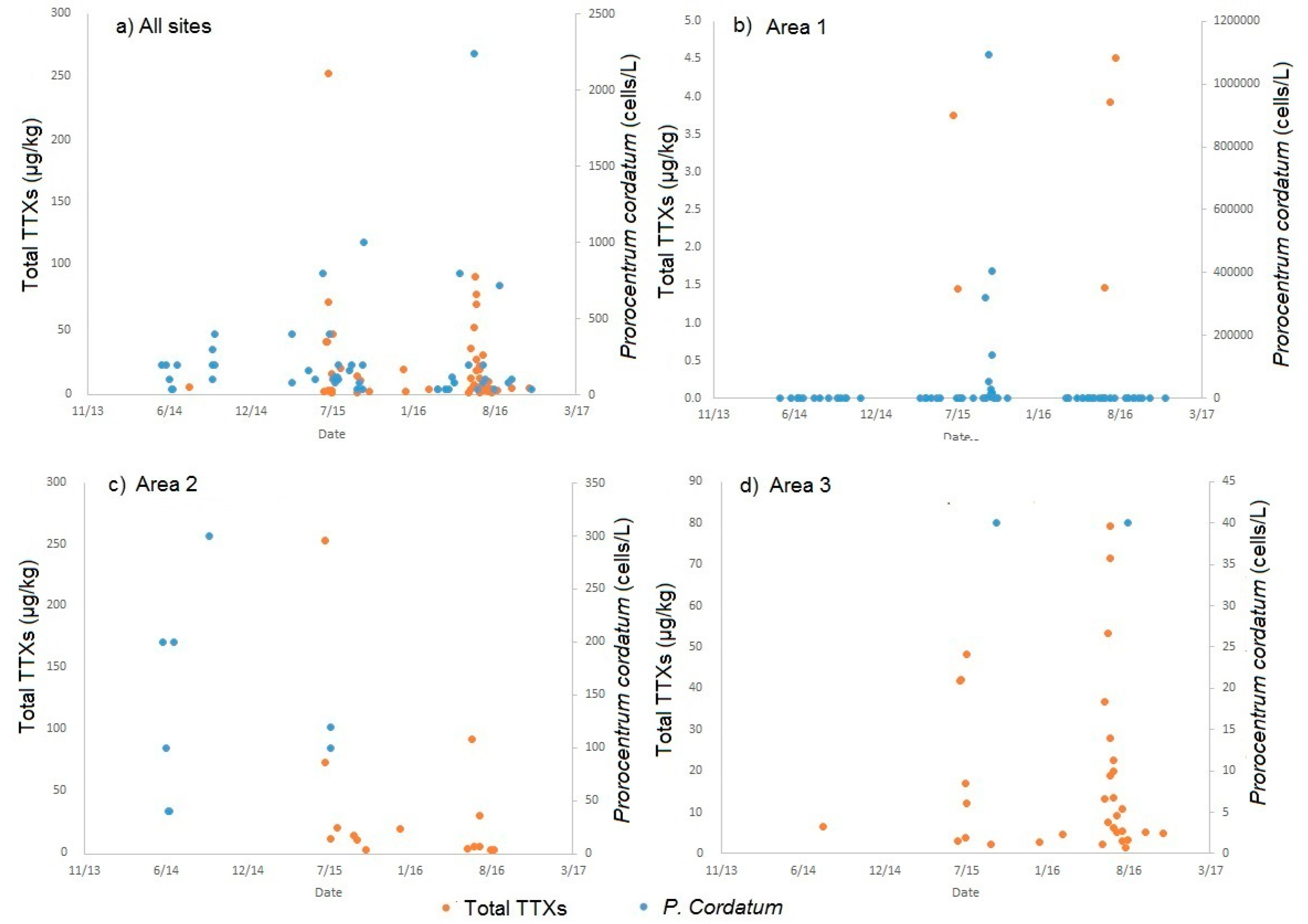
2.4. Links to Prorocentrum Cordatum
3. Discussion
3.1. Presence of TTXs in UK Shellfish
3.2. Potential Causative Factors
4. Materials and Methods
4.1. Reagents and Chemicals
4.2. Shellfish Samples
- Year 2014 samples: 603 OC samples and 13 scallop verification samples: Scotland 476 (6 scallop), England 120 (7 scallop), and Wales 20.
- Year 2015 samples: 408 OC samples and 48 scallop verification samples: Scotland 194 (4 scallop), England 254 (44 scallop), Wales 8.
- Year 2016 samples: 57 OC samples from Northern Ireland and 154 OC samples from selected sites in Southern England.
