Four New Briarane Diterpenoids from Taiwanese Gorgonian Junceella fragilis
Abstract
:1. Introduction
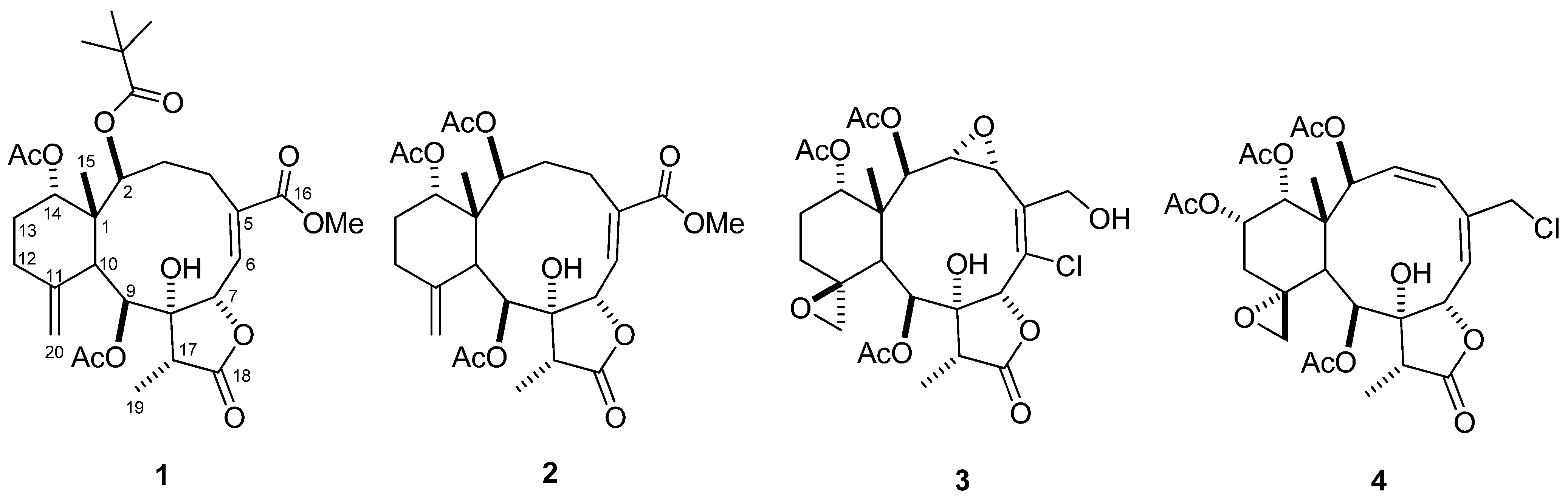
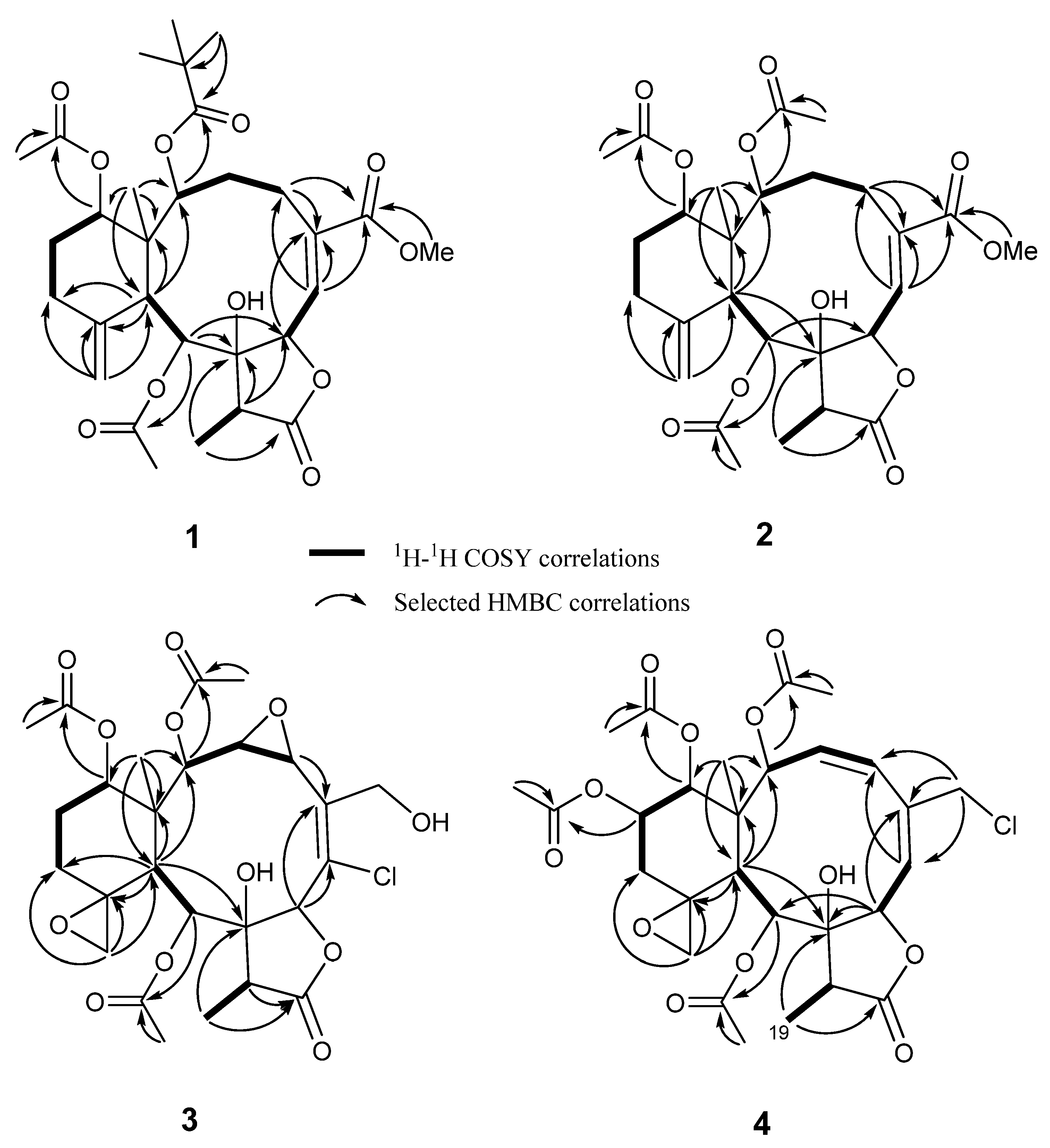
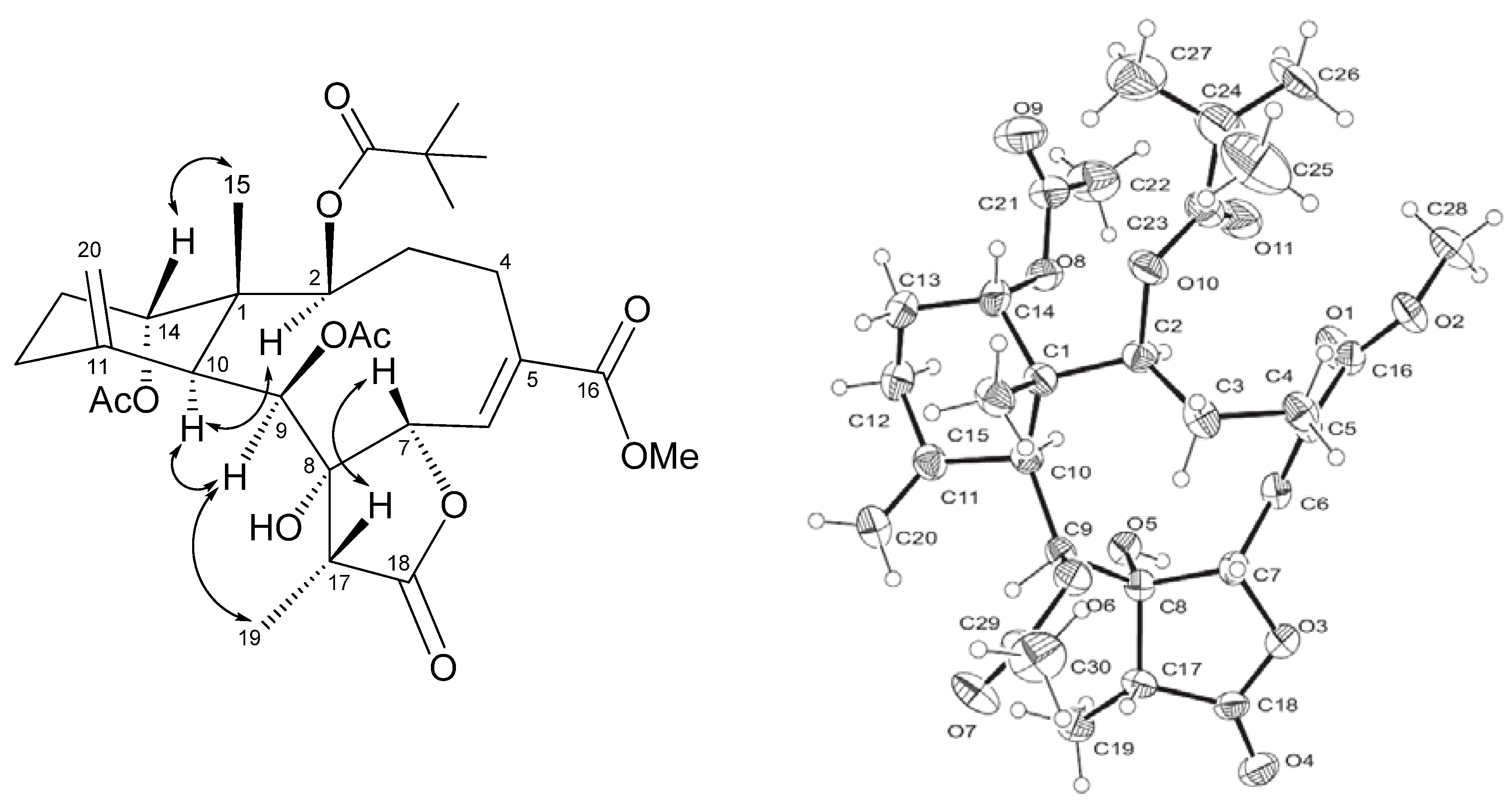
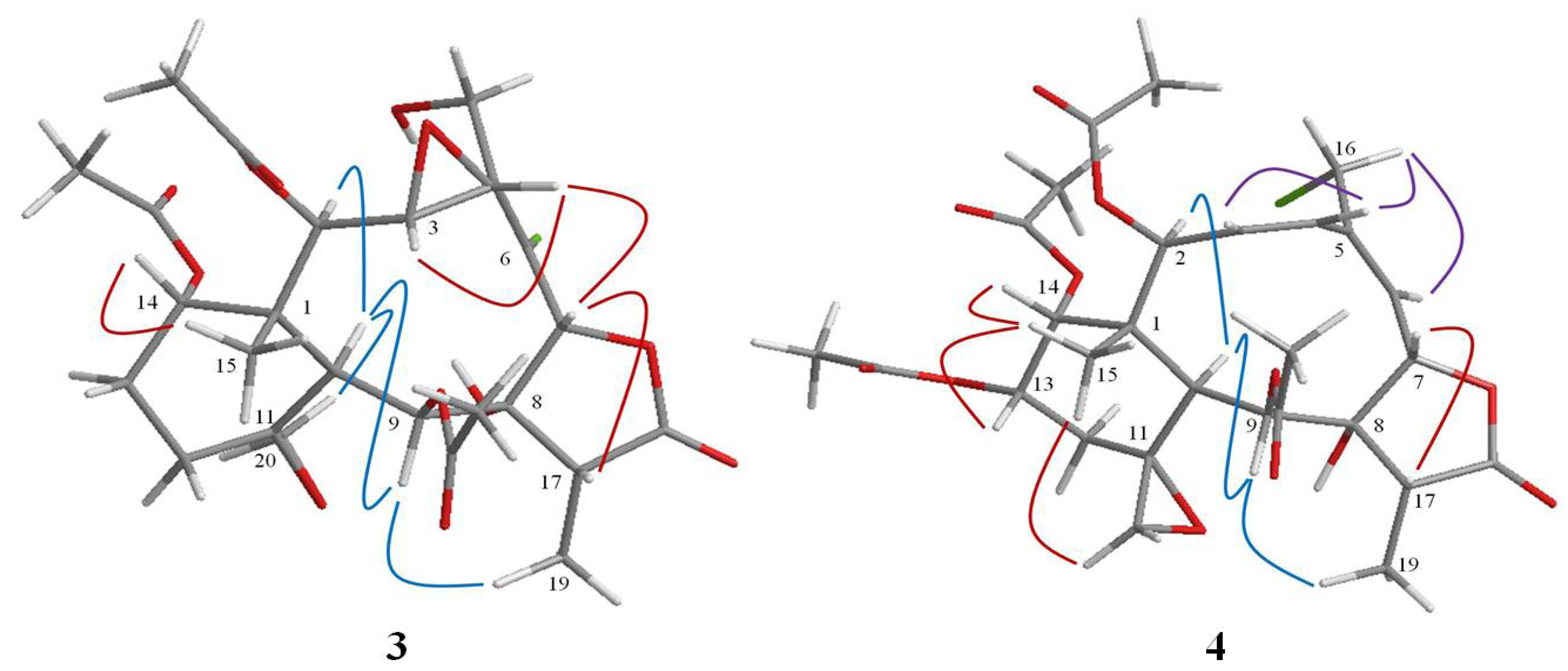
2. Results and Discussion
| No. | 1 | 2 | 3 | 4 |
|---|---|---|---|---|
| 2 | 5.11 (d, J = 7.6) | 5.00 (m) | 4.83 (d, J = 9.2) | 5.33 (d, J = 9.2) |
| 3 | 2.53 (m) | 2.48 (m) | 3.40 (dd, J = 9.6, 3.6) | 5.57 (dd, J = 10.4, 9.6) |
| 1.80 (m) | 1.81 (m) | |||
| 4 | 2.52 (m) | 2.81 (m) | 4.08 (d, J = 3.6) | 6.30 (d, J = 10.4) |
| 2.74 (m) | 2.51 (m) | |||
| 6 | 6.89 (d, J = 9.6) | 6.84 (d, J = 10.0) | - | 5.96 (d, J = 8.8) |
| 7 | 5.31 (d, J = 9.6) | 5.32 (d, J = 10.0) | 5.41 (s) | 4.91 (d, J = 8.8) |
| 9 | 5.60 (d, J = 2.8) | 5.56 (d, J = 3.5) | 5.64 (d, J = 8.0) | 4.68 (d, J = 5.2) |
| 10 | 3.38 (d, J = 2.8) | 3.27 (d, J = 3.5) | 2.49 (d, J = 8.0) | 3.02 (d, J = 5.2) |
| 12 | 2.23 (m) | 2.25 (m, 2H) | 2.18 (m) | 2.47 (td, J = 12.4, 1.6) |
| 1.80 (m) | - | 1.78 (m) | 1.32 (dd, J = 13.2, 3.6) | |
| 13 | 1.80 (m) | 1.81 (m, 2H) | 2.30 (m) | 4.95 (ddd, J = 12.8, 4.0, 2.8) |
| 1.40 (m) | 1.12 (m) | |||
| 14 | 4.64 (t, J = 2.8) | 4.71 (t, J = 3.5) | 4.88 (d, J = 5.2) | 5.18 (br s) |
| 15 | 1.08 (s) | 1.25 (s) | 1.24 (s) | 1.09 (s) |
| 16 | - | - | 4.57 (dd, J = 12.4, 8.8) | 4.56 (s, 2H) |
| - | - | 4.31 (dd, J = 12.4, 6.0) | - | |
| 17 | 2.63 (q, J = 7.2) | 2.60 (q, J = 7.0) | 2.26 (q, J = 7.2) | 2.26 (q, J = 6.8) |
