Sepia Ink Oligopeptide Induces Apoptosis in Prostate Cancer Cell Lines via Caspase-3 Activation and Elevation of Bax/Bcl-2 Ratio
Abstract
:1. Introduction
2. Results
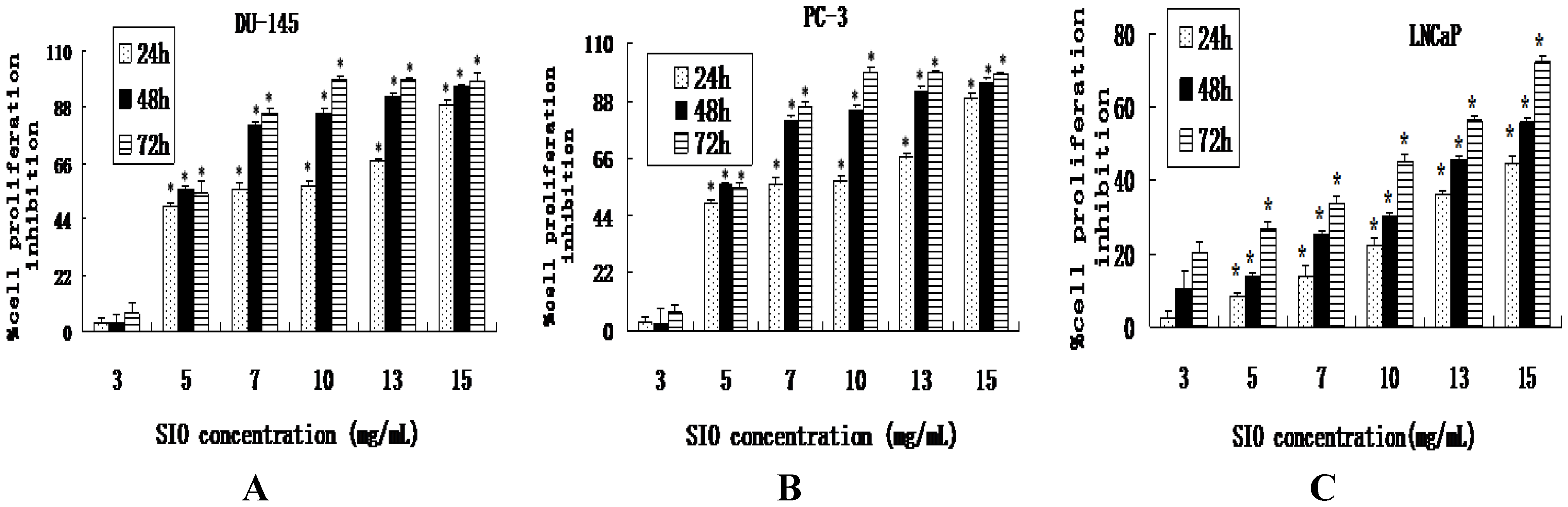
2.1. SIO Inhibits Cell Proliferation of DU-145, PC-3 and LNCaP Cell Lines
2.2. Morphologic Observation by Acridine Orange and Ethidium Bromide (AO/EB) Staining
 ) indicates viable cells; (
) indicates viable cells; (  ) indicates early apoptotic cells; (
) indicates early apoptotic cells; ( 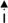 ) indicates late apoptotic cells. Each experiment was performed in triplicate (n = 3) and generated similar morphologic features. Original magnification 400×, bar = 50 μm.
) indicates late apoptotic cells. Each experiment was performed in triplicate (n = 3) and generated similar morphologic features. Original magnification 400×, bar = 50 μm.
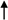 ) indicates viable cells; (
) indicates viable cells; (  ) indicates early apoptotic cells; (
) indicates early apoptotic cells; ( 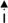 ) indicates late apoptotic cells. Each experiment was performed in triplicate (n = 3) and generated similar morphologic features. Original magnification 400×, bar = 50 μm.
