1. Introduction
Melanocytes are the major cell population in the skin epidermis which is responsible for melanin production and pigmentation of skin and hair. Melanin is synthesized in melanocytes upon ultraviolet irradiation and then transported to surrounding epidermal keratinocytes for absorption of energy and protection from sunburn. However, abnormal melanogenesis causes pigmentary disorders, such as hyperpigmentation (melasma and lentigines) or hypopigmentation (vitiligo and albinism), which are of clinical and cosmetic concerns. Melanogenic agents are commonly employed to modulate melanogenesis. Many widely used lightening compounds such as kojic acid or arbutin are isolated from natural sources or botanical extracts [
1]; few were of marine origin [
2], despite its tremendous biological and structural diversity [
3].
Melanogenesis is the process of pigment formation initiated by the rate-limiting enzyme tyrosinase that converts L-DOPA to dopaquinone before further oxidation to melanin. Activation of tyrosinase is associated with phosphorylation by protein kinase C-beta (PKC-β) and formation of a complex between phosphorylated tyrosinase and tyrosinase-related protein 1 (TYRP1) [
4]. In melanocytes, converging external stimuli stimulates adenylyl cyclase to raise cAMP that subsequently activates microphthalmia-associated transcription factor (MITF), a transcription factor for all melanogenic proteins, including tyrosinase (TYR), TYRP1, tyrosinase-related protein 2 (TYRP2), and PKC-β. MITF is thus a key factor linking both PKC-β- and cAMP-dependent pathways in the regulation of melanogenesis [
5]. Reduction of MITF binding to the promoters of these melanogenic proteins results in a decrease of cell pigmentation. Furthermore, MITF is also the master regulator for DNA replication, proliferation and genomic stability of melanoma cells and its reduction leads to DNA damage and defective cell replication [
6]. The precise function(s) of TYRP2 in melanogenesis, a DOPA-chrome tautomerase, remains largely unknown although it decreases cell sensitivity to oxidative stress and also modulates melanoma cell sensitivity to chemotherapy [
4,
7].
Reactive oxygen species (ROS) are not only by-products of normal cell metabolism but also act as signaling molecules in cell functions. An excessive ROS production causes damage of DNA, proteins and lipids, ultimately resulting in apoptosis and cell death. In melanocytes, intracellular ROS at sublethal concentrations induce melanogenesis [
8] through activation of apurinic/apyrimidinic endonuclease (APE) for oxidative DNA damage repair [
9,
10] and promotion of cell survival via MITF pathway [
9]. Interestingly, kojic acid, a widely used skin whitening agent, possesses both anti-tyrosinase and ROS scavenging activities [
11].
Geoditin A, an isomalabaricane triterpenoid isolated from many genera of marine sponges, has received special pharmaceutical attention because it inhibited cyclin-dependent kinase activity and subsequently suppressed tumor cell proliferation [
12,
13]. Our previous studies have also demonstrated geoditin A being a potent inducer of oxidative stress and apoptosis in HT29 cells [
14] and HL60 cells [
15]. It is the objective in this study to investigate if this oxidative stress might interfere with tyrosinase activity and melanogenesis in murine B16 melanoma cells.
2. Results and Discussion
Geoditin A (
Figure 1) is an isomalabaricane triterpene isolated from a South China Sea sponge,
Geodia japonica [
16]. This rare class of triterpenoids possesses potent tumor inhibitory activities [
17,
18], and thus received special attention for its potential for chemotherapeutic development [
19].
Figure 1.
Chemical structure of geoditin A.
Figure 1.
Chemical structure of geoditin A.
