The Promoting Effect of Ishige sinicola on Hair Growth
Abstract
:1. Introduction
2. Results
2.1. The Effect of I. sinicola Extract on the Hair-Fiber Elongation of Rat Vibrissa Follicle
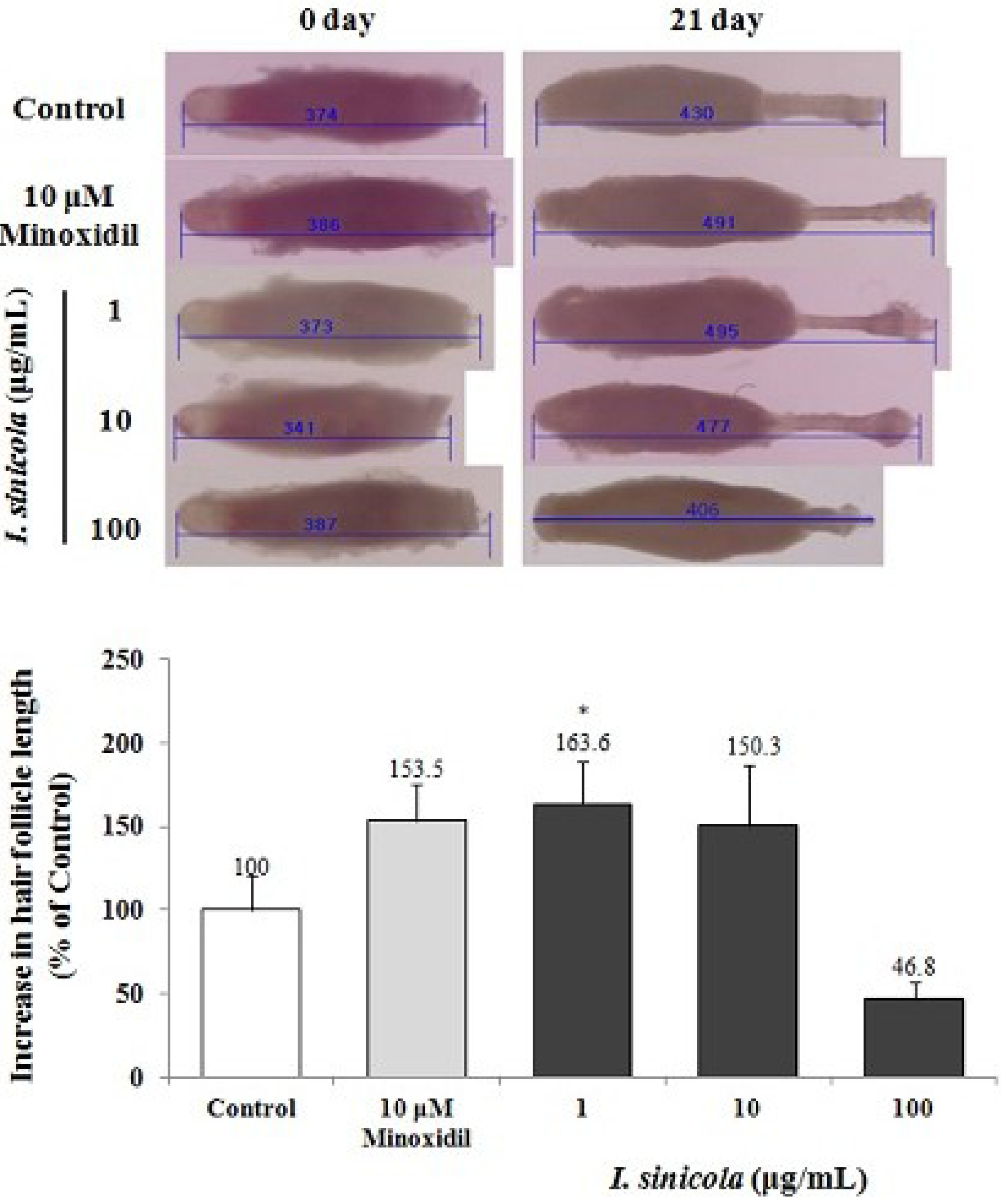
2.2. The Effect of I. sinicola Extract on the Anagen Induction in C57BL/6 Mice
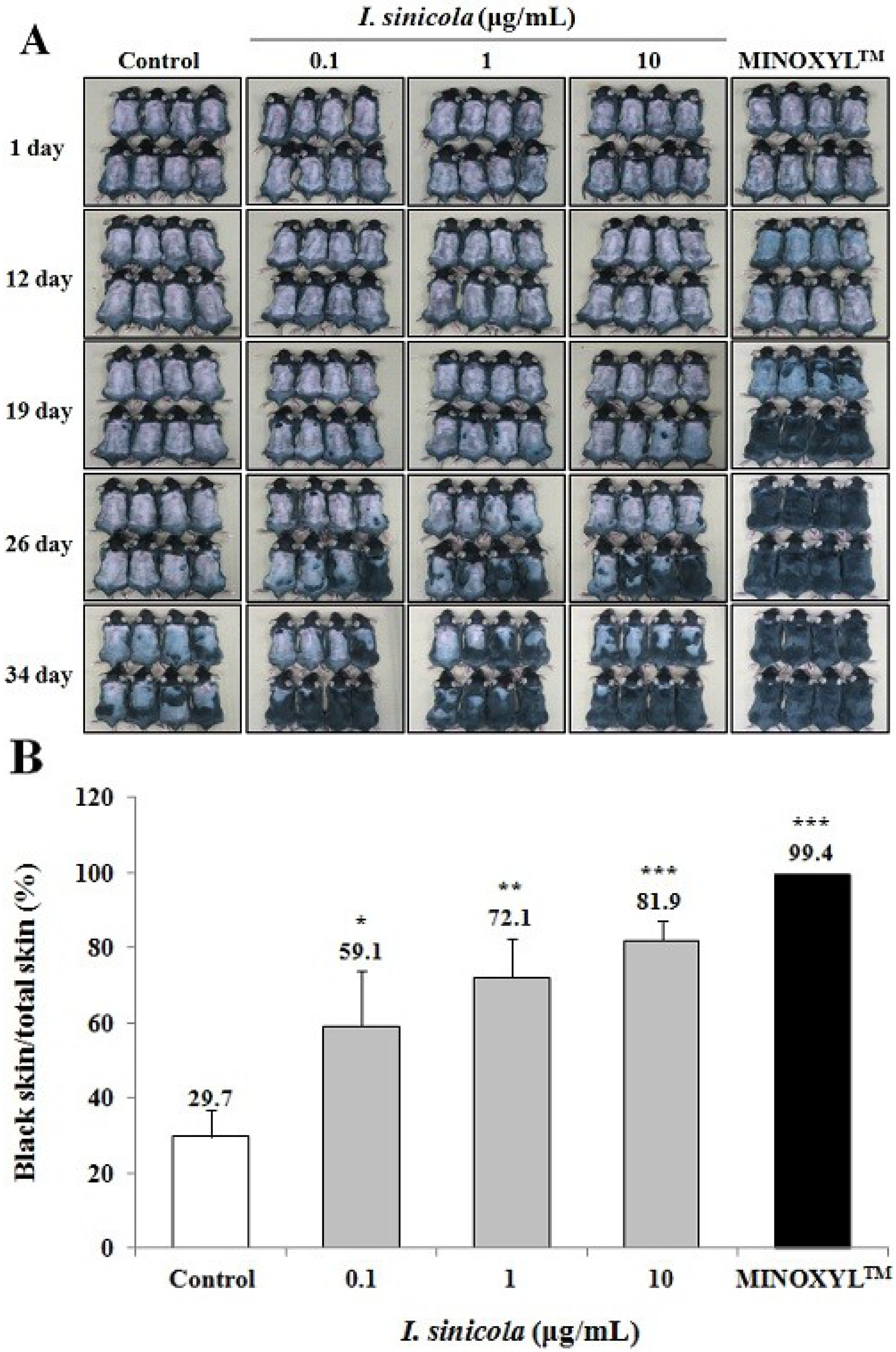
2.3. Effects of I. sinicola Extract on the 5α-Reductase Activity and the Proliferation of DPCs
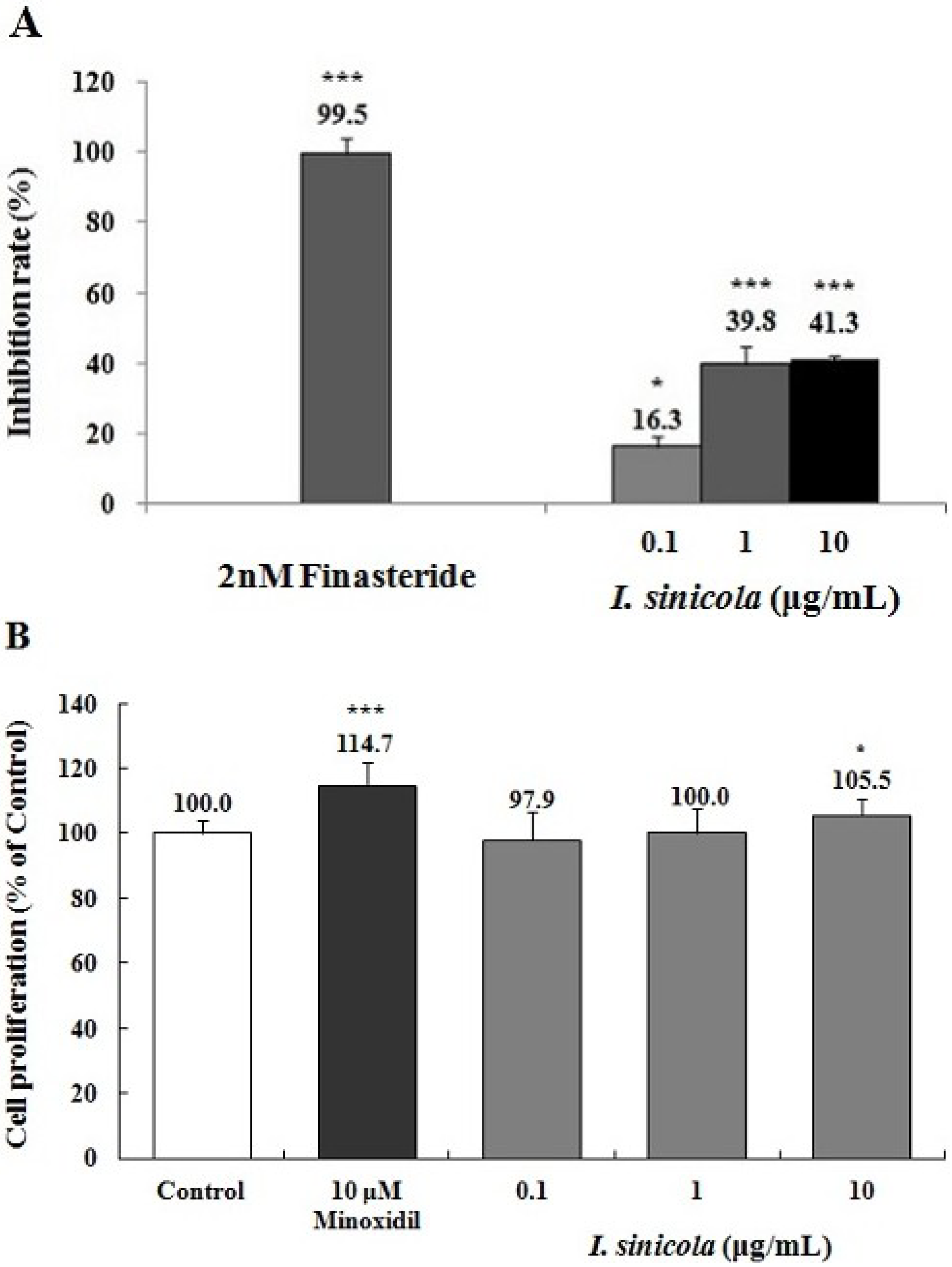
2.4. Effects of I. sinicola Extract on the Expression Levels of Cell Cycle-Associated Proteins and β-Catenin
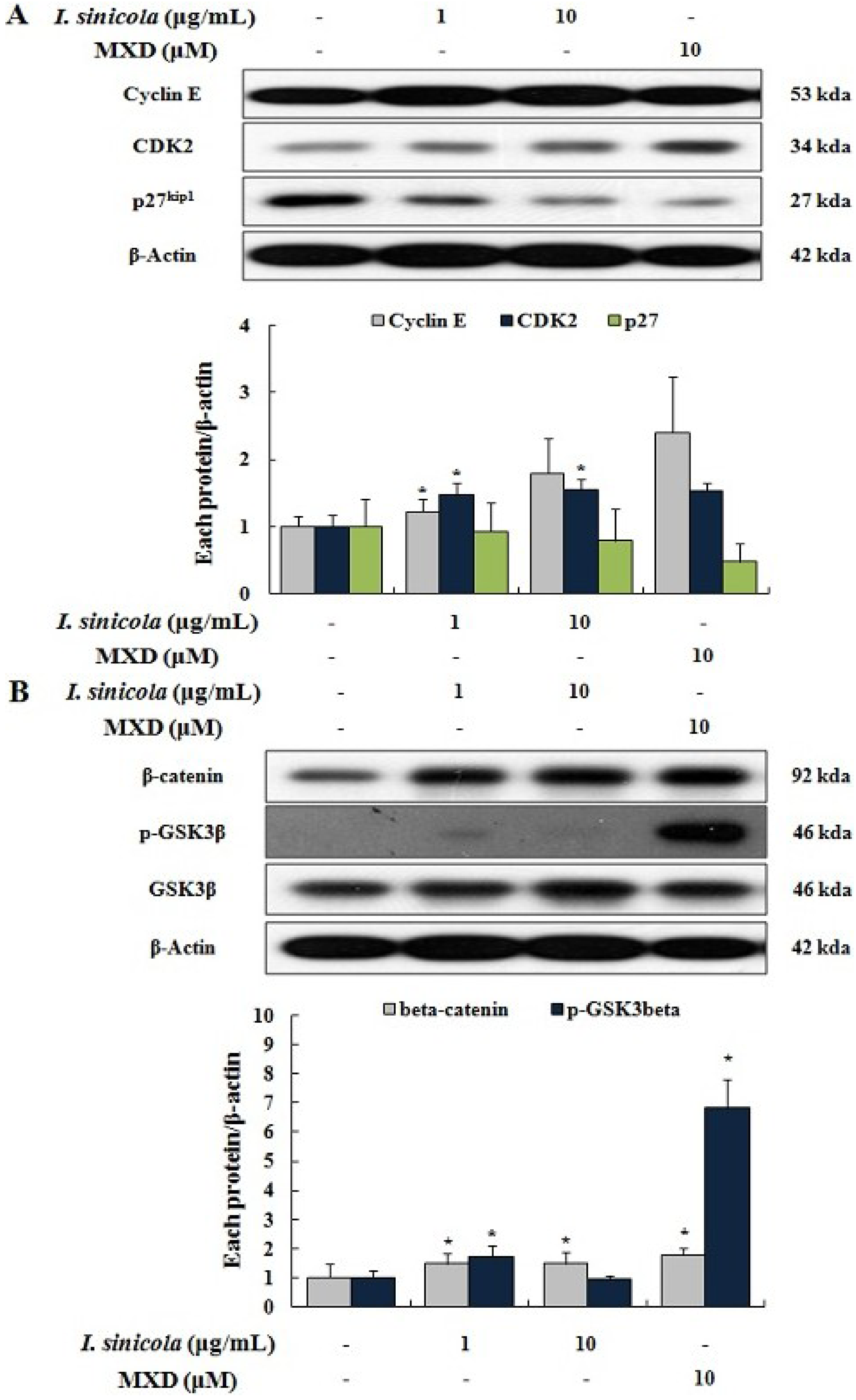
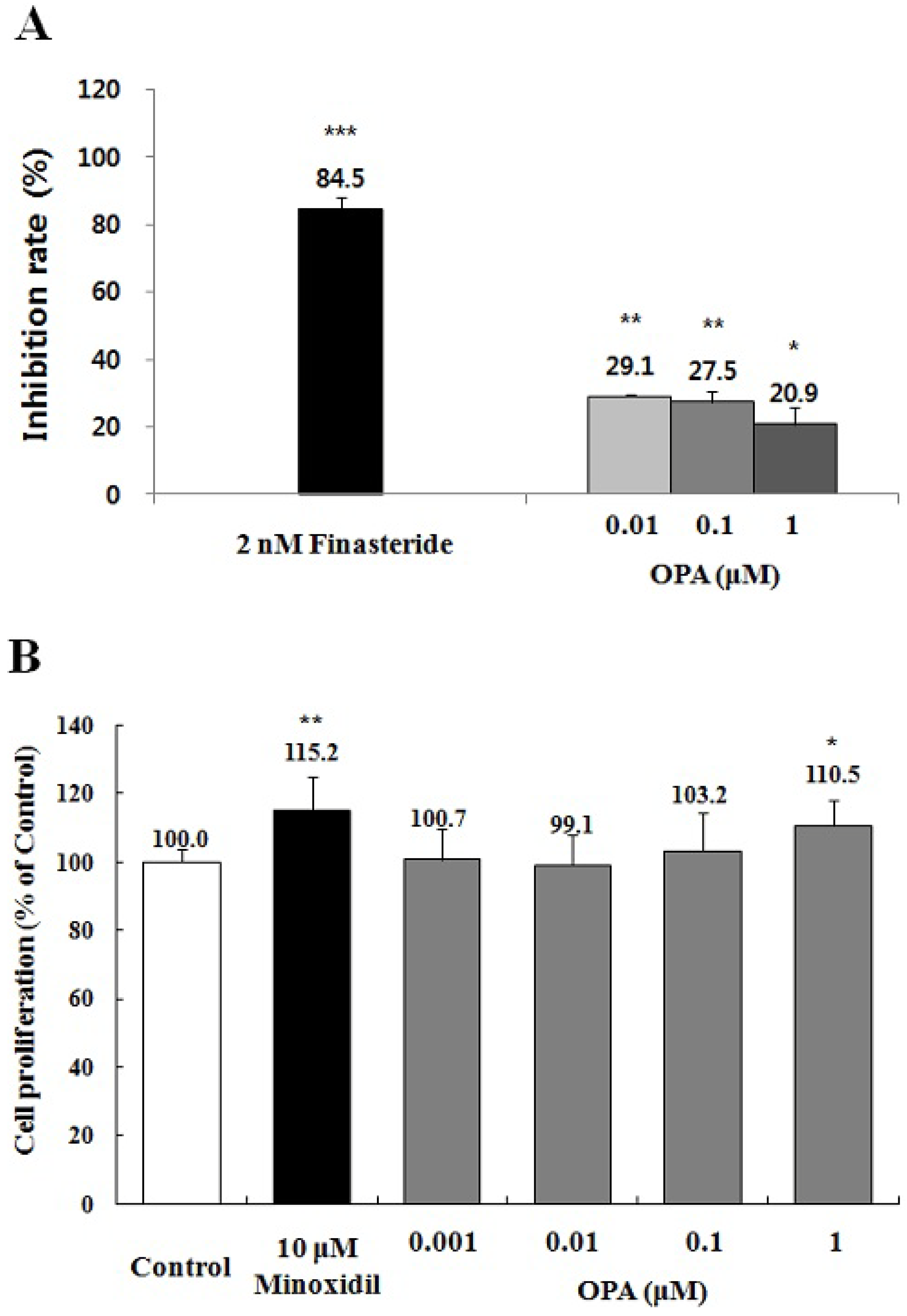
2.5. Effect of Octaphlorethol A on the 5α-Reductase Activity and the Proliferation of DPCs
3. Discussion
4. Experimental Section
4.1. The Preparation of I. sinicola Extract and Isolation of Octaphlorethol A
4.2. Animals
4.3. Isolation and Culture of Rat Vibrissa Follicles
4.4. Hair Growth Activity in Vivo
4.5. Assay for Prostatic 5α-Reductase Activity
4.6. Assay for the Proliferation of DPCs
4.7. Western Blot Analysis
4.8. Statistical Analysis
5. Conclusions
Acknowledgments
Conflict of Interest
References
- Price, V.H. Treatment of hair loss. N. Engl. J. Med. 1999, 341, 964. [Google Scholar] [CrossRef]
- Kaufman, K.D.; Olsen, E.A.; Whiting, D.; Savin, R.; DeVillez, R.; Bergfeld, W.; Price, V.H.; van Neste, D.; Roberts, J.L.; Hordinsky, M.; et al. Finasteride in the treatment of men with androgenetic alopecia. J. Am. Acad. Dermatol. 1998, 39, 578–589. [Google Scholar] [CrossRef]