) indicates late apoptotic cells. Each experiment was performed in triplicate (n = 3) and generated similar morphologic features. Original magnification 400×, bar = 50 μm.
2.3. SIO Induces Apoptosis in DU-145, PC-3 and LNCaP Cells Based on Flow Cytometry Analysis
2.4. Cell Cycle Analysis

| DU-145 (A) | |||||||
| SubG1 | G0/G1 | S | G2/M | ||||
| Control | 0 | 95 ± 2.3 | 1 ± 3.4 | 4 ± 2.9 | |||
| 5 mg/mL | 0 | 85 ± 2.2 | 8 ± 3.5 | 7 ± 2.6 | |||
| 10 mg/mL | 0 | 80 ± 1.5 | 12 ± 3.9 | 8 ± 3.6 | |||
| 15 mg/mL | 0 | 65 ± 1.9 | 19 ± 4.0 | 16 ± 2.4 | |||
| PC-3 (B) | |||||||
| SubG1 | G0/G1 | S | G2/M | ||||
| Control | 1 ± 4.3 | 50 ± 2.2 | 23 ± 2.3 | 26 ± 1.9 | |||
| 5 mg/mL | 3 ± 4.5 | 55 ± 3.0 | 20 ± 2.9 | 22 ± 2.1 | |||
| 10 mg/mL | 8 ± 3.9 | 58 ± 2.8 | 17 ± 3.4 | 17 ± 2.7 | |||
| 15 mg/mL | 24 ± 4.3 | 60 ± 1.9 | 8 ± 3.8 | 8 ± 3.5 | |||
| LNCaP (C) | |||||||
| SubG1 | G0/G1 | S | G2/M | ||||
| Control | 0 | 72 ± 1.2 | 3 ± 4.2 | 25 ± 4.5 | |||
| 5 mg/mL | 0 | 70 ± 1.7 | 7 ± 3.5 | 23 ± 4.3 | |||
| 10 mg/mL | 0 | 67 ± 2.1 | 7 ± 3.8 | 26 ± 2.9 | |||
| 15 mg/mL | 0 | 62 ± 2.5 | 9 ± 4.1 | 29 ± 3.5 | |||
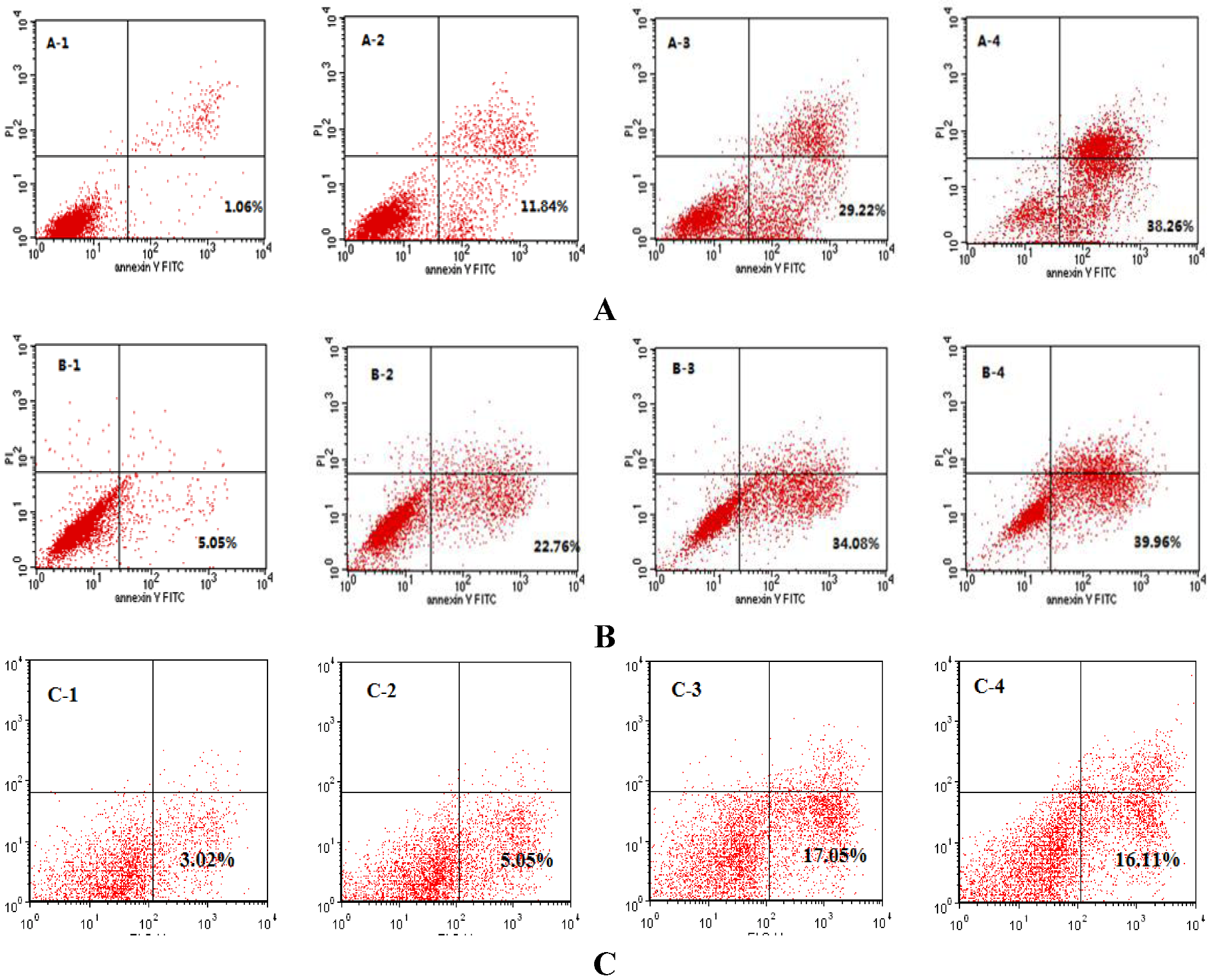
2.5. Western Blotting Results of Bcl-2, Bax and Caspase-3 in DU-145, PC-3 and LNCaP Cells Treated with SIO
3. Discussion
4. Experimental Section
4.1. Materials
4.2. DU-145. PC-3 and LNCaP Cells Culture
4.3. Anti-Proliferative Activity Using Cell Counting Kit-8(CCK-8) Assay

4.4. Morphologic Study with Fluorescence Microscope
4.5. Cell Apoptosis Analysis
4.6. Cell Cycle Analysis
4.7. Western Blot Analysis
4.8. Statistical Analysis
5. Conclusions
Acknowledgments
References
- Parkin, D.M.; Bray, F.; Ferlay, J.; Pisani, P. Global cancer statistics. CA Cancer J. Clin. 2011, 61, 69–90. [Google Scholar] [CrossRef]
- Baade, P.D.; Youlden, D.R.; Krnjacki, L.J. International epidemiology of prostate cancer: Geographical distribution and secular trends. Mol. Nutr. Food Res. 2009, 53, 171–184. [Google Scholar] [CrossRef]
- Bray, F.; Lortet-Tieulent, J.; Ferlay, J.; Forman, D.; Auvinen, A. Prostate cancer incidence and mortality t rends in 37 European countries: An overview. Eur. J. Cancer 2010, 46, 3040–3052. [Google Scholar] [CrossRef]
- Kvale, R.; Moller, B.; Angelsen, A.; Dahl, O.; Fosså, S.D.; Halvorsen, O.J.; Hoem, L.; Solberg, A.; Wahlqvist, R.; Bray, F. Regional trends in prostate cancer incidence, treatment with curative intent and mortality in Norway 1980-2007. Cancer Epidemiol. 2010, 34, 359–367. [Google Scholar]
- Hong, H.; Zhang, Y.; Sun, J.; Cai, W. Positron emission tomography imaging of prostate cancer. Amino Acids 2009, 39, 11–27. [Google Scholar]
- Braun, K.; Ehemann, V.; Wiessler, M.; Pipkorn, R.; Didinger, B.; Mueller, G.; Waldeck, W. High-resolution flow cytometry: A suitable tool for monitoring aneuploid prostate cancer cells after TMZ and TMZ-BioShuttle treatment. Int. J. Med. Sci. 2009, 6, 338–347. [Google Scholar]
- Peyromaure, M.; Valéri, A.; Rebillard, X.; Beuzeboc, P.; Soulié, M.; Salomon, L. Characteristics of prostate cancer in men less than 50-year-old. Prog. Urol. 2009, 19, 803–809. [Google Scholar] [CrossRef]
- Boiardi, A.; Silvan, A.; Milanesi, I.; Botturi, M.; Broggi, G. Primary glial tumor patients treated by combining cis-platin and etoposide. J. Neurooncol. 1991, 11, 165–170. [Google Scholar] [CrossRef]
- Jeremic, B.; Grujicic, D.; Jevremovic, S.; Stanisavljevic, B.; Milojevic, L.; Djuric, L.; Mijatovic, L. Carboplatin and etoposide chemotherapy regimen for recurrent malignant glioma: A phase II study. J. Clin. Oncol. 1992, 10, 1074–1077. [Google Scholar]
- White, L.; Sterling-Levis, K.; Fisher, R.; Tobias, V. Response of brain tumors to chemotherapy, evaluated in a clinically relevant xenograft model. J. Neurooncol. 1995, 25, 39–47. [Google Scholar] [CrossRef]
- Goel, S.; Mita, A.C.; Mita, M.; Rowinsky, E.K.; Chu, Q.S.; Wong, N.; Desjardins, C.; Fang, F.; Jansen, M.; Shuster, D.E.; et al. A phase I study of eribulin mesylate (E7389), a mechanistically novel inhibitor of microtubule dynamics, in patients with advanced solid malignancies. Clin. Cancer Res. 2009, 15, 4207–4212. [Google Scholar]
- Hadaschik, B.A.; Adomat, H.; Fazli, L.; Fradet, Y.; Andersen, R.J.; Gleave, M.E.; So, A.I. Intravesical chemotherapy of high-grade bladder cancer with HTI-286, A Synthetic analogue of the marine sponge product hemiasterlin. Clin. Cancer Res. 2008, 14, 1510–1518. [Google Scholar] [CrossRef]
- Den Brok, M.W.; Nuijen, B.; Meijer, D.M.; Millán, E.; Manada, C.; Beijnen, J.H. Pharmaceutical development of a parenteral lyophilised formulation of the investigational anticancer agent ES-285.HCl. PDA J. Pharm. Sci. Technol. 2005, 59, 246–257. [Google Scholar]
- Luesch, H.; Moore, R.E.; Paul, V.J.; Mooberry, S.L.; Corbett, T.H. Isolation of dolastatin 10 analogue from the marine cyanobacterium symploca species VP642 and total stereochemistry and biological evaluation of its analogue symplostatin 1. J. Nat. Prod. 2001, 64, 907–910. [Google Scholar] [CrossRef]
- Hoffman, M.A.; Blessing, J.A.; Lentz, S.S. Gynecologic Oncology Group Study. A phase II trial of dolastatin-10 in recurrent platinum-sensitive ovarian carcinoma: A Gynecologic Oncology Group study. Gynecol. Oncol. 2003, 89, 95–98. [Google Scholar] [CrossRef]
- Simmons, T.L.; Andrianasolo, E.; McPhail, K.; Flatt, P.; Gerwick, W.H. Marine natural products as anticancer drugs. Mol. Cancer Ther. 2005, 4, 333–342. [Google Scholar]
- Naraoka, T.; Chung, H.S.; Uchisawa, H.; Sasaki, J.; Matsue, H. Tyrosinase activity in antitumor compounds of squid ink. Food Sci. Technol. Res. 2000, 6, 171–175. [Google Scholar] [CrossRef]
- Zeng, M.J.; Zhang, D.M.; Chen, J.H. Research advances of antitumor peptides. Chin. J. Biochem. Pharm. 2007, 28, 139–141. [Google Scholar]
- Ding, G.F.; Huang, F.F.; Yang, Z.S.; Yu, D.; Yang, Y.F. Anticancer activity of an oligopeptide isolated from hydrolysates of Sepia ink. Chin. J. Nat. Med. 2011, 9, 151–155. [Google Scholar]
- Martin, S.J.; Green, D.R. Apoptosis and cancer: The failure of controls on cell death and cell survival. Crit. Rev. Oncol. Hematol. 1995, 8, 137–153. [Google Scholar] [CrossRef]
- Pan, G.; O’Rourke, K.; Chinnaiyan, A.M.; Gentz, R.; Ebner, R.; Ni, J.; Dixit, V.M. The receptor for the cytotoxic ligand TRAIL. Science 1997, 276, 111–113. [Google Scholar] [CrossRef]
- Zapata, J.M.; Pawlowski, K.; Haas, E.; Ware, C.F.; Godzik, A.; Reed, J.C. A diverse family of proteins containing tumor necrosis factor receptor-associated factor domains. J. Biol. Chem. 2001, 276, 24242–24252. [Google Scholar]
- Ashe, P.C.; Berry, M.D. Apoptotic signaling cascades. Prog. Neuropsychopharmacol. Biol. Psychiatry 2003, 27, 199–214. [Google Scholar] [CrossRef]
- Cory, S.; Adams, J.M. The Bcl2 family: Regulators of the cellular life-or-death switch. Nat. Rev. Cancer 2002, 2, 647–656. [Google Scholar] [CrossRef]
- Ghobrial, I.M.; Witzig, T.E.; Adjei, A.A. Targeting apoptosis pathways in cancer therapy. CA Cancer J. Clin. 2005, 55, 178–194. [Google Scholar] [CrossRef]
- Mirjolet, J.F.; Barberi-Heyob, M.; Didelot, C.; Peyrat, J.P.; Abecassis, J.; Millon, R.; Merlin, J.L. Bcl-2/Bax protein ratio predicts 5-fluorouracil sensitivity independently of p53 status. Br. J. Cancer 2000, 83, 1380–1386. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Huang, F.; Yang, Z.; Yu, D.; Wang, J.; Li, R.; Ding, G. Sepia Ink Oligopeptide Induces Apoptosis in Prostate Cancer Cell Lines via Caspase-3 Activation and Elevation of Bax/Bcl-2 Ratio. Mar. Drugs 2012, 10, 2153-2165. https://doi.org/10.3390/md10102153
Huang F, Yang Z, Yu D, Wang J, Li R, Ding G. Sepia Ink Oligopeptide Induces Apoptosis in Prostate Cancer Cell Lines via Caspase-3 Activation and Elevation of Bax/Bcl-2 Ratio. Marine Drugs. 2012; 10(10):2153-2165. https://doi.org/10.3390/md10102153
Chicago/Turabian StyleHuang, Fangfang, Zuisu Yang, Di Yu, Jiabin Wang, Rong Li, and Guofang Ding. 2012. "Sepia Ink Oligopeptide Induces Apoptosis in Prostate Cancer Cell Lines via Caspase-3 Activation and Elevation of Bax/Bcl-2 Ratio" Marine Drugs 10, no. 10: 2153-2165. https://doi.org/10.3390/md10102153