The values of IC
50 for cancer cell lines ranging from 0.1–20 µg/mL which were 2–3 folds higher than that for human fibroblasts (IC
50 = 60 μg/mL) [
14]. Our previous study has also demonstrated a potent cytotoxicity of geoditin A against human leukemia HL60 cells (IC
50 = 3 µg/mL) [
15], but mild to human colon carcinoma HT29 cells (IC
50 = 20 μg/mL) [
14]. The IC
50 for murine melanoma B16 cells in this study also falls in this range, with the value of ≈10 μg/mL (
Figure 2a). In addition to its cytotoxicity, geoditin A also interfered with L-DOPA conversion activity, a parameter indicative of melanogenesis, which decreased dramatically from 80% at 0.625 µg/mL to 25% at 5 µg/mL, with an EC
50 value of about 1 μg/mL. EC
50 value is defined as the effective concentration of geoditin A required to inhibit half maximal inhibition of L-DOPA conversion, and the ratio of L-DOPA conversion to cell viability is defined as melanogenic activity (
Figure 3a). Since an effective activity was achieved between 1.25 µg/mL to 5 µg/mL when most of the tested cells (≥80%) remained viable, the following study was therefore focused on this range.
Figure 2.
(a) Viability and melanogenic activity of geoditin A on murine melanoma B16 cells. Cells were incubated in 96-well plates with serial concentrations of geoditin A for 48 h, and their viability was assessed by sulforhodamine B (SRB) colorimetric assay. Another set of cells were lysed with 20 mM Tris base (pH 7) and reacted with L-DOPA for cellular tyrosinase activity (i.e., conversion of L-DOPA into dopaquinone) assay. Results were presented as mean and standard deviation of triplicates. (b) Melanin in cell lysates was pelleted and extracted with 1 M NaOH before the optical density absorbance of melanin solutions was determined at 490 nm. The melanin contents normalized to cellular protein content decrease in a dose-dependent manner. Results were presented as mean and standard deviation of triplicates.
Figure 2.
(a) Viability and melanogenic activity of geoditin A on murine melanoma B16 cells. Cells were incubated in 96-well plates with serial concentrations of geoditin A for 48 h, and their viability was assessed by sulforhodamine B (SRB) colorimetric assay. Another set of cells were lysed with 20 mM Tris base (pH 7) and reacted with L-DOPA for cellular tyrosinase activity (i.e., conversion of L-DOPA into dopaquinone) assay. Results were presented as mean and standard deviation of triplicates. (b) Melanin in cell lysates was pelleted and extracted with 1 M NaOH before the optical density absorbance of melanin solutions was determined at 490 nm. The melanin contents normalized to cellular protein content decrease in a dose-dependent manner. Results were presented as mean and standard deviation of triplicates.
Instead of using MTT cytotoxicity assay for HL60 cells and HT29 cells in previous studies, sulforhodamine B (SRB) colorimetric viability assay [
20] was used in this study because the absorbance wavelength of the melanin might interfere with that of methyl tetrazolium for MTT assay. In consistent with the prominent decrease of L-DOPA conversion activity in B16 cells after geoditin A treatment, levels of intracellular melanin content, the end product of this melanogenesis, also reduced in a dose-dependent manner (
Figure 2b).
Since geoditin A is an oxidative stressor and ROS is closely associated with melanogenesis, it is tempting to speculate that oxidative stress may interfere with pigmentation in B16 cells. This inhibitory effect can partially be attenuated by pre-treatment with 10 µM
N-acetylcysteine (NAC) (
Figure 3a), an oxidative scavenger also effectively reducing apoptosis in HT29 cells [
14]. Interestingly, no noticeable ROS production was measured until B16 cells were exposed to higher doses of geoditin A (≥10 µg/mL) thereby ROS production increased by about 3.5 folds (
Figure 3b), which suggested that ROS might not contribute to anti-melanogenic activity of geoditin A at doses ≤5 µg/mL. On the other hands, anti-melanogenesis was aggravated by SQ22536, an adenylate cyclase inhibitor (
Figure 3c); implicating that geoditin A-mediated melanogenesis is regulated through modulation of cAMP pathway. When excessive ROS production overwhelms cell tolerance at doses of geoditin A ≥10 μg/mL, cells committed to cell death (
Figure 2a), probably from apoptosis induced by oxidative stress as happened to HT29 cells and HL60 cells [
14,
15].
