Isolation and Structural Elucidation of Chondrosterins F–H from the Marine Fungus Chondrostereum sp.
Abstract
:1. Introduction
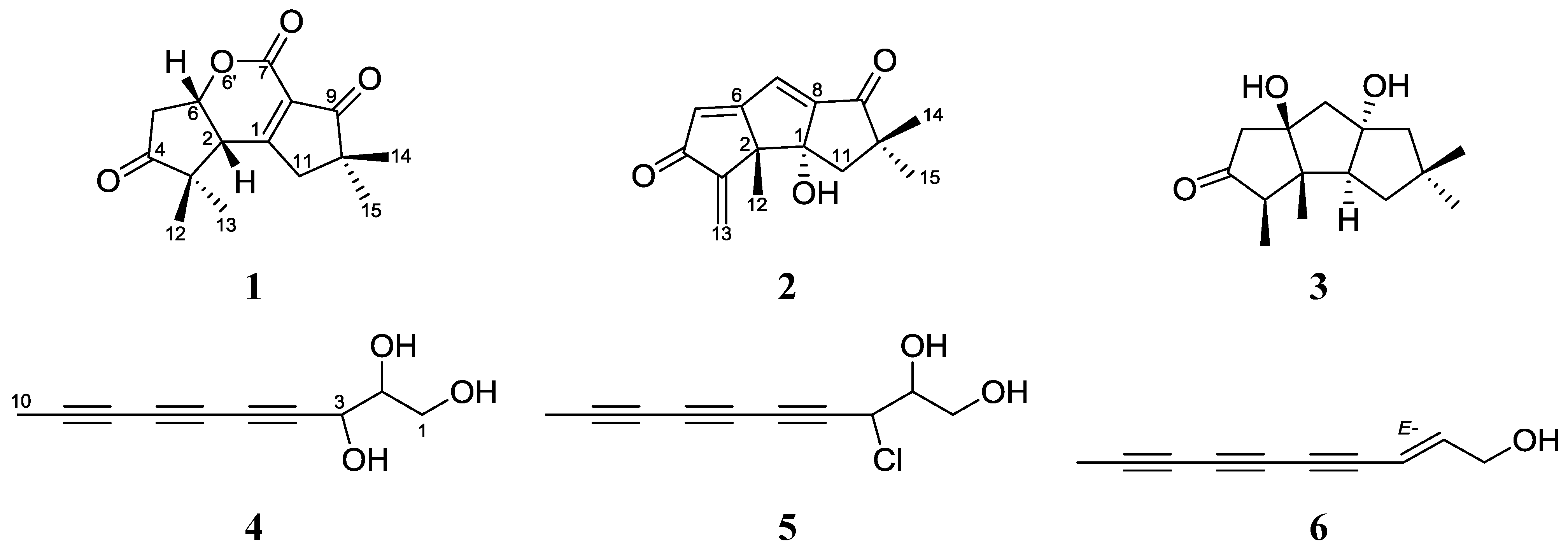
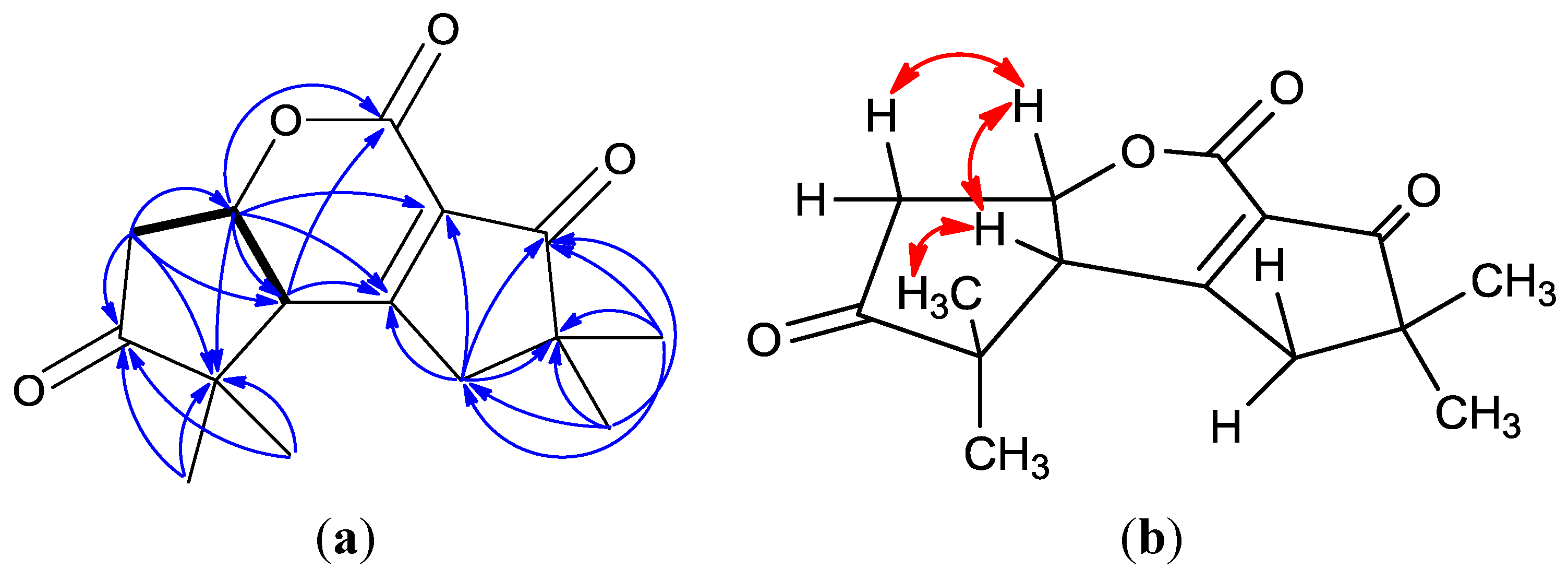
2. Results and Discussion
| Position | 1 | 2 | 3 | |||
|---|---|---|---|---|---|---|
| δC, type | δH, mult., ( J) | δC, type | δH, mult., ( J) | δC, type | δH, mult., ( J) | |
| 1 | 186.7, C | 83.6, C | 60.5, CH | 2.54, dd (12.0, 8.0) | ||
| 2 | 79.0, CH | 5.57, ddd (7.0, 3.0, 1.0) | 63.2, C | 54.5, C | ||
| 3 | 62.2, C | 147.5, C | 55.5, CH | 2.56, dd (7.0, 1.0) | ||
| 4 | 206.5, C | 195.3, C | 216.5, C | |||
| 5 | 31.3, CH2 | α: 2.72, dd (18.0, 10.0); | 128.4, CH | 6.39, s | 49.8, CH2 | α: 2.19, d (19.0); |
| β: 2.28, dd (18.0, 7.5) | β: 2.66, dd (19.0, 1.0) | |||||
| 6 | 52.2, CH | 3.43, ddd (10.0, 7.5, 7.0) | 184.3, C | 87.0, C | ||
| 7 | 174.7, C | 126.5, CH | 7.18, s | 56.8, CH2 | α: 2.38, d (15.0); | |
| β: 2.19, d (15.0) | ||||||
| 8 | 142.5, C | 157.3, C | 90.9, C | |||
| 9 | 206.0, C | 207.3, C | 58.8, CH2 | α: 1.94, d (14.0); | ||
| β: 1.77, dd (14.0, 3.0) | ||||||
| 10 | 51.0, C | 51.1, C | 40.1, C | |||
| 11 | 40.5, CH2 | α: 2.38, dd (19.0, 3.0); | 42.5, CH2 | 2.24, d (14.0); | 44.7, CH2 | α: 1.69, dd (12.0, 12.0); |
| β: 2.75, dd (19.0, 1.0) | 2.04, d (14.0) | β: 1.58, ddd (12.0, 8.0, 3.0) | ||||
| 12 | 23.5, CH3 | 1.49, s | 26.4, CH3 | 1.27, s | 11.3, CH3 | 0.83, s |
| 13 | 27.6, CH3 | 2.21, s | 116.6, CH2 | 6.18, s; | 8.2, CH3 | 1.02, d (7.0) |
| 5.39, s | ||||||
| 14 | 25.0, CH3 | 1.16, s | 27.3, CH3 | 1.40, s | 26.8, CH3 | 1.13, s |
| 15 | 25.3, CH3 | 1.24, s | 25.9, CH3 | 1.21, s | 29.6, CH3 | 1.07, s |
