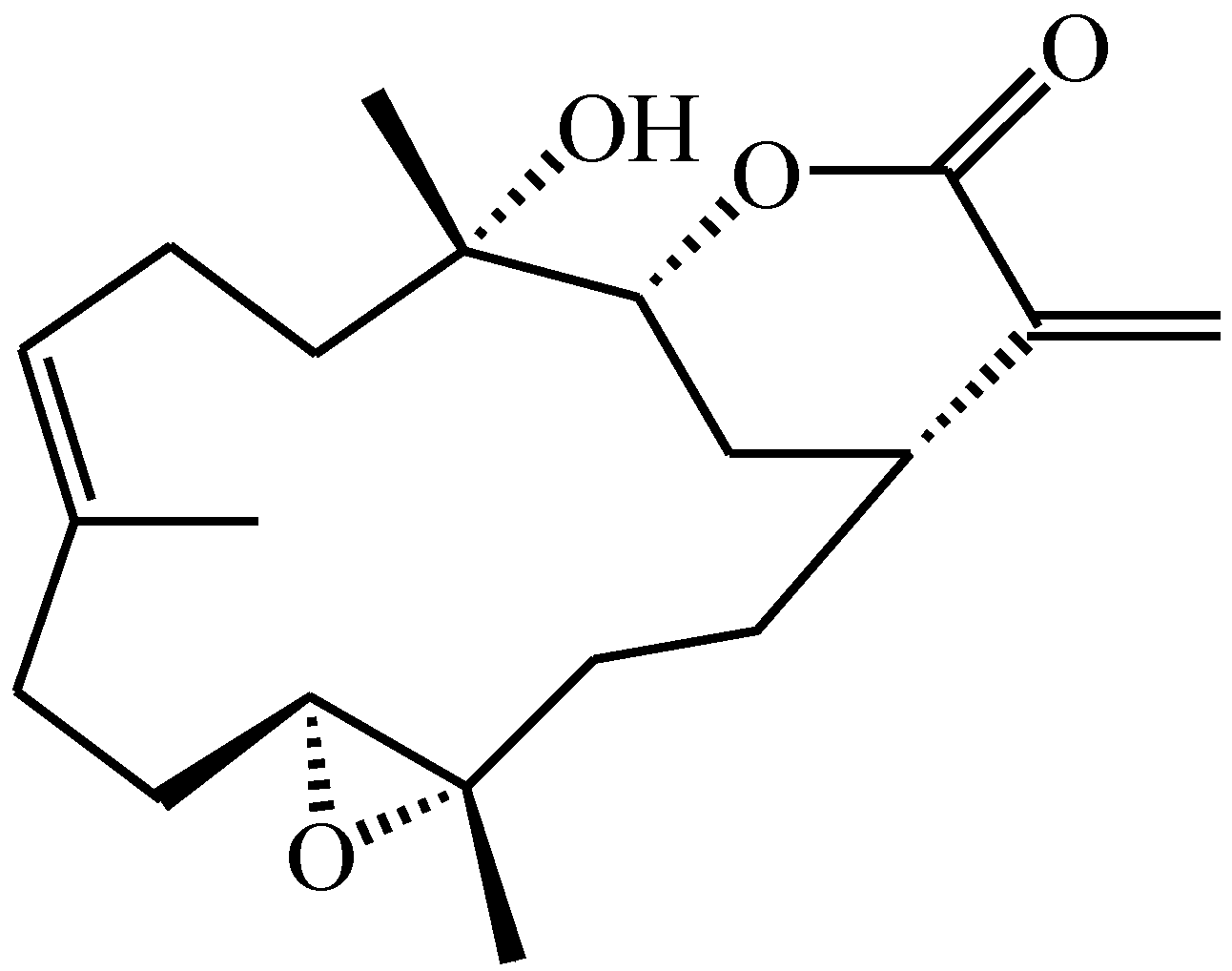
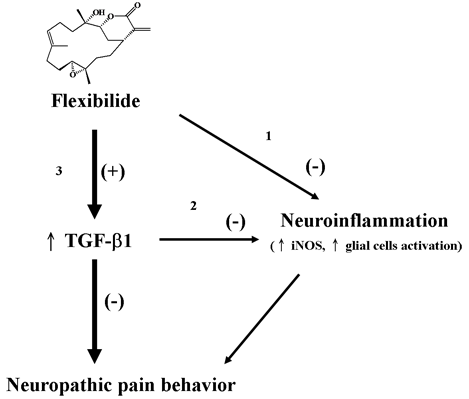Flexibilide Obtained from Cultured Soft Coral Has Anti-Neuroinflammatory and Analgesic Effects through the Upregulation of Spinal Transforming Growth Factor-β1 in Neuropathic Rats
Abstract
:1. Introduction
2. Results
2.1. Flexibilide Attenuates the CCI-Induced Thermal Hyperalgesia
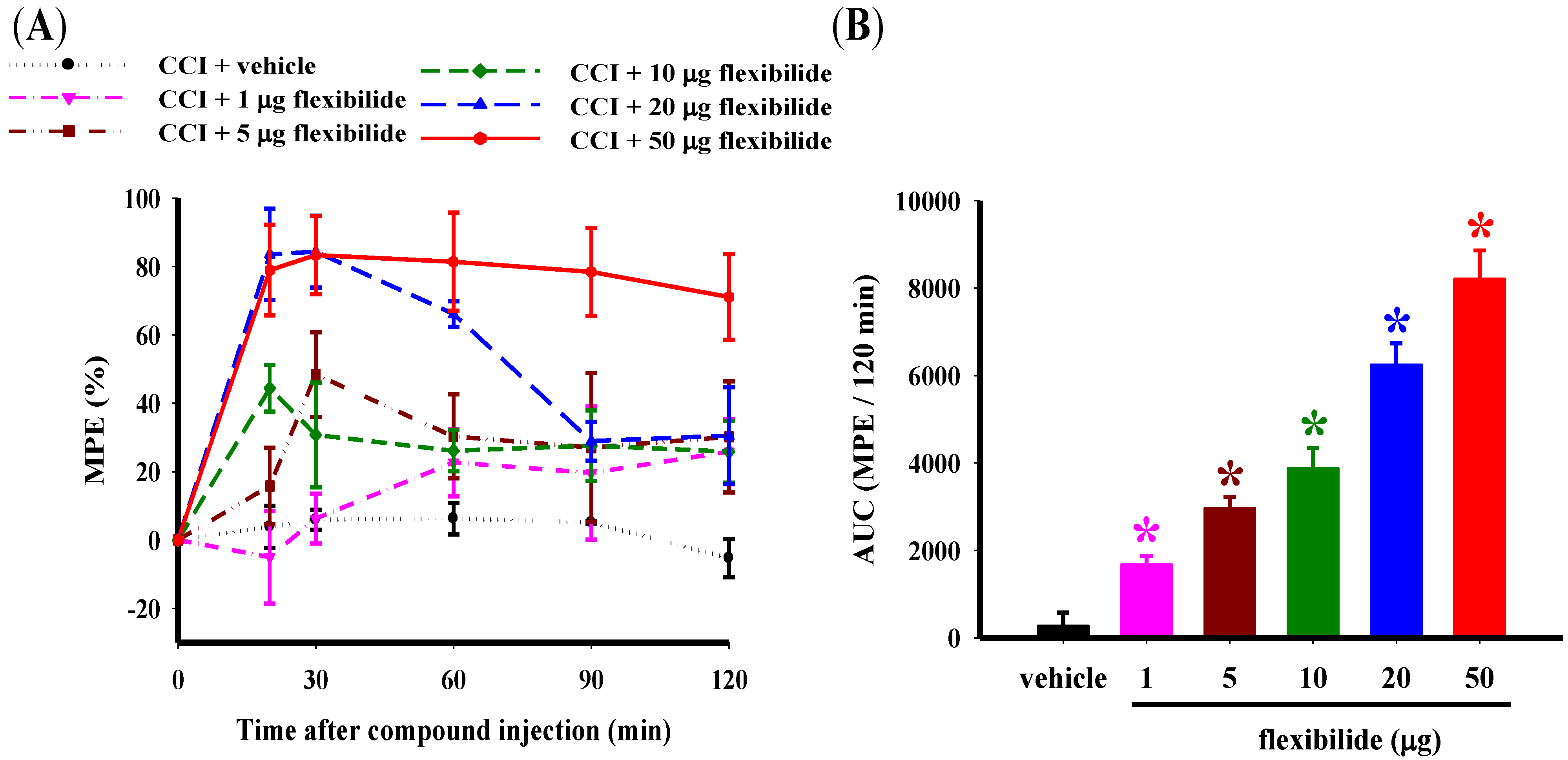
2.2. Prophylactic i.t. Flexibilide Prevents the Development of CCI-Induced Thermal Hyperalgesia and Weight-Bearing Deficits

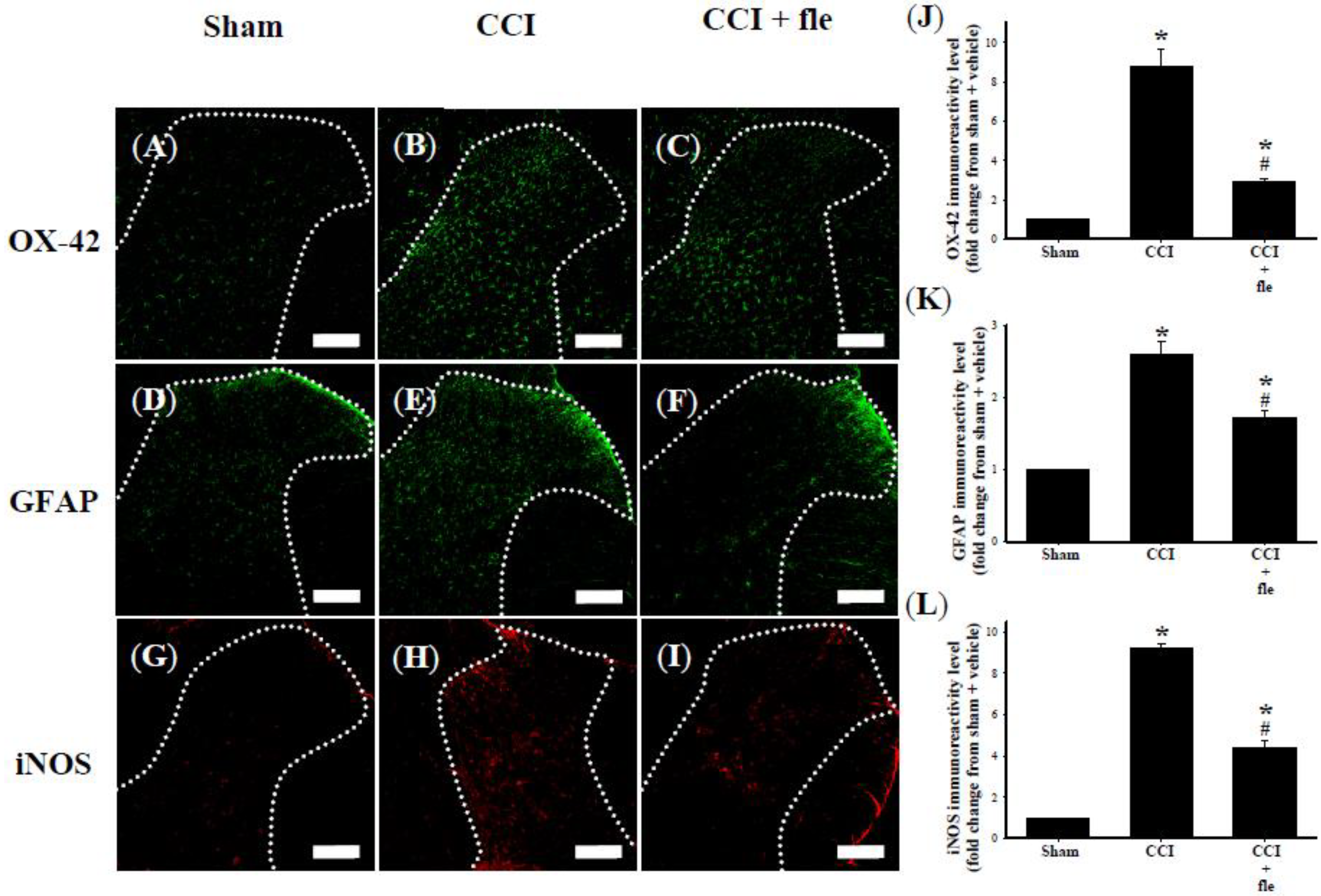
2.3. Flexibilide Inhibits CCI-Induced Spinal Neuroinflammation
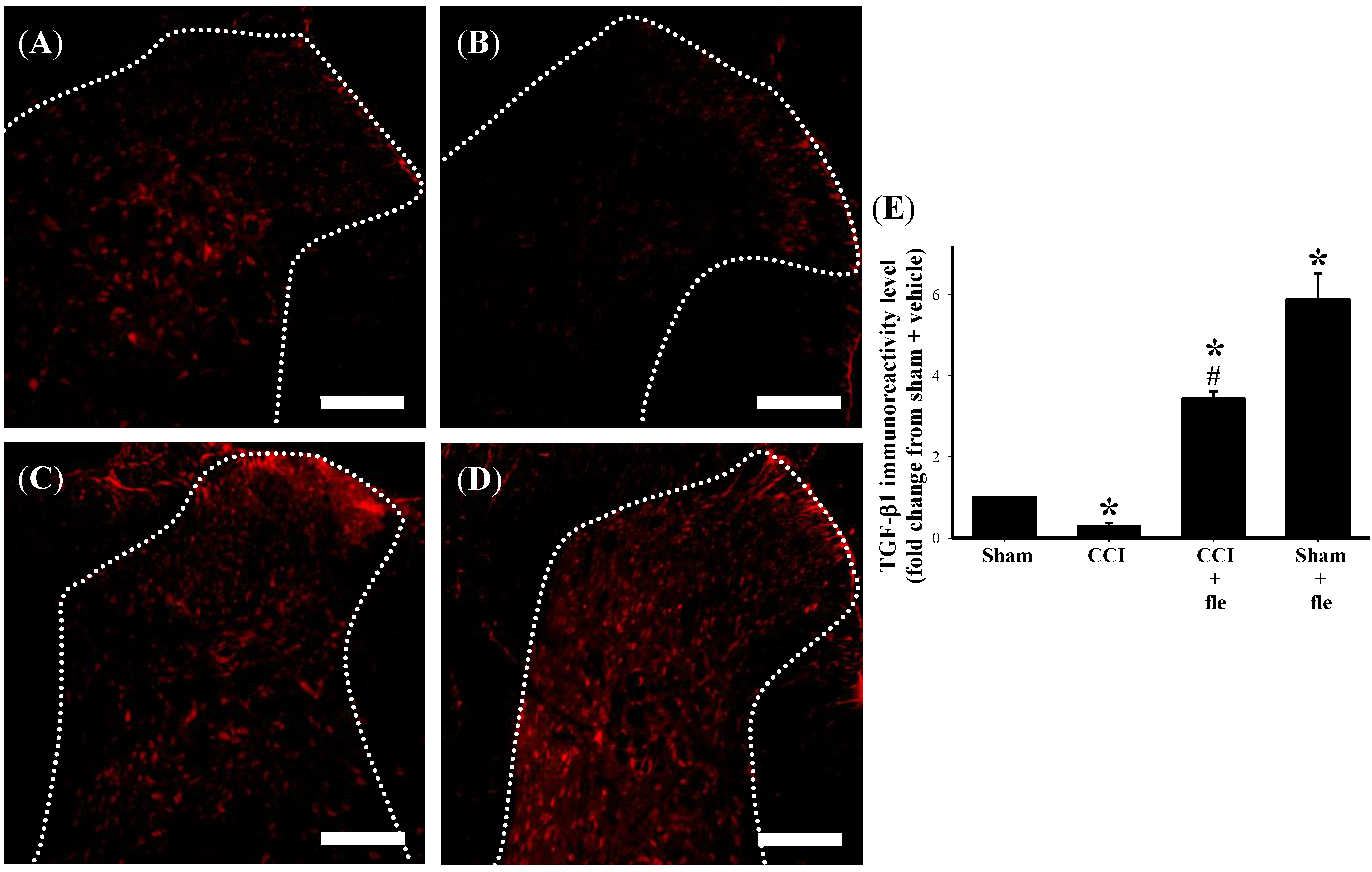
2.4. Flexibilide Attenuates the CCI-Induced Downregulation of TGF-β1

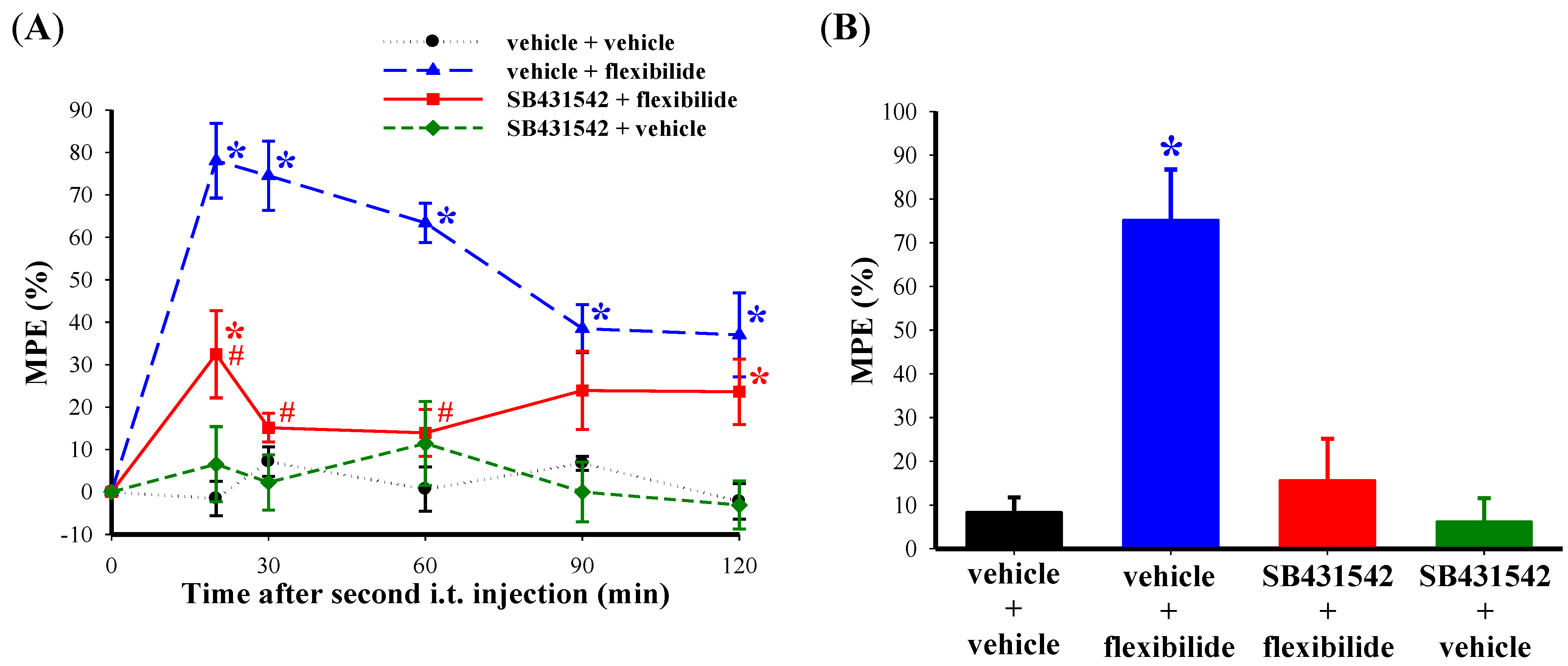
2.5. The Antinociceptive Effect of Flexibilide is Inhibited by a TGF-β Type I Receptor (TGF-βRI) Inhibitor
3. Discussion
3.1. Summary
3.2. Analgesic Effects of i.t. Flexibilide in Neuropathic Pain
3.3. The Effect of Flexibilide on Glial Cell Activation
3.4. The Role of TGF-β in the Spinal Neuroinflammation Induced Neuropathic Pain