Figure 3.
(a) Melanogenic activity (=ratio of L-DOPA conversion to viable cells) by geoditin A on B16 cells. Cells incubated in 96-well plates with geoditin A, ranging from 0.625 to 5 μg/mL, for 48 h was only slightly attenuated by pre-treatment with 10 µM N-acetylcysteine (oxidant scavenger). (b) Flow cytometric analysis revealed a low level of ROS production in cells treated with geoditin A at doses ≤5 µg/mL until an about 3.5-fold increase was measured at dose of 10 µg/mL whose production was greatly attenuated in the presence of 10 µM of NAC. Results were presented as mean and standard deviation of triplicates. (c) Inhibition of intracellular cyclic adenosine monophosphate (cAMP) by pre-incubation for 30 min with specific inhibitor SQ22536 (100 µM) further reduced melanogenic activity, indicating cAMP pathway is involved in geoditin-A mediated melanogenesis.
Figure 3.
(a) Melanogenic activity (=ratio of L-DOPA conversion to viable cells) by geoditin A on B16 cells. Cells incubated in 96-well plates with geoditin A, ranging from 0.625 to 5 μg/mL, for 48 h was only slightly attenuated by pre-treatment with 10 µM N-acetylcysteine (oxidant scavenger). (b) Flow cytometric analysis revealed a low level of ROS production in cells treated with geoditin A at doses ≤5 µg/mL until an about 3.5-fold increase was measured at dose of 10 µg/mL whose production was greatly attenuated in the presence of 10 µM of NAC. Results were presented as mean and standard deviation of triplicates. (c) Inhibition of intracellular cyclic adenosine monophosphate (cAMP) by pre-incubation for 30 min with specific inhibitor SQ22536 (100 µM) further reduced melanogenic activity, indicating cAMP pathway is involved in geoditin-A mediated melanogenesis.
Double-label immunofluorescence microscopy has demonstrated a dose-dependent decrease of tyrosinase (
Figure 4a–d) and cytosolic melanin (
Figure 4e–h) in B16 cells treated with geoditin A, particularly at dose of 5 μg/mL (
Figure 4d,h). No co-localization of tyrosinase with golgin-97, marker protein for the Golgi, indicate retention of tyrosinase in this organelle is unlikely (
Figure 4b–d).
Figure 4.
Dual immunofluorescence localization of tyrosinase with the Golgi marker in B16 cells. Cells were treated with serial concentrations of geoditin A for 48 h, fixed in 3% paraformaldehyde and incubated with an antibody against golgin-97 (a–d), the marker protein for the Golgi apparatus. Immunofluorescence was visualized by secondary antibody IgG conjugated to Alexa-488 (green) and Alexa-594 (red). Nuclei were counterstained with DAPI (blue). Typical Golgi stack (golgin-97, red) is shown at the juxtanuclear region while tyrosinase (green) in the cytoplasm of the untreated B16 cells (a) which was decreased in a dose-dependent manner (b, c, and d for 0.6, 1.25 and 5 μg/mL, respectively). Melanin granules also decreased in a dose-dependent manner after geoditin A treatment (e–h) under the phase contrast microscope (scale bar = 100 μm). No apoptotic bodies were found in DAPI-stained cells of all treatment groups (0.6–5 μg/mL)(b–d).
Figure 4.
