A Chronic Oral Toxicity Study of Marine Collagen Peptides Preparation from Chum Salmon (Oncorhynchus keta) Skin Using Sprague-Dawley Rat
Abstract
:1. Introduction
2. Results
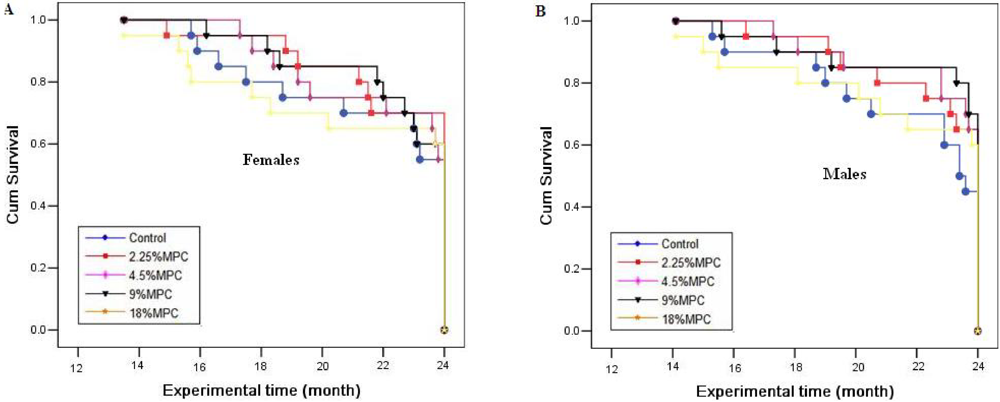
2.1. Survival Rates
2.2. General Condition, Bodyweights, Food Efficiency, Food Consumption and MCP Intake

| Sex | MCP (%) | n | 6 months Food efficiency (g/100 g) | n | 12 months Food efficiency (g/100 g) | n | 18 months Food efficiency (g/100 g) | n | 24 months Food efficiency (g/100 g) |
|---|---|---|---|---|---|---|---|---|---|
| Female | 0 | 20 | 3.54 ± 1.41 a | 20 | 2.04 ± 5.80 | 16 | −0.86 ± 5.82 | 10 | −0.02 ± 0.08 |
| 2.25 | 20 | 2.21 ± 1.50 | 20 | 2.74 ± 6.57 | 19 | 0.42 ± 3.15 | 10 | 0.04 ± 0.06 | |
| 4.5 | 20 | 2.74 ± 1.98 | 20 | 4.04 ± 2.49 | 18 | −0.35 ± 2.49 | 10 | 0.03 ± 0.05 | |
| 9 | 20 | 3.53 ± 2.24 | 20 | 3.50 ± 5.11 | 19 | 0.99 ± 2.22 | 10 | 0.01 ± 0.04 | |
| 18 | 20 | 2.74 ± 3.14 | 20 | 2.49 ± 1.78 | 17 | 0.39 ± 2.32 | 10 | 0.04 ± 0.05 | |
| Male | 0 | 20 | 2.13 ± 1.46 | 20 | 2.11 ± 3.00 | 18 | 0.76 ± 1.46 | 9 | −0.12 ± 0.30 |
| 2.25 | 20 | 3.05 ± 2.40 | 20 | 4.48 ± 1.66 | 19 | 0.57 ± 1.57 | 10 | −0.09 ± 0.14 | |
| 4.5 | 20 | 2.78 ± 2.86 | 20 | 2.79 ± 2.45 | 19 | 1.55 ± 2.82 | 10 | 0.00 ± 0.02 | |
| 9 | 20 | 3.33 ± 1.75 | 20 | 3.02 ± 2.44 | 18 | −0.50 ± 2.57 | 10 | −0.02 ± 0.07 | |
| 18 | 20 | 3.97 ± 2.06 | 20 | 3.54 ± 3.18 | 18 | 0.46 ± 1.53 | 10 | −0.05 ± 0.06 |
| Sex | MCP (%) | n | 12 months | n | 24 months | ||
|---|---|---|---|---|---|---|---|
| Food consumption a (g/kg·bw/day) | Intake of MCP b (g/kg·bw/day) | Food consumption a (g/kg·bw/day) | Intake of MCP b (g/kg·bw/day) | ||||
| Female | 0 | 20 | 65.4 ± 5.7 | 0.000 | 10 | 57.0 ± 3.9 | 0.000 |
| 2.25 | 20 | 63.7 ± 8.9 | 1.290 | 10 | 52.5 ± 4.1 | 1.063 | |
| 4.5 | 20 | 65.2 ± 9.2 | 2.641 | 10 | 54.7 ± 7.7 | 2.216 | |
| 9 | 20 | 61.4 ± 6.2 | 4.973 | 10 | 56.9 ± 4.9 | 4.609 | |
| 18 | 20 | 62.7 ± 7.2 | 10.157 | 10 | 53.0 ± 4.2 | 8.586 | |
