The Antiviral Activities and Mechanisms of Marine Polysaccharides: An Overview
Abstract
:1. Introduction
2. The Classification and Main Structure Features of Marine Polysaccharides
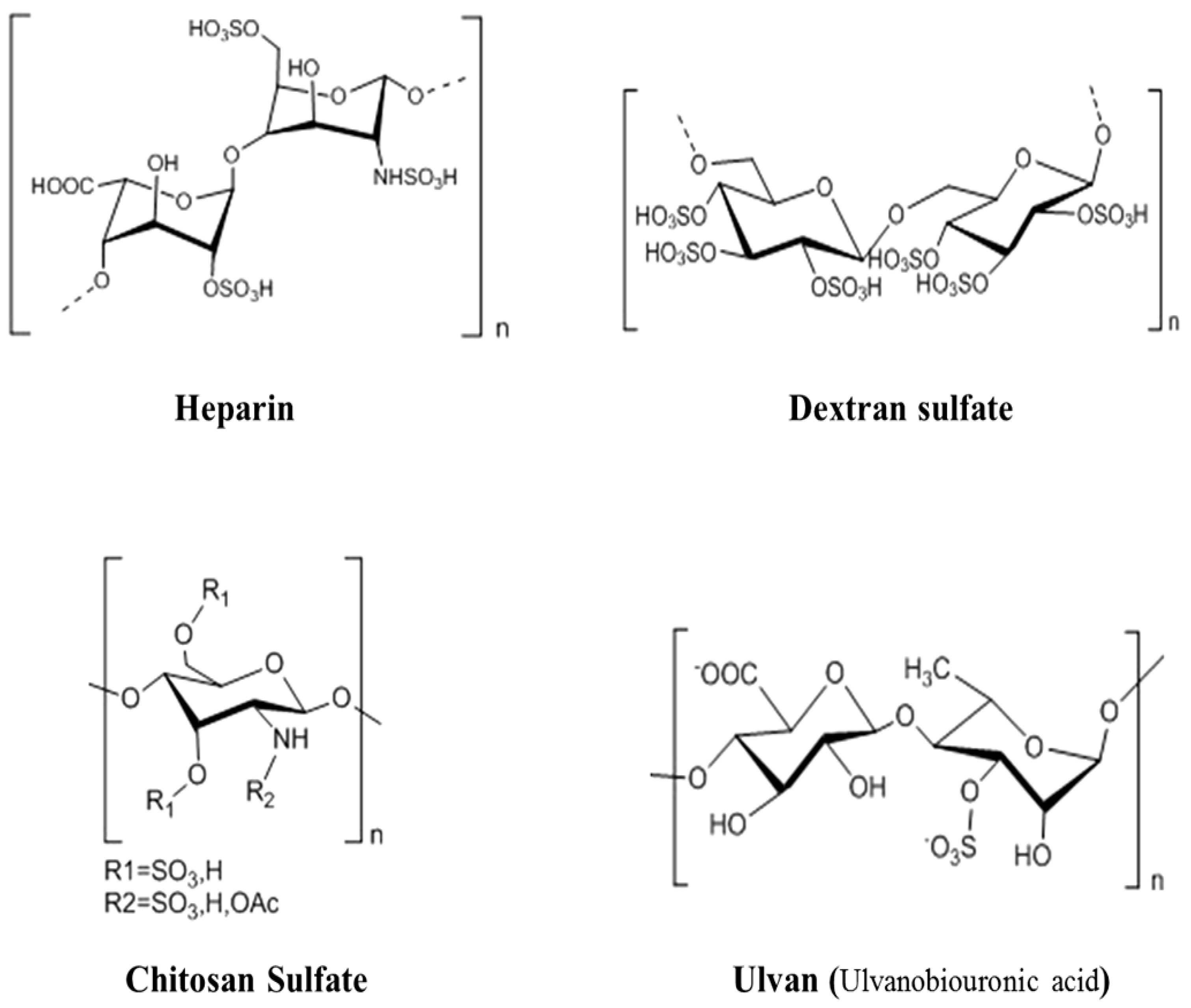
2.1. The Main Structure Features of Marine Animal Polysaccharides
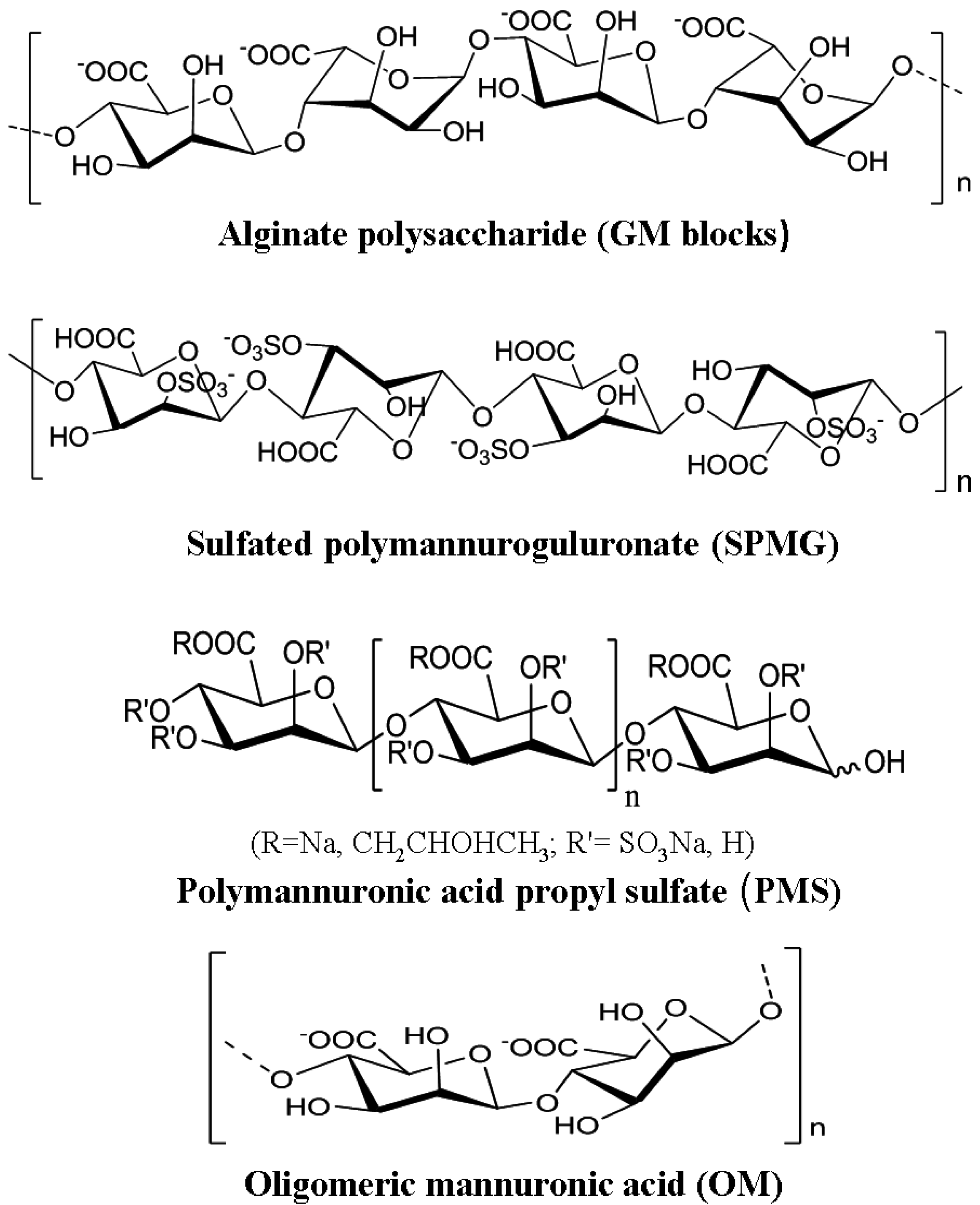
2.2. The Main Structure Features of Marine Plant Polysaccharides
2.3. The Main Structure Features of Marine Microbial Polysaccharides
5. Conclusions
Acknowledgments
References
- De Clercq, E. Antiviral drugs in current chemical reviews. J. Clin. Virol. 2004, 30, 115–133. [Google Scholar] [CrossRef]
- Laurienzo, P. Marine polysaccharides in pharmaceutical applications: An overview. Mar. Drugs 2010, 8, 2435–2465. [Google Scholar] [CrossRef]
- Jiao, G.L.; Yu, G.L.; Wang, W.; Zhao, X.L.; Zhang, J.Z.; Stephen, H.E. Properties of polysaccharides in several seaweeds from Atlantic Canada and their potential anti-influenza viral activities. J. Ocean Univ. China 2012, 11, 205–212. [Google Scholar] [CrossRef]
- Huang, Y.L.; Zheng, T.L. Studies on polysaccharides from marine organism, a review. Mar. Sci. 2004, 28, 58–61. [Google Scholar]
- Singh, K.; Ray, A.R. Biomedical applications of chitin, chitosan, and their derivative. J. Macromol. Sci. 2000, 40, 69–83. [Google Scholar] [CrossRef]
- Zierer, M.S.; Mourao, P.S. A wide diversity of sulfated polysaccharides are synthesized by different species of marine sponges. Carbohydr. Res. 2000, 328, 209–216. [Google Scholar] [CrossRef]
- Ngo, D.N.; Kim, M.M.; Kim, S.K. Chitin oligosaccharides inhibit oxidative stress in live cells. Carbohydr. Polym. 2008, 74, 228–234. [Google Scholar] [CrossRef]
- Burrows, F.; Louime, C.; Abazinge, M.; Onokpise, O. Extraction and evaluation of chitin from crub exoskeleton as a seed fungicide and plant growth enhancer. Am. Eurasian J. Agric. Environ. Sci. 2007, 2, 103–111. [Google Scholar]
- Kim, S.K.; Nghiep, N.D.; Rajapakse, N. Therapeutic prospectives of chitin, chitosan and their derivatives. J. Chitin Chitosan 2006, 11, 1–10. [Google Scholar]
- Jayakumar, R.; New, N.; Nagagama, H.; Furuike, T.; Tamura, H. Synthesis, characterization and biospecific degradation behavior of sulfated chitin. Macromol. Symp. 2008, 264, 163–167. [Google Scholar] [CrossRef]
- Suwan, J.; Zhang, Z.; Li, B.; Vongchan, P.; Meepowpan, P.; Zhang, F.; Mousa, S.A.; Mousa, S.; Premanode, B.; Kongtawelert, P.; et al. Sulfonation of papain-treated chitosan and its mechanism for anticoagulant activity. Carbohydr. Res. 2009, 344, 1190–1196. [Google Scholar] [CrossRef]
- He, Q.; Ao, Q.; Wang, A.; Gong, Y.; Zhao, N.; Zhang, X. In vitro cytotoxicity and protein drug release properties of chitosan/heparin microspheres. Tsinghua Sci. Technol. 2007, 12, 361–365. [Google Scholar] [CrossRef]
- Thierry, B.; Merhi, Y.; Silver, J.; Tabrizian, M. Biodegradable membrane-covered stent from chitosan-based polymers. J. Biomed. Mater. Res. 2005, 75, 556–566. [Google Scholar]
- Prabaharan, M.; Reis, R.L.; Mano, J.F. Carboxymethyl chitosan-graft-phosphatidylethanolamine: Amphiphilic matrices for controlled drug delivery. React. Funct. Polym. 2007, 67, 43–52. [Google Scholar] [CrossRef]