4.3. Sample Preparation and Toxin Analysis
4.4. Phytoplankton Collection and Analysis
4.5. Water Temperature Monitoring
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Tsuda, K.; Ikuma, S.; Kawamura, R.; Tachikawa, K.; Sakai, C.; Tamura, C.; Amakasu, D. Tetrodotoxin. VII. On the structures of Tetrodotoxin and its derivatives. Chem. Pharm. Bull. 1964, 12, 1357–1374. [Google Scholar] [CrossRef] [PubMed]
- Isbister, G.K.; Kiernan, M.C. Neurotoxic marine poisoning. Lancet Neurol. 2005, 4, 219–228. [Google Scholar] [CrossRef]
- Alcaraz, A.; Whiplle, R.E.; Gregg, H.R.; Anderson, B.D.; Grant, P.M. Analysis of Tetrodotoxin. Forensic Sci. Int. 1999, 99, 35–45. [Google Scholar] [CrossRef]
- Noguchi, T.; Miyazawa, K.; Daigo, K.; Arakawa, O. Paralytic shellfish poisoning (PSP toxin—And/or Tetrodotoxin-contaminated crabs and food poisoning by them. Toxin Rev. 2011, 30, 91–102. [Google Scholar] [CrossRef]
- Magarlamov, T.Y.; Melnikova, D.I.; Chernyshev, A.V. Tetrodotoxin-producing bacteria: Detection, distribution and migration of the toxin in aquatic systems. Toxins 2017, 9, 166. [Google Scholar] [CrossRef] [PubMed]
- Kodama, M.; Sato, S.; Sakamoto, S.; Ogata, T. Occurrence of Tetrodotoxin in Alexandirum tamarense, a causative dinoflagellate of paralytic shellfish poisoning. Toxicon 1996, 34, 1101–1105. [Google Scholar] [CrossRef]
- Vlamis, A.; Katikou, P.; Rodriguez, I.; Rey, V.; Alfonso, A.; Papazacharious, A.; Zacharaki, T.; Botana, A.M.; Botana, L.M. First detection of Tetrodotoxin in Greek Shellfish by UPLC-MS/MS potentially linked to the presence of the dinoflagellate Prorocentrum minimum. Toxins 2015, 7, 1779–1807. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, P.; Alfonso, A.; Alonso, E.; Rubiolo, J.A.; Roel, M.; Vlamis, A.; Katikou, P.; Jackson, S.A.; Lekha Menon, M.; Dobson, A.; et al. The association of bacterial C9-based TTX-like compounds with Prorocentrum minimum opens new uncertainties about shellfish seafood safety. Sci. Rep. 2017, 7, 40880. [Google Scholar] [CrossRef] [PubMed]
- Yasumoto, T.; Hashimoto, Y.; Bagnis, R.; Randall, J.E.; Banner, A.H. Toxicity of the Surgeon fishes. Bull. Jpn. Soc. Sci. Fish. 1971, 37, 724–734. [Google Scholar] [CrossRef]
- Yasumoto, T.; Endo, A.; Yasumura, D.; Nagai, H.; Murata, M.; Yotsu, M. Bacterial production of Tetrodotoxin and its derivatives. Toxicon 1988, 26, 50. [Google Scholar]
- Chau, R.; Kalaitzis, J.A.; Neilan, B.A. On the origin and biosynthesis of Tetrodotoxin. Aquat. Toxicol. 2011, 104, 61–72. [Google Scholar] [CrossRef] [PubMed]
- Hwang, P.-A.; Tsai, Y.-H.; Lin, H.-P.; Hwang, D.-F. Tetrodotoxin-binding proteins isolated from five species of toxic gastropods. Food Chem. 2007, 103, 1153–1158. [Google Scholar] [CrossRef]
- Arakawa, O.; Hwang, D.-F.; Taniyama, S.; Takatani, T. Toxins of pufferfish that cause human intoxications. In Coastal Environmental and Ecosystem Issues of the East China Sea; Ishimatsu, A., Lie, H.J., Eds.; TERRAPUB: Nagasaki, Japan, 2010; pp. 227–244. [Google Scholar]
- Miyazawa, K.; Higashiyama, M.; Hori, K.; Noguchi, T.; Ito, K.; Hashimoto, K. Distribution of tetrodotoxin in various organs of the starfish Astopecten polyacanthus. Mar. Biol. 1987, 96, 385–390. [Google Scholar] [CrossRef]
- Rodriguez, P.; Alfonso, A.; Vale, C.; Alfonso, C.; Vale, P.; Tellez, A.; Botana, L.M. First toxicity report of tetrodotoxin and 5,6,11-TrideoxyTTX in the Trumpet Shell Charonia lampas lampas in Europe. Anal. Chem. 2008, 80, 5622–5629. [Google Scholar] [CrossRef] [PubMed]
- Turner, A.D.; Powell, A.; Schofield, A.; Lees, D.N.; Baker-Austin, C. Detection of the pufferfish toxin Tetrodotoxin in European bivalves, England, 2013 to 2014. Eurosurveillance 2015, 20, 2–8. [Google Scholar] [CrossRef]
- Gerssen, A.; (Institute of Food Safety of Wageningen University (RIKILT), Wageningen, The Netherlands). Personal communication, 2017.
- Turner, A.D.; Boundy, M.J.; Dhanji-Rapkova, M. Development and single-laboratory validation of a liquid chromatography tandem mass spectrometry method for quantitation of Tetrodotoxin in mussels and oysters. J. AOAC Int. 2017, 100, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Turner, A.D.; Higgins, C.; Higman, W.; Hungerford, J. Potential threats posed by tetrodotoxins in UK waters: Examination of detection methodology used in their control. Mar. Drugs 2015, 13, 7357–7376. [Google Scholar] [CrossRef] [PubMed]
- EFSA. Scientific opinion on the risks for public health related to the presence of tetrodotoxin (TTX) and TTX analogues in marine bivalves and gastropods. EFSA J. 2017, 15, 65. [Google Scholar] [CrossRef]
- Silva, M.; Azavedo, J.; Rodriguez, P.; Alfonso, A.; Botana, L.M.; Vasconcelos, V. New gastropod vectors and tetrodotoxin potential expansion in temperate waters of the Atlantic Ocean. Mar. Drugs. 2012, 10, 712–726. [Google Scholar] [CrossRef] [PubMed]
- Nzoughet, J.K.; Campbell, K.; Barnes, P.; Cooper, K.M.; Chevallier, O.P.; Elliott, C.T. Comparison of sample preparation methods, validation of an UPLC–MS/MS procedure for the quantification of tetrodotoxin present in marine gastropods and analysis of pufferfish. Food Chem. 2013, 136, 1584–1589. [Google Scholar] [CrossRef] [PubMed]
- Turner, A.D.; Stubbs, B.; Coates, L.; Dhanji-Rapkova, M.; Hatfield, R.G.; Lewis, R.G.; Rowland-Pilgrim, S.; O’Neill, A.; Stubbs, P.; Ross, S.; et al. Variability of paralytic shellfish toxin occurrence in bivalve molluscs from Great Britain from official control monitoring as determined by pre-column oxidation liquid chromatography and implications for applying immunochemical tests. Harmful Algae 2014, 31, 87–99. [Google Scholar] [CrossRef] [PubMed]
- Lowe, J.A.; Howard, T.; Pardaens, A.; Tinker, J.; Jenkins, G.; Ridley, J.; Leake, J.; Holt, J.; Wakelin, S.; Wolf, J.; et al. UK Climate Projections Science Report: Marine and Coastal Projections; DEFRA (Department of Environment Fisheries and Rural Affairs): London, UK, 2009. [Google Scholar]
- Baker-Austin, C.; Trianes, J.A.; Salmenlinna, S.; Lofdahl, M.; Siitonen, A.; Taylorm, N.G.H.; Martinez-Urtaza, J. Heat wave-associated Vibriosis, Sweden and Finland, 2014. Emerg. Infect. Dis. 2016, 22, 1216–1220. [Google Scholar] [CrossRef] [PubMed]
- Noguchi, T.; Jeon, J.K.; Arakawa, O.; Sugita, H.; Deguchi, Y.; Shida, Y.; Hashimoto, K. Occurrence of tetrodotoxin and anhydrotetrodotoxin in Vibrio sp. isolated from the intestines of a xanthid crab, Atergatis floridus. J. Biochem. 1986, 99, 311–314. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Xie, L.; Xia, G.; Zhang, J.; Nie, Y.; Hu, J.; Wang, S.; Zhang, R. A new tetrodotoxin-producing actinomycete, Norcardiopsis dassonvillei, isolated from the ovaries of puffer fish Fugu rubripes. Toxicon 2005, 45, 851–859. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.-J.; Yu, R.-C.; Luo, X.; Zhou, M.-J.; Lin, X.