| Male | 0 | 20 | 51.7 ± 4.9 | 0.000 | 9 | 43.7 ± 3.9 | 0.000 |
| 2.25 | 20 | 51.3 ± 4.6 | 1.039 | 10 | 44.8 ± 3.2 | 0.907 | |
| 4.5 | 20 | 50.6 ± 5.8 | 2.049 | 10 | 44.4 ± 3.8 | 1.798 | |
| 9 | 20 | 50.7 ± 5.8 | 4.107 | 10 | 42.2 ± 3.4 | 3.418 | |
| 18 | 20 | 48.8 ± 3.6 | 7.906 | 10 | 41.1 ± 3.5 | 6.658 | |
2.3. Clinical Biochemical Parameters
2.4. Urinalysis
2.5. Relative Organ Weights
2.6. Histopathological Examination
| Months | Parameter | Female MCP (%) | Male MCP (%) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 0% | 2.25% | 4.5% | 9% | 18% | 0% | 2.25% | 4.5% | 9% | 18% | |||
| n | 10 | 10 | 10 | 10 | 10 | 10 | 10 | 10 | 10 | 10 | ||
| 12 | ALT (U/L) | 61.80 ± 12.17 a | 59.95 ± 16.43 | 67.79 ± 18.09 | 70.27 ± 11.39 | 68.93 ± 11.59 | 63.15 ± 16.78 | 58.40 ± 9.77 | 65.36 ± 14.71 | 58.07 ± 19.87 | 63.21 ± 16.88 | |
| AST (U/L) | 126.15 ± 38.96 | 137.70 ± 31.27 | 130.38 ± 28.35 | 134.67 ± 27.07 | 135.00 ± 43.59 | 151.10 ± 45.51 | 134.13 ± 37.31 | 163.00 ± 42.07 | 125.50 ± 45.85 | 123.64 ± 34.70 | ||
| ALB (g/L) | 37.96 ± 5.24 | 38.66 ± 2.41 | 39.49 ± 3.94 | 39.65 ± 3.12 | 39.54 ± 3.65 | 31.64 ± 2.03 | 31.55 ± 1.48 | 31.20 ± 2.88 | 29.37 ± 3.26 | 31.08 ± 2.01 | ||
| TP (g/L) | 80.00 ± 4.62 | 77.70 ± 5.11 | 80.81 ± 3.99 | 82.38 ± 5.39 | 81.20 ± 5.99 | 70.00 ± 2.81 | 68.06 ± 6.09 | 69.50 ± 4.45 | 65.87 ± 6.33 | 67.86 ± 2.71 | ||
| BUN (mmol/L) | 9.36 ± 2.57 | 8.14 ± 1.71 | 9.43 ± 1.82 | 9.93 ± 1.87 | 9.41 ± 1.38 | 6.09 ± 1.05 | 5.56 ± 0.87 | 6.73 ± 1.68 | 6.90 ± 2.68 | 5.86 ± 0.69 | ||
| CR (µmol/L) | 74.85 ± 13.63 | 78.65 ± 10.80 | 75.19 ± 11.86 | 83.07 ± 12.60 | 71.60 ± 12.11 | 68.85 ± 4.90 | 67.25 ± 3.00 | 73.86 ± 10.27 | 75.67 ± 16.39 | 68.57 ± 3.74 | ||
| TCHOL (mmol/L) | 2.57 ± 1.63 | 2.42 ± 0.56 | 2.43 ± 1.01 | 2.52 ± 0.39 | 2.51 ± 1.00 | 2.95 ± 1.44 | 2.31 ± 0.33 | 2.54 ± 1.36 | 2.67 ± 0.78 | 2.39 ± 0.67 | ||