- Burton, J.L.; Marshall, A. Hypertrichosis due to minoxidil. Br. J. Dermatol. 1979, 101, 593–595. [Google Scholar] [CrossRef]
- Gromley, G.J. Finasteride: A clinical review. Biomed. Pharmacother. 1995, 49, 319–324. [Google Scholar] [CrossRef]
- Kaufman, K.D.; Rotonda, J.; Shah, A.K.; Meehan, A.G. Long-term treatment with finasteride 1 mg decreases the likelihood of developing further visible hair loss in men with androgenetic alopecia (male pattern hair loss). Eur. J. Dermatol. 2008, 18, 400–406. [Google Scholar]
- Van Neste, D.; Fuh, V.; Sanchez-Pedreno, P.; Lopez-Bran, E.; Wolff, H.; Whiting, D.; Roberts, J.; Kopera, D.; Stene, J.J.; Calvieri, S.; et al. Finasteride increases anagen hair in men with androgenetic alopecia. Br. J. Dermatol. 2000, 143, 804–810. [Google Scholar] [CrossRef]
- Hamaoka, H.; Minakuchi, K.; Miyoshi, H.; Arase, S.; Chen, C.H.; Nakaya, Y. Effect of K+ channel openers on K+ channel in cultured human dermal papilla cells. J. Med. Invest. 1997, 44, 73–77. [Google Scholar]
- Shorter, K.; Farjo, N.P.; Picksley, S.M.; Randall, V.A. Human hair follicles contain two forms of ATP-sensitive potassium channels, only one of which is sensitive to minoxidil. FASEB J. 2008, 22, 1725–1736. [Google Scholar] [CrossRef]
- Lachgar, S.; Charveron, M.; Gall, Y.; Bonafe, J.L. Minoxidil upregulates the expression of vascular endothelial growth factor in human hair dermal papilla cells. Br. J. Dermatol. 1998, 138, 407–411. [Google Scholar] [CrossRef]
- Kwack, M.H.; Kang, B.M.; Kim, M.K.; Kim, J.C.; Sung, Y.K. Minoxidil activates beta-catenin pathway in human dermal papilla cells: A possible explanation for its anagen prolongation effect. J. Dermatol. Sci. 2011, 62, 154–159. [Google Scholar] [CrossRef]
- Stenn, K.S.; Paus, R. Controls of hair follicle cycling. Physiol. Rev. 2001, 81, 449–494. [Google Scholar]
- Han, J.H.; Kwon, O.S.; Chung, J.H.; Cho, K.H.; Eun, H.C.; Kim, K.H. Effect of minoxidil on proliferation and apoptosis in dermal papilla cells of human hair follicle. J. Dermatol. Sci. 2004, 34, 91–98. [Google Scholar] [CrossRef]
- Kim, S.C.; Kang, J.I.; Park, D.B.; Lee, Y.K.; Hyun, J.W.; Koh, Y.S.; Yoo, E.S.; Kim, J.A.; Kim, Y.H.; Kang, H.K. Promotion effect of Acankoreoside J, a lupane-triterpene in Acanthopanax koreanum, on hair growth. Arch. Pharm. Res. 2012, 35, 1495–1503. [Google Scholar] [CrossRef]
- Park, P.J.; Moon, B.S.; Lee, S.H.; Kim, S.N.; Kim, A.R.; Kim, H.J.; Park, W.S.; Choi, K.Y.; Cho, E.G.; Lee, T.R. Hair growth-promoting effect of Aconiti Ciliare Tuber extract mediated by the activation of Wnt/β-catenin signaling. Life Sci. 2012, 91, 935–943. [Google Scholar] [CrossRef]
- Park, S.; Shin, W.S.; Ho, J. Fructus panax ginseng extract promotes hair regeneration in C57BL/6 mice. J. Ethnopharmacol. 2011, 138, 340–344. [Google Scholar] [CrossRef]
- Ito, M.; Yang, Z.; Andl, T.; Cui, C.; Kim, N.; Millar, S.E.; Cotsarelis, G. Wnt-dependent de novo hair follicle regeneration in adult mouse skin after wounding. Nature 2007, 447, 316–320. [Google Scholar] [CrossRef]
- Ouji, Y.; Yoshikawa, M.; Shiroi, A.; Ishizaka, S. Promotion of hair follicle development and trichogenesis by Wnt-10b in cultured embryonic skin and in reconstituted skin. Biochem. Biophys. Res. Commun. 2006, 345, 581–587. [Google Scholar] [CrossRef]
- Rulifson, I.C.; Karnik, S.K.; Heiser, P.W.; Ten Berge, D.; Chen, H.; Gu, X.; Taketo, M.M.; Nusse, R.; Hebrok, M.; Kim, S.K. Wnt signaling regulates pancreatic beta cell proliferation. Proc. Natl. Acad. Sci. USA 2007, 104, 6247–6252. [Google Scholar] [CrossRef]
- Tetsu, O.; McCormick, F. Beta-catenin regulates expression of cyclin D1 in colon carcinoma cells. Nature 1999, 398, 422–426. [Google Scholar] [CrossRef]
- He, T.C.; Sparks, A.B.; Rago, C.; Hermeking, H.; Zawel, L.; da Costa, L.T.; Morin, P.J.; Vogelstein, B.; Kinzler, K.W. Identification of c-MYC as a target of the APC pathway. Science 1998, 281, 1509–1512. [Google Scholar] [CrossRef]
- Johnson, D.G.; Walker, C.L. Cyclins and cell cycle checkpoints. Annu. Rev. Pharmacol. Toxicol. 1999, 39, 295–312. [Google Scholar] [CrossRef]
- Sherr, C.J. Cancer cell cycles. Science 1996, 274, 1672–1677. [Google Scholar] [CrossRef]
- Choi, J.S.; Bae, H.J.; Kim, S.J.; Choi, I.S. In vitro antibacterial and anti-inflammatory properties of seaweed extracts against acne inducing bacteria, Propionibacterium acnes. J. Environ. Biol. 2011, 32, 313–318. [Google Scholar]
- Lee, S.H.; Kang, S.M.; Ko, S.C.; Lee, D.H.; Jeon, Y.J. Octaphlorethol A, a novel phenolic compound isolated from a brown alga, Ishige foliacea, increases glucose transporter 4-mediated glucose uptake in skeletal muscle cells. Biochem. Biophys. Res. Commun. 2012, 420, 576–581. [Google Scholar] [CrossRef]
- Müller-Röver, S.; Handjiski, B.; van der Veen, C.; Maurer, M.; Eichmüller, S.; Ling, G.; Hofmann, U.; Foitzik, K.; Mecklenburg, L.; Handjiski, B. A comprehensive guide for the accurate classification of murine hair follicles in distinct hair cycle stages. J. Invest. Dermatol. 2001, 117, 3–15. [Google Scholar] [CrossRef]
- Whiting, D.A. Male pattern hair loss: Current understanding. Int. J. Dermatol. 1998, 37, 561–566. [Google Scholar] [CrossRef]
- Trüeb, R.M. Molecular mechanisms of androgenetic alopecia. Exp. Gerontol. 2002, 37, 981–990. [Google Scholar] [CrossRef]
- Sherr, C.J.; Roberts, J.M. CDK inhibitors: Positive and negative regulators of G1-phase progression. Genes Dev. 1999, 13, 1501–1512. [Google Scholar] [CrossRef]
- Ouji, Y.; Yoshikawa, M.; Moriya, K.; Ishizaka, S. Effects of Wnt-10b on hair shaft growth in hair follicle cultures. Biochem. Biophys. Res. Commun. 2007, 359, 516–522. [Google Scholar] [CrossRef]
- Wangefjord, S.; Brändstedt, J.; Ericson Lindquist, K.; Nodin, B.; Jirström, K.; Eberhard, J. Associations of beta-catenin alterations and MSI screening status with expression of key cell cycle regulating proteins and survival from colorectal cancer. Diagn. Pathol. 2013, 8, 10. [Google Scholar]
- Hedgepeth, C.M.; Conrad, L.J.; Zhang, J.; Huang, H.C.; Lee, V.M.; Klein, P.S. Activation of the Wnt signaling pathway: A molecular mechanism for lithium action. Dev. Biol. 1997, 185, 82–91. [Google Scholar] [CrossRef]
- Philpott, M.P.; Green, M.R.; Kealy, T. Rat hair follicle growth in vitro. Br. J. Dermatol. 1992, 127, 600–607. [Google Scholar] [CrossRef]
- Philpott, M.P.; Kealey, T. Cyclical changes in rat vibrissa follicles maintained in vitro. J. Invest. Dermatol. 2000, 115, 1152–1155. [Google Scholar] [CrossRef]
- Kaufman, K.D. Androgen metabolism as it affects hair growth in androgenetic alopecia. Dermatol. Clin. 1996, 14, 697–711. [Google Scholar] [CrossRef]
- Jahoda, C.A.; Horne, K.A.; Oliver, R.F. Induction of hair growth by implantation of cultured dermal papilla cells. Nature 1984, 311, 560–562. [Google Scholar] [CrossRef]
- Elliott, K.; Stephenson, T.J.; Messenger, A.G. Differences in hair follicle dermal papilla volume are due to extracellular matrix volume and cell number: Implications for the control of hair follicle size and androgen responses. J. Invest. Dermatol. 1999, 113, 873–877. [Google Scholar] [CrossRef]
- Igata, M.; Motoshima, H.; Tsuruzoe, K.; Kojima, K.; Matsumura, T.; Kondo, T.; Taguchi, T.; Nakamaru, K.; Yano, M.; Kukidome, D. Adenosine monophosphate-activated protein kinase suppresses vascular smooth muscle cell proliferation through the inhibition of cell cycle progression. Circ. Res. 2005, 97, 837–844. [Google Scholar] [CrossRef]
- Prall, O.W.; Sarcevic, B.; Musgrove, E.A.; Watts, C.K.; Sutherland, R.L. Estrogen-induced activation of Cdk4 and Cdk2 during G1-S phase progression is accompanied by increased cyclin D1 expression and decreased cyclin-dependent kinase inhibitor association with cyclin E-Cdk2. J. Biol. Chem. 1997, 272, 10882–10894. [Google Scholar]
- Núñez, M.; Medina, V.; Cricco, G.; Croci, M.; Cocca, C.; Rivera, E.; Bergoc, R.; Martín, G. Glibenclamide inhibits cell growth by inducing G0/G1 arrest in the human breast cancer cell line MDA-MB-231. BMC Pharmacol. Toxicol. 2013, 14, 6. [Google Scholar] [CrossRef]
- Yamauchi, K.; Kurosaka, A. Inhibition of glycogen synthase kinase-3 enhances the expression of alkaline phosphatase and insulin-like growth factor-1 in human primary dermal papilla cell culture and maintains mouse hair bulbs in organ culture. Arch. Dermatol. Res. 2009, 301, 357–365. [Google Scholar] [CrossRef]
- Buhl, A.E.; Waldon, D.J.; Kawabe, T.T.; Holland, J.M. Minoxidil stimulates mouse vibrissae follicles in organ culture. J. Invest. Dermatol. 1989, 6, 129–131. [Google Scholar]
- Ohnemus, U.; Uenalan, M.; Conrad, F.; Handjiski, B.; Mecklenburg, L.; Nakamura, M.; Inzunza, J.; Gustafsson, J.A.; Paus, R. Hair cycle control by estrogens: Catagen induction via estrogen receptor (ER)-alpha is checked by ER beta signaling. Endocrinology 2005, 146, 1214–1225. [Google Scholar]
- Hirosumi, J.; Nakayama, O.; Fagan, T.; Sawada, K.; Chida, N.; Inami, M.; Takahashi, S.; Notsu, Y.; Okuhara, M.J. FK143, a novel nonsteroidal inhibitor of steroid 5-reductase: (1) In vitro effects on human and animal prostatic enzymes. J. Steroid Biochem. Mol. Biol. 1995, 52, 357–363. [Google Scholar] [CrossRef]
- Filsell, W.; Little, J.C.; Stones, A.J.; Granger, S.P.; Bayley, S.A. Transfection of rat dermal papilla cells with a gene encoding a temperature-sensitive polyomavirus large T antigen generates cell lines a differentiated phenotype. J. Cell Sci. 1994, 107, 1761–1772. [Google Scholar]
- Carmichael, J.; DeGraff, W.G.; Gazdar, A.F.; Minna, J.D.; Mitchell, J.B. Evaluation of a tetrazolium-based semiautomated colorimetric assay: Assessment of chemosensitivity testing. Cancer Res. 1987, 47, 936–942. [Google Scholar]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kang, J.-I.; Kim, E.-J.; Kim, M.-K.; Jeon, Y.-J.; Kang, S.-M.; Koh, Y.-S.; Yoo, E.-S.; Kang, H.-K. The Promoting Effect of Ishige sinicola on Hair Growth. Mar. Drugs 2013, 11, 1783-1799. https://doi.org/10.3390/md11061783
Kang J-I, Kim E-J, Kim M-K, Jeon Y-J, Kang S-M, Koh Y-S, Yoo E-S, Kang H-K. The Promoting Effect of Ishige sinicola on Hair Growth. Marine Drugs. 2013; 11(6):1783-1799. https://doi.org/10.3390/md11061783
Chicago/Turabian StyleKang, Jung-Il, Eun-JI Kim, Min-Kyoung Kim, You-Jin Jeon, Sung-Myung Kang, Young-Sang Koh, Eun-Sook Yoo, and Hee-Kyoung Kang. 2013. "The Promoting Effect of Ishige sinicola on Hair Growth" Marine Drugs 11, no. 6: 1783-1799. https://doi.org/10.3390/md11061783