- Gonzalez, R.P.; Leyva, A.; Moraes, M.O. Shark cartilage as source of antiangiogenic compounds: From basic to clinical research. Biol. Pharm. Bull. 2001, 24, 1097–1101. [Google Scholar] [CrossRef]
- Cimino, P.; Bifulco, G.; Casapullo, A.; Bruno, I.; Gomez-Paloma, L.; Riccio, R. Isolation and NMR characterization of rosacelose, a novel sulfated polysaccharide from the sponge Mixylla rosacea. Carbohydr. Res. 2001, 334, 39–47. [Google Scholar] [CrossRef]
- Huang, Y.L.; Zheng, Z.H.; Su, W.J.; Chen, F.; Wu, P.R.; Fang, J.R. Studies on the chemical composition of sea cucumber mensamar intercede III. Immunomodulative effects of PMI-1. Mar. Sci. Bull. 2001, 20, 88–91. [Google Scholar]
- Jiao, G.; Yu, G.; Zhang, J.; Ewart, S.H. Chemical structures and bioactivities of sulphated polysaccharides from marine algae. Mar. Drugs 2011, 9, 196–223. [Google Scholar] [CrossRef]
- Rioux, L.E.; Turgeon, S.L.; Beaulieu, M. Characterization of polysaccharides extracted from brown seaweeds. Carbohydr. Polym. 2007, 69, 530–537. [Google Scholar] [CrossRef]
- Mabeau, S.; Kloareg, B. Isolation and analysis of the cell walls of brown algae: Fucus spiralis, F. ceranoides, F. vesiculosus, F. serratus, Bifurcaria bifurcata and Laminaria digitata. J. Exp. Bot. 1986, 38, 1573–1580. [Google Scholar] [CrossRef]
- Lu, F.; Wei, X.; Zhao, R. Fucoidans: Structure and bioactivity. Molecules 2008, 13, 1671–1695. [Google Scholar] [CrossRef]
- Knutsen, S.H.; Myslabodski, D.E.; Larsen, B.; Usov, A.I. A modified system of nomenclature for red algal galactans. Bot. Mar. 1994, 37, 163–170. [Google Scholar]
- Liu, X.; Xu, F.; Shao, C.; She, Z.; Lin, Y.; Chan, W.L. Bioactive Metabolites from Marine Microorganisms. In Studies in Natural Products Chemistry; Atta-ur-Rahman, F.R.S., Ed.; Elsevier Science Publishers: Amsterdam, The Netherlands, 2008; pp. 197–310. [Google Scholar]
- Debbab, A.; Aly, A.H.; Lin, W.H.; Proksch, P. Bioactive compounds from marine bacteria and fungi. Microb. Biotechnol. 2010, 3, 544–563. [Google Scholar] [CrossRef]
- Waters, A.L.; Hill, R.T.; Place, A.R.; Hamann, M.T. The expanding role of marine microbes in pharmaceutical development. Curr. Opin. Biotechnol. 2010, 21, 780–786. [Google Scholar] [CrossRef]
- Thomas, T.R.A.; Kaulekar, D.P.; Lokabarathi, P.A. Marine drugs from sponge microbe association: A review. Mar. Drugs 2010, 8, 1417–1468. [Google Scholar] [CrossRef]
- Zheng, T.L.; Hong, H.S.; Wang, F.; Maskaoui, K.; Su, J.; Tian, Y. The distribution characters of bacteria β-glucosidase activity in the Taiwan Strait. Mar. Pollut. Bull. 2002, 45, 168–176. [Google Scholar] [CrossRef]
- Lee, J.B.; Hayashi, T.; Hayashi, K.; Sankawa, U. Structural analysis of calcium spirulan (Ca-SP)-derived oligosaccharides using electrospray ionization mass spectrometry. J. Nat. Prod. 2000, 63, 136–138. [Google Scholar] [CrossRef]
- Roger, O.; Kervarec, N.; Ratiskol, J.; Colliec-Jouault, S.; Chevolot, L. Structural studies of the main exopolysaccharide produced by the deep-sea bacterium Alteromonas infernus. Carbohydr. Res. 2004, 339, 2371–2380. [Google Scholar] [CrossRef]
- McCandless, E.L.; Craigie, J.S. Sulphated polysaccharides in red and brown algae. Planta 1979, 112, 201–212. [Google Scholar] [CrossRef]