| 19 | 1.19 (d, J = 7.2) | 1.19 (d, J = 7.0) | 1.22 (d, J = 7.2) | 1.12 (d, J = 6.8) |
| 20 | 5.02 (s) | 5.04 (s) | 2.98 (d, J = 4.4) | 3.52 (br s) |
| 4.98 (s) | 4.99 (s) | 2.80 (d, J = 4.0) | 2.72 (d, J = 2.4) | |
| 2-OCOCH3 | 1.92 (s) | 1.97 (s) | 2.10 (s) | 1.93 (s) |
| 9-OCOCH3 | 2.20 (s) | 2.23 (s) | 2.24 (s) | 2.16 (s) |
| 13-OCOCH3 | - | - | - | 2.07 (s) |
| 14-OCOCH3 | - | 1.93 (s) | 1.96 (s) | 1.95 (s) |
| 2-OCOC(CH3)3 | 1.38 (s, 9H) | - | - | - |
| 16-OCH3 | 3.81 (s) | 3.82 (s) | - | - |
| 8-OH | - | - | 5.86 br s | - |
| 16-OH | - | - | 3.72 (dd, J = 8.0, 6.0) | - |
| No. | 1 | 2 | 3 | 4 |
|---|---|---|---|---|
| 1 | 48.8 (s) | 47.4 (s) | 44.7 (s) | 46.4 (s) |
| 2 | 75.6(d) | 73.6 (d) | 75.9 (d) | 74.1 (d) |
| 3 | 32.3 (tH) | 30.9 (t) | 58.7 (d) | 131.7 (d) |
| 4 | 24.1 (t) | 22.8 (t) | 59.0 (d) | 128.0 (d) |
| 5 | 134.5 (s) | 134.2 (s) | 135.7 (s) | 139.9 (s) |
| 6 | 136.8 (d) | 138.1 (d) | 132.4 (d) | 126.0 (d) |
| 7 | 78.3 (d) | 77.5 (d) | 75.7 (d) | 78.4 (d) |
| 8 | 83.5 (s) | 83.5 (s) | 81.2 (s) | 80.8 (s) |
| 9 | 72.8 (d) | 72.8 (d) | 66.8 (d) | 64.2 (d) |
| 10 | 43.9 (d) | 43.2 (d) | 40.2 (d) | 37.4 (d) |
| 11 | 149.7 (s) | 150.5 (s) | 61.2 (s) | 58.1 (s) |
| 12 | 31.4 (t) | 29.4 (t) | 24.1 (t) | 34.2 (t) |
| 13 | 28.9 (t) | 27.3 (t) | 24.3 (t) | 67.6 (d) |
| 14 | 74.1 (d) | 74.8 (d) | 72.7 (d) | 73.7 (d) |
| 15 | 16.5 (q) | 15.0 (q) | 14.7 (q) | 14.3 (q) |
| 16 | 166.8 (s) | 168.0 (s) | 58.8 (s) | 44.6 (s) |