Dual immunofluorescence localization of tyrosinase with the Golgi marker in B16 cells. Cells were treated with serial concentrations of geoditin A for 48 h, fixed in 3% paraformaldehyde and incubated with an antibody against golgin-97 (a–d), the marker protein for the Golgi apparatus. Immunofluorescence was visualized by secondary antibody IgG conjugated to Alexa-488 (green) and Alexa-594 (red). Nuclei were counterstained with DAPI (blue). Typical Golgi stack (golgin-97, red) is shown at the juxtanuclear region while tyrosinase (green) in the cytoplasm of the untreated B16 cells (a) which was decreased in a dose-dependent manner (b, c, and d for 0.6, 1.25 and 5 μg/mL, respectively). Melanin granules also decreased in a dose-dependent manner after geoditin A treatment (e–h) under the phase contrast microscope (scale bar = 100 μm). No apoptotic bodies were found in DAPI-stained cells of all treatment groups (0.6–5 μg/mL)(b–d).
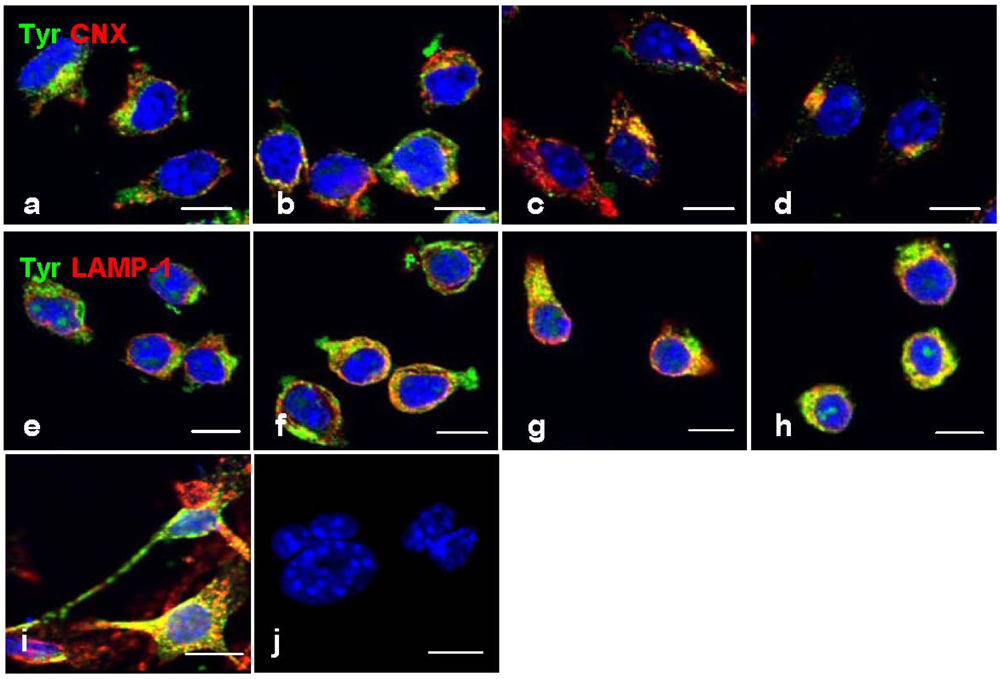
However, a strong immunofluorescence tyrosinase was co-localized with CNX (
Figure 5b–d) and LAMP-1 (
Figure 5f–h), not only indicating its retention in the ER and lysosomes, but also a possibility of improper trafficking of this enzyme to the lysosome for degradation. This phenomenon was also found in mouse melanocyte melan-a cells treated with progesterone (
Figure 5i) [
21], or B16 cells treated with inulavosin, a flavanol isolated from Inula nervosa [
22].
The initial catalytic reaction of melanogenesis (a physiological process of melanin biosynthesis) is regulated by the rate-limiting enzyme, tyrosinase, and its related proteins, TYRP1 and TYRP2 [
23]. Tyrosinase is activated when it is glycosylated, phosophorylated and forming a complex with TYRP1 in organelles along the melanogenic pathway, namely endoplasmic reticulum, the Golgi and the melanosome [
4,
24]. However, tyrosinase has common targeting motifs for melanosomes and lysosomes, making it possible to be mistargeted to lysosomes for degradation [
25]. Whether this is a result of trafficking defect or improper maturation of tyrosinase in geoditin A-treated cells remains unclear in this study, and further study is required to elucidate its underlying mechanism.