-T. Toxin-screening and identification of bacteria from highly toxic marine gastropod Nassarius semiplicatus. Toxicon 2008, 52, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Salvitti, L.; Wood, S.A.; Taylor, D.I.; McNabb, P.; Cary, S.C. First identification of tetrodotoxin in the flatworm Stylochoplana sp.; a source of TTX for the sea slug Pleurobranchaea maculate. Toxicon 2015, 95, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Powell, A.; Baker-Austin, C.; Wagley, S.; Bayley, A.; Hartnell, R. Isolation of Pandemic Vibrio parahaemolyticus from UK Water and Shellfish Produce. Microb. Ecol. 2013, 65, 924–927. [Google Scholar] [CrossRef] [PubMed]
- Tirado, M.C.; Clarke, R.; Jaykus, L.A.; McQuatters-Gollop, A.; Frank, J.M. Climate change and food safety: A review. Food Res. Int. 2010, 43, 1745–1765. [Google Scholar] [CrossRef]
- Pfeffer, C.S.; Hite, M.F.; Oliver, J.D. Ecology of Vibrio vulnificus in estuarine waters of eastern North Carolina. Appl. Environ. Microbiol. 2003, 69, 3526–3531. [Google Scholar] [CrossRef] [PubMed]
- Baker-Austin, C.; Trinanes, J.A.; Taylor, N.G.H.; Hartnell, R.; Siitonene, A.; Martinez-Urtaza, J. Ermerging Vibrio risk at high latitutes in response to ocean warming. Nat. Clim. Chang. 2012. [Google Scholar] [CrossRef]
- Vezzulli, L.; Colwell, R.R.; Pruzzo, C. Ocean warming and spread of pathogenic Vibrios in the aquatic environment. Microb. Ecol. 2013, 65, 817–825. [Google Scholar] [CrossRef] [PubMed]
- Vezzulli, L.; Brettar, I.; Pezzati, E.; Reid, P.C.; Colwell, R.R.; Hofle, M.G.; Pruzzo, C. Long-term effects of ocean warming on the prokaryotic community: Evidence from the vibrios. ISME J. 2012, 6, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Lima, F.P.; Wethey, D.S. Three decades of high-resolution coastal sea surface temperatures reveal more than warming. Nat. Commun. 2012, 3, 704–713. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, G.J.; Perry, M.C.; Prior, M.J. The Climate of the United Kingdom and Recent Trends; Report from Met Office Hadley Centre: Exeter, UK, 2008. Available online: http://ukclimateprojections.metoffice.gov.uk/media.jsp?mediaid=87933&filetype=pdf (accessed on 10 August 2017).
- Miraglia, M.; Marvin, H.J.P.; Kleter, G.A.; Battilani, P.; Brera, C.; Coni, E.; Cubadda, F.; Croci, L.; De Santis, B.; Dekkers, S.; et al. Climate change and food safety: An emerging issue with special focus on Europe. Food Chem. Toxicol. 2009, 47, 1009–1021. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, J.; Moore, S.K.; Kunkel, K.E.; Sun, L. A framework for examining climate-driven changes to the seasonality and geographicl range of coastal pathogens and harmful algae. Clim. Risk Manag. 2015, 8, 16–27. [Google Scholar] [CrossRef]
- Wells, M.L.; Trainer, V.L.; Smayda, T.J.; Karlson, B.S.O.; Trick, C.G.; Kudela, R.M.; Ishikawa, A.; Bernard, S.; Wulff, A.; Anderson, D.M.; et al. Harmful algal blooms and climate change: Learning from the past and present to forecast the future. Harmful Algae 2015, 49, 68–93. [Google Scholar] [CrossRef] [PubMed]
- Botana, L. A toxicological perspective to climate change: Aquatic toxins. Chem. Res. Toxicol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Swan, S.; (Scottish Association for Marine Science, Oban, UK). Personal communication, 2017.
- Turner, A.D.; Algoet, M.; Hatfield, R.; Norton, D.; Morris, S.; Reese, A.; Lees, D.N. Single laboratory validation of the AOAC HPLC method (2005.06) for mussels: Refinement and extension of the method to additional toxins. J. AOAC Int. 2009, 92, 190–207. [Google Scholar] [PubMed]
- Boundy, M.J.; Selwood, A.I.; Harwood, D.T.; McNabb, P.S.; Turner, A.D. Development of a sensitive and selective liquid chromatography-mass spectrometry method for high throughput analysis of paralytic shellfish toxins using graphitized carbon solid phase extraction. J. Chromatogr. A 2015, 1387, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Turner, A.D.; McNabb, P.S.; Harwood, D.T.; Selwood, A.I.; Boundy, M.J. Single-laboratory validation of a multitoxin ultra-performance LC-hydrophilic interaction LC-MS/MS method for quantitation of Paralytic Shellfish Toxins in bivalve shellfish. J. AOAC Int. 2015, 98, 609–621. [Google Scholar] [CrossRef] [PubMed]
- Franks, P.J.S.; Anderson, D.M. Sampling coastal dinoflagellate blooms: Equipment, strategies and data processing. In Biology, Epidemiology and Management of Pyrodinium bahamense Red Tides, Proceedings of the Management and Training Workshop, Bandar Seri Begawan, Brunei, 23–30 May 1989; Hallegraeff, G., Maclean, J.L., Eds.; Fisheries Department, Ministry of Development: Bandar Seri Begawan, Brunei; Makati, Philippines, 1989; pp. 235–254. [Google Scholar]
- Karlson, B.; Cusack, C.; Bresnan, E. Microscopic and Molecular methods for quantitative phytoplankton analysis. In Manuals and Guides; International Oceanographic Commission, UNESCO: Paris, France, 2010; Volume 55. [Google Scholar]

| Country | Year | Samples Tested | Results > LOR 1 | Results > 20 µg/kg | Maximum Concentration µg/kg | Dates for Positives (>LOR) |
|---|---|---|---|---|---|---|
| England | 2014 | 113 | 1 (0.9%) | 0 | 6 | na |
| 2015 | 210 | 21 (10%) | 6 (3%) | 253 | July–August | |
| 2016 | 154 | 33 (21%) | 8 (6%) | 93 | January–July | |
| Wales | 2014 | 20 | 0 | 0 | na | na |
| 2015 | 8 | 0 | 0 | na | na | |
| 2016 | 0 | 0 | 0 | na | na | |
| Scotland | 2014 | 470 | 2 (0.4%) | 1 (0.2%) | 26 | July |
| 2015 | 190 | 0 | 0 | na | na | |
| 2016 | 0 | 0 | 0 | na | na | |
| N. Ireland | 2014 | 0 | 0 | 0 | na | na |
| 2015 | 0 | 0 | 0 | na | na | |
| 2016 | 57 | 0 | 0 | na | na |
| Temperature | 0–4 °C | 5–9 °C | 10–14 °C | 15–19 °C | 20–24 °C | |
|---|---|---|---|---|---|---|
| England | Total | 0 | 25 | 86 | 185 | 26 |
| >LOR | 0 | 3 (12%) | 2 (2%) | 39 (21%) | 3 (12%) | |
| >20 µg/kg | 0 | 0 | 0 | 12 (7%) | 2 (8%) | |
| Scotland and Wales | Total | 3 | 19 | 184 | 51 | 0 |
| >LOR | 0 | 0 | 0 | 0 | 0 | |
| >20 µg/kg | 0 | 0 | 0 | 0 | 0 |
| All Sites | 0–5 m | 5–20 m | >20 m | River | Estuary | Open |
|---|---|---|---|---|---|---|
| 55 | 51 (93%) | 4 (7%) | 0 | 6 (11%) | 45 (82%) | 4 (7%) |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Turner, A.D.; Dhanji-Rapkova, M.; Coates, L.; Bickerstaff, L.; Milligan, S.; O’Neill, A.; Faulkner, D.; McEneny, H.; Baker-Austin, C.; Lees, D.N.; et al. Detection of Tetrodotoxin Shellfish Poisoning (TSP) Toxins and Causative Factors in Bivalve Molluscs from the UK. Mar. Drugs 2017, 15, 277. https://doi.org/10.3390/md15090277
Turner AD, Dhanji-Rapkova M, Coates L, Bickerstaff L, Milligan S, O’Neill A, Faulkner D, McEneny H, Baker-Austin C, Lees DN, et al. Detection of Tetrodotoxin Shellfish Poisoning (TSP) Toxins and Causative Factors in Bivalve Molluscs from the UK. Marine Drugs. 2017; 15(9):277. https://doi.org/10.3390/md15090277
Chicago/Turabian StyleTurner, Andrew D., Monika Dhanji-Rapkova, Lewis Coates, Lesley Bickerstaff, Steve Milligan, Alison O’Neill, Dermot Faulkner, Hugh McEneny, Craig Baker-Austin, David N. Lees, and et al. 2017. "Detection of Tetrodotoxin Shellfish Poisoning (TSP) Toxins and Causative Factors in Bivalve Molluscs from the UK" Marine Drugs 15, no. 9: 277. https://doi.org/10.3390/md15090277





