Identification and Functional Characterization of Genes Encoding Omega-3 Polyunsaturated Fatty Acid Biosynthetic Activities from Unicellular Microalgae
Abstract
:1. Introduction
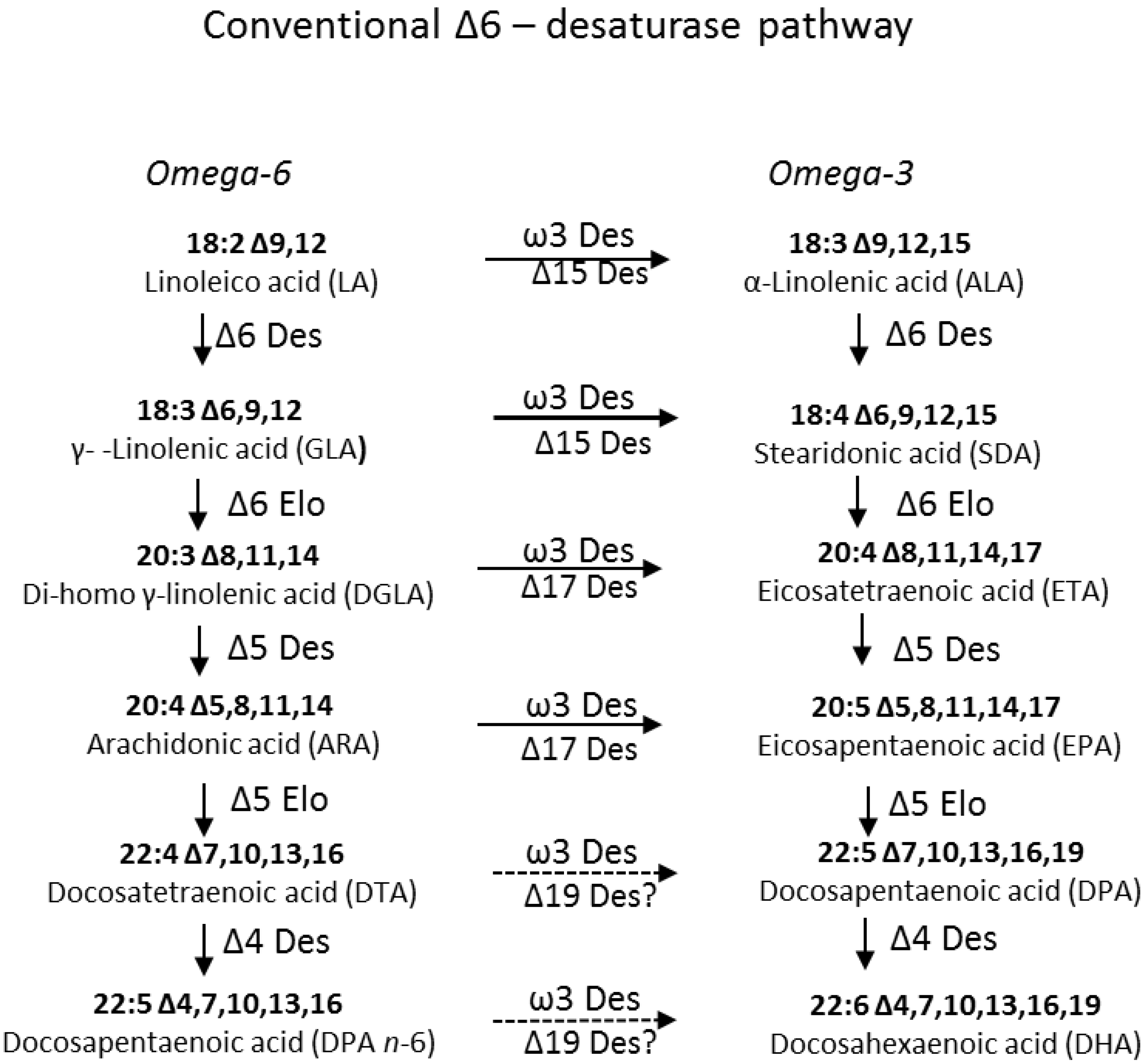
2. Results and Discussion
2.1. Identification and Functional Characterization of Ostreococcus RCC809 Genes for “Front-End” Desaturases
2.1.1. Fatty Acid Composition of Ostreococcus RCC809

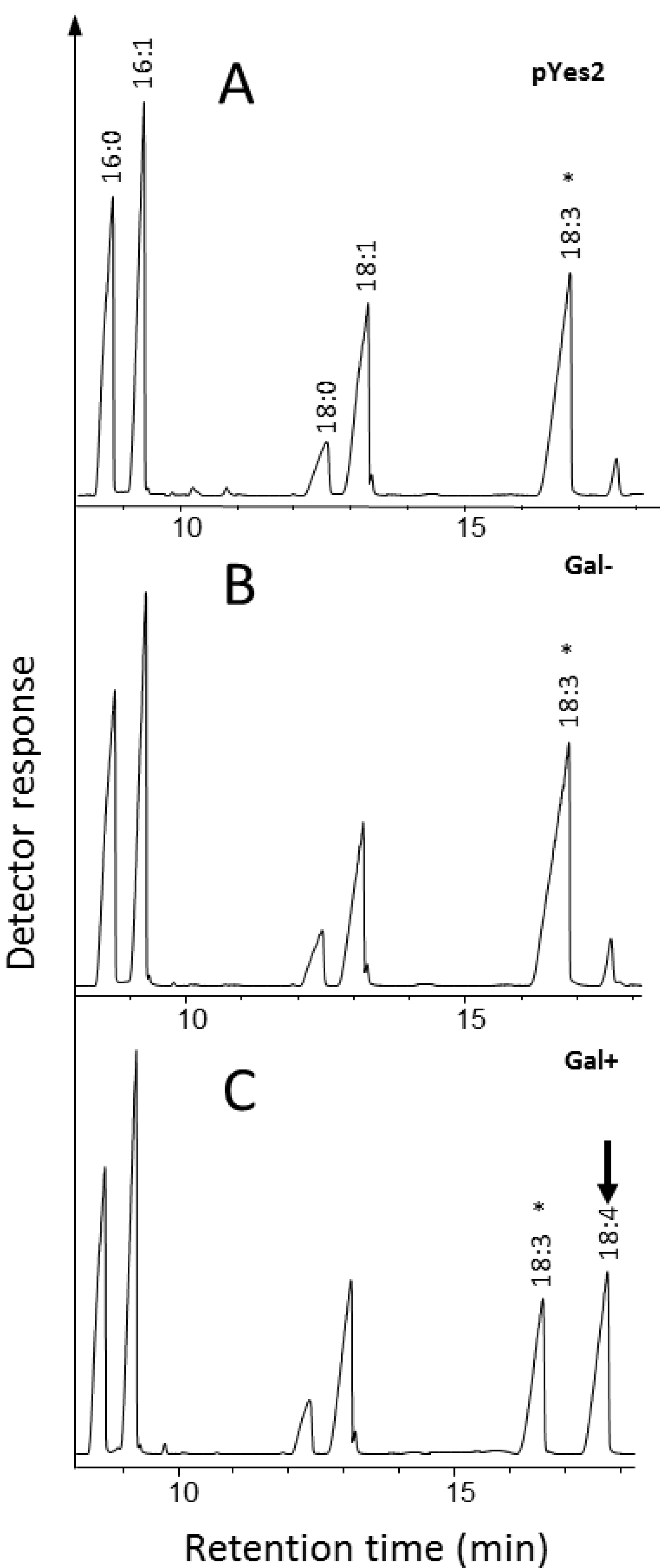
2.1.2. Identification and Functional Characterization in Yeast of a Putative Δ6-Desaturase from Ostreococcus RCC809
| Species | Protein ID | Amino Acids | Closest Match on Genbank, % Identity | Designation | Defined Function |
|---|---|---|---|---|---|
| Ostreococcus RCC809 | 59992 | 461 | Δ6-desaturase from O.lucimarinus (82%) Accession number: DAA34893.1 | Ost809D6 | C18 Δ6-desaturase |
| Ostreococcus RCC809 | 40461 | 459 | Δ4-desaturase from O. lucimarinus (85%) Accession number: XP_001415743.1 | Ost809D4 | C22 Δ4-desaturase |
| Construct | ||||||||
|---|---|---|---|---|---|---|---|---|
| Fatty Acid Composition (Molar %) ± SD | ||||||||
| Ost809D6 Gal − | Ost809D6 Gal + | Ost809D4 Gal − | Ost809D4 Gal + | FcElo6 Gal − | FcElo6 Gal + | FcElo6 Gal − | FcElo6 Gal + | |
| 16:0 | 22.3 ± 0.1 | 22.2 ± 0.4 | 32.8 ± 0.3 | 34.4 ± 0.2 | 24.5 ± 0.3 | 22.1 ± 0.1 | 20.1 ± 0.4 | 21.4 ± 0.1 |
| 16:1 | 25.4 ± 0.3 | 26.1 ± 0.3 | 38.9 ± 0.4 | 37.8 ± 0.3 | 26.8 ± 0.2 | 25.1 ± 0.3 | 22.3 ± 0.2 | 21.9 ± 0.3 |
| 18:0 | 3.7 ± 0.5 | 3.3 ± 0.3 | 7.9 ± 0.4 | 7.3 ± 0.5 | 3.7 ± 0.4 | 3.2 ± 0.3 | 3.7 ± 0.3 | 2.8 ± 0.4 |
| 18:1 | 13.4 ± 0.4 | 14.2 ± 0.4 | 17.8 ± 0.3 | 17.2 ± 0.4 | 15.3 ± 0.3 | 13.2 ± 0.5 | 3.4 ± 0.2 | 3.6 ± 0.3 |
| LA | ND | ND | ND | ND | ND | ND | ND | ND |
| GLA | ND | ND | ND | ND | 29.7 ± 0.2 | 22.3 ± 0.1 | 28.2 ± 0.4 | 18.6 ± 0.5 |
| ALA | 32.9 ± 0.2 | 15.7 ± 0.1 | ND | ND | ND | ND | ND | ND |
| SDA | 2.3 ± 0.6 | 18.5 ± 0.1 | ND | ND | ND | ND | 22.3 ± 0.3 | 12.2 ± 0.4 |
| 20:4n-3 | ND | ND | ND | ND | ND | ND | ND | 7.8 ± 0.3 |
| DGLA | ND | ND | ND | ND | ND | 14.1 ± 0.2 | ND | 11.7 ± 0.4 |
| DPA | ND | ND | 2.6 ± 0.3 | 2.8 ± 0.2 | ND | ND | ND | ND |
| DHA | ND | ND | ND | 0.5 ± 0.2 | ND | ND | ND | ND |
2.1.3. Identification and Functional Characterization of a Putative Δ4-Desaturase from Ostreococcus RCC809

2.2. Identification and Functional Characterization of a Putative Δ6-Elongase from Fragilariopsis cylindrus
2.2.1. Fatty Acid Composition of Fragilariopsis cylindrus
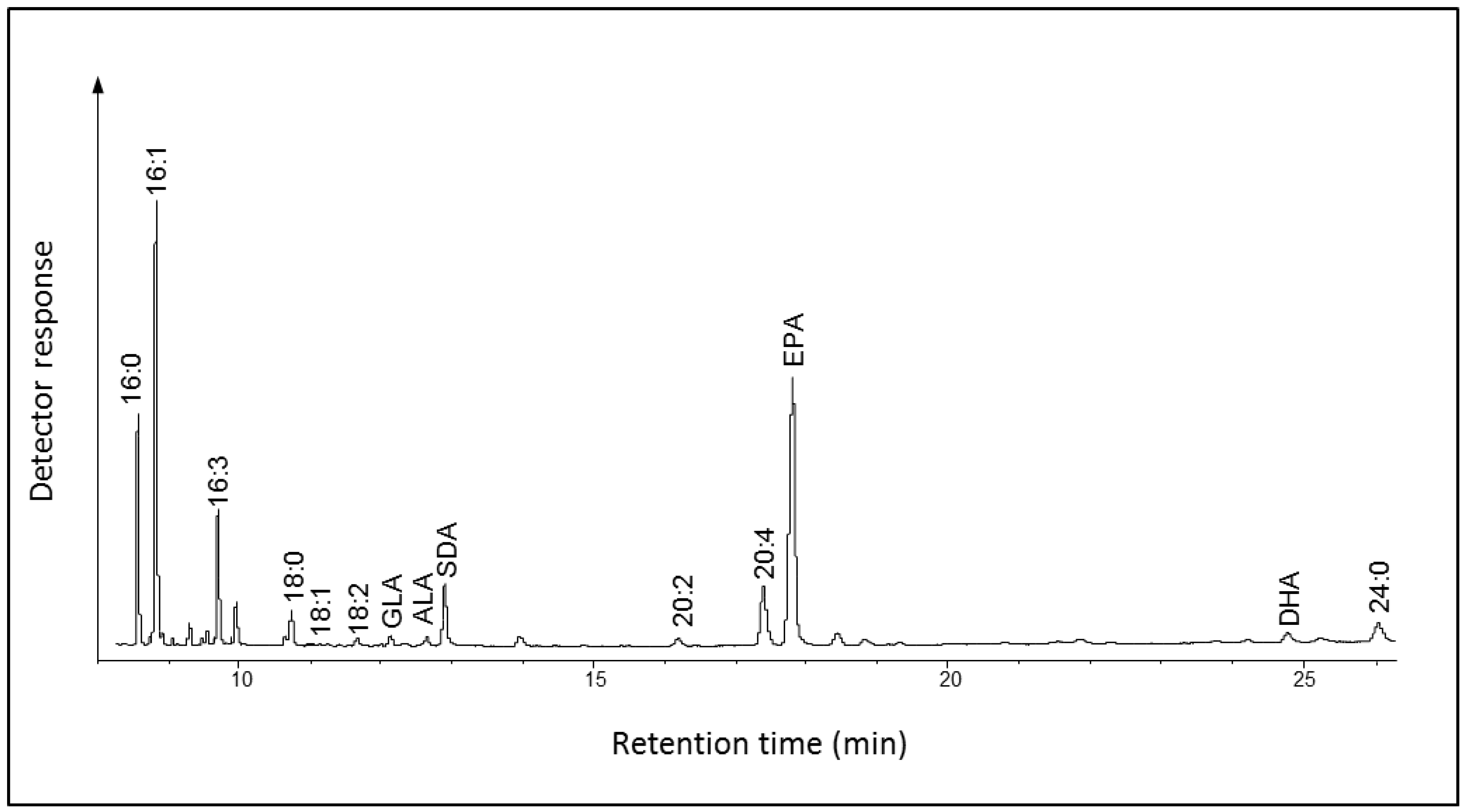
2.2.2. Functional Expression in Yeast of a Putative Elongase

3. Experimental Section
3.1. Growth and Harvesting of Microalgal Strains
3.2. Identification and Cloning of Putative PUFA Genes
3.3. Functional Expression in Yeast
3.4. Fatty Acid Analysis
