Mass Spectrometry-Based Metabolomics to Elucidate Functions in Marine Organisms and Ecosystems
Abstract
:Abbreviations
| AMDIS | Automated mass spectral deconvolution and identification system |
| AMS | Aerosol mass spectrometry |
| APCI | Atmospheric pressure chemical ionization |
| ARA | arachidonic acid |
| CCAP | Culture collection of algae and protozoa |
| DAD | Diode array detector |
| DESI | Desorption electrospray ionization |
| DHA | Docosahexaenoic acid |
| ECNI | Electron capture negative ionization |
| ELSD | Evaporative light scattering detector |
| EI | Electronic impact |
| FWS | Ectocarpus freshwater strain |
| GC | Gas chromatography |
| HS | Headspace |
| I3 | Integrated Infrastructure Initiative |
| ICP | Inductively coupled plasma |
| FP7 | Framework Programme 7 |
| FT-ICR | Fourier transform ion cyclotron resonance |
| LAESI | Laser ablation electrospray ionization |
| LC | Liquid chromatography |
| MALDI | Matrix assisted laser desorption ionization |
| MRM | Multiple reaction monitoring |
| MS | Mass spectrometry |
| NGS | Next Generation Sequencing |
| NICI | Negative ion chemical ionization |
| NMR | Nuclear magnetic resonance |
| PDMS | Polydimethylsiloxane |
| PUAs | Polyunsaturated aldehydes |
| PUFAs | Polyunsatured fatty acids |
| Q | quadrupole |
| SBSE | Stir bar sorptive extraction |
| SIMS | Secondary ion mass spectrometry |
| SPE | Solid phase extraction |
| SPME | Solid phase microextraction |
| SWS | Ectocarpus seawater strain |
| TD | Thermodesorption |
| TMS | Trimethylsilyl derivatives |
| TOF | Time of flight |
| UPLC | Ultra performance liquid chromatography |
| VHOCs | Volatile halogenated organic compounds |
| VOCs | Volatile organic compounds. |
1. Introduction
2. Overview of Mass Spectrometry (MS)-Based Metabolic Profiling/Metabolomics in Marine Organisms
2.1. Bacteria (Heterotrophic and Cyanobacteria)
2.2. Micro and Macroalgae
2.2.1. Diatoms
2.2.2. Macroalgae
2.3. Animals (Vertebrates and Invertebrates)
2.3.1. Vertebrates
2.3.2. Invertebrates
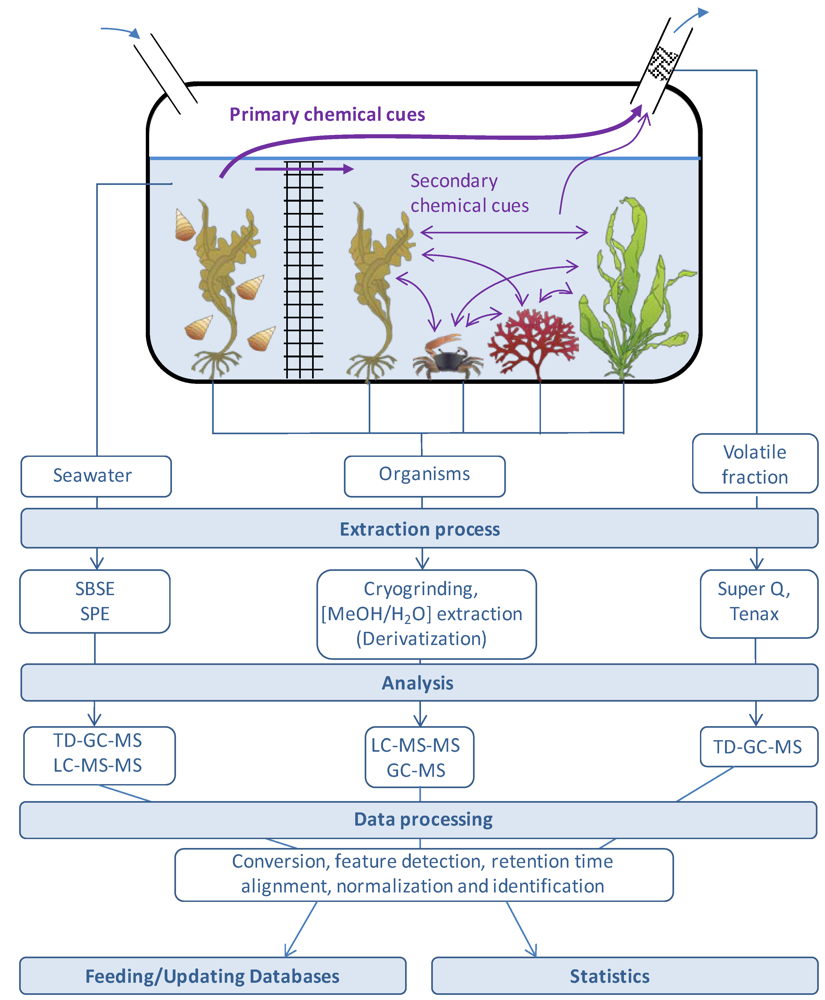
3. Metabolic Footprinting: Analysis of Seawater Using MS-Based Metabolite Profiling Techniques, a Powerful Tool in Chemical Ecology
4. Technical Challenges
4.1. Interferences
4.2. Extraction of Metabolites from Seawater
4.3. Data Treatment
4.4. Towards Metabolomics Database Dedicated to Marine (eco)Systems
4.5. Federation of Marine Biologists and Ecologists for a Better Integration and Promotion of Standard Initiatives
5. Metabolite Profiling for Integrative and Systems Biology
6. Conclusion and Perspectives
Acknowledgments
References
- Mora, C.; Tittensor, D.P.; Adl, S.; Simpson, A.G.B.; Worm, B. How many species are there on earth and in the ocean? PLoS Biol. 2011, 9, e1001127. [Google Scholar] [CrossRef]
- Blunt, J.W.; Copp, B.R.; Keyzers, R.A.; Munro, M.H.G.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2012, 29, 144–222. [Google Scholar]
- Gerwick, W.H.; Moore, B.S. Lessons from the past and charting the future of marine natural products drug discovery and chemical biology. Chem. Biol. 2012, 19, 85–98. [Google Scholar]
- Miller, M.G. Environmental metabolomics: A SWOT analysis (strengths, weaknesses, opportunities, and threats). J. Proteome Res. 2007, 6, 540–545. [Google Scholar] [CrossRef]
- Prince, E.K.; Poulson, K.L.; Myers, T.L.; Sieg, R.D.; Kubanek, J. Characterization of allelopathic compounds from the red tide dinoflagellate Karenia brevis. Harmful Algae 2010, 10, 39–48. [Google Scholar] [CrossRef]
- Viant, M.R.; Bearden, D.W.; Bundy, J.G.; Burton, I.W.; Collette, T.W.; Ekman, D.R.; Ezernieks, V.; Karakachi, T.K.; Lin, C.Y.; Rochfort, S.; et al. International NMR-based environmental metabolomics intercomparison exercise. Environ. Sci. Technol. 2009, 43, 219–225. [Google Scholar]
- Weckwerth, W. Metabolomics in systems biology. Annu. Rev. Plant Biol. 2003, 54, 669–689. [Google Scholar]
- Hollywood, K.; Brison, D.R.; Goodacre, R. Metabolomics: Current technologies and future trends. Proteomics 2006, 6, 4716–4723. [Google Scholar]
- Viant, M.R. Metabolomics of aquatic organisms: The new “omics” on the block. Mar. Ecol. Prog. Ser. 2007, 332, 301–306. [Google Scholar]
- Jamers, A.; Blust, R.; De Coen, W. Omics in algae: Paving the way for a systems biological understanding of algal stress phenomena? Aquat. Toxicol. 2009, 92, 114–121. [Google Scholar] [CrossRef]
- Lucas, M.; Laplaze, L.; Bennett, M.J. Plant systems biology: Network matters. Plant Cell Environ. 2011, 34, 535–553. [Google Scholar]
- Weckwerth, W. Green systems biology—From single genomes, proteomes and metabolomes to ecosystems research and biotechnology. J. Proteomics 2011, 75, 284–305. [Google Scholar]
- Fürch, T.; Preusse, M.; Tomasch, J.; Zech, H.; Wagner-Döbler, I.; Wittmann, C. Metabolic fluxes in the central carbon metabolism of Dinoroseobacter shibae and Phaeobacter gallaeciensis, two members of the marine Roseobacter clade. BMC Microbiol. 2009, 9. [Google Scholar]