3.5. The Impact of TGF-β on the Analgesic Effects of Flexibilide
3.6. The Relationship between Flexibilide, TGF-β1 and iNOS
3.7. Possible Mechanism of Flexibilide in Treating Neuropathic Pain

3.8. The Advantages of Flexibilide Obtained from Cultivated Soft Coral
4. Methods and Materials
4.1. Preparation of Flexibilide
4.2. Animals
4.3. I.t. Catheter Implantation and Induction of Peripheral Neuropathy
4.4. Effects of i.t. TGF-β1 Injection on CCI-Induced Gait Abnormalities and Nociception
- (1)
- To measure the antinociceptive effects of different doses of flexibilide on CCI-induced neuropathy, the following six treatments were administered at 14 days post-CCI:
- i.t. vehicle
- i.t. flexibilide (1 μg)
- i.t. flexibilide (5 μg)
- i.t. flexibilide (10 μg)
- i.t. flexibilide (20 μg)
- i.t. flexibilide (50 μg)
- (2)
- In order to explore the effect of preventative i.t. flexibilide treatment on the development of neuropathic pain, the following two treatment groups were used:
- i.t. vehicle (10 μg, twice daily for 4 weeks immediately after CCI surgery)
- i.t. flexibilide (10 μg, twice daily for 4 weeks immediately after CCI surgery)
- (3)
- In order to explore the effect of SB431542 (catalog no. S4317; Sigma Co., Ltd., St Louis, MO, USA), a selective inhibitor of the TGF-βRI [73], on the antinociceptive effects of flexibilide in CCI-induced neuropathy, the following four groups of rats were used at 14 days post-CCI:
- i.t. vehicle + vehicle (60 min after the first injection);
- i.t. vehicle + flexibilide (20 μg)
- i.t. SB431542 (2 μg) + flexibilide (20 μg)
- i.t. SB431542 (2 μg) + vehicle
4.5. Spinal Immunohistofluorescence
- (1)
- sham-operated plus i.t. vehicle (administered twice daily for 2 consecutive weeks immediately after sham surgery)
- (2)
- CCI plus i.t. vehicle (administered twice daily for 2 consecutive weeks immediately after CCI surgery)
- (3)
- CCI plus i.t. flexibilide (administered twice daily for 2 consecutive weeks immediately after CCI surgery)
- (4)
- sham-operated plus i.t. flexibilide (administered twice daily for 2 consecutive weeks immediately after sham operated).