- Lahaye, M. Developments on gelling algal galactans, their structure and physico-chemistry. J. Appl. Phycol. 2001, 13, 173–184. [Google Scholar] [CrossRef]
- Chiu, Y.H.; Chan, Y.L.; Tsai, L.W.; Li, T.L.; Wu, C.J. Prevention of human enterovirus 71 infection by kappa carrageenan. Antivir. Res. 2012, 95, 128–134. [Google Scholar] [CrossRef]
- Talarico, L.B.; Damonte, E.B. Interference in dengue virus adsorption and uncoating by carrageenans. Virology 2007, 363, 473–485. [Google Scholar] [CrossRef]
- Grassauer, A.; Weinmuellner, R.; Meier, C.; Pretsch, A.; Prieschl-Grassauer, E.; Unger, H. Iota-Carrageenan is a potent inhibitor of rhinovirus infection. Virol. J. 2008, 5, 107. [Google Scholar] [CrossRef]
- Carlucci, M.J.; Scolaro, L.A.; Noseda, M.D.; Cerezo, A.S.; Damonte, E.B. Protective effect of a natural carrageenan on genital herpes simplex virus infection in mice. Antivir. Res. 2004, 64, 137–141. [Google Scholar]
- Leibbrandt, A.; Meier, C.; König-Schuster, M.; Weinmüllner, R.; Kalthoff, D.; Pflugfelder, B.; Graf, P.; Frank-Gehrke, B.; Beer, M.; Fazekas, T.; et al. Iota-carrageenan is a potent inhibitor of influenza A virus infection. PLoS One 2010, 5, e14320. [Google Scholar]
- Talarico, L.B.; Noseda, M.D.; Ducatti, D.R.B.; Duarte, M.E.; Damonte, E.B. Differential inhibition of dengue virus infection in mammalian and mosquito cells by iota-carrageenan. J. Gen. Virol. 2011, 92, 1332–1342. [Google Scholar] [CrossRef]
- Buck, C.B.; Thompson, C.D.; Roberts, J.N.; Muller, M.; Lowy, D.R.; Schiller, J.T. Carrageenan is a potent inhibitor of papillomavirus infection. PLoS Pathog. 2006, 2, e69. [Google Scholar] [CrossRef]
- Yamada, T.; Ogamo, A.; Saito, T.; Uchiyama, H.; Nakagawa, Y. Preparation of O-acylated low-molecular-weight carrageenans with potent anti-HIV activity and low anticoagulant effect. Carbohydr. Polym. 2000, 41, 115–120. [Google Scholar] [CrossRef]
- Yamada, T.; Ogamo, A.; Saito, T.; Watanabe, J.; Uchiyama, H.; Nakagawa, Y. Preparation and anti-HIV activity of low-molecular-weight carrageenans and their sulfated derivatives. Carbohydr. Polym. 1997, 32, 51–55. [Google Scholar] [CrossRef]
- Ji, J.; Wang, L.C.; Wu, H.; Luan, H.M. Bio-function summary of marine oligosaccharides. Int. J. Biol. Sci. 2011, 3, 74–86. [Google Scholar]
- Tang, F.; Chen, F.; Li, F. Preparation and potential in vivo anti-influenza virus activity of low molecular-weight κ-carrageenans and their derivatives. J. Appl. Polym. Sci. 2012. [Google Scholar] [CrossRef]
- Wang, W.; Zhang, P.; Hao, C.; Zhang, X.E.; Cui, Z.Q.; Guan, H.S. In vitro inhibitory effect of carrageenan oligosaccharide on influenza A H1N1 virus. Antivir. Res. 2011, 92, 237–246. [Google Scholar] [CrossRef]
- Wang, W.; Zhang, P.; Yu, G.L.; Li, C.X.; Hao, C.; Qi, X.; Zhang, L.J.; Guan, H.S. Preparation and anti-influenza A virus activity of κ-carrageenan oligosaccharide and its sulphated derivatives. Food Chem. 2012, 133, 880–888. [Google Scholar] [CrossRef]
- Damonte, E.B.; Matulewicz, M.C.; Cerezo, A.S. Sulfated seaweed polysaccharides as antiviral agents. Curr. Med. Chem. 2004, 11, 2399–2419. [Google Scholar] [CrossRef]
- Carlucci, M.J.; Ciancia, M.; Matulewicz, M.C.; Cerezo, A.S.; Damonte, E.B. Antiherpetic activity and mode of action of natural carrageenans of diverse structural types. Antivir. Res. 1999, 43, 93–102. [Google Scholar]