- Shin, M.H.; Lee, D.Y.; Liu, K.H.; Fiehn, O.; Kim, K.H. Evaluation of sampling and extraction methodologies for the global metabolic profiling of Saccharophagus degradans. Anal. Chem. 2010, 82, 6660–6666. [Google Scholar]
- Shin, M.H.; Lee, D.Y.; Skogerson, K.; Wohlgemuth, G.; Choi, I.G.; Fiehn, O.; Kim, K.H. Global metabolic profiling of plant cell wall polysaccharide degradation by Saccharophagus degradans. Biotechnol. Bioeng. 2010, 105, 477–488. [Google Scholar] [CrossRef]
- Shin, M.H.; Lee, D.Y.; Wohlgemuth, G.; Choi, I.-G.; Fiehn, O.; Kim, K.H. Global metabolite profiling of agarose degradation by Saccharophagus degradans 2-40. New Biotechnol. 2010, 27, 156–168. [Google Scholar]
- Lee, D.Y.; Fiehn, O. High quality metabolomic data for Chlamydomonas reinhardtii. Plant Methods 2008, 4. [Google Scholar]
- Schneemann, I.; Wiese, J.; Kunz, A.L.; Imhoff, J.F. Genetic approach for the fast discovery of phenazine producing bacteria. Mar. Drugs 2011, 9, 772–789. [Google Scholar]
- Lee, J.S.; Kim, Y.-S.; Park, S.; Kim, J.; Kang, S.-J.; Lee, M.-H.; Ryu, S.; Choi, J.M.; Oh, T.-K.; Yoon, J.-H. Exceptional production of both prodigiosin and cycloprodigiosin as major metabolic constituents by a novel marine bacterium, Zooshikella rubidus S1-1. Appl. Environ. Microbiol. 2011, 77, 4967–4973. [Google Scholar]
- Vynne, N.G.; Månsson, M.; Nielsen, K.F.; Gram, L. Bioactivity, chemical profiling, and 16S rRNA-based phylogeny of Pseudoalteromonas strains collected on a global research cruise. Mar. Biotechnol. 2011, 13, 1062–1073. [Google Scholar] [CrossRef]
- Mansson, M.; Gram, L.; Larsen, T.O. Production of bioactive secondary metabolites by marine Vibrionaceae. Mar. Drugs 2011, 9, 1440–1468. [Google Scholar]
- Wietz, M.; Mansson, M.; Gotfredsen, C.H.; Larsen, T.O.; Gram, L. Antibacterial compounds from Marine Vibrionaceae isolated on a global expedition. Mar. Drugs 2010, 8, 2946–2960. [Google Scholar] [CrossRef]
- Brito-Echeverría, J.; Lucio, M.; López-López, A.; Antón, J.; Schmitt-Kopplin, P.; Rosselló-Móra, R. Response to adverse conditions in two strains of the extremely halophilic species Salinibacter ruber. Extremophiles 2011, 15, 379–389. [Google Scholar] [CrossRef]
- Eisenhut, M.; Huege, J.; Schwarz, D.; Bauwe, H.; Kopka, J.; Hagemann, M. Metabolome phenotyping of inorganic carbon limitation in cells of the wild type and photorespiratory mutants of the cyanobacterium Synechocystis sp. strain PCC 6803. Plant Physiol. 2008, 148, 2109–2120. [Google Scholar] [CrossRef]
- Krall, L.; Huege, J.; Catchpole, G.; Steinhauser, D.; Willmitzer, L. Assessment of sampling strategies for gas chromatography-mass spectrometry (GC-MS) based metabolomics of cyanobacteria. J.Chromatogr. B 2009, 877, 2952–2960. [Google Scholar]
- Yang, C.; Hua, Q.; Shimizu, K. Metabolic flux analysis in Synechocystis using isotope distribution from 13C-labeled glucose. Metab. Eng. 2002, 4, 202–216. [Google Scholar] [CrossRef]
- Baran, R.; Bowen, B.P.; Northern, T.R. Untargeted metabolic footprinting reveals a surprising breadth of metabolite uptake and release by Synechococcus sp. PCC 7002. Mol. BioSyst. 2011, 7, 3200–3206. [Google Scholar] [CrossRef]
- Esquenazi, E.; Coates, C.; Simmons, L.; Gonzalez, D.; Gerwick, W.H.H.; Dorrestein, P.C. Visualizing the spatial distribution of secondary metabolites produced by marine cyanobacteria and sponges via MALDI-TOF imaging. Mol. BioSyst. 2008, 4, 562–570. [Google Scholar]
- Esquenazi, E.; Jones, A.C.; Byrum, T.; Dorrestein, P.C.; Gerwick, W.H. Temporal dynamics of natural product biosynthesis in marine cyanobacteria. Proc. Natl. Acad. Sci. USA 2011, 108, 5226–5231. [Google Scholar]
- Engene, N.; Choi, H.; Esquenazi, E.; Rottacker, E.C.; Ellisman, M.H.; Dorrestein, P.C.; Gerwick, W.H. Underestimated biodiversity as a major explanation for the perceived rich secondary metabolite capacity of the cyanobacterial genus Lyngbya. Environ. Microbiol. 2011, 13, 1601–1610. [Google Scholar] [CrossRef]
- Esquenazi, E.; Daly, M.; Bahrainwala, T.; Gerwick, W.H.; Dorrestein, P.C. On mobility mass spectrometry enables the efficient detection and identification of halogenated natural products from cyanobacteria with minimal sample preparation. Bioorg. Med. Chem. 2011, 19, 6639–6644. [Google Scholar]
- Nappo, M.; Berkov, S.; Codina, C.; Avila, C.; Messina, P.; Zupo, V.; Bastida, J. Metabolite profiling of the benthic diatom Cocconeis scutellum by GC-MS. J. Appl. Phycol. 2009, 21, 295–306. [Google Scholar] [CrossRef]
- Vidoudez, C.; Pohnert, G. Growth phase-specific release of polyunsaturated aldehydes by the diatom Skeletonema marinoi. J. Plankton Res. 2008, 30, 1305–1313. [Google Scholar] [CrossRef]
- Vidoudez, C.; Pohnert, G. Comparative metabolomics of the diatom Skeletonema marinoi in different growth phases. Metabolomics 2011. [Google Scholar]