Figure 5.
Co-localization immunofluorescence of tyrosinase in ER and lysosomes. Cells were treated with geoditin A [untreated (a), 1.25 μg/mL (b), 2.5 μg/mL (c), 5 μg/mL (d) for 48 h], fixed with 3% paraformaldehyde and immunostained with antibodies against tyrosinase (green), CNX protein (red) in the ER (a–d) and LAMP-1 (red) in the lysosome (e–h). No retention of tyrosinase in the ER and lysosomes as depicted by two separated immunostains in cells without geoditin A treatment (a,e), but strong co-localization (yellow) was observed in cells with geoditin A treatment (b–d and f–h for CNX and LAMP-1 with tyrosinase in the ER and lysosomes, respectively). A co-localized tyrosinase and LAMP-1 was also shown in melan-a cells treated with progesterone for 4 days as a positive control (i).Apoptotic nuclei were observed in DAPI-stained cells treated with higher dose of geoditin A (10 μg/mL) for 48 h (scale bar = 100 μm).
Figure 5.
Co-localization immunofluorescence of tyrosinase in ER and lysosomes. Cells were treated with geoditin A [untreated (a), 1.25 μg/mL (b), 2.5 μg/mL (c), 5 μg/mL (d) for 48 h], fixed with 3% paraformaldehyde and immunostained with antibodies against tyrosinase (green), CNX protein (red) in the ER (a–d) and LAMP-1 (red) in the lysosome (e–h). No retention of tyrosinase in the ER and lysosomes as depicted by two separated immunostains in cells without geoditin A treatment (a,e), but strong co-localization (yellow) was observed in cells with geoditin A treatment (b–d and f–h for CNX and LAMP-1 with tyrosinase in the ER and lysosomes, respectively). A co-localized tyrosinase and LAMP-1 was also shown in melan-a cells treated with progesterone for 4 days as a positive control (i).Apoptotic nuclei were observed in DAPI-stained cells treated with higher dose of geoditin A (10 μg/mL) for 48 h (scale bar = 100 μm).
Members of melanogenic genes, including tyrosinase, TYRP1, and PKC-β, are regulated at their transcriptional levels by microphthalmia-associated transcription factor (MITF) [
5]. In addition to pigmentation, MITF also promotes pigment cell growth and differentiation [
26]. MITF expression is activated by agents that elevate the intracellular level of cAMP [
5] or apurinic/apyrimidinic endonuclease (APE-1/Ref-1) in response to ROS [
10], and thus strong oxidative stress and cAMP inhibitors impinging on MITF become targets for depigmentation. Resveratrol is a good example for post-translational regulation through cAMP pathway [
25].
The decrease of immunofluorescence tyrosinase in B16 cells (
Figure 4 and
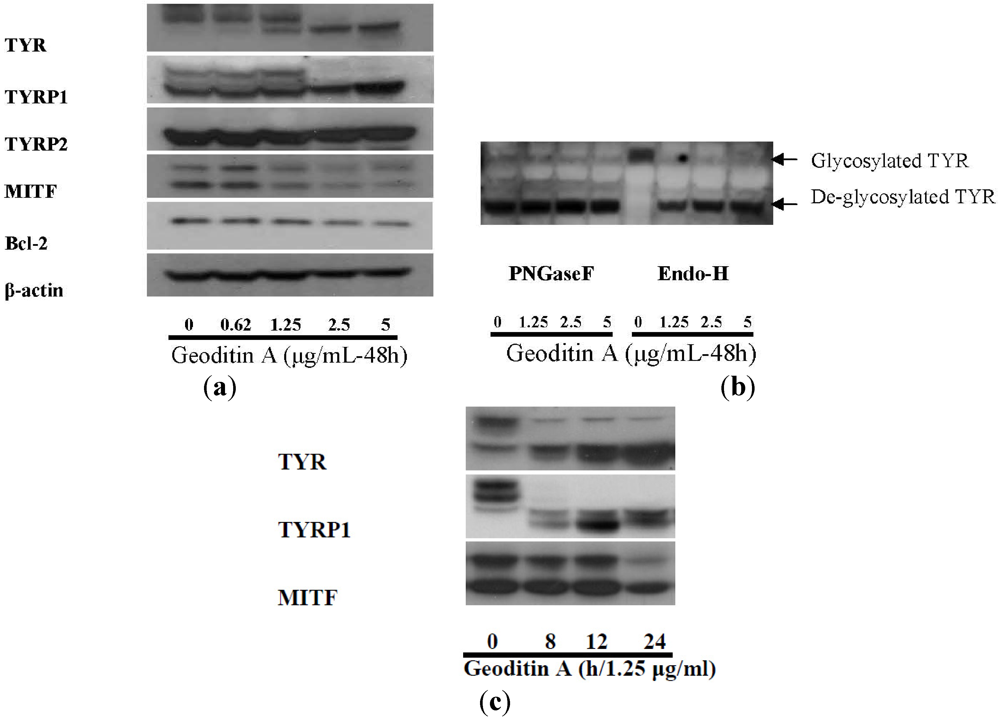
Figure 5) after geoditin A treatment is further confirmed by immunoblotting for melanogenic proteins (
Figure 6). One major band for tyrosinase (80 kD) was revealed in untreated B16 cells while a second band for protein of smaller size (about 70 kD) increased in a dose-dependent manner in geoditin A-treated cells (
Figure 6a). These bands represent different degrees of maturation of melanogenic proteins (tyrosinase and tyrosinase-related proteins) in the ER [
27]. Nascent tyrosinase is glycosylated and properly folded in the ER and completely matured in the Golgi before it is secreted to melanosomes for melanogenesis. Immature tyrosinase retains in the ER and bound to chaperones, such as calnexin (CNX) and calreticulin. Thus, our immunofluorescence analysis shows a reduced amount of tyrosinase in the Golgi and cytoplasm but it bound significantly with CNX, implicating retention of immature tyrosinase in the ER (
Figure 5b–d). Similar immunoblotting pattern was also found for TYRP1 protein. This decrease was not attributed to cell death because there were still about 80% viable cells at 5 μg/mL when the L-DOPA conversion activity had already reduced to only 30% of the untreated control (
Figure 2a).
Figure 6.
(a) Expression of melanogenic genes in geoditin A-treated B16 cells. Protein lysates of B16 cells treated with geoditin A, ranging from 0 to 5 μg/mL, for 48 h were subject to western blotting for examination of levels of melanogenic proteins (TYR, TYRP1, TYRP2 and MITF). Images represent results from at least three separated experiments. Equal amount of proteins analyzed were normalized with β-actin. (b) Glycosylation study of tyrosinase with EndoH and PGNase F digestion shows matured tyrosinase was present in untreated B16 cells while aberrant maturation of tyrosinase increased in a dose-dependent manner after treatment with geoditin A (1.25, 2.5 and 5 μg/mL) for 48 h. (c) Western blotting for kinetic analysis of melanogenic protein expression in B16 cells treated with 1.25 μg/mL geoditin A for 8–24 h. Two bands for tyrosinase and three bands for TYRP1 were present in B16 cells, proteins of larger size (~80 kD) represents mature tyrosinase/TYRP1 while bands of smaller size represent immature glycoforms of these two proteins. Immature TYR and TYRP1 were prominent after treatment of 1.25 g/mL geoditin A for 8 h, while decrease of MITF expression remained at untreated levels until 24 h, which is far behind the change of both TYR and TYRP1.
Figure 6.