- Girond, S.; Crance, J.M.; van Cuyck-Gandre, H.; Renaudet, J.; Deloince, R. Antiviral activity of carrageenan on hepatitis A virus replication in cell culture. Res. Virol. 1991, 142, 261–270. [Google Scholar] [CrossRef]
- Talarico, L.B.; Pujol, C.A.; Zibetti, R.G.; Faría, P.C.; Noseda, M.D.; Duarte, M.E.; Damonte, E.B. The antiviral activity of sulfated polysaccharides against dengue virus is dependent on virus serotype and host cell. Antivir. Res. 2005, 66, 103–110. [Google Scholar]
- Nelson, R.M.; Cecconi, O.; Roberts, W.G.; Aruffo, A.; Linhardt, R.J.; Bevilacqua, M.P. Heparin oligosaccharides bind L- and P-selectin and inhibit acute inflammation. Blood 1993, 82, 3253–3258. [Google Scholar]
- Douglas, M.S.; Ali, S.; Rix, D.A.; Zhang, J.G.; Kirby, J.A. Endothelial production of MCP-1: Modulation by heparin and consequences for mononuclear cell activation. Immunology 1997, 92, 512–518. [Google Scholar]
- Yu, L.; Garg, H.G.; Li, B.; Linhardt, R.J.; Hales, C.A. Antitumor effect of butanoylated heparin with low anticoagulant activity on lung cancer growth in mice and rats. Curr. Cancer Drug Targets 2010, 10, 229–241. [Google Scholar] [CrossRef]
- Copeland, R.; Balasubramaniam, A.; Tiwari, V.; Zhang, F.; Bridges, A.; Linhardt, R.J.; Shukla, D.; Liu, J. Using a 3-O-sulfated heparin octasaccharide to inhibit the entry of herpes simplex virus type 1. Biochemistry 2008, 47, 5774–5783. [Google Scholar]
- Ramos-Kuri, M.; Barron Romero, B.L.; Aguilar-Setien, A. Inhibition of three alpha herpes viruses (herpes simplex 1 and 2 and pseudo rabies virus) by heparin, heparan and other sulfated polyelectrolytes. Arch. Med. Res. 1996, 27, 43–48. [Google Scholar]
- Neyts, J.; Snoeck, R.; Schols, D.; Balzarini, J.; Esko, J.D.; van Schepdael, A.; de Clercq, E. Sulfated polymers inhibit the interaction of human cytomegalovirus with cell surface heparan sulfate. Virology 1992, 189, 48–58. [Google Scholar] [CrossRef]
- Okazaki, K.; Matsuzaki, T.; Sugahara, Y.; Okada, J.; Hasebe, M.; Iwamura, Y.; Ohnishi, M.; Kanno, T.; Shimizu, M.; Honda, E. BHV-1 adsorption is mediated by the interaction of glycoprotein gIII with heparinlike moiety on the cell surface. Virology 1991, 181, 666–670. [Google Scholar] [CrossRef]
- Kari, B.; Gehrz, R. A human cytomegalovirus glycoprotein complex designated gC-II is a major heparin-binding component of the envelope. J. Virol. 1992, 66, 1761–1764. [Google Scholar]
- Madeleine, L.M.; Reinhard, G. Dextran sulfate inhibits the fusion of influenza virus with model membranes, and suppresses influenza virus replication in vivo. Antivir. Res. 1990, 14, 39–50. [Google Scholar] [CrossRef]
- Witvrouw, M.; Schols, D.; Andrei, G.; Snoeck, R.; Hosoya, M.; Pauwels, R.; Balzarini, J.; de Clercq, E. Antiviral activity of low-MW dextran sulphate (derived from dextran MW 1000) compared to dextran sulphate samples of higher MW. Antivir. Chem. Chemother. 1991, 2, 171–179. [Google Scholar]
- Ivanova, V.; Rouseva, R.; Kolarova, M.; Serkedjieva, J.; Rachev, R.; Manolova, N. Isolation of a polysaccharide with antiviral effect from Ulva lactuca. Prep. Biochem. 1994, 24, 83–97. [Google Scholar] [CrossRef]