- Lang, I.; Hodac, L.; Friedl, T.; Feussner, I. Fatty acid profiles and their distribution patterns in microalgae: A comprehensive analysis of more than 2000 strains from the SAG culture collection. BMC Plant Biol. 2011, 11. [Google Scholar]
- d’Ippolito, G.; Lamari, N.; Montresor, M.; Romano, G.; Cutignano, A.; Gerecht, A.; Cimino, G.; Fontana, A. 15S-lipoxygenase metabolism in the marine diatom Pseudo-nitzschia delicatissima. New Phytol. 2009, 183, 1064–1071. [Google Scholar] [CrossRef]
- Gu, Q.; David, F.; Lynen, F.; Vanormelingen, P.; Vyverman, W.; Rumpel, K.; Xu, G.; Sandra, P. Evaluation of ionic liquid stationary phases for one dimensional gas chromatography-mass spectrometry and comprehensive two dimensional gas chromatographic analyses of fatty acids in marine biota. J. Chromatogr. A 2011, 1218, 3056–3063. [Google Scholar]
- Yan, X.; Chen, D.; Xu, J.; Zhou, C. Profiles of photosynthetic glycerolipids in three strains of Skeletonema determined by UPLC-Q-TOF-MS. J. Appl. Phycol. 2011, 23, 271–282. [Google Scholar] [CrossRef]
- Allen, A.E.; Dupont, C.L.; Oborník, M.; Horák, A.; Nunes-Nesi, A.; McCrow, J.P.; Zheng, H.; Johnson, D.A.; Hu, H.; Fernie, A.R.; et al. Evolution and metabolic significance of the urea cycle in photosynthetic diatoms. Nature 2011, 473, 203–207. [Google Scholar]
- Vanelslander, B.; Paul, C.; Grueneberg, J.; Prince, E.K.; Gillard, J.; Sabbe, K.; Pohnert, G.; Vyverman, W. Daily bursts of biogenic cyanogen bromide (BrCN) control biofilm formation around a marine benthic diatom. Proc. Natl. Acad. Sci. USA 2012, 109, 2412–2417. [Google Scholar]
- Nylund, G.M.; Weinberger, F.; Rempt, M.; Pohnert, G. Metabolomic assessment of induced and activated chemical defence in the invasive red alga Gracilaria vermiculophylla. PLoS One 2011, 6, e29359. [Google Scholar]
- Lion, U.; Wiesemeier, T.; Weinberger, F.; Beltran, J.; Flores, V.; Faugeron, S.; Correa, J.; Pohnert, G. Phospholipases and galactolipases trigger oxylipin-mediated wound-activated defence in the red alga Gracilaria chilensis against epiphytes. ChemBioChem 2006, 7, 457–462. [Google Scholar] [CrossRef]
- Bouarab, K.; Adas, F.; Gaquerel, E.; Kloareg, B.; Salaün, J.-P.; Potin, P. The innate immunity of a marine red alga involves oxylipins from both the eicosanoid and octadecanoid pathways. Plant Physiol. 2004, 135, 1838–1848. [Google Scholar]
- Gaquerel, E.; Hervé, C.; Labrière, C.; Boyen, C.; Potin, P.; Salaün, J.-P. Evidence for oxylipin synthesis and induction of a new polyunsaturated fatty acid hydroxylase activity in Chondrus crispus in response to methyljasmonate. Biochim. Biophys. Acta 2007, 1771, 565–575. [Google Scholar]
- Jiang, Z.D.; Ketchum, S.O.; Gerwick, W.H. 5-Lipoxygenase-derived oxylipins from the red alga Rhodymenia pertusa. Phytochemistry 2000, 53, 129–133. [Google Scholar]
- Yuan, Y.V.; Westcott, N.D.; Hu, C.; Kitts, D.D. Mycosporine-like amino acid composition of the edible red alga, Palmaria palmata (dulse) harvested from the west and east coasts of Grand Manan Island, New Brunswick. Food Chem. 2009, 112, 321–328. [Google Scholar] [CrossRef]
- Lane, A.L.; Nyadong, L.; Galhena, A.S.; Shearer, T.L.; Stout, E.P.; Parry, R.M.; Kwasnik, M.; Wang, M.D.; Hay, M.E.; Fernandez, F.M.; et al. Desorption electrospray ionization mass spectrometry reveals surface-mediated antifungal chemical defense of a tropical seaweed. Proc. Natl. Acad. Sci. USA 2009, 106, 7314–7319. [Google Scholar]
- Payo, D.A.; Colo, J.; Calumpong, H.; de Clerck, O. Variability of non-polar secondary metabolites in the red alga Portieria. Mar. Drugs 2011, 9, 2438–2468. [Google Scholar] [CrossRef] [Green Version]
- Kundel, M.; Thorenz, U.R.; Petersen, J.H.; Huang, R.-J.; Bings, N.H.; Hoffmann, T. Application of mass spectrometric techniques for the trace analysis of short-lived iodine-containing volatiles emitted by seaweed. Anal. Bioanal. Chem. 2012, 84, 1439–1445. [Google Scholar]
- La Barre, S.; Potin, P.; Leblanc, C.; Delage, L. The halogenated metabolism of brown algae (Phaeophyta), its biological importance and its environmental significance. Mar Drugs. 2010, 31, 988–1010. [Google Scholar]
- Verhaeghe, E.F.; Fraysse, A.; Guerquin-Kern, J.L.; Wu, T.D.; Devès, G.; Mioskowski, C.; Leblanc, C.; Ortega, R.; Ambroise, Y.; Potin, P. Microchemical imaging of iodine distribution in the brown alga Laminaria digitata suggests a new mechanism for its accumulation. J. Biol. Inorg. Chem. 2008, 13, 257–269. [Google Scholar] [CrossRef]
- Klejdus, B.; Lojková, L.; Plaza, M.; Šnóblová, M.; Štěrbová, D. Hyphenated technique for the extraction and determination of isoflavones in algae: Ultrasound-assisted supercritical fluid extraction followed by fast chromatography with tandem mass spectrometry. J. Chromatogr. A 2010, 1217, 7956–7965. [Google Scholar] [CrossRef]
- El Hattab, J.; Culioli, G.; Piovetti, L.; Chitour, S.E.; Valls, R. Comparison of various extraction methods for identification and determination of volatile metabolites from the brown alga Dictyopteris membranacea. J. Chromatogr. A 2007, 1143, 1–7. [Google Scholar] [CrossRef]
- Ritter, A.; Goulitquer, S.; Salaün, J.-P.; Tonon, T.; Correa, J.A.; Potin, P. Copper stress induces biosynthesis of octadecanoid and eicosanoid oxygenated derivatives in the brown algal kelp Laminaria digitata. New Phytol. 2008, 180, 809–821. [Google Scholar] [CrossRef]
- Goulitquer, S.; Ritter, A.; Thomas, F.; Ferec, C.; Salaün, J.-P.; Potin, P. Release of volatile aldehydes by the brown algal kelp Laminaria digitata in response to both biotic and abiotic stress. ChembioChem 2009, 10, 977–982. [Google Scholar] [CrossRef]
- Gravot, A.; Dittami, S.M.; Rousvoal, S.; Lugan, R.; Eggert, A.; Collén, J.; Boyen, C.; Bouchereau, A.; Tonon, T. Diurnal oscillations of metabolite abundances and gene analysis provide new insights into central metabolic processes of the brown alga Ectocarpus siliculosus. New Phytol. 2010, 188, 98–110. [Google Scholar] [CrossRef]
- Dittami, S.M.; Gravot, A.; Renault, D.; Goulitquer, S.; Eggert, A.; Bouchereau, A.; Boyen, C.; Tonon, T. Integrative analysis of metabolite and transcript abundance during the short-term response to saline and oxidative stress in the brown alga Ectocarpus siliculosus. Plant Cell Environ. 2011, 34, 629–642. [Google Scholar] [CrossRef]
- Tonon, T.; Eveillard, D.; Prigent, S.; Bourdon, J.; Potin, P.; Boyen, C.; Siegel, A. Toward systems biology in brown algae to explore acclimation and adaptation to the shore environment. OMICS 2011, 15, 883–892. [Google Scholar]
- Dembitsky, V.M.; Řezanková, H.; Řezanka, T.; Hanuš, L.O. Variability of the fatty acids of the marine green algae belonging to the genus Codium. Biochem. Syst. Ecol. 2003, 31, 1125–1145. [Google Scholar] [CrossRef]
- Akakabe, Y.; Matsui, K.; Kajiwara, T. 2,4-Decadienals are produced via. (R)-11-HPITE from arachidonic acid in marine green alga Ulva conglobata. Bioorg. Med. Chem. 2003, 11, 3607–3609. [Google Scholar] [CrossRef]
- Tal, O.; Haim, A.; Harel, O.; Gerchman, Y. Melatonin as an antioxidant and its semi-lunar rhythm in green macroalga Ulva sp. J. Exp. Bot. 2011, 62, 1903–1910. [Google Scholar] [CrossRef]
- van Ginneken, V.J.T.; Helsper, J.P.F.G.; de Visser, W.; van Keulen, H.; Brandenburg, W.A. Polyunsaturated fatty acids in various macroalgal species from north Atlantic and tropical seas. Lipids Health. Dis. 2011, 10. [Google Scholar]
- Kumari, P.; Reddy, C.R.K.; Jha, B. Comparative evaluation and selection of a method for lipid and fatty acid extraction from macroalgae. Anal. Biochem. 2011, 415, 134–144. [Google Scholar]
- Kumar, M.; Kumari, P.; Trivedi, N.; Shukla, M.K.; Gupta, V.; Reddy, C.R.K.; Jha, B. Minerals, PUFAs and antioxidant properties of some tropical seaweeds from Saurashtra coast of India. J. Appl. Phycol. 2011, 23, 797–810. [Google Scholar]
- del Carmen Alvarez, M.; Donarski, J.A.; Elliott, M.; Charlton, A.J. Evaluation of extraction methods for use with NMR-based metabolomics in the marine polychaete ragworm, Hediste diversicolor. Metabolomics 2010, 6, 541–549. [Google Scholar] [CrossRef]
- Schock, T.B.; Stancyk, D.A.; Thobodeaux, L.; Burnett, K.G.; Burnett, L.E.; Boroujerdi, A.F.B.; Bearden, D.W. Metabolomic analysis of Atlantic blue crab, Callinectes sapidus, hemolymph following oxidative stress. Metabolomics 2010, 6, 250–262. [Google Scholar] [CrossRef]
- Soanes, K.H.; Achenbach, J.C.; Burton, I.W.; Hui, J.P.M.; Penny, S.L.; Karakach, T.K. Molecular characterization of zebrafish embryogenesis via DNA microarrays and multiplatform time course metabolomics studies. J. Proteome Res. 2011, 10, 5102–5117. [Google Scholar]
- Karakash, T.B.; Huenupi, E.C.; Soo, E.C.; Walter, J.A.; Afonso, L.O.B. 1HNMR and mass spectrometric characterization of the metabolic response of juvenile Atlantic salmon (Salmo salar) to long-term handling stress. Metabolomics 2009, 5, 123–137. [Google Scholar] [CrossRef]
- Zhang, X.; Xu, H; Zhang, H.; Guo, Y.; Dai, Z.; Chen, X. Simultaneous determination of albendazole and its metabolites in fish muscle tissue by stable isotope dilution ultra-performance liquid chromatography tandem mass spectrometry. Anal. Bioanal. Chem. 2011, 401, 727–734. [Google Scholar] [CrossRef]
- Kim, J.W.; Isobe, T.; Ramaswamy, B.R.; Chang, K.-H.; Amano, A.; Miller, T.M.; Siringan, F.P.; Tanabe, S. Contamination and bioaccumulation of benzotriazole ultraviolet stabilizers in fish from Manila Bay, the Philippines using an ultra-fast liquid chromatography-tandem mass spectrometry Original. Chemosphere 2011, 85, 751–758. [Google Scholar]
- Oh, S.F.; Vickery, T.W.; Serhan, C.N. Chiral lipidomics of E-series resolvins: Aspirin and the biosynthesis of novel mediators. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2011, 1811, 737–747. [Google Scholar] [CrossRef]