| TG (mmol/L) | 4.14 ± 2.09 | 2.47 ± 1.64 * | 2.87 ± 2.11 * | 3.09 ± 1.65 | 3.22 ± 1.48 | 2.45 ± 1.35 | 2.03 ± 1.01 | 1.81 ± 1.01 | 2.13 ± 1.54 | 2.03 ± 0.79 | ||
| GLU (mmol/L) | 6.27 ± 0.69 | 6.27 ± 0.52 | 6.50 ± 0.37 | 6.66 ± 0.44 | 6.57 ± 0.40 | 6.07 ± 1.65 | 6.25 ± 0.41 | 6.04 ± 0.62 | 6.24 ± 0.31 | 6.39 ± 0.40 | ||
| 24 | n | 10 | 10 | 10 | 10 | 10 | 9 | 10 | 10 | 10 | 10 | |
| ALT (U/L) | 41.89 ± 13.93 | 41.30 ± 5.95 | 45.30 ± 12.28 | 43.17 ± 7.14 | 45.63 ± 15.77 | 43.50 ± 12.99 | 41.67 ± 10.97 | 41.25 ± 7.76 | 41.89 ± 8.34 | 43.50 ± 17.13 | ||
| AST (U/L) | 152.11 ± 26.94 | 181.70 ± 47.22 | 162.70 ± 30.79 | 153.17 ± 40.59 | 166.13 ± 57.35 | 164.60 ± 63.55 | 132.56 ± 43.02 | 163.25 ± 37.21 | 160.89 ± 28.94 | 133.71 ± 50.51 | ||
| ALB (g/L) | 37.39 ± 2.65 | 37.38 ± 2.64 | 35.00 ± 2.12 | 35.07 ± 1.73 | 35.94 ± 1.57 | 28.65 ± 2.47 | 28.19 ± 2.81 | 29.20 ± 0.82 | 29.29 ± 2.82 | 29.53 ± 1.45 | ||
| TP (g/L) | 75.11 ± 7.46 | 78.33 ± 5.10 | 73.80 ± 6.07 | 76.50 ± 4.97 | 74.50 ± 3.51 | 63.00 ± 4.55 | 64.11 ± 4.70 | 61.75 ± 3.86 | 69.44 ± 5.81 * | 67.43 ± 2.57 | ||
| BUN (mmol/L) | 6.76 ± 1.55 | 6.41 ± 1.35 | 6.64 ± 1.59 | 6.08 ± 2.15 | 6.21 ± 0.72 | 6.05 ± 1.46 | 7.01 ± 4.80 | 5.08 ± 0.83 | 5.83 ± 0.87 | 7.64 ± 4.88 | ||
| CR (µmol/L) | 67.22 ± 5.14 | 71.10 ± 5.32 | 66.90 ± 5.02 | 72.00 ± 5.34 | 71.25 ± 5.50 | 56.90 ± 6.45 | 58.43 ± 4.12 | 53.25 ± 2.63 | 61.10 ± 4.62 | 61.30 ± 3.01 | ||
| TCHOL (mmol/L) | 2.61 ± 0.58 | 2.49 ± 0.42 | 2.48 ± 0.35 | 2.79 ± 0.37 | 2.48 ± 0.45 | 4.02 ± 1.32 | 3.49 ± 0.76 | 2.47 ± 0.27 * | 2.98 ± 0.58 * | 3.08 ± 0.73 * | ||
| TG (mmol/L) | 4.33 ± 1.37 | 3.63 ± 0.88 | 2.48 ± 1.11 * | 2.96 ± 1.34 * | 3.42 ± 1.24 | 3.99 ± 2.07 | 2.67 ± 0.85 * | 2.21 ± 0.86 * | 2.31 ± 1.02 * | 2.35 ± 0.87 * | ||
| GLU (mmol/L) | 5.27 ± 0.46 | 5.06 ± 1.09 | 4.73 ± 0.82 | 5.58 ± 0.43 | 4.94 ± 1.15 | 4.82 ± 0.65 | 4.63 ± 0.93 | 5.18 ± 0.57 | 5.53 ± 0.69 | 5.37 ± 0.93 | ||