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Voigt, R.G.; Jensen, C.L.; Fraley, J.K.; Rozelle, J.C.; Brown, F.R.; Heird, W.C. Relationship between omega-3 long-chain polyunsaturated fatty acid status during early infancy and neurodevelopmental status at 1 year of age. J. Hum. Nutr. Diet 2000, 15, 111–120. [Google Scholar]
- Calder, P.C. N-3 polyunsaturated fatty acids and inflammation: from molecular biology to the clinic. Lipids 2003, 38, 343–352. [Google Scholar] [CrossRef]
- Haslam, R.P.; Ruiz-Lopez, N.; Eastmond, P.; Moloney, M.; Sayanova, O.; Napier, J.A. The modification of plant oil composition via metabolic engineering—better nutrition by design. Plant Biotechnol. J. 2013, 11, 157–168. [Google Scholar] [CrossRef]
- Ruiz-Lopez, N.; Haslam, R.P.; Usher, S.L.; Napier, J.A.; Sayanova, O. Reconstitution of EPA and DHA biosynthesis in Arabidopsis: Iterative metabolic engineering for the synthesis of n-3 LC-PUFAs in transgenic plants. Metab. Eng. 2013, 17, 30–41. [Google Scholar] [CrossRef]
- Petrie, J.R.; Shrestha, P.; Zhou, X.R.; Mansour, M.P.; Liu, Q.; Belide, S.; Nichols, P.D.; Singh, S.P. Metabolic Engineering Plant Seeds with Fish Oil-Like Levels of DHA. PLoS One 2012, 7, e49165. [Google Scholar] [CrossRef]
- Venegas-Calerón, M.; Sayanova, O.; Napier, J.A. An alternative to fish oils: Metabolic engineering of oil-seed crops to produce omega-3 long chain polyunsaturated fatty acids. Prog. Lipid Res. 2010, 49, 108–119. [Google Scholar] [CrossRef]
- Sayanova, O.V.; Beaudoin, F.; Michaelson, L.V.; Shewry, P.R.; Napier, J.A. Identification of Primula fatty acid Δ6-desaturases with n-3 substrate preferences. FEBS Lett. 2003, 542, 100–104. [Google Scholar] [CrossRef]
- Sayanova, O.; Haslam, R.; Venegas-Calerón, M.; Napier, J.A. Identification of Primula “front-end” desaturases with distinct n-6 or n-3 substrate preferences. Planta 2006, 224, 1269–1277. [Google Scholar] [CrossRef]
- García-Maroto, F.; Mañas-Fernández, A.; Garrido-Cárdenas, J.A.; Alonso, D.L. Substrate specificity of acyl-Δ6-desaturases from Continental versus Macaronesian Echium species. Phytochemistry 2006, 67, 540–544. [Google Scholar] [CrossRef]
- Hoffmann, M.; Wagner, M.; Abbadi, A.; Fulda, M.; Feussner, I. Metabolic engineering of omega3-very long chain polyunsaturated fatty acid production by an exclusively acyl-CoA-dependent pathway. J. Biol. Chem. 2008, 283, 22352–22362. [Google Scholar] [CrossRef]
- Petrie, J.R.; Shrestha, P.; Mansour, M.P.; Nichols, P.D.; Liu, Q.; Singh, S.P. Metabolic engineering of omega-3 long-chain polyunsaturated fatty acids in plants using an acyl-CoA Delta6-desaturase with omega-3 preference from the marine microalga Micromonas pusilla. Metab. Eng. 2010, 12, 233–240. [Google Scholar] [CrossRef]
- Petrie, J.R.; Liu, Q.; Mackenzie, A.; Shrestha, P.; Mansour, M.P.; Robert, S.S.; Frampton, D.F.; Blackburn, S.I.; Nichols, P.D.; Singh, S.P. Isolation and characterization of a high-efficiency desaturase and elongases from microalgae for transgenic LC-PUFA production. Mar. Biotechnol. 2010, 12, 430–438. [Google Scholar] [CrossRef]
- Demir-Hilton, E.; Sudek, S.; Cuvelier, M.L.; Gentemann, C.L.; Zehr, J.P.; Worden, Z. Global distribution patterns of distinct clades of the photosynthetic picoeukaryote Ostreococcus. ISME J. 2011, 5, 1095–1107. [Google Scholar] [CrossRef]
- Domergue, F.; Abbadi, A.; Zähringer, U.; Moreau, H.; Heinz, E. In vivo characterization of the first acyl-CoA Delta6-desaturase from a member of the plant kingdom, the microalga Ostreococcus tauri. Biochem. J. 2005, 389, 483–490. [Google Scholar]
- Meyer, A.; Kirsch, H.; Domergue, F.; Abbadi, A.; Sperling, P.; Bauer, J.; Cirpus, P.; Zank, T.K.; Moreau, H.; Roscoe, T.J.; et al. Novel fatty acid elongases and their use for the reconstitution of docosahexaenoic acid biosynthesis. J. Lipid Res. 2004, 45, 1899–1909. [Google Scholar] [CrossRef]
- Ahman, K.; Heilmann, M.; Feussner, I. Identification of a Δ4-desaturase from microalga Ostreococcus lucimarinus. Eur. J. Lipid Sci. Technol. 2011, 113, 832–840. [Google Scholar] [CrossRef]
- Wagner, M.; Hoppe, K.; Czabany, T.; Heilmann, M.; Daum, G.; Feussner, I.; Fulda, M. Identification and characterization of an acyl-CoA: Diacylglycerol acyltransferase 2 (DGAT2) gene from the microalga O. tauri. Plant Phys. Biochem. 2010, 48, 407–416. [Google Scholar] [CrossRef]
- Joint Genome Institute Ostreococcus sp. RCC809 2.0 genome project. Available online: http://genome.jgi-psf.org/OstRCC809_2/OstRCC809_2.home.html (accessed on 5 April 2012).