4.6. Spinal Western Blot Analysis
- (1)
- sham-operated plus i.t. vehicle
- (2)
- CCI plus i.t. vehicle
- (3)
- CCI plus i.t. flexibilide
- (4)
- sham-operated plus i.t. vehicle
- (5)
- sham-operated plus i.t. flexibilide
4.7. Data and Statistical Analyses
5. Conclusions
Supplementary Files
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Mayer, A.M.; Rodriguez, A.D.; Taglialatela-Scafati, O.; Fusetani, N. Marine pharmacology in 2009–2011: Marine compounds with antibacterial, antidiabetic, antifungal, anti-inflammatory, antiprotozoal, antituberculosis, and antiviral activities; affecting the immune and nervous systems, and other miscellaneous mechanisms of action. Mar. Drugs 2013, 11, 2510–2573. [Google Scholar] [CrossRef] [Green Version]
- Liu, Z.; Xu, P.; Yu, L.; Zeng, W. Microtubule-targeting anticancer agents from marine natural source. Anticancer Agents Med. Chem. 2014, 14, 409–417. [Google Scholar] [CrossRef]
- Shi, D.; Guo, S.; Jiang, B.; Guo, C.; Wang, T.; Zhang, L.; Li, J. HPN, a synthetic analogue of bromophenol from red alga Rhodomela confervoides: Synthesis and anti-diabetic effects in c57bl/ksj-db/db mice. Mar. Drugs 2013, 11, 350–362. [Google Scholar] [CrossRef]
- Chaves Lde, S.; Nicolau, L.A.; Silva, R.O.; Barros, F.C.; Freitas, A.L.; Aragao, K.S.; Ribeiro Rde, A.; Souza, M.H.; Barbosa, A.L.; Medeiros, J.V. Antiinflammatory and antinociceptive effects in mice of a sulfated polysaccharide fraction extracted from the marine red algae Gracilaria caudata. Immunopharmacol. Immunotoxicol. 2013, 35, 93–100. [Google Scholar] [CrossRef]
- Chatter, R.; Ben Othman, R.; Rabhi, S.; Kladi, M.; Tarhouni, S.; Vagias, C.; Roussis, V.; Guizani-Tabbane, L.; Kharrat, R. In vivo and in vitro anti-inflammatory activity of neorogioltriol, a new diterpene extracted from the red algae Laurencia glandulifera. Mar. Drugs 2011, 9, 1293–1306. [Google Scholar] [CrossRef]
- Huang, S.Y.; Chen, N.F.; Chen, W.F.; Hung, H.C.; Lee, H.P.; Lin, Y.Y.; Wang, H.M.; Sung, P.J.; Sheu, J.H.; Wen, Z.H. Sinularin from indigenous soft coral attenuates nociceptive responses and spinal neuroinflammation in carrageenan-induced inflammatory rat model. Mar. Drugs 2012, 10, 1899–1919. [Google Scholar] [CrossRef]
- Martins, A.; Vieira, H.; Gaspar, H.; Santos, S. Marketed marine natural products in the pharmaceutical and cosmeceutical industries: Tips for success. Mar. Drugs 2014, 12, 1066–1101. [Google Scholar] [CrossRef]
- Rahman, H.; Austin, B.; Mitchell, W.J.; Morris, P.C.; Jamieson, D.J.; Adams, D.R.; Spragg, A.M.; Schweizer, M. Novel anti-infective compounds from marine bacteria. Mar. Drugs 2010, 8, 498–518. [Google Scholar] [CrossRef]
- Essack, M.; Bajic, V.B.; Archer, J.A. Conotoxins that confer therapeutic possibilities. Mar. Drugs 2012, 10, 1244–1265. [Google Scholar] [CrossRef]
- Kijjoa, A.; Sawangwong, P. Drugs and cosmetics from the sea. Mar. Drugs 2004, 2, 73–82. [Google Scholar] [CrossRef]
- Harden, N.; Cohen, M. Unmet needs in the management of neuropathic pain. J. Pain Symptom Manag. 2003, 25, S12–S17. [Google Scholar] [CrossRef]
- Ellis, A.; Bennett, D.L. Neuroinflammation and the generation of neuropathic pain. Br. J. Anaesth. 2013, 111, 26–37. [Google Scholar] [CrossRef]
- Moalem, G.; Tracey, D.J. Immune and inflammatory mechanisms in neuropathic pain. Brain Res. Rev. 2006, 51, 240–264. [Google Scholar] [CrossRef]
- Sastre, M.; Klockgether, T.; Heneka, M.T. Contribution of inflammatory processes to Alzheimer’s disease: Molecular mechanisms. Int. J. Dev. Neurosci. 2006, 24, 167–176. [Google Scholar] [CrossRef]
- Block, M.L.; Hong, J.S. Microglia and inflammation-mediated neurodegeneration: Multiple triggers with a common mechanism. Prog. Neurobiol. 2005, 76, 77–98. [Google Scholar] [CrossRef]
- Ji, R.R.; Suter, M.R. p38 MAPK, microglial signaling, and neuropathic pain. Mol. Pain 2007, 3, 33. [Google Scholar] [CrossRef]
- Zhuang, Z.Y.; Gerner, P.; Woolf, C.J.; Ji, R.R. ERK is sequentially activated in neurons, microglia, and astrocytes by spinal nerve ligation and contributes to mechanical allodynia in this neuropathic pain model. Pain 2005, 114, 149–159. [Google Scholar] [CrossRef]
- Jha, M.K.; Jeon, S.; Suk, K. Glia as a link between neuroinflammation and neuropathic pain. Immune Netw. 2012, 12, 41–47. [Google Scholar] [CrossRef]
- Vallejo, R.; Tilley, D.M.; Vogel, L.; Benyamin, R. The role of glia and the immune system in the development and maintenance of neuropathic pain. Pain Pract. 2010, 10, 167–184. [Google Scholar] [CrossRef]
- Raghavendra, V.; Tanga, F.; DeLeo, J.A. Inhibition of microglial activation attenuates the development but not existing hypersensitivity in a rat model of neuropathy. J. Pharmacol. Exp. Ther. 2003, 306, 624–630. [Google Scholar] [CrossRef]
- Ji, R.R.; Kawasaki, Y.; Zhuang, Z.Y.; Wen, Y.R.; Decosterd, I. Possible role of spinal astrocytes in maintaining chronic pain sensitization: Review of current evidence with focus on bFGF/JNK pathway. Neuron Glia Biol. 2006, 2, 259–269. [Google Scholar] [CrossRef]
- Watkins, L.R.; Maier, S.F. Glia: A novel drug discovery target for clinical pain. Nat. Rev. Drug Discov. 2003, 2, 973–985. [Google Scholar] [CrossRef]