| Sex | MCP (%) | n | Brain (g/100 g·bw) | Heart (g/100 g·bw) | Lungs (g/100 g·bw) | Liver (g/100 g·bw) | Spleen (g/100 g·bw) | Kidneys (g/100 g·bw) | Adrenals (g/100 g·bw) | Ovaries (g/100 g·bw) | Testis (g/100 g·bw) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Female | 0 | 11 | 0.433 ± 0.195 a | 0.320 ± 0.070 | 0.354 ± 0.066 | 2.808 ± 0.819 | 0.196 ± 0.232 | 0.665 ± 0.182 | 0.034 ± 0.012 | 0.020 ± 0.010 | |
| 2.25 | 14 | 0.450 ± 0.157 | 0.404 ± 0.096 | 0.367 ± 0.139 | 2.638 ± 0.880 | 0.114 ± 0.059 | 0.606 ± 0.276 | 0.030 ± 0.018 | 0.026 ± 0.010 | ||
| 4.5 | 11 | 0.519 ± 0.275 | 0.381 ± 0.118 | 0.351 ± 0.110 | 2.387 ± 0.609 | 0.115 ± 0.046 | 0.629 ± 0.253 | 0.025 ± 0.006 | 0.019 ± 0.008 | ||
| 9 | 12 | 0.494 ± 0.240 | 0.430 ± 0.035 | 0.412 ± 0.094 | 2.573 ± 0.459 | 0.111 ± 0.016 | 0.734 ± 0.290 | 0.032 ± 0.007 | 0.026 ± 0.002 | ||
| 18 | 12 | 0.416 ± 0.099 | 0.350 ± 0.071 | 0.348 ± 0.094 | 2.423 ± 0.849 | 0.155 ± 0.093 | 0.659 ± 0.177 | 0.031 ± 0.007 | 0.023 ± 0.016 | ||
| Male | 0 | 9 | 0.281 ± 0.082 | 0.410 ± 0.059 | 0.391 ± 0.123 | 3.218 ± 1.216 | 0.129 ± 0.037 | 0.726 ± 0.214 | 0.022 ± 0.009 | 0.276 ± 0.067 | |
| 2.25 | 13 | 0.318 ± 0.046 | 0.433 ± 0.099 | 0.441 ± 0.079 | 2.761 ± 0.850 | 0.134 ± 0.040 | 0.770 ± 0.246 | 0.016 ± 0.011 | 0.384 ± 0.110 | ||
| 4.5 | 13 | 0.369 ± 0.150 | 0.471 ± 0.079 | 0.425 ± 0.028 | 2.468 ± 0.727 | 0.135 ± 0.087 | 0.637 ± 0.162 | 0.017 ± 0.013 | 0.397 ± 0.135 | ||
| 9 | 14 | 0.338 ± 0.099 | 0.490 ± 0.110 | 0.406 ± 0.111 | 2.870 ± 0.877 | 0.118 ± 0.072 | 0.762 ± 0.431 | 0.019 ± 0.008 | 0.380 ± 0.090 | ||
| 18 | 12 | 0.382 ± 0.133 | 0.465 ± 0.088 | 0.412 ± 0.090 | 3.199 ± 0.642 | 0.151 ± 0.052 | 0.833 ± 0.275 | 0.015 ± 0.004 | 0.301 ± 0.098 |
| Sites | Lesions | Female MCP (%) | Male MCP (%) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 0 | 2.25 | 4.5 | 9 | 18 | 0 | 2.25 | 4.5 | 9 | 18 | ||
| (n = 20) | (n = 20) | (n = 20) | (n = 20) | (n = 20) | (n = 20) | (n = 20) | (n = 20) | (n = 20) | (n = 20) | ||
| Brain | Compression | 2:00 AM | 2 | 2 | 1 | 1 | 2 | 1 | 1 | 2 | 2 |
| