Innovative Alternative Technologies to Extract Carotenoids from Microalgae and Seaweeds
Abstract
:1. Introduction
2. Extraction Technologies for Carotenoids
Conventional Extraction Methods
3. Nonconventional Extraction of Carotenoids
3.1. Electrotechnologies
3.1.1. Pulsed Electric Field (PEF)-Assisted Extraction
3.1.2. Moderate Electric Field (MEF)-Assisted Extraction
3.1.3. High Voltage Electric Discharges (HVED)-Assisted Extraction
3.2. Pressurized Liquid Extraction
3.3. Supercritical Fluid Extraction (SFE)
3.4. Subcritical Fluid Extraction
3.5. Microwave-Assisted Extraction
3.6. Ultrasound-Assisted Extraction
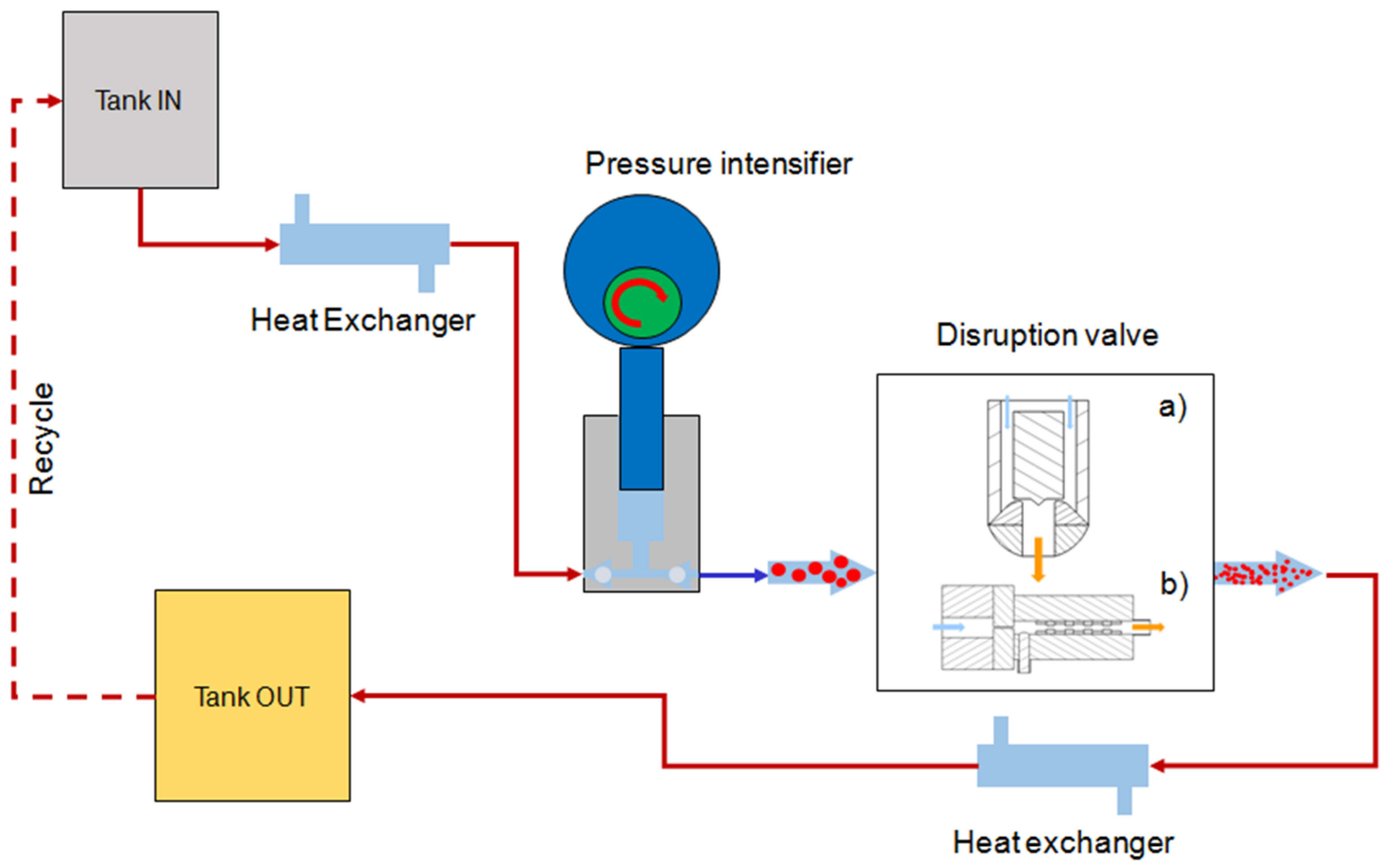
3.7. High Pressure Homogenization (HPH)
4. Conclusions and Future Perspectives
Acknowledgments
Conflicts of Interest
References
- Lorenz, R.T.; Cysewski, G.R. Commercial potential for Haematococcus microalgae as a natural source of astaxanthin. Trends Biotechnol. 2000, 18, 160–167. [Google Scholar] [CrossRef]
- Liaaen-Jensen, S. Basic carotenoid chemistry. In Carotenoids in Health and Disease; Krinsky, N.I., Mayne, S.T., Sies, H., Eds.; Marcel Dekker: New York, NY, USA, 2004; pp. 1–30. [Google Scholar]
- Ritz, T.; Damjanović, A.; Schulten, K. Light-harvesting and photoprotection by carotenoids: Structure-based calculations for photosynthetic antenna systems. In Photosynthesis: Mechanisms and Effects; Springer: Dordrecht, The Netherlands, 1998; pp. 487–490. [Google Scholar]
- Hammond, B.R.; Renzi, L.M. Carotenoids. Adv. Nutr. Int. Rev. J. 2013, 4, 474–476. [Google Scholar] [CrossRef] [PubMed]
- Krinsky, N.I.; Mayne, S.T.; Sies, H. Carotenoids in Health and Disease; Marcel Dekker: New York, NY, USA, 2004. [Google Scholar]
- Fiedor, J.; Burda, K. Potential role of carotenoids as antioxidants in human health and disease. Nutrients 2014, 6, 466–488. [Google Scholar] [CrossRef] [PubMed]
- Johnson, E.J. The role of carotenoids in human health. Nutr. Clin. Care 2002, 5, 56–65. [Google Scholar] [CrossRef] [PubMed]
- Vílchez, C.; Forján, E.; Cuaresma, M.; Bédmar, F.; Garbayo, I.; Vega, J.M. Marine carotenoids: Biological functions and commercial applications. Mar. Drugs 2011, 9, 319–333. [Google Scholar] [CrossRef] [PubMed]
- Guedes, A.C.; Amaro, H.M.; Malcata, F.X. Microalgae as Sources of Carotenoids. Mar. Drugs 2011, 9, 625–644. [Google Scholar] [CrossRef] [PubMed]
- MarketsandMarkets. Carotenoids Market by Type (Astaxanthin, Beta-Carotene, Canthaxanthin, Lutein, Lycopene, & Zeaxanthin), Source (Synthetic and Natural), Application (Supplements, Food, Feed, and Cosmetics), by Region—Global Trends and Forecasts to 2021. Available online: http://www.marketsandmarkets.com/Market-Reports/carotenoid-market-158421566.html (accessed on 24 September 2016).
- Rodriguez-Amaya, D.B. Food Carotenoids: Chemistry, Biology and Technology; Wiley-Blackwell: West Sussex, UK, 2016. [Google Scholar]
- Safafar, H.; Van Wagenen, J.; Møller, P.; Jacobsen, C. Carotenoids, phenolic compounds and tocopherols contribute to the antioxidative properties of some microalgae species grown on industrial wastewater. Mar. Drugs 2015, 13, 7339–7356. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grung, M.; D’Souza, F.M.L.; Borowitzka, M.; Liaaen-Jensen, S. Algal Carotenoids 51. Secondary carotenoids 2. Haematococcus pluvialis aplanospores as a source of (3S, 3′S)-astaxanthin esters. J. Appl. Phycol. 1992, 4, 165–171. [Google Scholar] [CrossRef]
- Minhas, A.K.; Hodgson, P.; Barrow, C.J.; Adholeya, A. A review on the assessment of stress conditions for simultaneous production of microalgal lipids and carotenoids. Front. Microbiol. 2016, 7, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Sun, Z.; Sun, P.; Chen, T.; Chen, F. Microalgal carotenoids: Beneficial effects and potential in human health. Food Funct. 2014, 5, 413–425. [Google Scholar] [CrossRef] [PubMed]
- Grama, B.S.; Delhaye, A.; Agathos, S.N.; Jeffryes, C. Industrial biotechnology of vitamins, biopigments, and antioxidants. In Industrial Biotechnology of Vitamins, Biopigments, and Antioxidants; Vandamme, E.J., Revuelta, J.L., Eds.; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2016; pp. 265–286. [Google Scholar]
- Boominathan, M.; Mahesh, A. Seaweed carotenoids for cancer therapeutics. In Handbook of Anticancer Drugs from Marine Origin; Kim, S.-K., Ed.; Springer: Cham, Switzerland, 2015; pp. 185–203. [Google Scholar]
- Billakanti, J.M.; Catchpole, O.J.; Fenton, T.A.; Mitchell, K.A.; MacKenzie, A.D. Enzyme-assisted extraction of fucoxanthin and lipids containing polyunsaturated fatty acids from Undaria pinnatifida using dimethyl ether and ethanol. Process Biochem. 2013, 48, 1999–2008. [Google Scholar] [CrossRef]
- Roh, M.K.; Uddin, M.S.; Chun, B.S. Extraction of fucoxanthin and polyphenol from Undaria pinnatifida using supercritical carbon dioxide with co-solvent. Biotechnol. Bioprocess Eng. 2008, 13, 724–729. [Google Scholar] [CrossRef]
- Quitain, A.T.; Kai, T.; Sasaki, M.; Goto, M. Supercritical carbon dioxide extraction of fucoxanthin from Undaria pinnatifida. J. Agric. Food Chem. 2013, 61, 5792–5797. [Google Scholar] [CrossRef] [PubMed]
- Xiao, X.; Si, X.; Yuan, Z.; Xu, X.; Li, G. Isolation of fucoxanthin from edible brown algae by microwave-assisted extraction coupled with high-speed countercurrent chromatography. J. Sep. Sci. 2012, 35, 2313–2317. [Google Scholar] [CrossRef] [PubMed]
- Piovan, A.; Seraglia, R.; Bresin, B.; Caniato, R.; Filippini, R. Fucoxanthin from Undaria pinnatifida: Photostability and coextractive effects. Molecules 2013, 18, 6298–6310. [Google Scholar] [CrossRef] [PubMed]
- Kanda, H.; Kamo, Y.; Machmudah, S.; Wahyudiono, E.Y.; Goto, M. Extraction of fucoxanthin from raw macroalgae excluding drying and cell wall disruption by liquefied dimethyl ether. Mar. Drugs 2014, 12, 2383–2396. [Google Scholar] [CrossRef] [PubMed]