- Yan, X.; Xu, J.; Chen, J.; Chen, D.; Xu, S.; Luo, Q.; Wang, Y. Lipidomics focusing on serum polar lipids reveals species dependent stress resistance of fish under tropical storm. Metabolomics 2012, 8, 299–309. [Google Scholar]
- Weijs, L.; Das, K.; Siebert, U.; van Elk, N.; Jauniaux, T.; Neels, H.; Blust, R.; Covaci, A. Concentrations of chlorinated and brominated contaminants and their metabolites in serum of harbour seals and harbour porpoises. Environ. Int. 2009, 35, 842–850. [Google Scholar]
- Weijs, L.; Losada, S.; Das, K.; Roosens, L.; Reijnders, P.J.; Santos, J.F.; Neels, H.; Blust, R.; Covaci, A. Biomagnification of naturally-produced methoxylated polybrominated diphenyl ethers (MeO-PBDEs) in harbour seals and harbour porpoises from the southern North Sea. Environ. Int. 2009, 35, 893–899. [Google Scholar]
- Connor, K.M.; Gracey, A.Y. High-resolution analysis of metabolic cycles in the intertidal mussel Mytilus californianus. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2012, 302, R103–R111. [Google Scholar] [CrossRef]
- Spann, N.; Aldridge, D.C.; Griffin, J.L.; Jones, O.A. Size-dependent effects of low level cadmium and zinc exposure on the metabolome of the Asian clam, Corbicula fluminea. Aquat. Toxicol. 2011, 105, 589–599. [Google Scholar] [CrossRef]
- Beach, D.G.; Quilliam, M.A.; Hellou, J. Analysis of pyrene metabolites in marine snails by liquid chromatography using fluorescence and mass spectrometry detection. J. Chromatogr. B 2009, 877, 2142–2152. [Google Scholar]
- Ivanešivić, J.; Thomas, O.P.; Lejeusne, C.; Chevaldonné, P.; Pérez, T. Metabolic fingerprinting as an indicator of biodiversity: towards understanding inter-specific relationships among Homoscleromorpha sponges. Metabolomics 2011, 7, 289–304. [Google Scholar] [CrossRef]
- Simmons, T.L.; Coates, R.C.; Clark, B.R.; Engene, N.; Gonzalez, D.; Esquenazi, E.; Dorrestein, P.C.; Gerwick, W.H. Biosynthetic origin of natural products isolated from marine microorganism-invertebrate assemblages. Proc. Natl. Acad. Sci. USA 2008, 105, 4587–4594. [Google Scholar]
- Shrestha, B.; Vertes, A. In situ metabolic profiling of single cells by laser ablation electrospray ionization mass spectrometry. Anal. Chem. 2009, 81, 8265–8271. [Google Scholar] [CrossRef]
- Lakshmi, V.; Kumar, R. Metabolites from Sinularia species. Nat. Prod. Res. 2009, 23, 801–850. [Google Scholar] [CrossRef]
- Sarma, N.S.; Krishna, M.S.; Pasha, S.G.; Rao, T.S.P.; Venkateswarlu, Y.; Parameswaran, P.S. Marine metabolites: The sterols of soft coral. Chem. Rev. 2009, 109, 2803–2828. [Google Scholar]
- Ivanchina, N.V.; Kicha, A.A.; Stonik, V.A. Steroid glycosides from marine organisms. Steroids 2011, 76, 425–454. [Google Scholar]
- Li, Y.; Pattenden, G. Perspectives on the structural and biosynthetic interrelationships between oxygenated furanocembranoids and their polycyclic congeners found in corals. Nat. Prod. Rep. 2011, 28, 1269–1310. [Google Scholar]
- Li, C.; Hill, R.W.; Jones, D. Determination of betaine metabolites and dimethylsulfoniopropionate in coral tissues using liquid chromatography-time-of-flight mass spectrometry and stable isotope-labeled internal standards. J. Chromatogr. B 2010, 878, 1809–1816. [Google Scholar]
- Berrue, F.; Withers, S.T.; Haltli, B.; Withers, J.; Kerr, R.G. Chemical screening method for the rapid identification of microbial sources of marine invertebrate-associated metabolites. Mar. Drugs 2011, 9, 369–381. [Google Scholar]
- Cheng, S.-Y.; Chen, P.-W.; Chen, H.-P.; Wang, S.-K.; Duh, C.-Y. New cembranolides from the Dongsha atoll soft coral Lobophytum durum. Mar. Drugs 2011, 9, 1307–1318. [Google Scholar] [CrossRef]
- Kell, D.B.; Brown, M.; Davey, H.; Dunn, W.B.; Spasic, I.; Oliver, S.G. Metabolic footprinting and systems biology: the medium is the message. Nat. Rev. Microbiol. 2005, 3, 557–565. [Google Scholar]
- Hay, M. Marine Chemical ecology: Chemical signals and cues structure marine populations, communities, and ecosystems. Annu. Rev. Mar. Sci. 2009, 1, 193–212. [Google Scholar] [CrossRef]
- Barofsky, A.; Vidoudez, C.; Pohnert, G. Metabolic profiling reveals growth stage variability in diatom exudates. Limnol. Oceanogr. Methods 2009, 7, 382–390. [Google Scholar]
- Barofsky, A.; Simonelli, P.; Vidoudez, C.; Troedsson, C.; Nejstgaard, C.; Jakobsen, H.H.; Pohnert, G. Growth phase of the diatom Skeletonema marinoi influences the metabolic profile of the cells and the selective feeding of the copepod Calanus spp. J. Plankton Res. 2010, 32, 263–272. [Google Scholar] [CrossRef]
- Lane, A.L.; Kubanek, J. Secondary Metabolite Defenses Against Pathogens and Biofoulers. In Algal Chemical Ecology; Amsler, C.D., Ed.; Springer-Verlag: Berlin, Germany, 2008; pp. 229–243. [Google Scholar]
- Thomas, F.; Cosse, A.; Goulitquer, S.; Raimund, S.; Morin, P.; Valero, M.; Leblanc, C.; Potin, P. Waterborne signaling primes the expression of elicitor-induced genes and buffers the oxidative responses in the brown alga Laminaria digitata. PLoS One 2011, 6, e21475. [Google Scholar]
- Pohnert, G. Chemical noise in the silent ocean. J. Plankton Res. 2010, 32, 141–144. [Google Scholar]
- Prince, E.K.; Pohnert, G. Searching for signals in the noise: Metabolomics in chemical ecology. Anal. Bioanal. Chem. 2010, 396, 193–197. [Google Scholar]
- Keller, B.O.; Sui, J.; Young, A.B.; Whittal, R.M. Interferences and contaminants encountered in modern mass spectrometry. Anal. Chim. Acta 2008, 627, 71–81. [Google Scholar]
- Nguyen, K.T.T.; Scapolla, C.; Di Carro, M.; Magi, E. Rapid and selective determination of UV filters in seawater by liquid chromatography-tandem mass spectrometry combined with stir bar sorptive extraction. Talanta 2011, 85, 2375–2384. [Google Scholar]
- Takaichi, S. Carotenoids in algae: Distributions, biosyntheses and functions. Mar. Drugs 2011, 9, 1101–1118. [Google Scholar]
- Buskirk, A.D.; Hettick, J.M.; Chipinda, I.; Law, B.F.; Siegel, P.D.; Slaven, J.E.; Green, B.J.; Beezhold, D.H. Fungal pigments inhibit the matrix-assisted laser desorption/ionization time-of-flight mass spectrometry analysis of darkly pigmented fungi. Anal. Biochem. 2011, 411, 122–128. [Google Scholar]
- Colombini, M.P.; Modugno, F.; Giacomelli, A. Two procedures for suppressing interference from inorganic pigments in the analysis by gas chromatography-mass spectrometry of proteinaceous binders in paintings. J. Chromatogr. A 1999, 846, 101–111. [Google Scholar]
- Onofrejová, L.; Vasickova, J.; Klejdus, B.; Stratil, P.; Misurcova, L.; Kracmar, S.; Kopecky, J.; Vacek, J. Bioactive phenols in algae: The application of pressurized-liquid and solid-phase extraction techniques. J. Pharm. Biomed. Anal. 2010, 51, 464–470. [Google Scholar]
- Békri, K.; Saint-Louis, R.; Pelletier, E. Determination of tributyltin and 4-hydroxybutyldibutyltin chlorides in seawater by liquid chromatography with atmospheric pressure chemical ionization-mass spectrometry. Anal. Chim. Acta 2006, 578, 203–212. [Google Scholar]
- Wu, J.; Qian, X.; Yang, Z.; Zhang, L. Study on the matrix effect in the determination of selected pharmaceutical residues in seawater by solid-phase extraction and ultra-high-performance liquid chromatography-electrospray ionization low-energy collision-induced dissociation tandem mass spectrometry. J. Chromatogr. A 2010, 1217, 1471–1475. [Google Scholar] [CrossRef]
- Chou, C.-C.; Lee, M.-R. Determination of organotin compounds in water by headspace solid phase microextraction with gas chromatography-mass spectrometry. J. Chromatogr. A 2005, 1064, 1–8. [Google Scholar] [CrossRef]
- Smith, C.A.; Want, E.J.; O’Maille, G.; Abagyan, R.; Siuzdak, G. XCMS: Processing mass spectrometry data for metabolite profiling using nonlinear peak alignment, matching, and identification. Anal. Chem. 2006, 78, 779–787. [Google Scholar]
- Lommen, A. MetAlign: An interface-driven, versatile metabolomics tool for hyphenated full-scan MS data pre-processing. Anal. Chem. 2009, 81, 3079–3086. [Google Scholar]
- Pluskal, T.; Castillo, S.; Villar-Briones, A.; Orešič, M. MZmine 2: Modular framework for processing, visualizing, and analyzing mass spectrometry-based molecular profile data. BMC Bioinformatics 2010, 11. [Google Scholar]
- Xia, J.; Psychogios, N.; Young, N.; Wishart, D.S. MetaboAnalyst: A web server for metabolomic data analysis and interpretation. Nucleic Acids Res. 2009, 37, W652–W660. [Google Scholar]
- Kastenmüller, G.; Römisch-Margl, W.; Wägele, B.; Altmaier, E.; Suhre, K. MetaP-server: A web-based metabolomics data analysis tool. J. Biomed. Biotechnol. 2011. [Google Scholar]
- Xia, J.; Sinelnikov, I.V.; Wishart, D.S. MetATT: A web-based metabolomics tool for analyzing time-series and two-factor datasets. Bioinformatics 2011, 27, 2455–2456. [Google Scholar]
- Carroll, A.J.; Badger, M.R.; Millar, A.H. The MetabolomeExpress Project: Enabling web-based processing, analysis and transparent dissemination of GC/MS metabolomics datasets. BMC Bioinformatics 2010, 11. [Google Scholar]
- Stein, S.E. An integrated method for spectrum extraction and compound identification from gas chromatography/mass spectrometry data. J. Am. Soc. Mass Spectrom. 1999, 10, 770–781. [Google Scholar]
- Styczynski, M.P.; Moxley, J.F.; Tong, L.V.; Walther, J.L.; Jensen, K.L.; Stephanopoulos, G.N. Systematic identification of conserved metabolites in GC/MS data for metabolomics and biomarker discovery. Anal. Chem. 2007, 79, 966–973. [Google Scholar]
- Cock, J.M.; Coelho, S.M. Algal models in plant biology. J. Exp. Bot. 2011, 62, 2425–2430. [Google Scholar]
- West, J.; Kraft, G. Ectocarpus siliculosus (Dillwyn) Lyngb. from Hopkins River Falls, Victoria—The first record of a freshwater brown alga in Australia. Muelleria 1996, 9, 29–33. [Google Scholar]
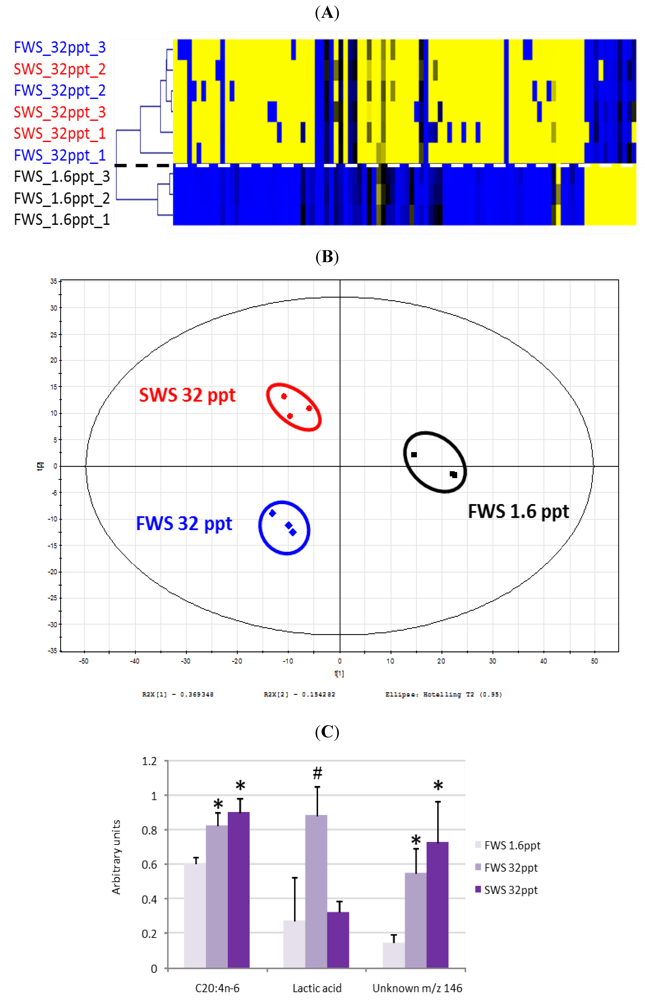
- Dittami, S.M.; Gravot, A.; Goulitquer, S.; Rousvoal, S.; Peters, A.F.; Bouchereau, A.; Boyen, C.; Tonon, T. Towards deciphering dynamic changes and evolutionary mechanisms involved in the adaptation to low salinities in Ectocarpus (brown algae). Plant J. 2012. [Google Scholar]
- Wishart, D.S. Advances in metabolite identification. Bioanalysis 2011, 3, 1769–1782. [Google Scholar]
- Fabris, M.; Matthijs, M.; Rombauts, S.; Vyverman, V.; Goossens, A.; Baart, G.J.E. The metabolic blueprint of Phaeodactylum tricornutum reveals a eukaryotic Entner-Doudoroff glycolytic pathway. Plant J. 2012. [Google Scholar]
- Caspi, R.; Altman, T.; Dale, J.M.; Dreher, K.; Fulcher, C.A.; Gilham, F.; Kaipa, P.; Karthikeyan, A.S.; Kothari, A.; Krummenacker, M.; et al. The MetaCyc database of metabolic pathways and enzymes and the BioCyc collection of pathway/genome databases. Nucleic Acids Res. 2010, 38, D473–D479. [Google Scholar]
- Fahy, E.; Sud, M.; Cotter, D.; Subramaniam, S. LIPID MAPS online tools for lipid research. Nucleic Acids Res. 2007, 35, W606–W612. [Google Scholar]
- Hashimoto, K.; Goto, S.; Kanehisa, M. KEGG GLYCAN and glycome informatics. Tanpakushitsu Kakusan Koso 2008, 53, 1698–1702. [Google Scholar]
- Knox, C.; Law, V.; Jewison, T.; Liu, P.; Ly, S.; Frolkis, A.; Pon, A.; Banco, K.; Mak, C.; Neveu, V.; et al. DrugBank 3.0: A comprehensive resource for “omics” research on drugs. Nucleic Acids Res. 2011, 39, D1035–D1041. [Google Scholar]
- Degtyarenko, K.; de Matos, P.; Ennis, M.; Hastings, J.; Zbinden, M.; Mc-Naught, A.; Alcántara, R.; Darsow, M.; Guedj, M.; Ashburner, M. ChEBI: A database and ontology for chemical entities of biological interest. Nucleic Acids Res. 2008, 36, D344–D350. [Google Scholar]
- Little, J.L.; Williams, A.J.; Pshenichnov, A.; Tkachenko, V. Identification of “known unknowns” utilizing accurate mass data and ChemSpider. J. Am. Soc. Mass Spectrom. 2012, 23, 179–185. [Google Scholar]
- Cui, Q.; Lewis, I.A.; Hegeman, A.D.; Anderson, M.E.; Li, J.; Schulte, C.F.; Westler, W.M.; Eghbalnia, H.R.; Sussman, M.R.; Markley, J.L. Metabolite identification via the madison metabolomics consortium database. Nat. Biotechnol. 2008, 26, 162–164. [Google Scholar]
- Wang, Y.; Xiao, J.; Suzek, T.O.; Zhang, J.; Wang, J.; Bryant, S.H. PubChem: A public information system for analyzing bioactivities of small molecules. Nucleic Acids Res. 2009, 37, W623–W633. [Google Scholar]
- Wishart, D.S.; Knox, C.; Guo, A.C.; Eisner, R.; Young, N.; Gautam, B.; Hau, D.D.; Psychogios, N.; Dong, E.; Bouatra, S.; et al. HMDB: A knowledgebase for the human metabolome. Nucleic Acids Res. 2009, 37, D603–D610. [Google Scholar]
- Matthews, L.; Gopinath, G.; Gillespie, M.; Caudy, M.; Croft, D.; de Bono, B.; Garapati, P.; Hemish, J.; Hermjakob, H.; Jassal, B.; et al. Reactome knowledgebase of human biological pathways and processes. Nucleic Acids Res. 2009, 37, D619–D622. [Google Scholar]
- Shinbo, Y.; Nakamura, Y.; Altaf-Ul-Amin, M.; Asahi, H.; Kurokawa, K.; Arita, M.; Saito, K.; Ohta, D.; Shibata, D.; Kanaya, S. KNApSAcK: A Comprehensive Species-Metabolite Relationship Database. In Biotechnology in Agriculture and Forestry; Nagata, T., Lörz, H., Widholm, J.M., Eds.; Springer-Verlag: Berlin Heidelberg, Germany, 2006; Volume 57, pp. 165–181. [Google Scholar]
- Horai, H.; Arita, M.; Kanaya, S.; Nihei, Y.; Ikeda, T.; Suwa, K.; Ojima, Y.; Tanaka, K.; Tanaka, S.; Aoshima, K.; et al. MassBank: A public repository for sharing mass spectral data for life sciences. J. Mass Spectrom. 2010, 45, 703–714. [Google Scholar] [CrossRef]
- Kind, T.; Wohlgemuth, G.; Lee, D.Y.; Lu, Y.; Palazoglu, M.; Shahbaz, S.; Fiehn, O. FiehnLib: Mass spectral and retention index libraries for metabolomics based on quadrupole and time-of-flight gas chromatography/mass spectrometry. Anal. Chem. 2009, 81, 10038–10048. [Google Scholar]
- Kopka, J.; Schauer, N.; Krueger, S.; Birkemeyer, C.; Usadel, B.; Bergmuller, E.; Dormann, P.; Weckwerth, W.; Gibon, Y.; Stitt, M.; et al. [email protected]: The golm metabolome database. Bioinformatics 2005, 21, 1635–1638. [Google Scholar]
- Stein, S.E. Chemical substructure identification by mass spectral library searching. J. Am. Soc. Mass Spectrom. 1995, 6, 644–655. [Google Scholar]
- Fernie, A.R.; Aharoni, A.; Willmitzer, L.; Stitt, M.; Tohge, T.; Kopka, J.; Carroll, A.J.; Saito, K.; Fraser, P.D.; DeLuca, V. Recommendations for reporting metabolite data. Plant Cell 2011, 23, 2477–2482. [Google Scholar]
- Davis, G.D.J.; Vasanthi, A.H.R. Seaweed metabolite database (SWMD): A database of natural compounds from marine algae. Bioinformation 2011, 5, 361–364. [Google Scholar]
- MarinLit Database, version vpc 12.5, University of Canterbury: Christchurch, New Zealand, 2008.
- Nosengo, N. Marine biology network launches into choppy waters. Nature 2011, 470, 444–445. [Google Scholar]
- La Barre, S.L.; Weinberger, F.; Kervarec, N.; Potin, P. Monitoring defensive responses in macroalgae-limitations and perspectives. Phytochem. Rev. 2004, 3, 371–379. [Google Scholar]
- Billoud, B.; Le Bail, A.; Charrier, B. A stochastic 1D nearest-neighbour automaton models early development of the brown alga Ectocarpus siliculosus. Funct. Plant Biol. 2008, 35, 1014–1024. [Google Scholar] [CrossRef]
- Le Bail, A.; Billoud, B.; LePanse, S.; Chevinesse, S.; Charrier, B. ETOILE regulates developmental patterning in the filamentous brown alga Ectocarpus siliculosus. Plant Cell 2011, 23, 1666–1678. [Google Scholar] [CrossRef]
- Ben-Tabou de-Leon, S.; Davidson, E.H. Experimentally based sea urchin gene regulatory network and the causal explanation of developmental phenomenology. Wiley Interdiscip. Rev. Syst. Biol. Med. 2009, 1, 237–246. [Google Scholar]
- Bellé, R.; Prigent, S.; Siegel, A.; Cormier, P. Model of cap-dependent translation initiation in sea urchin: A step towards the eukaryotic translation regulation network. Mol. Reprod. Dev. 2010, 77, 257–264. [Google Scholar]
- Zhang, Y.; Thiele, I.; Weekes, D.; Li, Z.; Jaroszewski, L.; Ginalski, K.; Deacon, A.M.; Wooley, J.; Lesley, S.A.; Wilson, I.A.; et al. Three-dimensional structural view of the central metabolic network of Thermotoga maritima. Science 2009, 325, 1544–1549. [Google Scholar]
- Kim, H.U.; Kim, S.Y.; Jeong, H.; Kim, T.Y.; Kim, J.J.; Choy, H.E.; Yi, K.Y.; Rhee, J.H.; Lee, S.Y. Integrative genome-scale metabolic analysis of Vibrio vulnificus for drug targeting and discovery. Mol. Syst. Biol. 2011, 7, 460. [Google Scholar]
- Li, S.Z.; Pozhitkov, A.; Ryan, R.A.; Manning, C.S.; Brown-Peterson, N.; Brouwer, M. Constructing a fish metabolic network model. Genome Biol. 2010, 11. [Google Scholar]
- Sorokina, O.; Corellou, F.; Dauvillée, D.; Sorokin, A.; Goryanin, I.; Ball, S.; Bouget, F.-Y.; Millar, A.J. Microarray data can predict diurnal changes of starch content in the picoalga Ostreococcus. BMC Syst. Biol. 2011, 5. [Google Scholar]
- Sauer, U. Metabolic networks in motion: 13C-based flux analysis. Mol. Syst. Biol. 2006, 2. [Google Scholar]
- Tang, Y.J.; Martin, H.G.; Myers, S.; Rodriguez, S.; Baidoo, E.E.; Keasling, J.D. Advances in analysis of microbial metabolic fluxes via. (13)C isotopic labeling. Mass Spectrom. Rev. 2009, 28, 362–375. [Google Scholar] [CrossRef]
- Chikayama, E.; Suto, M.; Nishihara, T.; Shinozaki, K.; Kikuchi, J. Systematic NMR analysis of stable isotope labeled metabolite mixtures in plant and animal systems: Coarse grained views of metabolic pathways. PLoS One 2008, 3, e3805. [Google Scholar]
- Saito, K.; Matsuda, F. Metabolomics for functional genomics, systems biology, and biotechnology. Annu. Rev. Plant Biol. 2010, 61, 463–489. [Google Scholar] [CrossRef]
- Tcherkez, G.; Mahé, A.; Hodges, M. 12C/13C fractionations in plant primary metabolism. Trends Plant Sci. 2011, 16, 499–506. [Google Scholar]
- Bondu, S.; Cerantola, S.; Kervarec, N.; Deslandes, E. Impact of the salt stress on the photosynthetic carbon flux and 13C-label distribution within floridoside and digeneaside in Solieria chordalis. Phytochemistry 2009, 70, 173–184. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Goulitquer, S.; Potin, P.; Tonon, T. Mass Spectrometry-Based Metabolomics to Elucidate Functions in Marine Organisms and Ecosystems. Mar. Drugs 2012, 10, 849-880. https://doi.org/10.3390/md10040849
Goulitquer S, Potin P, Tonon T. Mass Spectrometry-Based Metabolomics to Elucidate Functions in Marine Organisms and Ecosystems. Marine Drugs. 2012; 10(4):849-880. https://doi.org/10.3390/md10040849
Chicago/Turabian StyleGoulitquer, Sophie, Philippe Potin, and Thierry Tonon. 2012. "Mass Spectrometry-Based Metabolomics to Elucidate Functions in Marine Organisms and Ecosystems" Marine Drugs 10, no. 4: 849-880. https://doi.org/10.3390/md10040849