- Tonon, T.; Sayanova, O.; Michaelson, L.V.; Qing, R.; Harvey, D.; Larson, T.R.; Li, Y.; Napier, J.A.; Graham, I.A. Fatty acid desaturases from the microalga Thalassiosira pseudonana. FEBS J. 2005, 272, 3401–3412. [Google Scholar] [CrossRef]
- Sayanova, O.; Haslam, R.; Venegas-Calerón, M.; Ruiz-Lopez, N.; Worthy, C.; Rooks, P.; Allen, M.J.; Napier, J.A. Identification and functional characterization of genes encoding the omega-3 polyunsaturated fatty acid biosynthetic pathway from the coccolithophore Emiliania huxleyi. Phytochemistry 2011, 72, 594–600. [Google Scholar] [CrossRef]
- Joint Genome Institute Fragilariopsis cylindrus genome project. Available online: http://genome.jgi-psf.org/Fracy1/Fracy1.home.html (accessed on 10 May 2012).
- Harrison, P.J.; Waters, R.E.; Taylor, F.J.R. A broad spectrum artificial seawater medium for coastal and open ocean phytoplankton. J. Phycol. 1980, 16, 28–35. [Google Scholar]
- Berges, J.A.; Franklin, D.J.; Harrison, P.J. Evolution of an artificial seawater medium: improvements in enriched seawater, artificial water over the past two decades. J. Phycol. 2001, 37, 1138–1145. [Google Scholar] [CrossRef]
- Morel, F.M.M.; Rueter, J.G.; Anderson, D.M.; Guillard, R.R.L. Aquil: A chemically defined phytoplankton culture medium for trace metal studies. J. Phycol. 1979, 15, 135–141. [Google Scholar]
- Price, N.M.; Harrison, G.I.; Hering, J.G.; Hudson, R.J.; Nirel, P.M.V.; Palenik, B.; Morel, F.M.M. Preparation and chemistry of the artificial algal culture medium Aquil. Biol. Oceanogr. 1989, 6, 443–461. [Google Scholar]
- Sunda, W.G.; Price, N.M.; Morel, F.M.M. Trace metal ion buffers and their use in culture studies. In Algal Culturing Techniques; Anderson, R.A., Ed.; Academic Press: Amsterdam, The Netherlands, 2005; pp. 35–63. [Google Scholar]
- Sayanova, O.; Smith, M.A.; Lapinskas, P.; Stobart, A.K.; Dobson, G.; Christie, W.W.; Shewry, P.R.; Napier, J.A. Expression of a borage desaturase cDNA containing an N-terminal cytochrome b5 domain results in the accumulation of high levels of delta6-desaturated fatty acids in transgenic tobacco. Proc. Natl. Acad. Sci. USA 1997, 94, 4211–4216. [Google Scholar] [CrossRef]
Supplementary Files
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Vaezi, R.; Napier, J.A.; Sayanova, O. Identification and Functional Characterization of Genes Encoding Omega-3 Polyunsaturated Fatty Acid Biosynthetic Activities from Unicellular Microalgae. Mar. Drugs 2013, 11, 5116-5129. https://doi.org/10.3390/md11125116
Vaezi R, Napier JA, Sayanova O. Identification and Functional Characterization of Genes Encoding Omega-3 Polyunsaturated Fatty Acid Biosynthetic Activities from Unicellular Microalgae. Marine Drugs. 2013; 11(12):5116-5129. https://doi.org/10.3390/md11125116
Chicago/Turabian StyleVaezi, Royah, Johnathan A. Napier, and Olga Sayanova. 2013. "Identification and Functional Characterization of Genes Encoding Omega-3 Polyunsaturated Fatty Acid Biosynthetic Activities from Unicellular Microalgae" Marine Drugs 11, no. 12: 5116-5129. https://doi.org/10.3390/md11125116