- Zhang, X.; Xu, Y.; Wang, J.; Zhou, Q.; Pu, S.; Jiang, W.; Du, D. The effect of intrathecal administration of glial activation inhibitors on dorsal horn BDNF overexpression and hind paw mechanical allodynia in spinal nerve ligated rats. J. Neural Transm. 2012, 119, 329–336. [Google Scholar] [CrossRef]
- Ledeboer, A.; Sloane, E.M.; Milligan, E.D.; Frank, M.G.; Mahony, J.H.; Maier, S.F.; Watkins, L.R. Minocycline attenuates mechanical allodynia and proinflammatory cytokine expression in rat models of pain facilitation. Pain 2005, 115, 71–83. [Google Scholar] [CrossRef]
- Jean, Y.H.; Chen, W.F.; Sung, C.S.; Duh, C.Y.; Huang, S.Y.; Lin, C.S.; Tai, M.H.; Tzeng, S.F.; Wen, Z.H. Capnellene, a natural marine compound derived from soft coral, attenuates chronic constriction injury-induced neuropathic pain in rats. Br. J. Pharmacol. 2009, 158, 713–725. [Google Scholar] [CrossRef]
- Lin, Y.C.; Huang, S.Y.; Jean, Y.H.; Chen, W.F.; Sung, C.S.; Kao, E.S.; Wang, H.M.; Chakraborty, C.; Duh, C.Y.; Wen, Z.H. Intrathecal lemnalol, a natural marine compound obtained from Formosan soft coral, attenuates nociceptive responses and the activity of spinal glial cells in neuropathic rats. Behav. Pharmacol. 2011, 22, 739–750. [Google Scholar] [CrossRef]
- Sweitzer, S.M.; Schubert, P.; DeLeo, J.A. Propentofylline, a glial modulating agent, exhibits antiallodynic properties in a rat model of neuropathic pain. J. Pharmacol. Exp. Ther. 2001, 297, 1210–1217. [Google Scholar]
- Levy, D.; Hoke, A.; Zochodne, D.W. Local expression of inducible nitric oxide synthase in an animal model of neuropathic pain. Neurosci. Lett. 1999, 260, 207–209. [Google Scholar] [CrossRef]
- Verge, V.M.; Xu, Z.; Xu, X.J.; Wiesenfeld-Hallin, Z.; Hokfelt, T. Marked increase in nitric oxide synthase mRNA in rat dorsal root ganglia after peripheral axotomy: In situ hybridization and functional studies. Proc. Natl. Acad. Sci. USA 1992, 89, 11617–11621. [Google Scholar] [CrossRef]
- Kitto, K.F.; Haley, J.E.; Wilcox, G.L. Involvement of nitric oxide in spinally mediated hyperalgesia in the mouse. Neurosci. Lett. 1992, 148, 1–5. [Google Scholar] [CrossRef]
- Sousa, A.M.; Prado, W.A. The dual effect of a nitric oxide donor in nociception. Brain Res. 2001, 897, 9–19. [Google Scholar] [CrossRef]
- Masue, T.; Dohi, S.; Asano, T.; Shimonaka, H. Spinal antinociceptive effect of epidural nonsteroidal antiinflammatory drugs on nitric oxide-induced hyperalgesia in rats. Anesthesiology 1999, 91, 198–206. [Google Scholar] [CrossRef]
- Hervera, A.; Leanez, S.; Negrete, R.; Motterlini, R.; Pol, O. Carbon monoxide reduces neuropathic pain and spinal microglial activation by inhibiting nitric oxide synthesis in mice. PLoS One 2012, 7, e43693. [Google Scholar]
- Lawrence, D.A. Transforming growth factor-beta: A general review. Eur. Cytokine Netw. 1996, 7, 363–374. [Google Scholar]
- Grande, J.P. Role of transforming growth factor-beta in tissue injury and repair. Proc. Soc. Exp. Biol. Med. 1997, 214, 27–40. [Google Scholar] [CrossRef]
- Dobolyi, A.; Vincze, C.; Pal, G.; Lovas, G. The neuroprotective functions of transforming growth factor beta proteins. Int. J. Mol. Sci. 2012, 13, 8219–8258. [Google Scholar] [CrossRef] [Green Version]
- Letterio, J.J.; Roberts, A.B. TGF-beta: A critical modulator of immune cell function. Clin. Immunol. Immunopathol. 1997, 84, 244–250. [Google Scholar] [CrossRef]
- Chen, W.; Jin, W.; Cook, M.; Weiner, H.L.; Wahl, S.M. Oral delivery of group a streptococcal cell walls augments circulating TGF-beta and suppresses streptococcal cell wall arthritis. J. Immunol. 1998, 161, 6297–6304. [Google Scholar]
- Jin, Y.X.; Xu, L.Y.; Guo, H.; Ishikawa, M.; Link, H.; Xiao, B.G. TGF-beta1 inhibits protracted-relapsing experimental autoimmune encephalomyelitis by activating dendritic cells. J. Autoimmun. 2000, 14, 213–220. [Google Scholar] [CrossRef]
- Jones, L.L.; Kreutzberg, G.W.; Raivich, G. Transforming growth factor beta’s 1, 2 and 3 inhibit proliferation of ramified microglia on an astrocyte monolayer. Brain Res. 1998, 795, 301–306. [Google Scholar] [CrossRef]
- Makwana, M.; Jones, L.L.; Cuthill, D.; Heuer, H.; Bohatschek, M.; Hristova, M.; Friedrichsen, S.; Ormsby, I.; Bueringer, D.; Koppius, A.; et al. Endogenous transforming growth factor beta 1 suppresses inflammation and promotes survival in adult CNS. J. Neurosci. 2007, 27, 11201–11213. [Google Scholar] [CrossRef]
- Vergeli, M.; Mazzanti, B.; Ballerini, C.; Gran, B.; Amaducci, L.; Massacesi, L. Transforming growth factor-beta 1 inhibits the proliferation of rat astrocytes induced by serum and growth factors. J. Neurosci. Res. 1995, 40, 127–133. [Google Scholar] [CrossRef]
- Echeverry, S.; Shi, X.Q.; Haw, A.; Liu, H.; Zhang, Z.W.; Zhang, J. Transforming growth factor-beta1 impairs neuropathic pain through pleiotropic effects. Mol. Pain 2009, 5, 16. [Google Scholar] [CrossRef]
- Chen, N.F.; Huang, S.Y.; Chen, W.F.; Chen, C.H.; Lu, C.H.; Chen, C.L.; Yang, S.N.; Wang, H.M.; Wen, Z.H. TGF-beta1 attenuates spinal neuroinflammation and the excitatory amino acid system in rats with neuropathic pain. J. Pain 2013, 14, 1671–1685. [Google Scholar] [CrossRef]
- Weinheimer, A.J.; Matson, J.A.; Hossain, M.B.; van der Helm, D. Marine anticancer agents: Sinularin and dihydrosinularin, new cembranolides from the soft coral, Sinularia flexibilis. Tetrahedron Lett. 1977, 18, 2923–2926. [Google Scholar] [CrossRef]
- Kazlauskas, R.; Murphy, P.T.; Wells, R.J.; Schonholzer, P.; Coll, J.C. Cembranoid constituents from an Australian collection of the soft coral Sinularia flexibilis. Aust. J. Chem. 1978, 31, 1817–1824. [Google Scholar] [CrossRef]
- Buckle, P.J.; Baldo, B.A.; Taylor, K.M. The anti-inflammatory activity of marine natural products—6-n-tridecylsalicylic acid, flexibilide and dendalone 3-hydroxybutyrate. Agents Actions 1980, 10, 361–367. [Google Scholar] [CrossRef]
- Woolf, C.J.; Mannion, R.J. Neuropathic pain: Aetiology, symptoms, mechanisms, and management. Lancet 1999, 353, 1959–1964. [Google Scholar] [CrossRef]
- Zimmermann, M. Pathobiology of neuropathic pain. Eur. J. Pharmacol. 2001, 429, 23–37. [Google Scholar]
- Baron, R. Mechanisms of disease: Neuropathic pain—A clinical perspective. Nat. Clin. Pract. Neurol. 2006, 2, 95–106. [Google Scholar] [CrossRef]
- Kauppila, T.; Kontinen, V.K.; Pertovaara, A. Weight bearing of the limb as a confounding factor in assessment of mechanical allodynia in the rat. Pain 1998, 74, 55–59. [Google Scholar] [CrossRef]
- Wang, L.X.; Wang, Z.J. Animal and cellular models of chronic pain. Adv. Drug Deliv. Rev. 2003, 55, 949–965. [Google Scholar]
- Milligan, E.D.; Watkins, L.R. Pathological and protective roles of glia in chronic pain. Nat. Rev. Neurosci. 2009, 10, 23–36. [Google Scholar] [CrossRef]
- Watkins, L.R.; Milligan, E.D.; Maier, S.F. Glial activation: A driving force for pathological pain. Trends Neurosci. 2001, 24, 450–455. [Google Scholar] [CrossRef]
- Cao, H.; Zhang, Y.Q. Spinal glial activation contributes to pathological pain states. Neurosci. Biobehav. Rev. 2008, 32, 972–983. [Google Scholar] [CrossRef]
- Coyle, D.E. Partial peripheral nerve injury leads to activation of astroglia and microglia which parallels the development of allodynic behavior. Glia 1998, 23, 75–83. [Google Scholar] [CrossRef]
- Raghavendra, V.; Tanga, F.Y.; DeLeo, J.A. Complete freunds adjuvant-induced peripheral inflammation evokes glial activation and proinflammatory cytokine expression in the CNS. Eur. J. Neurosci. 2004, 20, 467–473. [Google Scholar] [CrossRef]
- Shibata, K.; Sugawara, T.; Fujishita, K.; Shinozaki, Y.; Matsukawa, T.; Suzuki, T.; Koizumi, S. The astrocyte-targeted therapy by Bushi for the neuropathic pain in mice. PLoS One 2011, 6, e23510. [Google Scholar]
- Mika, J.; Osikowicz, M.; Rojewska, E.; Korostynski, M.; Wawrzczak-Bargiela, A.; Przewlocki, R.; Przewlocka, B. Differential activation of spinal microglial and astroglial cells in a mouse model of peripheral neuropathic pain. Eur. J. Pharmacol. 2009, 623, 65–72. [Google Scholar] [CrossRef]
- Dennler, S.; Goumans, M.J.; ten Dijke, P. Transforming growth factor beta signal transduction. J. Leukoc. Biol. 2002, 71, 731–740. [Google Scholar]
- Massague, J. TGF-beta signal transduction. Annu. Rev. Biochem. 1998, 67, 753–791. [Google Scholar] [CrossRef]
- Derynck, R.; Feng, X.H. TGF-beta receptor signaling. Biochim. Biophys. Acta 1997, 1333, F105–F150. [Google Scholar]
- Wrana, J.L.; Attisano, L.; Wieser, R.; Ventura, F.; Massague, J. Mechanism of activation of the TGF-beta receptor. Nature 1994, 370, 341–347. [Google Scholar] [CrossRef]
- Imai, K.; Takeshita, A.; Hanazawa, S. TGF-beta inhibits lipopolysaccharide-stimulated activity of c-Jun N-terminal kinase in mouse macrophages. FEBS Lett. 1999, 456, 375–378. [Google Scholar] [CrossRef]
- Chung, S.W.; Kwon, M.Y.; Kang, Y.H.; Chung, H.T.; Lee, S.J.; Kim, H.P.; Perrella, M.A. Transforming growth factor-beta1 suppression of endotoxin-induced heme oxygenase-1 in macrophages involves activation of Smad2 and downregulation of Ets-2. J. Cell. Physiol. 2012, 227, 351–360. [Google Scholar] [CrossRef]
- Imai, K.; Takeshita, A.; Hanazawa, S. Transforming growth factor-beta inhibits lipopolysaccharide-stimulated expression of inflammatory cytokines in mouse macrophages through downregulation of activation protein 1 and CD14 receptor expression. Infect. Immun. 2000, 68, 2418–2423. [Google Scholar] [CrossRef]
- Hausmann, E.H.; Hao, S.Y.; Pace, J.L.; Parmely, M.J. Transforming growth factor beta 1 and gamma interferon provide opposing signals to lipopolysaccharide-activated mouse macrophages. Infect. Immun. 1994, 62, 3625–3632. [Google Scholar]
- Suzumura, A.; Sawada, M.; Yamamoto, H.; Marunouchi, T. Transforming growth factor-beta suppresses activation and proliferation of microglia in vitro. J. Immunol. 1993, 151, 2150–2158. [Google Scholar]
- Prud’homme, G.J.; Piccirillo, C.A. The inhibitory effects of transforming growth factor-beta-1 (TGF-beta1) in autoimmune diseases. J. Autoimmun. 2000, 14, 23–42. [Google Scholar] [CrossRef]
- Ma, M.; Ma, Y.; Yi, X.; Guo, R.; Zhu, W.; Fan, X.; Xu, G.; Frey, W.H., II; Liu, X. Intranasal delivery of transforming growth factor-beta1 in mice after stroke reduces infarct volume and increases neurogenesis in the subventricular zone. BMC Neurosci. 2008, 9, 117. [Google Scholar] [CrossRef]
- Kuruvilla, A.P.; Shah, R.; Hochwald, G.M.; Liggitt, H.D.; Palladino, M.A.; Thorbecke, G.J. Protective effect of transforming growth factor beta 1 on experimental autoimmune diseases in mice. Proc. Natl. Acad. Sci. USA 1991, 88, 2918–2921. [Google Scholar] [CrossRef]
- Caraci, F.; Battaglia, G.; Bruno, V.; Bosco, P.; Carbonaro, V.; Giuffrida, M.L.; Drago, F.; Sortino, M.A.; Nicoletti, F.; Copani, A. TGF-beta1 pathway as a new target for neuroprotection in Alzheimer’s disease. CNS Neurosci. Ther. 2011, 17, 237–249. [Google Scholar] [CrossRef]
- Caraci, F.; Battaglia, G.; Busceti, C.; Biagioni, F.; Mastroiacovo, F.; Bosco, P.; Drago, F.; Nicoletti, F.; Sortino, M.A.; Copani, A. TGF-beta 1 protects against Abeta-neurotoxicity via the phosphatidylinositol-3-kinase pathway. Neurobiol. Dis. 2008, 30, 234–242. [Google Scholar] [CrossRef]
- Lai, Y.L.; Smith, P.M.; Lamm, W.J.; Hildebrandt, J. Sampling and analysis of cerebrospinal fluid for chronic studies in awake rats. J. Appl. Physiol. 1983, 54, 1754–1757. [Google Scholar]
- Inman, G.J.; Nicolas, F.J.; Callahan, J.F.; Harling, J.D.; Gaster, L.M.; Reith, A.D.; Laping, N.J.; Hill, C.S. SB-431542 is a potent and specific inhibitor of transforming growth factor-beta superfamily type I activin receptor-like kinase (ALK) receptors ALK4, ALK5, and ALK7. Mol. Pharmacol. 2002, 62, 65–74. [Google Scholar] [CrossRef]
- Guzik, T.J.; Korbut, R.; Adamek-Guzik, T. Nitric oxide and superoxide in inflammation and immune regulation. J. Physiol. Pharmacol. 2003, 54, 469–487. [Google Scholar]
- Roos, D.; van Bruggen, R.; Meischl, C. Oxidative killing of microbes by neutrophils. Microbes Infect. 2003, 5, 1307–1315. [Google Scholar] [CrossRef]
- Witko-Sarsat, V.; Rieu, P.; Descamps-Latscha, B.; Lesavre, P.; Halbwachs-Mecarelli, L. Neutrophils: Molecules, functions and pathophysiological aspects. Lab. Investig. 2000, 80, 617–653. [Google Scholar] [CrossRef]
- Hu, S.; Sheng, W.S.; Peterson, P.K.; Chao, C.C. Differential regulation by cytokines of human astrocyte nitric oxide production. Glia 1995, 15, 491–494. [Google Scholar] [CrossRef]
- Vilhardt, F. Microglia: Phagocyte and glia cell. Int. J. Biochem. Cell Biol. 2005, 37, 17–21. [Google Scholar] [CrossRef]
- Ledeboer, A.; Breve, J.J.; Poole, S.; Tilders, F.J.; van Dam, A.M. Interleukin-10, interleukin-4, and transforming growth factor-beta differentially regulate lipopolysaccharide-induced production of pro-inflammatory cytokines and nitric oxide in co-cultures of rat astroglial and microglial cells. Glia 2000, 30, 134–142. [Google Scholar] [CrossRef]
- Minghetti, L.; Levi, G. Microglia as effector cells in brain damage and repair: Focus on prostanoids and nitric oxide. Prog. Neurobiol. 1998, 54, 99–125. [Google Scholar] [CrossRef]
- Bredt, D.S.; Snyder, S.H. Nitric oxide: A physiologic messenger molecule. Annu. Rev. Biochem. 1994, 63, 175–195. [Google Scholar] [CrossRef]
- Levy, D.; Zochodne, D.W. No pain: Potential roles of nitric oxide in neuropathic pain. Pain Pract. 2004, 4, 11–18. [Google Scholar] [CrossRef]
- Lin, Q.; Palecek, J.; Paleckova, V.; Peng, Y.B.; Wu, J.; Cui, M.; Willis, W.D. Nitric oxide mediates the central sensitization of primate spinothalamic tract neurons. J. Neurophysiol. 1999, 81, 1075–1085. [Google Scholar]
- Tassorelli, C.; Greco, R.; Wang, D.; Sandrini, M.; Sandrini, G.; Nappi, G. Nitroglycerin induces hyperalgesia in rats—A time-course study. Eur. J. Pharmacol. 2003, 464, 159–162. [Google Scholar] [CrossRef]
- Takano, Y.; Kuno, Y.; Sato, E.; Takano, M.; Sato, I. Hyperalgesia induced by intrathecal administration of nitroglycerin involves NMDA receptor activation in the spinal cord. Masui 1997, 46, 1354–1361. [Google Scholar]
- Lui, P.W.; Lee, C.H. Preemptive effects of intrathecal cyclooxygenase inhibitor or nitric oxide synthase inhibitor on thermal hypersensitivity following peripheral nerve injury. Life Sci. 2004, 75, 2527–2538. [Google Scholar] [CrossRef]
- Motoyoshi-Yamashiro, A.; Tamura, M.; Moriyama, M.; Takano, K.; Kawabe, K.; Nakajima, H.; Katoh-Semba, R.; Furuichi, T.; Nakamura, Y. Activation of cultured astrocytes by amphotericin B: Stimulation of NO and cytokines production and changes in neurotrophic factors production. Neurochem. Int. 2013, 63, 93–100. [Google Scholar] [CrossRef]
- Sun, X.C.; Chen, W.N.; Li, S.Q.; Cai, J.S.; Li, W.B.; Xian, X.H.; Hu, Y.Y.; Zhang, M.; Li, Q.J. Fluorocitrate, an inhibitor of glial metabolism, inhibits the up-regulation of NOS expression, activity and no production in the spinal cord induced by formalin test in rats. Neurochem. Res. 2009, 34, 351–359. [Google Scholar] [CrossRef]
- Vodovotz, Y.; Geiser, A.G.; Chesler, L.; Letterio, J.J.; Campbell, A.; Lucia, M.S.; Sporn, M.B.; Roberts, A.B. Spontaneously increased production of nitric oxide and aberrant expression of the inducible nitric oxide synthase in vivo in the transforming growth factor beta 1 null mouse. J. Exp. Med. 1996, 183, 2337–2342. [Google Scholar] [CrossRef]
- Molinski, T.F.; Dalisay, D.S.; Lievens, S.L.; Saludes, J.P. Drug development from marine natural products. Nat. Rev. Drug Discov. 2009, 8, 69–85. [Google Scholar]
- Su, J.H.; Lin, Y.F.; Lu, Y.; Yeh, H.C.; Wang, W.H.; Fan, T.Y.; Sheu, J.H. Oxygenated cembranoids from the cultured and wild-type soft corals Sinularia flexibilis. Chem. Pharm. Bull. (Tokyo) 2009, 57, 1189–1192. [Google Scholar] [CrossRef]
- Lu, Y.; Huang, C.Y.; Lin, Y.F.; Wen, Z.H.; Su, J.H.; Kuo, Y.H.; Chiang, M.Y.; Sheu, J.H. Anti-inflammatory cembranoids from the soft corals Sinularia querciformis and Sinularia granosa. J. Nat. Prod. 2008, 71, 1754–1759. [Google Scholar] [CrossRef]
- Lu, Y.; Su, J.H.; Huang, C.Y.; Liu, Y.C.; Kuo, Y.H.; Wen, Z.H.; Hsu, C.H.; Sheu, J.H. Cembranoids from the soft corals Sinularia granosa and Sinularia querciformis. Chem. Pharm. Bull. (Tokyo) 2010, 58, 464–466. [Google Scholar] [CrossRef]
- Yaksh, T.L.; Rudy, T.A. Chronic catheterization of the spinal subarachnoid space. Physiol. Behav. 1976, 17, 1031–1036. [Google Scholar]
- Bennett, G.J.; Xie, Y.K. A peripheral mononeuropathy in rat that produces disorders of pain sensation like those seen in man. Pain 1988, 33, 87–107. [Google Scholar] [CrossRef]
- Wen, Z.H.; Chao, C.H.; Wu, M.H.; Sheu, J.H. A neuroprotective sulfone of marine origin and the in vivo anti-inflammatory activity of an analogue. Eur. J. Med. Chem. 2010, 45, 5998–6004. [Google Scholar] [CrossRef]
- Basso, D.M.; Beattie, M.S.; Bresnahan, J.C. A sensitive and reliable locomotor rating scale for open field testing in rats. J. Neurotrauma 1995, 12, 1–21. [Google Scholar] [CrossRef]
- Hains, B.C.; Waxman, S.G. Activated microglia contribute to the maintenance of chronic pain after spinal cord injury. J. Neurosci. 2006, 26, 4308–4317. [Google Scholar] [CrossRef]
- Hargreaves, K.; Dubner, R.; Brown, F.; Flores, C.; Joris, J. A new and sensitive method for measuring thermal nociception in cutaneous hyperalgesia. Pain 1988, 32, 77–88. [Google Scholar]
- Wen, Z.H.; Tang, C.C.; Chang, Y.C.; Huang, S.Y.; Hsieh, S.P.; Lee, C.H.; Huang, G.S.; Ng, H.F.; Neoh, C.A.; Hsieh, C.S.; et al. Glucosamine sulfate reduces experimental osteoarthritis and nociception in rats: Association with changes of mitogen-activated protein kinase in chondrocytes. Osteoarthr. Cartil. 2010, 18, 1192–1202. [Google Scholar] [CrossRef]
- Kobayashi, K.; Imaizumi, R.; Sumichika, H.; Tanaka, H.; Goda, M.; Fukunari, A.; Komatsu, H. Sodium iodoacetate-induced experimental osteoarthritis and associated pain model in rats. J. Vet. Med. Sci. 2003, 65, 1195–1199. [Google Scholar] [CrossRef]
- Rowland, M.; Tozer, T.N. Assessment of AUC. In Clinical Pharmacokinetics: Concepts and Applications, 3rd ed.; Balado, D., Ed.; Lippincott Williams and Wilkins: Philadelphia, PA, USA, 1995; pp. 469–470. [Google Scholar]
- Sung, B.; Lim, G.; Mao, J. Altered expression and uptake activity of spinal glutamate transporters after nerve injury contribute to the pathogenesis of neuropathic pain in rats. J. Neurosci. 2003, 23, 2899–2910. [Google Scholar]
- Jean, Y.H.; Chen, W.F.; Duh, C.Y.; Huang, S.Y.; Hsu, C.H.; Lin, C.S.; Sung, C.S.; Chen, I.M.; Wen, Z.H. Inducible nitric oxide synthase and cyclooxygenase-2 participate in anti-inflammatory and analgesic effects of the natural marine compound lemnalol from Formosan soft coral Lemnalia cervicorni. Eur. J. Pharmacol. 2008, 578, 323–331. [Google Scholar] [CrossRef]
- Chen, W.F.; Sung, C.S.; Jean, Y.H.; Su, T.M.; Wang, H.C.; Ho, J.T.; Huang, S.Y.; Lin, C.S.; Wen, Z.H. Suppressive effects of intrathecal granulocyte colony-stimulating factor on excessive release of excitatory amino acids in the spinal cerebrospinal fluid of rats with cord ischemia: Role of glutamate transporters. Neuroscience 2010, 165, 1217–1232. [Google Scholar] [CrossRef]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Chen, N.-F.; Huang, S.-Y.; Lu, C.-H.; Chen, C.-L.; Feng, C.-W.; Chen, C.-H.; Hung, H.-C.; Lin, Y.-Y.; Sung, P.-J.; Sung, C.-S.; et al. Flexibilide Obtained from Cultured Soft Coral Has Anti-Neuroinflammatory and Analgesic Effects through the Upregulation of Spinal Transforming Growth Factor-β1 in Neuropathic Rats. Mar. Drugs 2014, 12, 3792-3817. https://doi.org/10.3390/md12073792
Chen N-F, Huang S-Y, Lu C-H, Chen C-L, Feng C-W, Chen C-H, Hung H-C, Lin Y-Y, Sung P-J, Sung C-S, et al. Flexibilide Obtained from Cultured Soft Coral Has Anti-Neuroinflammatory and Analgesic Effects through the Upregulation of Spinal Transforming Growth Factor-β1 in Neuropathic Rats. Marine Drugs. 2014; 12(7):3792-3817. https://doi.org/10.3390/md12073792
Chicago/Turabian StyleChen, Nan-Fu, Shi-Ying Huang, Ching-Hsiang Lu, Chun-Lin Chen, Chien-Wei Feng, Chun-Hong Chen, Han-Chun Hung, Yen-You Lin, Ping-Jyun Sung, Chun-Sung Sung, and et al. 2014. "Flexibilide Obtained from Cultured Soft Coral Has Anti-Neuroinflammatory and Analgesic Effects through the Upregulation of Spinal Transforming Growth Factor-β1 in Neuropathic Rats" Marine Drugs 12, no. 7: 3792-3817. https://doi.org/10.3390/md12073792