- Yan, X.; Chuda, Y.; Suzuki, M.; Nagata, T. Fucoxanthin as the major antioxidant in Hijikia fusiformis, a common edible seaweed. Biosci. Biotechnol. Biochem. 1999, 63, 605–607. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.-J.; Wang, G.-C.; Zhang, M.; Tseng, C.K. Isolation of fucoxanthin from the rhizoid of Laminaria japonica Aresch. J. Integr. Plant Biol. 2005, 47, 1009–1015. [Google Scholar] [CrossRef]
- Saravana, P.S.; Getachew, A.T.; Cho, Y.-J.; Choi, J.H.; Park, Y.B.; Woo, H.C.; Chun, B.S. Influence of co-solvents on fucoxanthin and phlorotannin recovery from brown seaweed using supercritical CO2. J. Supercrit. Fluids 2016, in press. [Google Scholar] [CrossRef]
- Heo, S.-J.; Jeon, Y.-J. Protective effect of fucoxanthin isolated from Sargassum siliquastrum on UV-B induced cell damage. J. Photochem. Photobiol. B 2009, 95, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Noviendri, D.; Jaswir, I.; Salleh, H.M.; Taher, M.; Miyashita, K.; Ramli, N. Fucoxanthin extraction and fatty acid analysis of Sargassum binderi and S. duplicatum. J. Med. Plants Res. 2011, 5, 2405–2412. [Google Scholar]
- Conde, E.; Moure, A.; Domínguez, H. Supercritical CO2 extraction of fatty acids, phenolics and fucoxanthin from freeze-dried Sargassum muticum. J. Appl. Phycol. 2014, 2, 957–964. [Google Scholar] [CrossRef]
- Nomura, M.; Kamogawa, H.; Susanto, E.; Kawagoe, C.; Yasui, H.; Saga, N.; Hosokawa, M.; Miyashita, K. Seasonal variations of total lipids, fatty acid composition, and fucoxanthin contents of Sargassum horneri (Turner) and Cystoseira hakodatensis (Yendo) from the northern seashore of Japan. J. Appl. Phycol. 2013, 25, 1159–1169. [Google Scholar] [CrossRef]
- Afolayan, A.F.; Bolton, J.J.; Lategan, C.A.; Smith, P.J.; Beukes, D.R. Fucoxanthin, tetraprenylated toluquinone and toluhydroquinone metabolites from Sargassum heterophyllum inhibit the in vitro growth of the malaria parasite Plasmodium falciparum. Z. Naturforschung C J. Biosci. 2008, 63, 848–852. [Google Scholar] [CrossRef]
- Heo, S.-J.; Ko, S.-C.; Kang, S.-M.; Kang, H.-S.; Kim, J.-P.; Kim, S.-H.; Lee, K.-W.; Cho, M.-G.; Jeon, Y.-J. Cytoprotective effect of fucoxanthin isolated from brown algae Sargassum siliquastrum against H2O2-induced cell damage. Eur. Food Res. Technol. 2008, 228, 145–151. [Google Scholar] [CrossRef]
- Zaragozá, M.C.; López, D.; P Sáiz, M.; Poquet, M.; Pérez, J.; Puig-Parellada, P.; Màrmol, F.; Simonetti, P.; Gardana, C.; Lerat, Y.; et al. Toxicity and antioxidant activity in vitro and in vivo of two Fucus vesiculosus extracts. J. Agric. Food Chem. 2008, 56, 7773–7780. [Google Scholar] [CrossRef] [PubMed]
- Peng, J.; Yuan, J.-P.; Wu, C.-F.; Wang, J.-H. Fucoxanthin, a marine carotenoid present in brown seaweeds and diatoms: Metabolism and bioactivities relevant to human health. Mar. Drugs 2011, 9, 1806–1828. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.-K.; Pangestuti, R. Biological activities and potential health benefits of fucoxanthin derived from marine brown algae. Adv. Food Nutr. Res. 2011, 64, 111–128. [Google Scholar] [PubMed]
- Zhang, H.; Tang, Y.; Zhang, Y.; Zhang, S.; Qu, J.; Wang, X.; Kong, R.; Han, C.; Liu, Z.; Zhang, H.; et al. Fucoxanthin: A promising medicinal and nutritional ingredient. Evid. Based Complement. Altern. Med. 2015, 2015, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Gammone, M.A.; D’Orazio, N. Anti-obesity activity of the marine carotenoid fucoxanthin. Mar. Drugs 2015, 13, 2196–2214. [Google Scholar] [CrossRef] [PubMed]
- Pangestuti, R.; Kim, S.-K. Carotenoids, bioactive metabolites derived from seaweeds. In Springer Handbook of Marine Biotechnology; Kim, S.-K., Ed.; Springer: Berlin/Heidelberg, Germany, 2015; pp. 816–821. [Google Scholar]
- Siegel, B.Z.; Siegel, S.M. The chemical composition of algal cell walls. CRC Crit. Rev. Microbiol. 1973, 3, 1–26. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Sevilla, J.M.; Acién Fernández, F.G.; Molina Grima, E. Biotechnological production of lutein and its applications. Appl. Microbiol. Biotechnol. 2010, 86, 27–40. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, C.R.; Das, L.; Bhagawati, B.; Goswami, B.C. A comparative study of carotenoid extraction from algae in different solvent systems. Asian J. Plant Sci. Res. 2012, 2, 546–549. [Google Scholar]
- Mendes-Pinto, M.M.; Raposo, M.F.J.; Bowen, J.; Young, A.J.; Morais, R. Evaluation of different cell disruption processes on encysted cells of Haematococcus pluvialis: Effects on astaxanthin recovery and implications for bio-availability. J. Appl. Phycol. 2001, 13, 19–24. [Google Scholar] [CrossRef]
- Mojaat, M.; Foucault, A.; Pruvost, J.; Legrand, J. Optimal selection of organic solvents for biocompatible extraction of beta-carotene from Dunaliella salina. J. Biotechnol. 2008, 133, 433–441. [Google Scholar] [CrossRef] [PubMed]
- Hejazi, M.A.; de Lamarliere, C.; Rocha, J.M.S.; Vermuë, M.; Tramper, J.; Wijffels, R.H. Selective extraction of carotenoids from the microalga Dunaliella salina with retention of viability. Biotechnol. Bioeng. 2002, 79, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Castro-Puyana, M.; Herrero, M.; Urreta, I.; Mendiola, J.A.; Cifuentes, A.; Ibáñez, E.; Suárez-Alvarez, S. Optimization of clean extraction methods to isolate carotenoids from the microalga Neochloris oleoabundans and subsequent chemical characterization using liquid chromatography tandem mass spectrometry. Anal. Bioanal. Chem. 2013, 405, 4607–4616. [Google Scholar] [CrossRef] [PubMed]
- Castro-Puyana, M.; Herrero, M.; Mendiola, J.A.; Suárez-Alvarez, S.; Cifuentes, A.; Ibáñez, E. Extraction of new bioactives from neochloris oleoabundans using pressurized technologies and food grade solvents. In Proceedings of the III Iberoamerican Conference on Supercritical Fluids Cartagena de Indias (Combodia), Cartagena, Colombia, 1–5 April 2013.
- Golberg, A.; Sack, M.; Teissie, J.; Pataro, G.; Pliquett, U.; Saulis, G.; Stefan, T.; Miklavcic, D.; Vorobiev, E.; Frey, W. Energy-efficient biomass processing with pulsed electric fields for bioeconomy and sustainable development. Biotechnol. Biofuels 2016, 9, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Donsi, F.; Ferrari, G.; Pataro, G. Applications of pulsed electric field treatments for the enhancement of mass transfer from vegetable tissue. Food Eng. Rev. 2010, 2, 109–130. [Google Scholar] [CrossRef]
- Kulshrestha, S.; Sastry, S. Frequency and voltage effects on enhanced diffusion during moderate electric field (MEF) treatment. Innov. Food Sci. Emerg. Technol. 2003, 4, 189–194. [Google Scholar] [CrossRef]
- Vorobiev, E.; Lebovka, N. Enhanced extraction from solid foods and biosuspensions by pulsed electrical energy. Food Eng. Rev. 2010, 2, 95–108. [Google Scholar] [CrossRef]
- Poojary, M.M.; Roohinejad, S.; Barba, F.J.; Koubaa, M.; Puértolas, E.; Jambrak, A.R.; Greiner, R.; Oey, I. Application of pulsed electric field treatment for food waste recovery operations. In Handbook of Electroporation; Miklavcic, D., Ed.; Springer: Cham, Switzerland, 2017; in press. [Google Scholar]
- Raso, J.; Frey, W.; Ferrari, G.; Pataro, G.; Knorr, D.; Teissie, J.; Miklavčič, D. Recommendations guidelines on the key information to be reported in studies of application of PEF technology in food and biotechnological processes. Innov. Food Sci. Emerg. Technol. 2016, 37, 312–321. [Google Scholar] [CrossRef]
- Töpfl, S. Pulsed Electric Fields (PEF) for Permeabilization of Cell Membranes in Food- and Bioprocessing. Applications, Process and Equipment Design and Cost Analysis. Ph.D. Thesis, Technische Universität Berlin, Berlin, Germany, 2006. [Google Scholar]
- Grimi, N.; Dubois, A.; Marchal, L.; Jubeau, S.; Lebovka, N.I.; Vorobiev, E. Selective extraction from microalgae Nannochloropsis sp. using different methods of cell disruption. Bioresour. Technol. 2014, 153, 254–259. [Google Scholar] [CrossRef] [PubMed]
- Scholz, M.J.; Weiss, T.L.; Jinkerson, R.E.; Jing, J.; Roth, R.; Goodenough, U.; Posewitz, M.C.; Gerken, H.G. Ultrastructure and composition of the Nannochloropsis gaditana cell wall. Eukaryot. Cell 2014, 13, 1450–1464. [Google Scholar] [CrossRef] [PubMed]
- Luengo, E.; Martínez, J.M.; Bordetas, A.; Álvarez, I.; Raso, J. Influence of the treatment medium temperature on lutein extraction assisted by pulsed electric fields from Chlorella vulgaris. Innov. Food Sci. Emerg. Technol. 2015, 29, 15–22. [Google Scholar] [CrossRef]
- Kotnik, T.; Kramar, P.; Pucihar, G.; Miklavčič, D.; Tarek, M. Cell membrane electroporation—Part 1: The phenomenon. IEEE Electr. Insul. Mag. 2012, 28, 14–23. [Google Scholar] [CrossRef]
- Esser, A.T.; Smith, K.C.; Gowrishankar, T.R.; Vasilkoski, Z.; Weaver, J.C. Mechanisms for the intracellular manipulation of organelles by conventional electroporation. Biophys. J. 2010, 98, 2506–2514. [Google Scholar] [CrossRef] [PubMed]
- Schoenbach, K.H.; Beebe, S.J.; Buescher, E.S. Intracellular effect of ultrashort electrical pulses. Bioelectromagnetics 2001, 22, 440–448. [Google Scholar] [CrossRef] [PubMed]
- Luengo, E.; Condón-Abanto, S.; Álvarez, I.; Raso, J. Effect of pulsed electric field treatments on permeabilization and extraction of pigments from Chlorella vulgaris. J. Membr. Biol. 2014, 247, 1269–1277. [Google Scholar] [CrossRef] [PubMed]
- Luengo, E.; Martínez, J.M.; Coustets, M.; Álvarez, I.; Teissié, J.; Rols, M.-P.; Raso, J. A comparative study on the effects of millisecond- and microsecond-pulsed electric field treatments on the permeabilization and extraction of pigments from Chlorella vulgaris. J. Membr. Biol. 2015, 248, 883–891. [Google Scholar] [CrossRef] [PubMed]
- Parniakov, O.; Barba, F.J.; Grimi, N.; Marchal, L.; Jubeau, S.; Lebovka, N.; Vorobiev, E. Pulsed electric field assisted extraction of nutritionally valuable compounds from microalgae Nannochloropsis spp. using the binary mixture of organic solvents and water. Innov. Food Sci. Emerg. Technol. 2015, 27, 79–85. [Google Scholar] [CrossRef]
- Parniakov, O.; Barba, F.J.; Grimi, N.; Marchal, L.; Jubeau, S.; Lebovka, N.; Vorobiev, E. Pulsed electric field and pH assisted selective extraction of intracellular components from microalgae Nannochloropsis. Algal Res. 2015, 8, 128–134. [Google Scholar] [CrossRef]
- Jaeschke, D.P.; Menegol, T.; Rech, R.; Mercali, G.D.; Marczak, L.D.F. Carotenoid and lipid extraction from Heterochlorella luteoviridis using moderate electric field and ethanol. Process Biochem. 2016, 51, 1636–1643. [Google Scholar] [CrossRef]
- Postma, P.R.; Pataro, G.; Capitoli, M.; Barbosa, M.J.; Wijffels, R.H.; Eppink, M.H.M.; Olivieri, G.; Ferrari, G. Selective extraction of intracellular components from the microalga Chlorella vulgaris by combined pulsed electric field-temperature treatment. Bioresour. Technol. 2016, 203, 80–88. [Google Scholar] [CrossRef] [PubMed]
- Lebovka, N.I.; Praporscic, I.; Vorobiev, E. Effect of moderate thermal and pulsed electric field treatments on textural properties of carrots, potatoes and apples. Innov. Food Sci. Emerg. Technol. 2004, 5, 9–16. [Google Scholar] [CrossRef]
- Lebovka, N.I.; Praporscic, I.; Ghnimi, S.; Vorobiev, E. Temperature enhanced electroporation under the pulsed electric field treatment of food tissue. J. Food Eng. 2005, 69, 177–184. [Google Scholar] [CrossRef]
- Kulshrestha, S.; Sarang, S.; Loghavi, L.; Sastry, S. Moderate electrothermal treatments of cellular tissues. In Electrotechnologies for Extraction from Food Plants and Biomaterials; Vorobiev, E., Lebovka, N., Eds.; Springer: New York, NY, USA, 2009; pp. 83–94. [Google Scholar]
- Kusnadi, C.; Sastry, S.K. Effect of moderate electric fields on salt diffusion into vegetable tissue. J. Food Eng. 2012, 110, 329–336. [Google Scholar] [CrossRef]
- Nezammahalleh, H.; Ghanati, F.; Adams, T.A.; Nosrati, M.; Shojaosadati, S.A. Effect of moderate static electric field on the growth and metabolism of Chlorella vulgaris. Bioresour. Technol. 2016, 218, 700–711. [Google Scholar] [CrossRef] [PubMed]
- Richter, B.E.; Jones, B.A.; Ezzell, J.L.; Porter, N.L. Accelerated solvent extraction: A technique for sample preparation. Anal. Chem. 1996, 68, 1033–1039. [Google Scholar] [CrossRef]
- Plaza, M.; Turner, C. Pressurized hot water extraction of bioactives. TrAC Trends Anal. Chem. 2015, 71, 39–54. [Google Scholar] [CrossRef]
- Osorio-Tobón, J.F.; Meireles, M.A.A.; Osorio-Tobón, J.F.; Meireles, M.A.A. Recent applications of pressurized fluid extraction: Curcuminoids extraction with pressurized liquids. Food Public Health 2013, 3, 289–303. [Google Scholar] [CrossRef]
- Santos, D.T.; Veggi, P.C.; Meireles, M.A.A. Optimization and economic evaluation of pressurized liquid extraction of phenolic compounds from jabuticaba skins. J. Food Eng. 2012, 108, 444–452. [Google Scholar] [CrossRef]
- Gao, Q.; Haglund, P.; Pommer, L.; Jansson, S. Evaluation of solvent for pressurized liquid extraction of PCDD, PCDF, PCN, PCBz, PCPh and PAH in torrefied woody biomass. Fuel 2015, 154, 52–58. [Google Scholar] [CrossRef]
- Li, P.; Li, S.P.; Lao, S.C.; Fu, C.M.; Kan, K.K.W.; Wang, Y.T. Optimization of pressurized liquid extraction for Z-ligustilide, Z-butylidenephthalide and ferulic acid in Angelica sinensis. J. Pharm. Biomed. Anal. 2006, 40, 1073–1079. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, J.; Theegala, C. Optimizing a continuous flow lipid extraction system (CFLES) used for extracting microalgal lipids. GCB Bioenergy 2013, 5, 327–337. [Google Scholar] [CrossRef]
- Hu, J.; Guo, Z.; Glasius, M.; Kristensen, K.; Xiao, L.; Xu, X. Pressurized liquid extraction of ginger (Zingiber officinale Roscoe) with bioethanol: An efficient and sustainable approach. J. Chromatogr. A 2011, 1218, 5765–5773. [Google Scholar] [CrossRef] [PubMed]
- Aliakbarian, B.; Casazza, A.A.; Perego, P. Valorization of olive oil solid waste using high pressure–high temperature reactor. Food Chem. 2011, 128, 704–710. [Google Scholar] [CrossRef]
- Ben Hamissa, A.M.; Seffen, M.; Aliakbarian, B.; Casazza, A.A.; Perego, P.; Converti, A. Phenolics extraction from Agave americana (L.) leaves using high-temperature, high-pressure reactor. Food Bioprod. Process. 2012, 90, 17–21. [Google Scholar] [CrossRef]
- Casazza, A.A.; Aliakbarian, B.; Sannita, E.; Perego, P. High-pressure high-temperature extraction of phenolic compounds from grape skins. Int. J. Food Sci. Technol. 2012, 47, 399–405. [Google Scholar] [CrossRef]
- Latoui, M.; Aliakbarian, B.; Casazza, A.A.; Seffen, M.; Converti, A.; Perego, P. Extraction of phenolic compounds from Vitex agnus-castus L. Food Bioprod. Process. 2012, 90, 748–754. [Google Scholar] [CrossRef]
- Lopresto, C.G.; Petrillo, F.; Casazza, A.A.; Aliakbarian, B.; Perego, P.; Calabrò, V. A non-conventional method to extract D-limonene from waste lemon peels and comparison with traditional Soxhlet extraction. Sep. Purif. Technol. 2014, 137, 13–20. [Google Scholar] [CrossRef]
- Moreau, R.A.; Powell, M.J.; Singh, V. Pressurized liquid extraction of polar and nonpolar lipids in corn and oats with hexane, methylene chloride, isopropanol, and ethanol. J. Am. Oil Chem. Soc. 2003, 80, 1063–1067. [Google Scholar] [CrossRef]
- Jeannotte, R.; Hamel, C.; Jabaji, S.; Whalen, J.K. Comparison of solvent mixtures for pressurized solvent extraction of soil fatty acid biomarkers. Talanta 2008, 77, 195–199. [Google Scholar] [CrossRef] [PubMed]
- Bermejo, D.V.; Luna, P.; Manic, M.S.; Najdanovic-Visak, V.; Reglero, G.; Fornari, T. Extraction of caffeine from natural matter using a bio-renewable agrochemical solvent. Food Bioprod. Process. 2013, 91, 303–309. [Google Scholar] [CrossRef]
- Wu, H.; Chen, M.; Fan, Y.; Elsebaei, F.; Zhu, Y. Determination of rutin and quercetin in Chinese herbal medicine by ionic liquid-based pressurized liquid extraction–liquid chromatography–chemiluminescence detection. Talanta 2012, 88, 222–229. [Google Scholar] [CrossRef] [PubMed]
- Choi, M.P.K.; Chan, K.K.C.; Leung, H.W.; Huie, C.W. Pressurized liquid extraction of active ingredients (ginsenosides) from medicinal plants using non-ionic surfactant solutions. J. Chromatogr. A 2003, 983, 153–162. [Google Scholar] [CrossRef]
- Mustafa, A.; Turner, C. Pressurized liquid extraction as a green approach in food and herbal plants extraction: A review. Anal. Chim. Acta 2011, 703, 8–18. [Google Scholar] [CrossRef] [PubMed]
- Ong, E.S.; Cheong, J.S. H.; Goh, D. Pressurized hot water extraction of bioactive or marker compounds in botanicals and medicinal plant materials. J. Chromatogr. A 2006, 1112, 92–102. [Google Scholar] [CrossRef] [PubMed]
- Denery, J.R.; Dragull, K.; Tang, C.; Li, Q.X. Pressurized fluid extraction of carotenoids from Haematococcus pluvialis and Dunaliella salina and kavalactones from Piper methysticum. Anal. Chim. Acta 2004, 501, 175–181. [Google Scholar] [CrossRef]
- Herrero, M.; Jaime, L.; Martin-Alvarez, P.J.; Cifuentes, A.; Ibanez, E. Optimization of the extraction of antioxidants from Dunaliella salina microalga by pressurized liquids. J. Agric. Food Chem. 2006, 54, 5597–5603. [Google Scholar] [CrossRef] [PubMed]
- Plaza, M.; Santoyo, S.; Jaime, L.; García-Blairsy Reina, G.; Herrero, M.; Señoráns, F.J.; Ibáñez, E. Screening for bioactive compounds from algae. J. Pharm. Biomed. Anal. 2010, 51, 450–455. [Google Scholar] [CrossRef] [PubMed]
- Cha, K.H.; Kang, S.W.; Kim, C.Y.; Um, B.H.; Na, Y.R.; Pan, C.H. Effect of pressurized liquids on extraction of antioxidants from Chlorella vulgaris. J. Agric. Food Chem. 2010, 58, 4756–4761. [Google Scholar] [CrossRef] [PubMed]
- Cha, K.H.; Lee, H.J.; Koo, S.Y.; Song, D.G.; Lee, D.U.; Pan, C.H. Optimization of pressurized liquid extraction of carotenoids and chlorophylls from chlorella vulgaris. J. Agric. Food Chem. 2010, 58, 793–797. [Google Scholar] [CrossRef] [PubMed]
- Jaime, L.; Rodríguez-Meizoso, I.; Cifuentes, A.; Santoyo, S.; Suarez, S.; Ibáñez, E.; Señorans, F.J. Pressurized liquids as an alternative process to antioxidant carotenoids’ extraction from Haematococcus pluvialis microalgae. LWT—Food Sci. Technol. 2010, 43, 105–112. [Google Scholar] [CrossRef] [Green Version]
- Plaza, M.; Santoyo, S.; Jaime, L.; Avalo, B.; Cifuentes, A.; Reglero, G.; García-Blairsy Reina, G.; Señorans, F.J.; Ibáñez, E. Comprehensive characterization of the functional activities of pressurized liquid and ultrasound-assisted extracts from Chlorella vulgaris. LWT—Food Sci. Technol. 2012, 46, 245–253. [Google Scholar] [CrossRef]
- Kim, S.M.; Jung, Y.J.; Kwon, O.N.; Cha, K.H.; Um, B.H.; Chung, D.; Pan, C.H. A potential commercial source of fucoxanthin extracted from the microalga Phaeodactylum tricornutum. Appl. Biochem. Biotechnol. 2012, 166, 1843–1855. [Google Scholar] [CrossRef] [PubMed]
- Shang, Y.F.; Kim, S.M.; Lee, W.J.; Um, B.H. Pressurized liquid method for fucoxanthin extraction from Eisenia bicyclis (Kjellman) Setchell. J. Biosci. Bioeng. 2011, 111, 237–241. [Google Scholar] [CrossRef] [PubMed]
- Koo, S.Y.; Cha, K.H.; Song, D.G.; Chung, D.; Pan, C.H. Optimization of pressurized liquid extraction of zeaxanthin from Chlorella ellipsoidea. J. Appl. Phycol. 2012, 24, 725–730. [Google Scholar] [CrossRef]
- Taucher, J.; Baer, S.; Schwerna, P.; Hofmann, D.; Hümmer, M.; Buchholz, R.; Becker, A. Cell disruption and pressurized liquid extraction of carotenoids from microalgae. Thermodyn. Catal. 2016, 7, 1–7. [Google Scholar] [CrossRef]
- Jeon, Y.; Wijesinghe, W.A.J.P.; Kim, S. Enzyme-assisted extraction and recovery of bioactive components from seaweeds. In Handbook of Marinemacroalgae: Biotechnology and Applied Phycology, 1st ed.; Kim, S.-K., Ed.; John Wiley & Sons, Ltd.: New York, NY, USA, 2012; pp. 221–228. [Google Scholar]
- Lang, Q.; Wai, C.M. Supercritical fluid extraction in herbal and natural product studies—A practical review. Talanta 2001, 53, 771–782. [Google Scholar] [CrossRef]
- Zougagh, M.; Valcárcel, M.; Rı́os, A. Supercritical fluid extraction: A critical review of its analytical usefulness. TrAC Trends Anal. Chem. 2004, 23, 399–405. [Google Scholar] [CrossRef]
- Talisic, G.C.; Yumang, A.N.; Salta, M.T.S. Supercritical Fluid Extraction of β-carotene from D. Salina algae using C2H6 and C2H2. In Proceedings of the 2012 International Conference on Geological and Environmental Sciences, Jeju Island, Korea, 29–30 June 2012; pp. 30–34.
- Krichnavaruk, S.; Shotipruk, A.; Goto, M.; Pavasant, P. Supercritical carbon dioxide extraction of astaxanthin from Haematococcus pluvialis with vegetable oils as co-solvent. Bioresour. Technol. 2008, 99, 5556–5560. [Google Scholar] [CrossRef] [PubMed]
- Ruen-ngam, D.; Shotipruk, A.; Pavasant, P.; Machmudah, S.; Goto, M. Selective extraction of lutein from alcohol treated Chlorella vulgaris by supercritical CO2. Chem. Eng. Technol. 2012, 35, 255–260. [Google Scholar] [CrossRef]
- Macías-Sánchez, M.D.; Serrano, C.M.; Rodríguez, M.R.; Martínez de la Ossa, E. Kinetics of the supercritical fluid extraction of carotenoids from microalgae with CO2 and ethanol as cosolvent. Chem. Eng. J. 2009, 150, 104–113. [Google Scholar] [CrossRef]
- Bustamante, A.; Roberts, P.; Aravena, R.; Del Valle, J.M. Supercritical extraction of astaxanthin from H. pluvialis using ethanol-modified CO2. Experiments and modeling. In Proceedings of the 11th International Conference of Eng Food, Athens, Greece, 22–26 May 2011.
- Aravena, R.I.; del Valle, J.M. Effect of microalgae preconditioning on supercritical CO2 extraction of astaxanthin from Haematococcus pluvialis. In Proceedings of the 10th International Symposium of Supercritical Fluids, San Francisco, CA, USA, 13–16 May 2012.
- Yen, H.W.; Chiang, W.C.; Sun, C.H. Supercritical fluid extraction of lutein from Scenedesmus cultured in an autotrophical photobioreactor. J. Taiwan Inst. Chem. Eng. 2012, 43, 53–57. [Google Scholar] [CrossRef]
- Macías-Sánchez, M.D.; Fernandez-Sevilla, J.M.; Fernández, F.G.A.; García, M.C.C.; Grima, E.M. Supercritical fluid extraction of carotenoids from Scenedesmus almeriensis. Food Chem. 2010, 123, 928–935. [Google Scholar] [CrossRef]
- Kitada, K.; Machmudah, S.; Sasaki, M.; Goto, M.; Nakashima, Y.; Kumamoto, S.; Hasegawa, T. Supercritical CO2 extraction of pigment components with pharmaceutical importance from Chlorella vulgaris. J. Chem. Technol. Biotechnol. 2009, 84, 657–661. [Google Scholar] [CrossRef]