- Vo, T.-S.; Kim, S.-K. Potential anti-HIV agents from marine resources: An overview. Mar. Drugs 2010, 8, 2871–2892. [Google Scholar] [CrossRef]
- Xin, X.L.; Geng, M.Y.; Guan, H.S.; Li, Z.L. Study on the mechanism of inhibitory action of 911 on replication of HIV-1 in vitro. Chin. J.Mar. Drugs 2000, 19, 15–18. [Google Scholar]
- Xin, X.L.; Ding, H.; Geng, M.Y.; Liang, P.F.; Li, Y.X.; Guan, H.S. Studies of the anti-AIDS effects of marine polysaccharide drug 911 and its related mechanisms of action. Chin. J. Mar. Drugs 2000, 6, 4–8. [Google Scholar]
- Geng, M.Y.; Li, F.C.; Xin, X.L.; Li, J.; Yan, Z.W.; Guan, H.S. The potential molecular targets of marine sulfated polymannuroguluronate interfering with HIV-1 entry Interaction between SPMG and HIV-1 gp120 and CD4 molecule. Antivir. Res. 2003, 59, 127–135. [Google Scholar]
- Miao, B.C.; Geng, M.Y.; Li, J.; Li, F.; Chen, H.; Guan, H.S.; Ding, J. Sulfated polymannuroguluronate, a novel anti-acquired immune deficiency syndrome (AIDS) drug candidate, targeting CD4 in lymphocyte. Biochem. Pharmacol. 2004, 68, 641–649. [Google Scholar] [CrossRef]
- Liu, H.Y.; Geng, M.Y.; Xin, X.L.; Li, F.C.; Chen, H.X.; Guan, H.S.; Ding, J. Multiple and multivalent interactions of novel anti-AIDS drug candidates, sulfated polymannuronate (SPMG)-derived oligosaccharides, with gp120 and their anti-HIV activitie. Glycobiology 2005, 15, 501–510. [Google Scholar]
- Jiang, B.F.; Xu, X.F.; Li, L.; Yuan, W. Study on “911” anti-HBV effect in HepG2.2.15 cells culture. Mod. Prev. Med. 2003, 30, 517–518. [Google Scholar]
- Queiroz, K.C.S.; Medeiros, V.P.; Queiroz, L.S.; Abreu, L.R.D.; Rocha, H.A.O.; Ferreira, C.V.; Juca, M.B.; Aoyama, H.; Leite, E.L. Inhibition of reverse transcriptase activity of HIV by polysaccharides of brown algae. Biomed. Pharmacother. 2008, 62, 303–307. [Google Scholar] [CrossRef]
- Hidari, K.I.P.J.; Takahashi, N.; Arihara, M.; Nagaoka, M.; Morita, K.; Suzuki, T. Structure and anti-dengue virus activity of sulfated polysaccharide from a marine alga. Biochem. Biophys. Res. Commun. 2008, 376, 91–95. [Google Scholar] [CrossRef]
- Akamatsu, E.; Shimanaga, M.; Kamei, Y. Isolation of an anti-influenza virus substance, MC26 from a marine brown alga Sargassum piluliferum and its antiviral activity against influenza virus. Coast. Bioenviron. 2003, 1, 29–34. [Google Scholar]
- Muto, S.; Niimura, K.; Oohara, M.; Oguchi, Y.; Matsunaga, K.; Hirose, K.; Kakuchi, J.; Sugita, N.; Furusho, T. Polysaccharides from marine algae and antiviral drugs containing the same as active ingredients. Eur. Patent EP295956, 21 December 1988. [Google Scholar]
- Yu, N.; Liu, S.; Han, J.J.; Sun, F.S. The depressive effect of glycosaminoglycan from scallop on type-I herpes simplex virus. Acta Acad. Med. Qingdao Univ. 2008, 2, 111–114. [Google Scholar]
- Zhang, H.F.; Li, J.B.; Hou, G.; Huang, D.N. Study on the inhibition effects of Perna viridis polysaccharides on influenza virus reproduction in MDCK cell cultures. Mod. Med. J. China 2008, 5, 4–7. [Google Scholar]
- Wu, S.M.; Xu, W.M.; Xu, Z.; Lin, F.; Sheng, L. Antiviral effect of oyster extract compound capsule in ducks infected by DHBV. Chin. Pharm. J. 1996, 31, 304–307. [Google Scholar]
- Woo, E.R.; Kim, W.S.; Kim, Y.S. Virus-cell fusion inhibitory activity for the polysaccharides from various Korean edible clams. Arch. Pharm. Res. 2001, 24, 514–517. [Google Scholar] [CrossRef]
- Davis, R.; Zivanovic, S.; D’Souza, D.H.; Davidson, M.P. Effectiveness of chitosan on the inactivation of enteric viral surrogates. Food Microbiol. 2012, 32, 57–62. [Google Scholar] [CrossRef]
- Pospieszny, H.; Chirkov, S.; Atabekov, J. Induction of antiviral resistance in plants by chitosan. Plant Sci. 1991, 79, 63–68. [Google Scholar] [CrossRef]
- Davydova, V.N.; Nagorskaia, V.P.; Gorbach, V.I.; Kalitnik, A.A.; Reunov, A.V.; Solov’eva, T.F.; Ermak, I.M. Chitosan antiviral activity: Dependence on structure and depolymerization method. Appl. Biochem. Microbiol. 2011, 47, 113–118. [Google Scholar]
- Chirkov, S.N. The antiviral activity of chitosan (review). Appl. Biochem. Microbiol. 2002, 38, 1–8. [Google Scholar] [CrossRef]
- Kulikov, S.N.; Chirkov, S.N.; Il’ina, A.V.; Lopatin, S.A.; Varlamov, V.P. Effect of the molecular weight of chitosan on its antiviral activity in plants. Prik. Biokhim. Mikrobiol. 2006, 42, 224–228. [Google Scholar]
- Sosa, M.A.; Fazely, F.; Koch, J.A.; Vercellotti, S.V.; Ruprecht, R.M. N-carboxymethylchitosan-N,O-sulfate as an anti-HIV-1 agent. Biochem. Biophys. Res. Commun. 1991, 174, 489–496. [Google Scholar] [CrossRef]
- Nishimura, S.I.; Kai, H.; Shinada, K.; Yoshida, T.; Tokura, S.; Kurita, K.; Nakashima, H.; Yamamoto, N.; Uryu, T. Regioselective syntheses of sulfated polysaccharides: Specific anti-HIV-1 activity of novel chitin sulfates. Carbohydr. Res. 1998, 306, 427–433. [Google Scholar] [CrossRef]
- Dou, J.L.; Tan, C.Y.; Du, Y.G.; Bai, X.F.; Wang, K.Y.; Ma, X.J. Effects of chitooligosaccharides on rabbit neutrophils in vitro. Carbohydr. Polym. 2007, 69, 209–213. [Google Scholar] [CrossRef]
- Jeon, Y.J.; Kim, S.K. Continuous production of chitooligosaccharides using a dual reactor system. Process Biochem. 2000, 35, 623–632. [Google Scholar] [CrossRef]
- Jeon, Y.J.; Kim, S.K. Production of chitooligosaccharides using ultrafiltration membrane reactor and their antibacterial activity. Carbohydr. Polym. 2000, 41, 133–141. [Google Scholar] [CrossRef]
- Artan, M.; Karadeniz, F.; Karagozlu, M.Z.; Kim, M.M.; Kim, S.K. Anti-HIV-1 activity of low molecular weight sulfated chitooligosaccharides. Carbohydr. Res. 2010, 345, 656–662. [Google Scholar] [CrossRef]
- Pospieszny, H.; Atabekov, J.G. Effect of chitosan on the hypersensitive reaction of bean to alfalfa mosaic virus. Plant Sci. 1989, 62, 29–31. [Google Scholar] [CrossRef]
- Guan, H.S. New drug—Polymannuronic acid propyl sulfates. China Patent 93100608.2, 6 July 1994. [Google Scholar]
- Guan, H.S. New drug—polymeric mannuronic acids sulfate. China Patent 95110396.2, 2 October 1996. [Google Scholar]
- Wang, W.; Yu, G.L.; Hao, C.; Guan, H.S. The application of one kind of oligomeric mannuronic acid in the preparation of anti-influenza A H1N1 virus drugs. China Patent 201110408962.7, 9 December 2011. [Google Scholar]
- Wang, W.; Li, C.X.; Guan, H.S.; Yu, G.L.; Wang, S.X. The application of polymannuronic acid propyl sulfate in the preparation of drugs against influenza A (H1N1) virus. China Patent 201210201876.3, 18 June 2012. [Google Scholar]