| 17 | 44.7 (d) | 42.9 (d) | 42.9 (d) | 43.8 (d) |
| 18 | 174.4 (s) | 175.4 (s) | 174.8 (s) | 175.2 (s) |
| 19 | 8.5 (q) | 6.6 (q) | 6.4 (q) | 6.3 (q) |
| 20 | 112.5 (d) | 112.7 (d) | 58.4 (t) | 50.1 (t) |
| 2-OCOCH3 | - | 170.0 (s) | 171.2 (s) | 170.0 (s) |
| 2-OCOCH3 | - | 20.9 (q) | 20.9 (q) | 20.8 (q) |
| 9-OCOCH3 | 168.3 (s) | 169.3 (s) | 169.2 (s) | 170.2 (s) |
| 9-OCOCH3 | 23.3 (q) | 21.7 (q) | 21.9 (q) | 21.5 (q) |
| 13-OCOCH3 | - | - | - | 170.2 (s) |
| 13-OCOCH3 | - | - | - | 21.0 (q) |
| 14-OCOCH3 | 169.6 (s) | 170.5 (s) | 170.2 (s) | 170.0 (s) |
| 14-OCOCH3 | 23.0 (q) | 21.2 (q) | 21.0 (q) | 21.3 (q) |
| 2-OCOC(CH3)3 | 174.7 (s) | - | - | - |
| 2-OCOC(CH3)3 | - a | - | - | - |
| 2-OCOC(CH3)3 | 28.0 (q) | - | - | - |
| 16-OCH3 | 53.6 (q) | 52.5 (q) | - | - |
| Compound | Superoxide anion | Elastase release |
|---|---|---|
| Inhibition (%) | Inhibition (%) | |
| 1 | 32.5 ± 1.5 *** | 35.6 ± 3.2 * |
| 2 | 28.7 ± 3.4 * | 34.1 ± 2.9 ** |
| 3 | 9.70 ± 1.3 ** | 16.0 ± 5.3 * |
| 4 | 5.80 ± 3.0 | −4.5 ± 3.4 |
3. Experimental Section
3.1. General Experimental Procedures
3.2. Animal Material
3.3. Extraction and Isolation
3.4. Single Crystal X-ray Structure Determination of Frajunolide P (1)
3.5. Anti-Inflammatory Assays
3.5.1. Human Neutrophils Elastase Release
3.5.2. Human Neutrophil Superoxide Generation
4. Conclusions
Acknowledgments
References
- Sung, P.J.; Sheu, J.H.; Xu, J.P. Survey of briarane-type diterpenoids of marine origin. Heterocycles 2002, 56, 535–579. [Google Scholar] [CrossRef]