- Nobre, B.; Marcelo, F.; Passos, R.; Beirão, L.; Palavra, A.; Gouveia, L.; Mendes, R. Supercritical carbon dioxide extraction of astaxanthin and other carotenoids from the microalga Haematococcus pluvialis. Eur. Food Res. Technol. 2006, 223, 787–790. [Google Scholar] [CrossRef]
- Macı́as-Sánchez, M.D.; Mantell, C.; Rodrı́guez, M.; de la Ossa, E.J.M.; Lubián, L.M.; Montero, O. Supercritical fluid extraction of carotenoids and chlorophyll a from Nannochloropsis gaditana. J. Food Eng. 2005, 66, 245–251. [Google Scholar] [CrossRef]
- Macías-Sánchez, M.D.; Mantell, C.; Rodríguez, M.; de la Ossa, E.J.M.; Lubián, L.M.; Montero, O. Supercritical fluid extraction of carotenoids and chlorophyll a from Synechococcus sp. J. Supercrit. Fluids 2007, 39, 323–329. [Google Scholar] [CrossRef]
- Macías-Sánchez, M.D.; Mantell, C.; Rodríguez, M.; de la Ossa, E.J.M.; Lubián, L.M.; Montero, O. Comparison of supercritical fluid and ultrasound-assisted extraction of carotenoids and chlorophyll a from Dunaliella salina. Talanta 2009, 77, 948–952. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, L.C.; Serrano, C.M.; Rodríguez, M.R.; de la Ossa, E.J.M.; Lubián, L.M. Extraction of carotenoids and fatty acids from microalgae using supercritical technology. Am. J. Anal. Chem. 2012, 3, 877–883. [Google Scholar] [CrossRef]
- Mendes, R.L.; Nobre, B.P.; Cardoso, M.T.; Pereira, A.P.; Palavra, A.F. Supercritical carbon dioxide extraction of compounds with pharmaceutical importance from microalgae. Inorg. Chim. Acta 2003, 356, 328–334. [Google Scholar] [CrossRef]
- Machmudah, S.; Shotipruk, A.; Goto, M.; Sasaki, M.; Hirose, T. Extraction of astaxanthin from Haematococcus p luvialis using supercritical CO2 and ethanol as entrainer. Ind. Eng. Chem. Res. 2006, 45, 3652–3657. [Google Scholar] [CrossRef]
- Wang, L.; Yang, B.; Yan, B.; Yao, X. Supercritical fluid extraction of astaxanthin from Haematococcus pluvialis and its antioxidant potential in sunflower oil. Innov. Food Sci. Emerg. Technol. 2012, 13, 120–127. [Google Scholar] [CrossRef]
- Valderrama, J.O.; Perrut, M.; Majewski, W. Extraction of astaxantine and phycocyanine from microalgae with supercritical carbon dioxide. J. Chem. Eng. Data 2003, 48, 827–830. [Google Scholar] [CrossRef]
- Macías-Sánchez, M.D.; Mantell Serrano, C.; Rodríguez Rodríguez, M.; de la Ossa, E.J.M.; Lubián, L.M.; Montero, O. Extraction of carotenoids and chlorophyll from microalgae with supercritical carbon dioxide and ethanol as cosolvent. J. Sep. Sci. 2008, 31, 1352–1362. [Google Scholar] [CrossRef] [PubMed]
- Liau, B.C.; Shen, C.T.; Liang, F.P.; Hong, S.E.; Hsu, S.L.; Jong, T.T.; Chang, C.M.J. Supercritical fluids extraction and anti-solvent purification of carotenoids from microalgae and associated bioactivity. J. Supercrit. Fluids 2010, 55, 169–175. [Google Scholar] [CrossRef]
- Fujii, K. Process integration of supercritical carbon dioxide extraction and acid treatment for astaxanthin extraction from a vegetative microalga. Food Bioprod. Process. 2012, 90, 762–766. [Google Scholar] [CrossRef]
- Esquivel-Hernández, D.; López, V.; Rodríguez-Rodríguez, J.; Alemán-Nava, G.; Cuéllar-Bermúdez, S.; Rostro-Alanis, M.; Parra-Saldívar, R. Supercritical carbon dioxide and microwave-assisted extraction of functional lipophilic compounds from Arthrospira platensis. Int. J. Mol. Sci. 2016, 17, 658. [Google Scholar] [CrossRef] [PubMed]
- Ota, M.; Watanabe, H.; Kato, Y.; Watanabe, M.; Sato, Y.; Smith, R.L.; Inomata, H. Carotenoid production from Chlorococcum littorale in photoautotrophic cultures with downstream supercritical fluid processing. J. Sep. Sci. 2009, 32, 2327–2335. [Google Scholar] [CrossRef] [PubMed]
- Liau, B.-C.; Hong, S.-E.; Chang, L.-P.; Shen, C.-T.; Li, Y.-C.; Wu, Y.-P.; Jong, T.-T.; Shieh, C.-J.; Hsu, S.-L.; Chang, C.-M.J. Separation of sight-protecting zeaxanthin from Nannochloropsis oculata by using supercritical fluids extraction coupled with elution chromatography. Sep. Purif. Technol. 2011, 78, 1–8. [Google Scholar] [CrossRef]
- Goto, M.; Kanda, H.; Wahyudiono; Machmudah, S. Extraction of carotenoids and lipids from algae by supercritical CO2 and subcritical dimethyl ether. J. Supercrit. Fluids 2015, 96, 245–251. [Google Scholar] [CrossRef]
- Fahmy, T.M.; Paulaitis, M.E.; Johnson, D.M.; McNally, M.E.P. Modifier effects in the supercritical fluid extraction of solutes from clay, soil, and plant materials. Anal. Chem. 1993, 65, 1462–1469. [Google Scholar] [CrossRef]
- Ritter, D.C.; Campbell, A.G. Supercritical carbon dioxide extraction of southern pine and ponderosa pine. Wood Fiber Sci. 1991, 23, 98–113. [Google Scholar]
- Gouveia, L.; Nobre, B.P.; Marcelo, F.M.; Mrejen, S.; Cardoso, M.T.; Palavra, A.F.; Mendes, R.L. Functional food oil coloured by pigments extracted from microalgae with supercritical CO2. Food Chem. 2007, 101, 717–723. [Google Scholar] [CrossRef]
- Thana, P.; Machmudah, S.; Goto, M.; Sasaki, M.; Pavasant, P.; Shotipruk, A. Response surface methodology to supercritical carbon dioxide extraction of astaxanthin from Haematococcus pluvialis. Bioresour. Technol. 2008, 99, 3110–3115. [Google Scholar] [CrossRef] [PubMed]
- Montero, O.; Macías-Sánchez, M.D.; Lama, C.M.; Lubián, L.M.; Mantell, C.; Rodríguez, M.; de la Ossa, E.J.M. Supercritical CO2 extraction of β-carotene from a marine strain of the Cyanobacterium Synechococcus species. J. Agric. Food Chem. 2005, 53, 9701–9707. [Google Scholar] [CrossRef] [PubMed]
- Jaime, L.; Mendiola, J.A.; Ibáñez, E.; Martin-Álvarez, P.J.; Cifuentes, A.; Reglero, G.; Señoráns, F.J. β-carotene isomer composition of sub- and supercritical carbon dioxide extracts. Antioxidant activity measurement. J. Agric. Food Chem. 2007, 55, 10585–10590. [Google Scholar] [CrossRef] [PubMed]
- Nobre, B.P.; Villalobos, F.; Barragán, B.E.; Oliveira, A.C.; Batista, A.P.; Marques, P.A.S.S.; Mendes, R.L.; Sovová, H.; Palavra, A.F.; Gouveia, L. A biorefinery from Nannochloropsis sp. microalga—Extraction of oils and pigments. Production of biohydrogen from the leftover biomass. Bioresour. Technol. 2013, 135, 128–136. [Google Scholar] [CrossRef] [PubMed]
- Sivagnanam, S.P.; Yin, S.; Choi, J.H.; Park, Y.B.; Woo, H.C.; Chun, B.S. Biological properties of fucoxanthin in oil recovered from two brown seaweeds using supercritical CO2 extraction. Mar. Drugs 2015, 13, 3422–3442. [Google Scholar] [CrossRef] [PubMed]
- Fan, X.D.; Hou, Y.; Huang, X.X.; Qiu, T.Q.; Jiang, J.G. Ultrasound-enhanced subcritical CO2 extraction of lutein from Chlorella pyrenoidosa. J. Agric. Food Chem. 2015, 63, 4597–4605. [Google Scholar] [CrossRef] [PubMed]
- King, J.W.; Srinivas, K.; Zhang, D. Advances in Critical Fluid Processing. In Alternatives to Conventional Food Processing; Proctor, A., Ed.; The Royal Society of Chemistry: Cambridge, UK, 2010; pp. 93–144. [Google Scholar]
- Semelsberger, T.A.; Borup, R.L.; Greene, H.L. Dimethyl ether (DME) as an alternative fuel. J. Power Sources 2006, 156, 497–511. [Google Scholar] [CrossRef]
- Kanda, H.; Li, P. Simple extraction method of green crude from natural blue-green microalgae by dimethyl ether. Fuel 2011, 90, 1264–1266. [Google Scholar] [CrossRef]
- Lu, J.; Feng, X.; Han, Y.; Xue, C. Optimization of subcritical fluid extraction of carotenoids and chlorophyll a from Laminaria japonica Aresch by response surface methodology. J. Sci. Food Agric. 2014, 94, 139–145. [Google Scholar] [CrossRef] [PubMed]
- Chemat, F.; Cravotto, G. (Eds.) Microwave-Assisted Extraction for Bioactive Compounds; Food Engineering Series; Springer: Boston, MA, USA, 2013.
- Pasquet, V.; Chérouvrier, J.-R.; Farhat, F.; Thiéry, V.; Piot, J.-M.; Bérard, J.-B.; Kaas, R.; Serive, B.; Patrice, T.; Cadoret, J.-P.; et al. Study on the microalgal pigments extraction process: Performance of microwave assisted extraction. Process Biochem. 2011, 46, 59–67. [Google Scholar] [CrossRef]
- Ruen-ngam, D.; Shotipruk, A.; Pavasant, P. Comparison of extraction methods for recovery of astaxanthin from Haematococcus pluvialis. Sep. Sci. Technol. 2011, 46, 64–70. [Google Scholar] [CrossRef]
- Poojary, M.M.; Passamonti, P. Optimization of extraction of high purity all-trans-lycopene from tomato pulp waste. Food Chem. 2015, 188, 84–91. [Google Scholar] [CrossRef] [PubMed]