| 1α-OH | 2.05, brs | |||||
| 6β-OH | 1.54, s | |||||
| 8α-OH | 1.74, s | |||||
| Position | 4 a, δC, type | 5 b, δC, type | 5 c, δC, type | 6 c, δC, type |
|---|---|---|---|---|
| 1 | 62.2, CH2 | 62.6, CH2 | 62.8, CH2 | 62.7, CH2 |
| 2 | 74.1, CH | 74.6, CH | 74.6, CH | 146.8, CH |
| 3 | 63.2, CH | 51.1, CH | 49.9, CH | 108.4, CH |
| 4–9 d | 79.6, C | 80.2, C | 78.3, C | 78.2, C |
| 78.4, C | 73.4, C | 73.4, C | 75.2, C | |
| 68.3, C | 71.0, C | 70.6, C | 73.6, C | |
| 63.8, C | 64.9, C | 66.3, C | 67.2, C | |
| 62.5, C | 63.7, C | 64.6, C | 64.9, C | |
| 58.7, C | 57.8, C | 57.8, C | 59.0, C | |
| 10 | 3.8, CH3 | 4.0, CH3 | 4.8, CH3 | 4.6, CH3 |
| Position | 4 a | 5 b | 5 c | 6 c |
|---|---|---|---|---|
| 1 | 3.41, ddd (11.0, 11.0, 5.5); | 3.45, ddd (11.2, 6.0, 4.8); | 3.85, d (4.5) | 4.25, dd (4.5, 2.0) |
| 3.37, ddd (11.0, 5.5, 5.5) | 3.39, ddd (11.2, 6.0, 4.8) | |||
| 2 | 3.46, ddd (10.0, 5.0, 5.0) | 3.70, dddd (6.0, 6.0, 6.0, 4.0) | 3.95, dt (6.0, 4.5) | 6.46, dt (16.0, 4.5) |
| 3 | 4.29, t (6.0) | 4.99, d (4.0) | 4.74, d (6.0) | 5.84, dt (16.0, 2.0) |
| 10 | 2.03, s | 2.04, s | 1.99, s | 1.98, s |
| 1-OH | 4.53, t (5.4) | 4.90, t (4.8) | 1.91, brs | 1.51, brs |
| 2-OH | 5.01, d (5.5) | 5.74, d (6.0) | 1.91, brs | |
| 3-OH | 5.68, d (6.5) |
| Cancer cell line | IC50 (μg/mL) |
|---|---|
| Human nasopharyngeal carcinoma cell line CNE1 | 8.33 |
| Human nasopharyngeal carcinoma cell line CNE2 | 6.07 |
| Human nasopharyngeal carcinoma cell line SUNE1 | 3.99 |
| Human lung cancer cell line A549 | 12.37 |
| Human colon cancer cell line Lovo | 2.16 |
| Human epidermoid carcinoma cell line KB | 28.55 |
| Human hepatic cancer cell line Bel7402 | 23.36 |
| Human breast cancer cell line MCF-7 | 4.57 |
3. Experimental Section
3.1. General Experimental Procedures
3.2. Fungal Strain and Culture Method
3.3. Extraction and Isolation
3.4. Cytotoxicity Assay
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Li, H.J.; Xie, Y.L.; Xie, Z.L.; Chen, Y.; Lam, C.K.; Lan, W.J. Chondrosterins A–E, triquinane-type sesquiterpenoids from soft coral-associated fungus Chondrostereum sp. Mar. Drugs 2012, 10, 627–638. [Google Scholar] [CrossRef]
- Li, H.J.; Lan, W.J.; Lam, C.K.; Yang, F.; Zhu, X.F. Hirsutane sesquiterpenoids from the marine-derived fungus Chondrostereum sp. Chem. Biodivers. 2011, 8, 317–324. [Google Scholar]
- Yang, F.; Gao, Y.H.; Wu, K.W.; Deng, R.; Li, D.D.; Wei, Z.X.; Jiang, S.; Wu, X.Q.; Feng, G.K.; Li, H.J.; Zhu, X.F. A novel sesquiterpene hirsutanol A induces autophagical cell death in human hepatocellular carcinoma cells by increasing reactive oxygen species. Chin. J. Cancer 2010, 29, 655–660. [Google Scholar] [CrossRef]
- Yang, F.; Chen, W.D.; Deng, R.; Li, D.D.; Wu, K.W.; Feng, G.K.; Li, H.J.; Zhu, X.F. Hirsutanol A induces apoptosis and autophagy via reactive oxygen species accumulation in breast cancer MCF-7 cells. J. Pharmacol. Sci. 2012, 119, 214–220. [Google Scholar]
- Takazawa, H.; Kashino, S. Incarnal. A new antibacterial sesquiterpene from basidiomycetes. Chem. Pharm. Bull. 1991, 39, 555–557. [Google Scholar] [CrossRef]
- Amouzou, E.; Ayer, W.A.; Browne, L.M. Antifungal sesquiterpenoids from an arthroconidial fungus. J. Nat. Prod. 1989, 52, 1042–1054. [Google Scholar] [CrossRef]
