Identification of Three Genes Encoding for the Late Acyltransferases of Lipid A in Cronobacter sakazakii
Abstract
:1. Introduction
2. Results and Discussion
2.1. Three Genes Encoding Late Acyltransferases Were Found in C. sakazakii BAA894
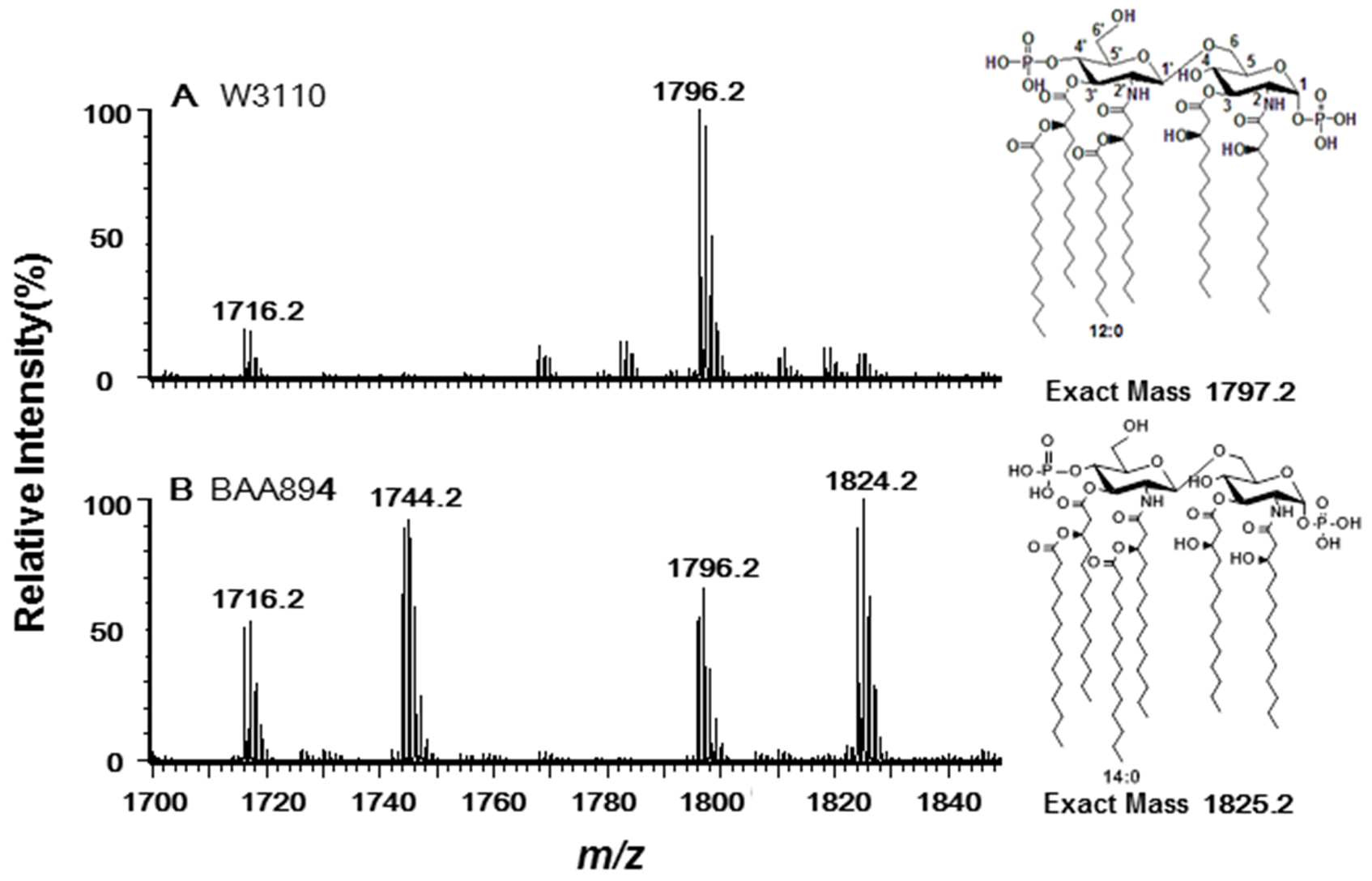

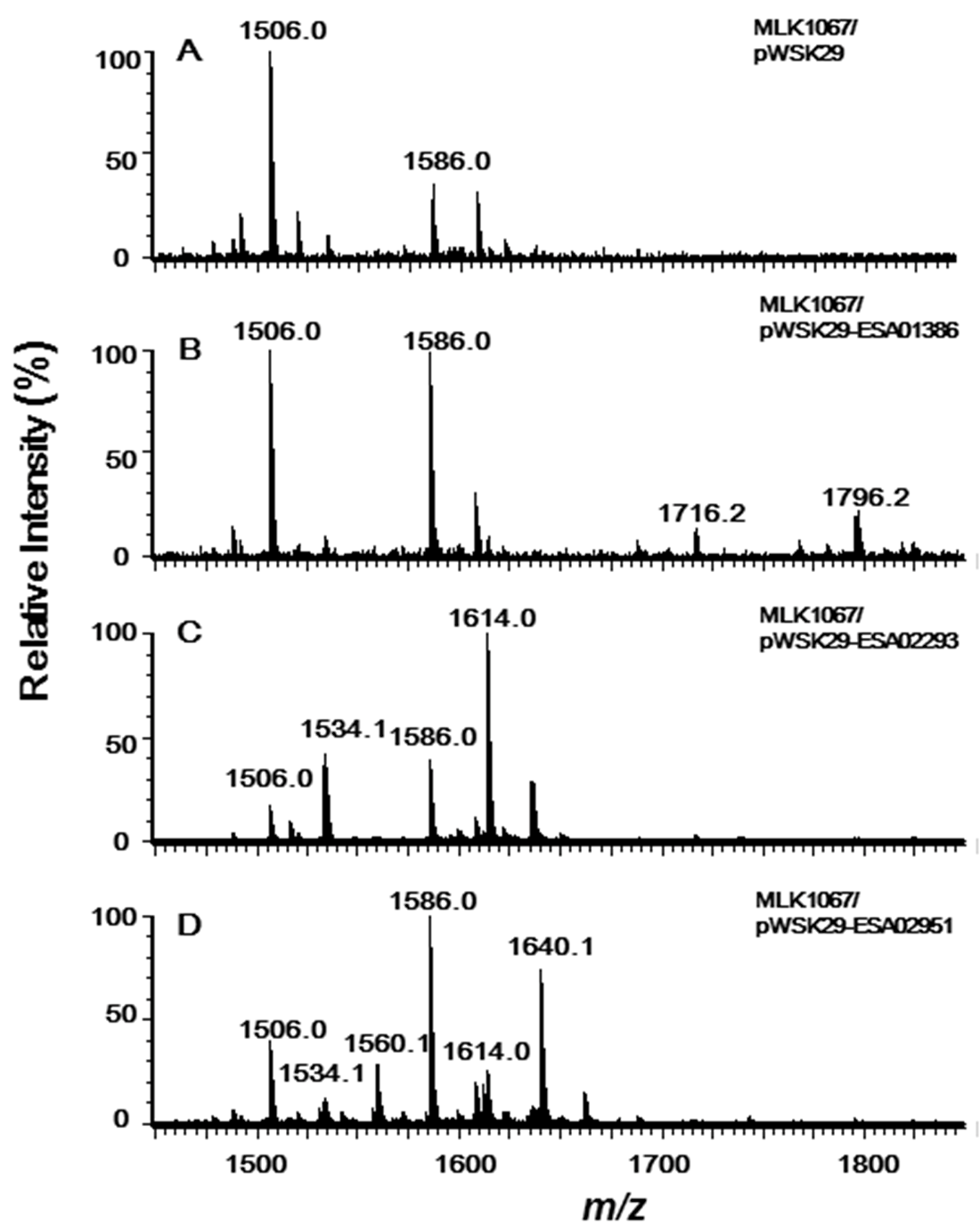
2.2. Acyltransferases Encoded by ESA02293 and ESA02952 Are Homologous to E. coli LpxL and LpxP, Respectively

2.3. Acyltransferase Encoded by ESA01386 Is the Homologue of E. coli LpxM
3. Experimental Section
3.1. DNA Preparation and PCR Techniques
3.2. Construction of Plasmids Containing Genes ESA02293, ESA02951 or ESA01386
| Strains or oligonucleotides | Relevant characteristic or sequence | Source or purpose |
|---|---|---|
| Strains | ||
| ATCC BAA894 | Wild-type C. sakazakii | ATCC |
| W3110 | Wide-type E. coli | ATCC |
| MKV15b | W3110 lpxL::Tn10 lpxM::Ωcam lpxP::kan | [17] |
| MKV15b/pWSK29 | MKV15b harboring pWSK29 | This work |
| MKV15b/pWSK29-ESA02293 | MKV15b harboring pWSK29-ESA02293 | This work |
| MKV15b/pWSK29-ESA02951 | MKV15b harboring pWSK29-ESA02951 | This work |
| MKV15b/pWSK29-ESA01386 | MKV15b harboring pWSK29-ESA01386 | This work |
| MLK1067 | W3110 lpxM::Ωcam | [16] |
| MLK1067/pWSK29 | MLK1067 harboring pWSK29 | This work |
| MLK1067/pWSK29-ESA01386 | MLK1067 harboring pWSK29-ESA01386 | This work |
| MLK1067/pWSK29-ESA02293 | MLK1067 harboring pWSK29-ESA02293 | This work |
| MLK1067/pWSK29-ESA02951 | MLK1067 harboring pWSK29-ESA02951 | This work |
| Oligonucleotides | ||
| F-ESA02293 | GCTCTAGAATGACGCATTTACCGCAATT | Forward primer for cloning ESA-02293 |
| R-ESA02293 | CGGGATCCTTAGTAGCGGGACGGCACGC | Reverse primer for cloning ESA-02293 |
| F-ESA02951 | GCTCTAGAATGAATCACGTCACGCCTTTTTC | Forward primer for cloning ESA-02951 |
| R-ESA02951 | CGGGATCCTCAGGTATAGAGCGACGCTTC | Reverse primer for cloning ESA-02951 |
| F-ESA01386 | GCTCTAGAATGGACACAAAAAAAACAAAAAGTG | Forward primer for cloning ESA-01386 |
| R-ESA01386 | CGGGATCCTTATTTCGGATAAAGATCTTTGC | Reverse primer for cloning ESA-01386 |
3.3. Lipid A Isolation from Different E. coli Strains
3.4. Mass Spectrometry Analysis
4. Conclusions
Acknowledgments
- Samples Availability: Available from the authors.
References
- Wang, X.; Quinn, P.J. Lipopolysaccharide: Biosynthetic pathway and structure modification. Prog. Lipid Res. 2010, 49, 97–107. [Google Scholar] [CrossRef]
- Raetz, C.R.; Garrett, T.A.; Reynolds, C.M.; Shaw, W.A.; Moore, J.D.; Smith, D.C.; Ribeiro, A.A.; Murphy, R.C.; Ulevitch, R.J.; Fearns, C.; et al. Kdo2-Lipid A of Escherichia coli, a defined endotoxin that activates macrophages via TLR-4. J. Lipid Res. 2006, 47, 1097–1111. [Google Scholar] [CrossRef]
- Miller, S.I.; Ernst, R.K.; Bader, M.W. LPS, TLR4 and infectious disease diversity. Nat. Rev. Microbiol. 2005, 3, 36–46. [Google Scholar] [CrossRef]
- Triantafilou, M.; Triantafilou, K. The dynamics of LPS recognition: Complex orchestration of multiple receptors. J. Endotoxin Res. 2005, 11, 5–11. [Google Scholar]
- Chen, J.; Tao, G.; Wang, X. Construction of an Escherichia coli mutant producing monophosphoryl lipid A. Biotechnol. Lett. 2011, 33, 1013–1019. [Google Scholar] [CrossRef]
- Zhang, C.; Li, Y.; Tao, G.; Li, Y.; Wang, X. Characterization of lipid A Cronobacter sakazakii. Eur. J. Mass Spectrom. 2010, 16, 531–538. [Google Scholar] [CrossRef]
- Raetz, C.R.; Guan, Z.; Ingram, B.O.; Six, D.A.; Song, F.; Wang, X.; Zhao, J. Discovery of new biosynthetic pathways: The lipid A story. J. Lipid Res. 2009, 50, S103–S108. [Google Scholar]
- Williams, A.H.; Raetz, C.R. Structural basis for the acyl chain selectivity and mechanism of UDP-N-acetylglucosamine acyltransferase. Proc. Natl. Acad. Sci. USA 2007, 104, 13543–13550. [Google Scholar]
- Buetow, L.; Smith, T.K.; Dawson, A.; Fyffe, S.; Hunter, W.N. Structure and reactivity of LpxD, the N-acyltransferase of lipid A biosynthesis. Proc. Natl. Acad. Sci. USA 2007, 104, 4321–4326. [Google Scholar]
- Li, Y.; Powell, D.A.; Shaffer, S.A.; Rasko, D.A.; Pelletier, M.R.; Leszyk, J.D.; Scott, A.J.; Masoudi, A.; Goodlett, D.R.; Wang, X.; et al. LPS remodeling is an evolved survival strategy for bacteria. Proc. Natl. Acad. Sci. USA 2012, 109, 8716–8721. [Google Scholar]
- McLendon, M.K.; Schilling, B.; Hunt, J.R.; Apicella, M.A.; Gibson, B.W. Identification of LpxL, a late acyltransferase of Francisella tularensis. Infect. Immun. 2007, 75, 5518–5531. [Google Scholar]
- Clementz, T.; Bednarski, J.J.; Raetz, C.R. Function of the htrB high temperature requirement gene of Escherchia coli in the acylation of lipid A: HtrB catalyzed incorporation of laurate. J. Biol. Chem. 1996, 271, 12095–12102. [Google Scholar]
- Geurtsen, J.; Angevaare, E.; Janssen, M.; Hamstra, H.J.; ten Hove, J.; de Haan, A.; Kuipers, B.; Tommassen, J.; van der Ley, P. A novel secondary acyl chain in the lipopolysaccharide of Bordetella pertussis required for efficient infection of human macrophages. J. Biol. Chem. 2007, 282, 37875–37884. [Google Scholar]
- Carty, S.M.; Sreekumar, K.R.; Raetz, C.R. Effect of cold shock on lipid A biosynthesis in Escherichia coli. Induction At 12 degrees C of an acyltransferase specific for palmitoleoyl-acyl carrier protein. J. Biol. Chem. 1999, 274, 9677–9685. [Google Scholar]
- Vorachek-Warren, M.K.; Carty, S.M.; Lin, S.; Cotter, R.J.; Raetz, C.R. An Escherichia coli mutant lacking the cold shock-induced palmitoleoyltransferase of lipid A biosynthesis: Absence of unsaturated acyl chains and antibiotic hypersensitivity at 12 degrees C. J. Biol. Chem. 2002, 277, 14186–14193. [Google Scholar]
- Brozek, K.A.; Raetz, C.R. Biosynthesis of lipid A in Escherichia coli. Acyl carrier protein-dependent incorporation of laurate and myristate. J. Biol. Chem. 1990, 265, 15410–15417. [Google Scholar]
- Vorachek-Warren, M.K.; Ramirez, S.; Cotter, R.J.; Raetz, C.R. A triple mutant of Escherichia coli lacking secondary acyl chains on lipid A. J. Biol. Chem. 2002, 277, 14194–14205. [Google Scholar]
- Metzger, L.E.; Raetz, C.R. An alternative route for UDP-diacylglucosamine hydrolysis in bacterial lipid A biosynthesis. Biochemistry 2010, 49, 6715–6726. [Google Scholar] [CrossRef]
- Bligh, E.G.; Dyer, W.J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 1959, 37, 911–917. [Google Scholar] [CrossRef]
- MassLynx Software, version 4.1, Waters Corporation: Milford, MA, USA, 2005.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Cai, L.; Li, Y.; Tao, G.; Guo, W.; Zhang, C.; Wang, X. Identification of Three Genes Encoding for the Late Acyltransferases of Lipid A in Cronobacter sakazakii. Mar. Drugs 2013, 11, 377-386. https://doi.org/10.3390/md11020377
Cai L, Li Y, Tao G, Guo W, Zhang C, Wang X. Identification of Three Genes Encoding for the Late Acyltransferases of Lipid A in Cronobacter sakazakii. Marine Drugs. 2013; 11(2):377-386. https://doi.org/10.3390/md11020377
Chicago/Turabian StyleCai, Liping, Yanyan Li, Guanjun Tao, Wen Guo, Chan Zhang, and Xiaoyuan Wang. 2013. "Identification of Three Genes Encoding for the Late Acyltransferases of Lipid A in Cronobacter sakazakii" Marine Drugs 11, no. 2: 377-386. https://doi.org/10.3390/md11020377