(a) Expression of melanogenic genes in geoditin A-treated B16 cells. Protein lysates of B16 cells treated with geoditin A, ranging from 0 to 5 μg/mL, for 48 h were subject to western blotting for examination of levels of melanogenic proteins (TYR, TYRP1, TYRP2 and MITF). Images represent results from at least three separated experiments. Equal amount of proteins analyzed were normalized with β-actin. (b) Glycosylation study of tyrosinase with EndoH and PGNase F digestion shows matured tyrosinase was present in untreated B16 cells while aberrant maturation of tyrosinase increased in a dose-dependent manner after treatment with geoditin A (1.25, 2.5 and 5 μg/mL) for 48 h. (c) Western blotting for kinetic analysis of melanogenic protein expression in B16 cells treated with 1.25 μg/mL geoditin A for 8–24 h. Two bands for tyrosinase and three bands for TYRP1 were present in B16 cells, proteins of larger size (~80 kD) represents mature tyrosinase/TYRP1 while bands of smaller size represent immature glycoforms of these two proteins. Immature TYR and TYRP1 were prominent after treatment of 1.25 g/mL geoditin A for 8 h, while decrease of MITF expression remained at untreated levels until 24 h, which is far behind the change of both TYR and TYRP1.
![Marinedrugs 10 00465 g006]()
The
N-glycan processing of tyrosinase in the ER is noteworthy because the immature form increased with the expense of its mature forms but the total amount of these proteins did not change significantly (
Figure 6). The oligosaccharide added to tyrosinase in the ER can be cleaved by endoglycosidase H (EndoH), while those added in both ER and the Golgi can be cleaved by peptide
N-glycosidase F (PNGase F) [
25]. The analysis of tyrosinase pattern by these two endoglycosidases demonstrates a strong band with higher molecular weight for mature tyrosinase but an absence of immature tyrosinase only in untreated cells. On the other hand, immature and EndoH-sensitive glycoforms of tyrosinase increased in B16 cells after geoditin A treatment (
Figure 6b), implicating aberrant glycosylation was induced by geoditin A that affected melanogenesis of B16 cells.
Since MITF is a master transcription factor for members of melanogenic proteins, its down-regulation should have a direct inhibition on the expression of TYR and TYRP1. However, a kinetic analysis of expression of these three proteins in cells treated with 1.25 μg/mL geoditin A (≈EC
50) for 8–24 h has revealed a significant reduction of expression of TYR and TYRP1 at 8 h which preceded the decrease of MITF at 12–24 h, indicating the decrease of TYR and TYRP1 is independent of MITF, and two separated pathways might be involved in their decrease (
Figure 6c).
Apart from melanogenic transcription of tyrosinase, MITF also regulates melanocytic proliferation and survival by up-regulating expression of anti-apoptotic bcl-2 gene as well as a number of survival genes including Pax3, c-kit, Sox9, Sox10 and LEF1 [
10]. A depletion of MITF causes defective replication and cell senescence and death [
6]. B16 cells died with increasing doses of geoditin A when the production of oxidants were overwhelmed (
Figure 3b) and the level of anti-apoptotic Bcl-2 protein decreased in a dose-dependent manner (
Figure 6a). Cells probably died by an apoptotic mechanism (
Figure 5j).
Of the four marine sponge isomalabaricane triterpenes, namely stellettins A and B, and geoditins A and B, tested in HL60 and HT29 cells, the carbonyl group at C-3 position of geoditin A was responsible for its strong apoptotic induction activity [
18]. However doses of geoditin A inhibiting tyrosinase activity (IC
50 ≈ 1 μg/mL) far precede those for cell death/apoptosis (≥10 μg/mL), implicating a machinery other than apoptosis induction by the carbonyl group may contribute to the down-regulation of MITF protein and tyrosinase activity. Experiment is being conducted to elucidate the molecular mechanism underlying the anti-tyrosinase activity elicited by geoditin A.