- Poojary, M.M.; Passamonti, P. Extraction of lycopene from tomato processing waste: Kinetics and modelling. Food Chem. 2015, 173, 943–950. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Chen, G.; Zhao, G.; Hu, X. Optimization of microwave-assisted extraction of astaxanthin from Haematococcus pluvialis by response surface methodology and antioxidant activities of the extracts. Sep. Sci. Technol. 2009, 44, 243–262. [Google Scholar] [CrossRef]
- Meullemiestre, A.; Breil, C.; Abert-Vian, M.; Chemat, F. Innovative techniques and alternative solvents for extraction of microbial oils. In Modern Techniques and Solvents for the Extraction of Microbial Oils; Springer: New York, NY, USA, 2015; pp. 19–42. [Google Scholar]
- Juliano, P.; Augustin, M.A.; Xu, X.-Q.; Mawson, R.; Knoerzer, K. Advances in high frequency ultrasound separation of particulates from biomass. Ultrason. Sonochem. 2016, in press. [Google Scholar] [CrossRef] [PubMed]
- Leong, T.; Johansson, L.; Juliano, P.; McArthur, S.L.; Manasseh, R. Ultrasonic separation of particulate fluids in small and large scale systems: A Review. Ind. Eng. Chem. Res. 2013, 52, 16555–16576. [Google Scholar] [CrossRef]
- Dolatowski, Z.J.; Stasiak, D.M. Ultrasonically Assisted Diffusion Processes. In Enhancing Extraction Processes in the Food Industry; Lebovka, F., Vorobiev, N., Chemat, E., Eds.; CRC Press: Boca Raton, FL, USA, 2012; pp. 123–144. [Google Scholar]
- Chemat, F.; Rombaut, N.; Sicaire, A.-G.; Meullemiestre, A.; Fabiano-Tixier, A.-S.; Abert-Vian, M. Ultrasound assisted extraction of food and natural products. Mechanisms, techniques, combinations, protocols and applications. A review. Ultrason. Sonochem. 2017, 34, 540–560. [Google Scholar] [CrossRef] [PubMed]
- Gerde, J.A.; Montalbo-Lomboy, M.; Yao, L.; Grewell, D.; Wang, T. Evaluation of microalgae cell disruption by ultrasonic treatment. Bioresour. Technol. 2012, 125, 175–181. [Google Scholar] [CrossRef] [PubMed]
- Zou, T.B.; Jia, Q.; Li, H.W.; Wang, C.X.; Wu, H.F. Response surface methodology for ultrasound-assisted extraction of astaxanthin from Haematococcus pluvialis. Mar. Drugs 2013, 11, 1644–1655. [Google Scholar] [CrossRef] [PubMed]
- Deenu, A.; Naruenartwongsakul, S.; Kim, S.M. Optimization and economic evaluation of ultrasound extraction of lutein from Chlorella vulgaris. Biotechnol. Bioprocess Eng. 2013, 18, 1151–1162. [Google Scholar] [CrossRef]
- Yamamoto, K.; King, P.M.; Wu, X.; Mason, T.J.; Joyce, E.M. Effect of ultrasonic frequency and power on the disruption of algal cells. Ultrason. Sonochem. 2015, 24, 165–171. [Google Scholar] [CrossRef] [PubMed]
- Dey, S.; Rathod, V.K. Ultrasound assisted extraction of β-carotene from Spirulina platensis. Ultrason. Sonochem. 2013, 20, 271–276. [Google Scholar] [CrossRef] [PubMed]
- Gerken, H.G.; Donohoe, B.; Knoshaug, E.P. Enzymatic cell wall degradation of Chlorella vulgaris and other microalgae for biofuels production. Planta 2013, 237, 239–253. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, D.B.; Flores, É.M.M.; Barin, J.S.; Mercadante, A.Z.; Jacob-Lopes, E.; Zepka, L.Q. Production of carotenoids from microalgae cultivated using agroindustrial wastes. Food Res. Int. 2014, 65, 144–148. [Google Scholar] [CrossRef]
- Leong, T.; Knoerzer, K.; Trujillo, F.J.; Johansson, L.; Manasseh, R.; Barbosa-Cánovas, G.V.; Juliano, P. Megasonic separation of food droplets and particles: Design considerations. Food Eng. Rev. 2015, 7, 298–320. [Google Scholar] [CrossRef]
- Bosma, R.; van Spronsen, W.A.; Tramper, J.; Wijffels, R.H. Ultrasound, a new separation technique to harvest microalgae. J. Appl. Phycol. 2003, 15, 143–153. [Google Scholar] [CrossRef]
- Donsì, F.; Ferrari, G.; Lenza, E.; Maresca, P. Main factors regulating microbial inactivation by high-pressure homogenization: Operating parameters and scale of operation. Chem. Eng. Sci. 2009, 64, 520–532. [Google Scholar] [CrossRef]
- Donsì, F.; Ferrari, G.; Maresca, P. High-Pressure Homogenization for Food Sanitization. In Global Issues in Food Science and Technology; Barbosa-Cánovas, G., Mortimer, A., Lineback, D., Spiess, W., Buckle, K., Colonna, P., Eds.; Elesvier: Cambridge, MA, USA, 2009; pp. 309–352. [Google Scholar]
- Donsì, F.; Annunziata, M.; Ferrari, G. Microbial inactivation by high pressure homogenization: Effect of the disruption valve geometry. J. Food Eng. 2013, 115, 362–370. [Google Scholar] [CrossRef]
- Donsì, F.; Sessa, M.; Ferrari, G. Nanometric-size delivery systems for bioactive compounds for the nutraceutical and food industries. In Bio-Nanotechnology: A Revolution in Food, Biomedical and Health Sciences; Bagchi, D., Bagchi, M., Moriyama, H., Shahidi, F., Eds.; Blackwell Publishing Ltd.: Oxford, UK, 2013; pp. 619–666. [Google Scholar]
- Yap, B.H.J.; Dumsday, G.J.; Scales, P.J.; Martin, G.J.O. Energy evaluation of algal cell disruption by high pressure homogenisation. Bioresour. Technol. 2015, 184, 280–285. [Google Scholar] [CrossRef] [PubMed]
- Samarasinghe, N.; Fernando, S.; Lacey, R.; Faulkner, W.B. Algal cell rupture using high pressure homogenization as a prelude to oil extraction. Renew. Energy 2012, 48, 300–308. [Google Scholar] [CrossRef]
- Samarasinghe, N.; Fernando, S.; Faulkner, B. Effect of high pressure homogenization on aqueous phase solvent extraction of lipids from Nannochloris oculata microalgae. J. Energy Nat. Resour. 2012, 1, 1–7. [Google Scholar] [CrossRef]
- Olaizola, M. Commercial production of astaxanthin from Haematococcus pluvialis using 25,000-liter outdoor photobioreactors. J. Appl. Phycol. 2000, 12, 499–506. [Google Scholar] [CrossRef]
- Clarke, A.; Prescott, T.; Khan, A.; Olabi, A.G. Causes of breakage and disruption in a homogeniser. Appl. Energy 2010, 87, 3680–3690. [Google Scholar] [CrossRef] [Green Version]
- Lee, A.K.; Lewis, D.M.; Ashman, P.J. Disruption of microalgal cells for the extraction of lipids for biofuels: Processes and specific energy requirements. Biomass Bioenergy 2012, 46, 89–101. [Google Scholar] [CrossRef]
- Xie, Y.; Ho, S.H.; Chen, C.N.N.; Chen, C.Y.; Jing, K.; Ng, I.S.; Chen, J.; Chang, J.S.; Lu, Y. Disruption of thermo-tolerant Desmodesmus sp. F51 in high pressure homogenization as a prelude to carotenoids extraction. Biochem. Eng. J. 2016, 109, 243–251. [Google Scholar] [CrossRef]
- Mulchandani, K.; Kar, J.R.; Singhal, R.S. Extraction of lipids from Chlorella saccharophila using high-pressure homogenization followed by three phase partitioning. Appl. Biochem. Biotechnol. 2015, 176, 1613–1626. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.G.; Kang, D.H.; Lee, D.B.; Lee, H.Y. Pretreatment for simultaneous production of total lipids and fermentable sugars from marine alga, Chlorella sp. Appl. Biochem. Biotechnol. 2013, 171, 1143–1158. [Google Scholar] [CrossRef] [PubMed]
- Olmstead, I.L.D.; Kentish, S.E.; Scales, P.J.; Martin, G.J.O. Low solvent, low temperature method for extracting biodiesel lipids from concentrated microalgal biomass. Bioresour. Technol. 2013, 148, 615–619. [Google Scholar] [CrossRef] [PubMed]
- Choi, W.Y.; Lee, H.Y. Effective production of bioenergy from marine Chlorella sp. by high-pressure homogenization. Biotechnol. Biotechnol. Equip. 2016, 30, 81–89. [Google Scholar] [CrossRef]
- Halim, R.; Rupasinghe, T.W.T.; Tull, D.L.; Webley, P.A. Mechanical cell disruption for lipid extraction from microalgal biomass. Bioresour. Technol. 2013, 140, 53–63. [Google Scholar] [CrossRef] [PubMed]
- Spiden, E.M.; Yap, B.H.J.; Hill, D.R.A.; Kentish, S.E.; Scales, P.J.; Martin, G.J.O. Quantitative evaluation of the ease of rupture of industrially promising microalgae by high pressure homogenization. Bioresour. Technol. 2013, 140, 165–171. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Li, Y.; Hu, X.; Su, W.; Zhong, M. Combined enzymatic and mechanical cell disruption and lipid extraction of green alga Neochloris oleoabundans. Int. J. Mol. Sci. 2015, 16, 7707–7722. [Google Scholar] [CrossRef] [PubMed]
- Shene, C.; Monsalve, M.T.; Vergara, D.; Lienqueo, M.E.; Rubilar, M. High pressure homogenization of Nannochloropsis oculata for the extraction of intracellular components: Effect of process conditions and culture age. Eur. J. Lipid Sci. Technol. 2016, 118, 631–639. [Google Scholar] [CrossRef]
- Cho, S.-C.; Choi, W.-Y.; Oh, S.-H.; Lee, C.-G.; Seo, Y.-C.; Kim, J.-S.; Song, C.-H.; Kim, G.-V.; Lee, S.-Y.; Kang, D.-H.; et al. Enhancement of lipid extraction from marine microalga, Scenedesmus associated with high-pressure homogenization process. J. Biomed. Biotechnol. 2012, 2012, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Jubeau, S.; Marchal, L.; Pruvost, J.; Jaouen, P.; Legrand, J.; Fleurence, J. High pressure disruption: A two-step treatment for selective extraction of intracellular components from the microalga Porphyridium cruentum. J. Appl. Phycol. 2013, 25, 983–989. [Google Scholar] [CrossRef]
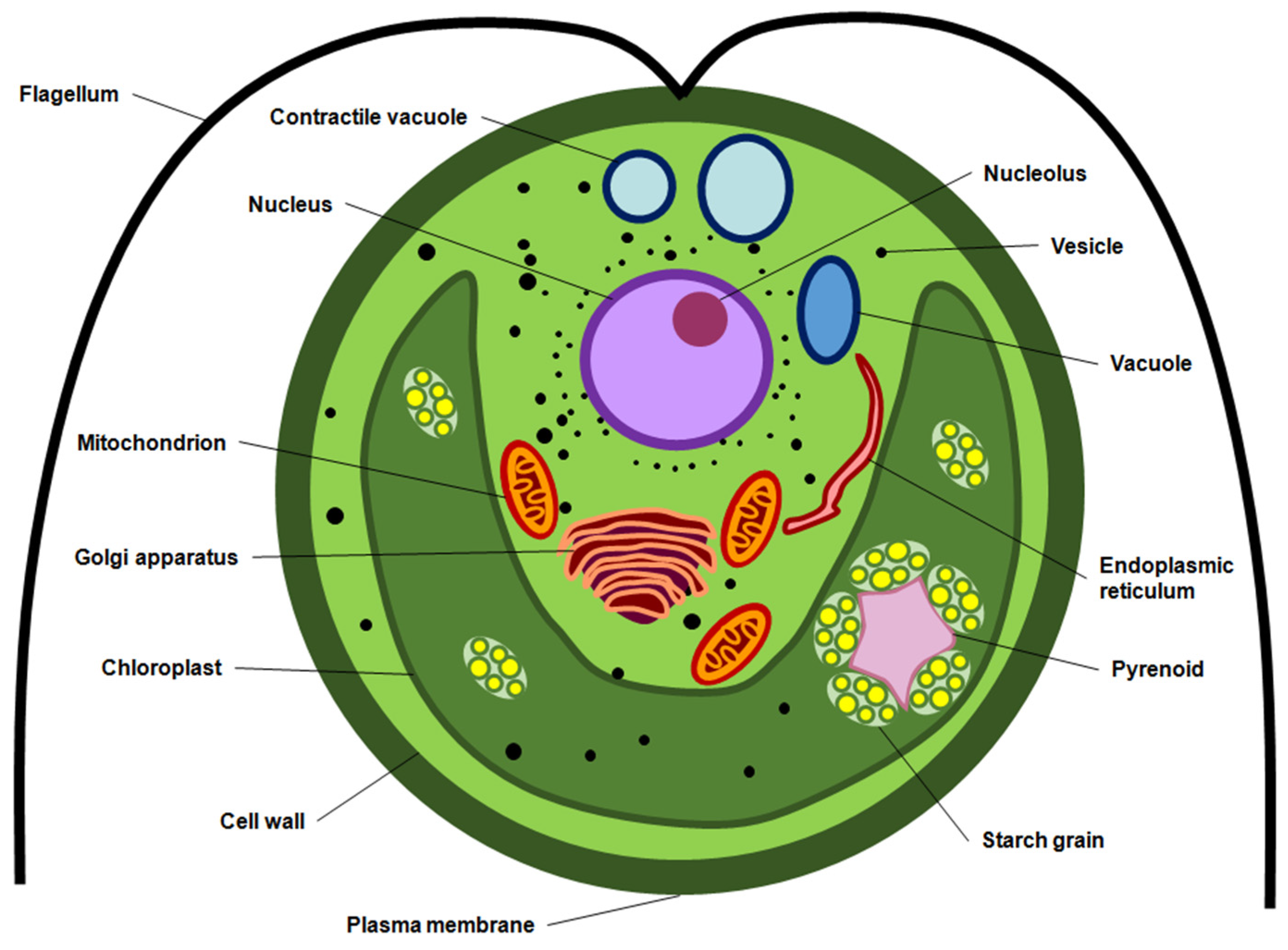

| Microalgae | Biomass Concentration | Electrical Treatment | Extraction Conditions | Carotenoid Yield | Notes | Reference |
|---|---|---|---|---|---|---|
| MEF | ||||||
| Heterochlorella luteoviridis | 4 g dry biomass/100 mL 25% ethanol solution | 0–180 V, 60 Hz, 10 min, <35 °C | 25%–75% ethanol, 50 min, 30 °C | Total carotenoids 1.21 mg/g dw | MEF induced a reversible electroporation improving the extraction efficiency. Xanthophylls all-trans-lutein and all-trans-zeaxanthin were the major carotenoids extracted. | [64] |
| PEF | ||||||
| Chlorella vulgaris | ~3% dw | 15 kV/cm, 100 kJ/kg | N/A a | Total carotenoids 525% recovery compared with the conventional ball milling homogenization process | Antioxidant activity of the extract was increased by almost 100%. | [53] |
| Chlorella vulgaris | 109 CFU/mL in McIlvaine buffer (pH 7) | 10–25 kV/cm 0.6–93 kJ/L of culture | 96% ethanol, 20 °C, 0–1 h | Total carotenoids: ~0.82 mg/g dw and 1.04 mg/g dw after, respectively, 0 h and 1 h of incubation after PEF treatment | Extraction yield significantly increased after 1 h of the application of PEF, likely caused by the plasmolysis of the chloroplast during the incubation time. | [60] |
| Chlorella vulgaris | 2 × 108 CFU/mL in McIlvaine buffer (pH 7) | Millisecond range: 1–40 ms pulses, 3.5–5 kV/cm 9–150 kJ/L of culture Microsecond range: 3 μs pulses 10–25 kV/cm 1.5–93 kJ/L | 96% ethanol 20 °C, 0–1 h | Total carotenoids: ~1.06 mg/L after 0 h and 1 h of incubation in the ms range; 1.09 mg/L and 1.58 mg/L after, respectively, 0 h and 1 h of incubation in the μs range | PEF in the ms range at a lower electric field strength created irreversible alterations, while in the μs range the defects were a dynamic structure along the post-pulse time. Higher energy efficiency of treatment in the μs range than in the ms range. | [61] |
| Chlorella vulgaris | 109 CFU/mL in McIlvaine buffer (pH 7) | 10–40 °C 10–25 kV/cm 1.5–93 kJ/L of culture | 96% ethanol, 20 °C, 0–1 h | Lutein up to 0.753 mg/g dw | Increasing temperature increased the sensitivity of microalgae cells to irreversible electroporation, and decreased the total specific energy required to obtain a given extraction yield. PEF treatment did not cause pigment degradation. | [56] |
| Spirulina platensis | ~3% dw | 15 kV/cm, 100 kJ/kg | N/A a | Total carotenoids 150% recovery compared with the conventional ball milling homogenization process | Antioxidant activity of the extract was increased by almost 100% | [53] |
| Nannochloropsis sp. | 1% (w/w) in distilled water | 20 kV/cm, 1–4 ms, 13.3–53.1 kJ/kg | N/A b | N/A c | PEF allowed selective extraction of water-soluble ionic components and water-soluble proteins, but was ineffective for extraction of pigments. | [54] |
| Nannochloropsis sp. | 1% (w/w) in distilled water | 20 kV/cm, 0.01–6 ms, 13.3–53.1 kJ/kg | Distilled water, up to 3 h, 50 °C, pH = 8.5–11 | Total carotenoids: ~0.04 mg/g dw after PEF (pH 8.5); ~0.2 mg/g dw after PEF (pH 8.5) + extraction at pH 11 | Extraction efficiency after PEF (pH 8.5) was comparable with that of the aqueous extraction at pH 11. PEF (pH 8.5) treatment was more efficient than PEF (pH 11) treatment. Supplementary extraction at pH = 11 allowed a noticeable increase of the concentrations yield. PEF extracts showed high purity. | [63] |
| Nannochloropsis sp. | 1% (w/w) in distilled water | 20 kV/cm, 0.01–6 ms, 13.3–53.1 kJ/kg | Aqueous DMSO, ethanol solutions: 0%, 30%, 50%, and 100%; 20 °C; 240 min | KPEF d ≈3.0 at 50% DMSO KPEF d ≈ 2.4 at 30% EtOH | High levels of extracted proteins at the first step with water, and noticeable enhancement of extraction of pigments at the second step with binary mixtures. The two-stage PEF-assisted procedure allowed effective extraction using less concentrated mixtures of organic solvents with water. | [62] |
| HVED | ||||||
| Nannochloropsis sp. | 1% (w/w) in distilled water | 40 kV/cm, 1–4 ms, 13.3–53.1 kJ/kg | N/A a | N/A c | Noticeably agglomeration of microalgae cells in the HVED-treated suspensions. Higher pigment recovery than PEF, but less than UAE and HPH. | [54] |
| Microalga/Seaweed | Extraction Condition | Carotenoid Yield | Notes | Reference | ||||
|---|---|---|---|---|---|---|---|---|
| Solvent a | Pretreatment | P b (bar) | T c (°C) | T d (h) | ||||
| Microalgae | ||||||||
| Haematococcus pluvialis | CO2 and 9.4% ethanol | Crushing and then grinding in dry ice | 300 | 60 | – | Astaxanthin >97% recovery | Co-solvent enhanced the recovery slightly | [122] |
| Synechococcus sp. | CO2 | Freeze drying | 500 | 60 | 4 | Total carotenoids 2.76 mg/g dw e | Optimal extraction conditions for β-carotene was 50 °C, 358 bar; for β-cryptoxanthin was 59 °C, 454 bar; and for zeaxanthin was 60 °C, 500 bar. | [134] |
| Haematococcus pluvialis | CO2 | Drying (powder form) | 70 | 500 | 4 | Astaxanthin 23.04 mg/g dw e | Pressure, extraction time, and the interaction between temperature and pressure had significant effect on astaxanthin yield. | [133] |
| Dunaliella salina | CO2 | Homogenization | 60 | 400 | 3 | Total carotenoids 12.17 mg/g algae dw | SFE was more selective than the UAE. | [117] |
| Chlorella vulgaris | CO2 and 7.5% ethanol | – | 80 | 500 | 3 | Lutein ~≥1.8 mg/g algae; β-carotene ~≥0.2 mg/g | Supercritical CO2 has high selectivity for lutein extraction, however the yield was lower than Soxhlet extraction; ethanol was better co-solvent than acetone. | [113] |
| Chlorococcum littorale | CO2 and 10 mol % ethanol | Freeze drying | 60 | 300 | 1–3 | Total carotenoids 0.094%–0.21% dw | Co-solvent enhanced the recovery slightly. | [127] |
| Scenedesmus sp. | CO2 and ethanol (30 mol %) | Freeze drying and milling | 70 | 400 | 1 | Lutein 2.210 mg/g algae e | Higher temperature lead to increased impurity. | [111] |
| Nannochloropsis gaditana | CO2 | Freeze drying (powder form) | 60 | 400 | 3 | Total carotenoids 0.343 mg/g algae dw e | Higher temperature lead to degradation. | [115] |
| Haematococcus pluvialis | CO2 and 5% ethanol | – | 70 | 550 | 4 | Astaxanthin 77.9% recovery with respect to 34.3 mg/g dw total content found in the sample using Soxhlet extraction | Astaxanthin yield increased with increasing cosolvent concentration up to 5% (v/v) ethanol. | [120] |
| Haematococcus pluvialis | CO2 and 10% ethanol | Freeze drying and ball milling | 60 | 300 | – | Carotenoid recovery 92%; esterified astaxanthin ~75%; lutein >90%; astaxanthin >90%; β-carotene >90%; and canthaxanthin ~85% | Crushing improved the recovery significantly. | [114] |
| Chlorella vulgaris | CO2 and 5% ethanol | Crushing | 40 | 300 | – | Total carotenoids up to 0.299% | Crushing increased pigments recovery. | [132] |
| Dunaliella salina | CO2 | Freeze drying | 9.8 | 443 | 1.6 | Total carotenoids 6.72% (predicted) | Higher yields were obtained at high pressures and low temperatures. | [135] |
| Nannochloropsis sp. | CO2 and 20% ethanol | Ball milling | 40 | 300 | >1 | – | Co-solvent increased the yield. | [136] |
| Scenedesmus almeriensis | CO2 | Freeze drying (powder form) | 60 | 400 | 5 | Lutein 0.0466 mg/g dw e β-carotene 1.5 mg/g dw e | Recovery was lower compared with conventional acetone extraction. | [112] |
| Synechococcus sp. | CO2 | Freeze drying | 50 | 300 | 3 | Total carotenoids 1.511 mg/g algae dw e | The highest carotenoids/chlorophylls selectivity was obtained at 200 bar and 60 °C. | [116] |
| Nannochloropsis oculata | CO2 and 16.7 wt % ethanol | – | 50 | 350 | – | Total carotenoids 7.61 mg/g dw | Anti-solvent precipitation of carotenoids allowed pure Zeaxanthin. | [124] |
| Nannochloropsis oculata | CO2 and ethanol | Grinding and freeze drying | 50 | 350 | – | Zeaxanthin 13.17 mg/g | Ethanol as a co solvent increased the yield, and was efficient than dichloromethane, toluene and soybean oil | [128] |
| Monoraphidium sp. | CO2 and ethanol | Freeze drying | 60 | 200 | 1 | Astaxanthin 2.02 mg/g dw | Ethanol as a co-solvent improved astaxanthin yield. | [125] |
| Chlorella vulgaris | CO2 and ethanol | Pretreatment process using alcohol as elution solvent | 40 | 400 | 0.75 | Lutein 1.78% recovery based on 7.9 mg/g obtained in Soxhlet extraction | Ethanol as an elution solvent removed chlorophyll a, b and β-carotene and improved selectivity of lutein | [107] |
| Haematococcus pluvialis | CO2 and 10% olive oil | Drying | 70 | 400 | 5 | Asthaxanthin 51% recovery | Olive oil co-solvent lead to a recovery comparable to ethanol as a co-solvent. | [106] |
| Nannochloropsis gaditana | CO2 and 5% ethanol | Freeze drying | 40–60 | 100–500 | 3 | Carotenoid yield up to 0.3% | Extraction kinetics was studies. | [108] |
| Synechococcus sp. | Carotenoid yield up to 0.12% | |||||||
| Dunaliella salina | Carotenoid yield up to 1.3% | |||||||
| Nannochloropsis gaditana | CO2 and 5% ethanol | Freeze drying | 60 | 500 | 3 | Total carotenoids 2.893 mg/g algae dw e | Supercritical extraction process with co-solvent was more selective than conventional methanol extraction. | [123] |
| Synechococcus sp. | 50 | 300 | Total carotenoids 1.860 mg/g algae dw e | |||||
| Dunaliella salina | Total carotenoids 9.629 mg/g algae dw e | |||||||
| Haematococcus pluvialis | CO2 and 2.3 mL/g sample ethanol | Freeze drying (powder form) | 65 | 435 | 3.5 | Astaxanthin recovery of 87.42% from sample containing 2.26% astaxanthin. | Increasing co-solvent amount resulted in an improving astaxanthin yield. | [121] |
| Synechococcus sp. | CO2 and ethanol | – | 40 and 60 | 400 and 200 | 3 | β-carotene 0.70 mg/g algae dw at 40 °C 400 bar e Zeaxanthin 0.70 mg/g algae dw at 60 °C 200 bar | CO2 with ethanol simultaneously extracted β-carotene and zeaxanthin. | [118] |
| Arthrospira platensis f | CO2 and 26.7% ethanol | Air drying and milling | 60 | 150 | 0.83 | Total carotenoids 283 mg/g algae e | MAE resulted in better extraction yield than SFE. | [126] |
| Seaweeds | ||||||||
| Undaria pinnatifida | CO2 and ethanol | Freeze drying | 50 | 200 | 1 | Fucoxanthin 7.53 mg/g dw | Yield was dependent on pressure and temperature combination. | [19] |
| Undaria pinnatifida | CO2 | Milling and microwave-assisted cell disruption | 40 | 400 | 3 | Fucoxanthin 38.5 mg/g e | MW pretreatment increased fucoxanthin yield. | [20] |
| Sargassum muticum | CO2 and ethanol | Freeze drying and comminutating | 50 | 200 | 1 | Fucoxanthin ~0.12 mg/g algae dw | Use of co-solvent increased fucoxanthin yield by 90 times. | [29] |
| Undaria pinnatifida | CO2 and 3.23% ethanol | Drying | 60 | 400 | 3 | Fucoxanthin 0.9945 mg/g dw e | Use of co-solvent increased fucoxanthin yield by 10 times. | [23] |
| Undaria pinnatifida | CO2 | Drying | 60 | 400 | 2.5 | Fucoxanthin ~0.058 mg/g dw e | Pressure, temperature and extraction time affected fucoxanthin recovery. | [129] |
| Saccharina japonica | CO2 and ethanol | Freeze drying and grinding | 45 | 250 | 2 | Fucoxanthin 0.41 mg/g dw e | SFE process extracted a similar content of fucoxanthin as when acetone–methanol conventional extraction was used. | [137] |
| Sargassum horneri | Fucoxanthin 0.77 mg/g dw e | |||||||
| Saccharina japonica | CO2 and 2% sunflower oil | Freeze drying | 50.62 | 200 | 2 | Total carotenoids 2.391 mg/g dw e; fucoxanthin 1.421 mg/g dw e | Sunflower oil as a co-solvent found to be the most effective, than soybean oil, canola oil, ethanol, and water. | [26] |
| Extraction Method | Temperature (°C) | Pressure (MPa) | Ultrasound Power (W) | Time (h) | Lutein Yield (μg/g) |
|---|---|---|---|---|---|
| SE | 43 | 0.1 | 0 | 18 | 546.4 |
| SWE | 150 | 5 | 0 | 1/3 | 0 |
| SCE | 50 | 25 | 0 | 4 | 393.3 |
| SCCE | 27 | 21 | 0 | 4 | 422.9 |
| SCCE with pretreatment | 27 | 21 | 0 | 4 (+3 h pretreatment) | 921.5 |
| USCCE with pretreatment | 27 | 21 | 1000 | 4 (+3 h pretreatment) | 1240.1 |
| Extraction Techniques | Time (h) | Temperature (°C) | Pressure (MPa) | Fucoxanthin Yield (μg/g) |
|---|---|---|---|---|
| Ethanol (Soxhlet) | 12 | 78 | ND | 50 |
| Liquefied DME | 0.72 | 25 | ND | 390 |
| Supercritical CO2 | 3 | 60 | 40 | 60.12 |
| 3 | 70 | 40 | 59.51 | |
| Supercritical CO2 with entrainer (3.23%) | 3 | 60 | 40 | 994.53 |
| Microalgae | Extraction Condition | Carotenoid Yield/Recovery | Notes | Reference | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Solvent a | Cell Concentration (g Cells Dry Weight/L) | Pretreatment | f b (kHz) | P c (W) | t d (min) | E e (kJ/kg) | T f(°C) | ||||
| Chlamydomonas reinhardtii | Water | 1.5 | Frozen cells with glycerol, thawing and suspension in artificial seawater | 20 | 2200 | 0.17 or 0.5 min at various amplitudes | 0–450 | N/A | Carotenoids—0.3 carotenoids/mg cells | 91%–95% disruption; 80 kJ/kg regardless of cell concentration | [154] |
| Chlorella pyrenoidosa | Subcritical CO2 at 5–35 MPa | N/A | no treatment, ethanol soaking or enzymatic pretreatment | 20–24 | 0–19 W/cm2; 0–1000 W 15–45 kg/h, time 0–6 h, | 15–33 | Lutein—87–124 mg lutein/100 g Chlorella | Ultrasound-enhanced subcritical CO2 extraction | [138] | ||
| Haematococcus pluvialis | Ethanol and ethyl acetate | 50 | From dried algae | 40 | 200 | 10–20 | 120–240 | 30–50 | Astaxanthin—27 mg/g | US led to higher astaxanthin compared with conventional treatment | [155] |
| Chlorella vulgaris | Ethanol (90%) | N/A (31 mL solid/g solvent) | With or without enzymatic pre-treatment, 50 °C | 35 | 56 W/cm2 | 60–240 | N/A | 37 | Lutein—3.16–3.36 mg/g wet weight | Highest ultrasound-based extraction was with enzymatic pre-treatment | [156] |
| Cylindrotheca closterium (bacillariophyte) | Acetone | N/A, 30 mL | Freeze dried | N/A | 4.3–12.2 | 3–15 | 25–350 | 8.5 | Fucoxanthin 3.5–4.5 g/mg | - | [144] |
| Dunaliella salina | Water | N/A | None | 20, 580, 864 and 1146 | 32.3, 3, 20, 60 | 30 | 5.4 | 15–20 | Carotenoids (yield not reported) | Inactivation efficiency 20 < 580 = 864 < 1146 kHz | [157] |
| Dunaliella tertiolecta (chlorophyte) | Water | 30 mL | None | N/A | 4.3–12.2 | 3–15 | 25–350 | 8.5 | β-carotene—5 mg/g | [144] | |
| Haematococcus pluvialis | Methanol, ethanol, acetonitrile, acetone | 0.1 g/30 mL | None | 38.5 | 18.4 | 0–90 | 2000 | 30–60 | Astaxanthin—73% recovery | 55%–60% yield increase of astaxanthin after US | [145] |
| Spirulina platensis | n-heptane, diethyl ether and hexane | 10–60 g/L solvent | Spray dried spirulina mixed with methanol and kept fat various times | 20 | 50–165 W (167 W/cm2) | 8 min with cycling | 220 kWh/m3 | 10–50 | β-carotene—0.8–1.0 mg/g | Extraction had variable increase with acoustic intensity. | [158] |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Poojary, M.M.; Barba, F.J.; Aliakbarian, B.; Donsì, F.; Pataro, G.; Dias, D.A.; Juliano, P. Innovative Alternative Technologies to Extract Carotenoids from Microalgae and Seaweeds. Mar. Drugs 2016, 14, 214. https://doi.org/10.3390/md14110214
Poojary MM, Barba FJ, Aliakbarian B, Donsì F, Pataro G, Dias DA, Juliano P. Innovative Alternative Technologies to Extract Carotenoids from Microalgae and Seaweeds. Marine Drugs. 2016; 14(11):214. https://doi.org/10.3390/md14110214
Chicago/Turabian StylePoojary, Mahesha M., Francisco J. Barba, Bahar Aliakbarian, Francesco Donsì, Gianpiero Pataro, Daniel A. Dias, and Pablo Juliano. 2016. "Innovative Alternative Technologies to Extract Carotenoids from Microalgae and Seaweeds" Marine Drugs 14, no. 11: 214. https://doi.org/10.3390/md14110214
APA StylePoojary, M. M., Barba, F. J., Aliakbarian, B., Donsì, F., Pataro, G., Dias, D. A., & Juliano, P. (2016). Innovative Alternative Technologies to Extract Carotenoids from Microalgae and Seaweeds. Marine Drugs, 14(11), 214. https://doi.org/10.3390/md14110214