- Kanekiyo, K.; Hayashi, K.; Takenaka, H.; Lee, J.B.; Hayashi, T. Anti-herpes simplex virus target of an acidic polysaccharide, nostoflan, from the edible blue-green alga Nostoc flagelliforme. Biol. Pharm. Bull. 2007, 30, 1573–1575. [Google Scholar] [CrossRef]
- Kim, M.; Yim, J.H.; Kim, S.Y.; Kim, H.S.; Lee, W.G.; Kim, S.J.; Kang, P.S.; Lee, C.K. In vitro inhibition of influenza A virus infection by marine microalga-derived sulfated polysaccharide p-KG03. Antivir. Res. 2012, 93, 253–259. [Google Scholar] [CrossRef]
- Carlucci, M.J.; Scolaro, L.A.; Damonte, E.B. Herpes simplex virus type 1variants arising after selection with an antiviral carageenan: Lack of correlation between drug susceptibility and synphenotype. J. Med. Virol. 2002, 68, 92–98. [Google Scholar] [CrossRef]
- Harden, E.A.; Falshaw, R.; Carnachan, S.M.; Kern, E.R.; Prichard, M.N. Virucidal activity of polysaccharide extracts from four algal species against herpes simplex virus. Antivir. Res. 2009, 83, 282–289. [Google Scholar] [CrossRef]
- Su, X.W.; Zivanovic, S.; D’Souza, D.H. Effect of chitosan on the infectivity of murine norovirus, feline calicivirus, and bacteriophage MS2. J. Food Protect. 2009, 72, 2623–2628. [Google Scholar]
- Mazumder, S.; Ghosal, P.K.; Pujol, C.A.; Carlucci, M.J.; Damonte, E.B.; Ray, B. Isolation, chemical investigation and antiviral activity of polysaccharides from Gracilaria corticata (Gracilariaceae, Rhodophyta). Int. J. Biol. Macromol. 2002, 31, 87–95. [Google Scholar] [CrossRef]
- Carlucci, M.J.; Pujol, C.A.; Ciancia, M.; Noseda, M.D.; Matulewicz, M.C.; Damonte, E.B.; Cerezo, A.S. Antiherpetic and anticoagulant properties of carrageenans from the red seaweed Gigartina skottsbergii and their cyclized derivatives: Correlation between structure and biological activity. Int. J. Biol. Macromol. 1997, 20, 97–105. [Google Scholar] [CrossRef]
- Carlucci, M.J.; Scolaro, L.A.; Damonte, E.B. Inhibitory action of natural carrageenans on herpes simplex virus infection of mouse astrocytes. Chemotheraphy 1999, 45, 429–436. [Google Scholar] [CrossRef]
- Trinchero, J.; Ponce, N.M.; Córdoba, O.L.; Flores, M.L.; Pampuro, S.; Stortz, C.A.; Salomón, H.; Turk, G. Antiretroviral activity of fucoidans extracted from the brown seaweed Adenocystis utricularis. Phytother. Res. 2009, 23, 707–712. [Google Scholar] [CrossRef]
- Feldman, S.C.; Reynaldi, S.; Stortz, C.A.; Cerezo, A.S.; Damonte, E.B. Antiviral properties of fucoidan fractions from Leathesia difformis. Phytomedcine 1999, 6, 335–340. [Google Scholar] [CrossRef]
- Majczak, G.A.H.; Richartz, R.R.T.B.; Duarte, M.E.R.; Noseda, M.D. Antiherpetic Activity of Heterofucans Isolated from Sargassum stenophyllum (Fucales, Phaeophyta). In Proceedings of the 17th International Seaweed Symposium, Cape Town, South Africa, 28 January–2 February 2001; Chapman, A.R.O., Anderson, R.J., Vreeland, V.J., Davison, I.R., Eds.; Oxford University Press: Oxford, UK, 2003; pp. 169–174. [Google Scholar]
- Ponce, N.M.A.; Pujol, C.A.; Damonte, E.B.; Flores, M.L.; Stortz, C.A. Fucoidans from the brown seaweed Adenocystis utricularis: Extraction methods, antiviral activity and structural studies. Carbohydr. Res. 2003, 338, 153–165. [Google Scholar] [CrossRef]