- Sung, P.J.; Chang, P.C.; Fang, L.S.; Sheu, J.H.; Chen, W.C.; Chen, Y.P.; Lin, M.R. Briarane-relatedditerpenoids Part II. Heterocycles 2005, 65, 195–204. [Google Scholar] [CrossRef]
- Sung, P.J.; Gwo, H.H.; Fan, T.Y.; Li, J.J.; Dong, J.; Han, C.C.; Wu, S.L.; Fang, L.S. Natural product chemistry of gorgonian corals of the genus Junceella. Biochem. Syst. Ecol. 2004, 32, 185–196. [Google Scholar] [CrossRef]
- Rodriguez, J.; Nieto, R.M.; Jimenez, C. New briarane stecholide diterpenes from the Indonesian gorgonian Briareum sp. J. Nat. Prod. 1998, 61, 313–317. [Google Scholar] [CrossRef]
- Sung, P.J.; Su, J.H.; Duh, C.Y.; Chiang, M.Y.; Sheu, J.H. Briaexcavatolides K–N, new briarane diterpenes from the gorgonian Briareum excavatum. J. Nat. Prod. 2001, 64, 318–323. [Google Scholar] [CrossRef]
- Cardellina, J.H., II; James, T.R., Jr.; Chen, M.H.M.; Clardy, J. Structure of brianthein W, from the soft coral Briareum polyanthes. J. Org. Chem. 1984, 49, 3398–3399. [Google Scholar] [CrossRef]
- Shin, J.; Park, M.; Fenical, M. The junceellolides, new anti-inflammatory diterpenoids of the briarane class from the Chinese gorgonian Junceella fragilis. Tetrahedron 1989, 45, 1633–1638. [Google Scholar] [CrossRef]
- Groweiss, A.; Look, S.A.; Fenical, W. Solenolides, new anti-inflammatory and antiviral diterpenoids from a marine octocoral of the genus Solenopodium. J. Org. Chem. 1988, 53, 2401–2406. [Google Scholar] [CrossRef]
- Isaacs, S.; Carmely, S.; Kashman, Y. Juncins A–F, six new briarane diterpenoids from the gorgonian Junceella juncea. J. Nat. Prod. 1990, 53, 596–602. [Google Scholar] [CrossRef]
- He, H.Y.; Faulkner, D.J. New chlorinated diterpenes from the gorgonian Junceella gemmacea. Tetrahedron 1991, 47, 3271–3280. [Google Scholar] [CrossRef]
- Shen, Y.C.; Chen, Y.H.; Hwang, T.L.; Guh, J.H.; Khalil, A.T. Four new briarane diterpenoids from the gorgonian coral Junceella fragilis. Helv. Chim. Acta 2007, 90, 1391–1398. [Google Scholar] [CrossRef]
- Liaw, C.C.; Shen, Y.C.; Lin, Y.S.; Hwang, T.L.; Kuo, Y.H.; Khalil, A.T. Frajunolides E–K, briarane diterpenes from Junceella fragilis. J. Nat. Prod. 2008, 71, 1551–1556. [Google Scholar] [CrossRef]
- Liaw, C.C.; Kuo, Y.H.; Lin, Y.S.; Hwang, T.L.; Shen, Y.C. Frajunolides L–O, four new 8-hydroxybriarane diterpenoids from the gorgonian Junceella fragilis. Mar. Drugs 2011, 9, 1477–1486. [Google Scholar] [CrossRef]
- Shen, Y.C.; Lin, Y.C.; Chiang, M.Y. Juncenolide A, a new briarane from the Taiwanese gorgonian Junceella juncea. J. Nat. Prod. 2002, 65, 54–56. [Google Scholar] [CrossRef]
- Shen, Y.C.; Lin, Y.C.; Ko, C.L.; Wang, L.T. New briarane from the Taiwanese gorgonian Junceella juncea. J. Nat. Prod. 2003, 66, 302–305. [Google Scholar] [CrossRef]
- Shen, Y.C.; Lin, Y.C.; Huang, Y.L. Juncenolide E, a new briarane from Taiwanese gorgonian Junceella juncea. J. Chin. Chem. Soc. 2003, 50, 1267–1270. [Google Scholar]
- Lin, Y.C.; Huang, Y.L.; Khalil, A.T.; Chen, M.H.; Shen, Y.C. Juncenolides F and G, two new briarane diterpenoids from Taiwanese gorgonian Junceella juncea. Chem. Pharm. Bull. 2005, 53, 128–130. [Google Scholar] [CrossRef]
- Chang, J.Y.; Liaw, C.C.; Fazary, A.E.; Hwang, T.L.; Shen, Y.C. New briarane diterpenoids from the gorgonian coral Junceella juncea. Mar. Drugs 2012, 10, 1321–1330. [Google Scholar] [CrossRef]
- Rodriguez, A.D.; Ramirez, C.; Cobar, O.M. Briareins C–L, 10 new briarane diterpenoids from the common caribbean gorgonian Briareum asbestinum. J. Nat. Prod. 1996, 59, 15–22. [Google Scholar] [CrossRef]
- Mohamadi, F.; Richards, N.G.J.; Guida, W.C.; Liskamp, R.; Lipton, M.; Caufield, C.; Chang, G.; Hendrickson, T.; Still, W.C. Macromodel—An integrated software system for modeling organic and bioorganic molecules using molecular mechanics. J. Comput. Chem. 1990, 11, 440–462. [Google Scholar] [CrossRef]
- Sheu, J.H.; Chen, Y.P.; Hwang, T.L.; Chiang, M.Y.; Fang, L.S.; Sung, P.J. Junceellolides J–L, 11,20-epoxybriaranes from the gorgonian coral Junceella fragilis. J. Nat. Prod. 2006, 69, 269–273. [Google Scholar] [CrossRef]
- Subrahmanyam, C.; Kulatheeswaran, R.; Ward, R.S. Briarane diterpenes from the Indian ocean gorgonian Gorgonella umbraculum. J. Nat. Prod. 1998, 61, 1120–1122. [Google Scholar] [CrossRef]
- Zhang, W.; Guo, Y.W.; Mollo, E.; Cimio, G. Junceellonoids A and B, two new briarane diterpenoids from the Chinese gorgonian Junceella fragilis Ridley. Helv. Chim. Acta 2004, 87, 2341–2345. [Google Scholar] [CrossRef]
- Qi, S.H.; Zhang, S.; Huang, H.; Xiao, Z.H.; Huang, J.S.; Li, Q.X. New briaranes from the South China Sea gorgonian Junceella juncea. J. Nat. Prod. 2004, 67, 1907–1910. [Google Scholar] [CrossRef]
- Hwang, T.L.; Yeh, S.H.; Leu, Y.L.; Chern, C.Y.; Hsu, H.C. Inhibition of superoxide anion and eastase rlease in human nutrophils by 3′-isopropoxychalcone via a cAMP-dependent pathway. Br. J. Pharmacol. 2006, 148, 78–87. [Google Scholar]
Supplementary Files
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Liaw, C.-C.; Lin, Y.-C.; Lin, Y.-S.; Chen, C.-H.; Hwang, T.-L.; Shen, Y.-C. Four New Briarane Diterpenoids from Taiwanese Gorgonian Junceella fragilis. Mar. Drugs 2013, 11, 2042-2053. https://doi.org/10.3390/md11062042
Liaw C-C, Lin Y-C, Lin Y-S, Chen C-H, Hwang T-L, Shen Y-C. Four New Briarane Diterpenoids from Taiwanese Gorgonian Junceella fragilis. Marine Drugs. 2013; 11(6):2042-2053. https://doi.org/10.3390/md11062042
Chicago/Turabian StyleLiaw, Chia-Ching, Yu-Chi Lin, Yun-Sheng Lin, Chung-Hsiung Chen, Tsong-Long Hwang, and Ya-Ching Shen. 2013. "Four New Briarane Diterpenoids from Taiwanese Gorgonian Junceella fragilis" Marine Drugs 11, no. 6: 2042-2053. https://doi.org/10.3390/md11062042
APA StyleLiaw, C.-C., Lin, Y.-C., Lin, Y.-S., Chen, C.-H., Hwang, T.-L., & Shen, Y.-C. (2013). Four New Briarane Diterpenoids from Taiwanese Gorgonian Junceella fragilis. Marine Drugs, 11(6), 2042-2053. https://doi.org/10.3390/md11062042