- Hellwig, V.; Dasenbrock, J.; Schumann, S.; Steglich, W.; Leonhardt, K.; Anke, T. New triquinane-type sesquiterpenoids from Macrocystidia cucumis (basidiomycetes). Eur. J. Org. Chem. 1998, 1998, 73–79. [Google Scholar]
- Hearn, M.T.W.; Turner, J.L. The carbon-13 nuclear magnetic resonance spectra of the antibiotic polyacetylenic nitrile, diatretyne 2 (7-cyanohept-trans-2-ene-4,6-diynoic acid), and related compounds. J. Chem. Soc. Perkin Trans. 2 1976, 1976, 1027–1029. [Google Scholar] [CrossRef]
- Hodge, P.; Jones, E.R.H.; Lowe, G. Natural acetylenes. XXII. Trans-Dehydromatricaria ester as a biosynthetic precursor of some fungal polyacetylenes. J. Chem. Soc. Perkin Trans. 1 1966, 1966, 1216–1219. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Li, H.-J.; Chen, T.; Xie, Y.-L.; Chen, W.-D.; Zhu, X.-F.; Lan, W.-J. Isolation and Structural Elucidation of Chondrosterins F–H from the Marine Fungus Chondrostereum sp. Mar. Drugs 2013, 11, 551-558. https://doi.org/10.3390/md11020551
Li H-J, Chen T, Xie Y-L, Chen W-D, Zhu X-F, Lan W-J. Isolation and Structural Elucidation of Chondrosterins F–H from the Marine Fungus Chondrostereum sp. Marine Drugs. 2013; 11(2):551-558. https://doi.org/10.3390/md11020551
Chicago/Turabian StyleLi, Hou-Jin, Ting Chen, Ying-Lu Xie, Wen-Dan Chen, Xiao-Feng Zhu, and Wen-Jian Lan. 2013. "Isolation and Structural Elucidation of Chondrosterins F–H from the Marine Fungus Chondrostereum sp." Marine Drugs 11, no. 2: 551-558. https://doi.org/10.3390/md11020551
APA StyleLi, H.-J., Chen, T., Xie, Y.-L., Chen, W.-D., Zhu, X.-F., & Lan, W.-J. (2013). Isolation and Structural Elucidation of Chondrosterins F–H from the Marine Fungus Chondrostereum sp. Marine Drugs, 11(2), 551-558. https://doi.org/10.3390/md11020551