- Baba, M.; Snoeck, R.; Pauwels, R.; de Clercq, E. Sulfated polysaccharides are potent and selective inhibitors of various enveloped viruses, including herpes simplex virus, cytomegalovirus, vesicular stomatitis virus, and human immunodeficiency virus. Antimicrob. Agents Chemother. 1988, 32, 1742–1745. [Google Scholar] [CrossRef]
- Mercer, J.; Schelhaas, M.; Helenius, A. Virus entry by endocytosis. Annu. Rev. Biochem. 2010, 79, 803–833. [Google Scholar] [CrossRef]
- Talarico, L.B.; Duarte, M.E.R.; Zibetti, R.G.M.; Noseda, M.D.; Damonte, E.B. An algal-derived DL-galactan hybrid is an efficient preventing agent for in vitro dengue virus infection. Planta Med. 2007, 73, 1464–1468. [Google Scholar] [CrossRef]
- González, M.E.; Alarcón, B.; Carrasco, L. Polysaccharides as antiviral agents: Antiviral activity of carrageenan. Antimicrob. Agents Chemother. 1987, 31, 1388–1393. [Google Scholar] [CrossRef]
- Turner, E.V.; Sonnenfeld, G. Interferon induction by the immunomodulating polyanion Lambda carrageenan. Infect. Immun. 1979, 25, 467–469. [Google Scholar]
- Zhou, G.; Sun, Y.P.; Xin, H.; Zhang, Y.; Li, Z.; Xu, Z. In vivo antitumor and immunomodulation activities of different molecular weight lambda-carrageenans from Chondrus ocellatus. Pharmacol. Res. 2004, 50, 47–53. [Google Scholar] [CrossRef]
- Yuan, H.M.; Song, J.M.; Li, X.G.; Li, X.G.; Li, N.; Dai, J.C. Immunomodulation and antitumor activity of κ-carrageenan oligosaccharides. Cancer Lett. 2006, 243, 228–234. [Google Scholar] [CrossRef]
- Kim, J.-K.; Cho, M.L.; Karnjanapratum, S.; Shin, I.-S.; You, S.G. In vitro and in vivo immunomodulatory activity of sulfated polysaccharides from Enteromorpha prolifera. Int. J. Biol. Macromol. 2011, 49, 1051–1058. [Google Scholar] [CrossRef]
- Patel, S. Therapeutic importance of sulfated polysaccharides from seaweeds: Updating the recent findings. 3 Biotech 2012, 2, 171–185. [Google Scholar] [CrossRef]
- Witvrouw, M.; de Clercq, E. Sulfated polysaccharides extracted from sea algae as potential antiviral drugs. Gen. Pharm. 1997, 29, 497–511. [Google Scholar] [CrossRef]
- Ghosh, T.; Chattopadhyay, K.; Marschall, M.; Karmakar, P.; Mandal, P.; Ray, B. Focus on antivirally active sulfated polysaccharides: From structure-activity analysis to clinical evaluation. Glycobiology 2009, 19, 2–15. [Google Scholar]
- Samples Availability: Available from the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wang, W.; Wang, S.-X.; Guan, H.-S. The Antiviral Activities and Mechanisms of Marine Polysaccharides: An Overview. Mar. Drugs 2012, 10, 2795-2816. https://doi.org/10.3390/md10122795
Wang W, Wang S-X, Guan H-S. The Antiviral Activities and Mechanisms of Marine Polysaccharides: An Overview. Marine Drugs. 2012; 10(12):2795-2816. https://doi.org/10.3390/md10122795
Chicago/Turabian StyleWang, Wei, Shi-Xin Wang, and Hua-Shi Guan. 2012. "The Antiviral Activities and Mechanisms of Marine Polysaccharides: An Overview" Marine Drugs 10, no. 12: 2795-2816. https://doi.org/10.3390/md10122795
APA StyleWang, W., Wang, S.-X., & Guan, H.-S. (2012). The Antiviral Activities and Mechanisms of Marine Polysaccharides: An Overview. Marine Drugs, 10(12), 2795-2816. https://doi.org/10.3390/md10122795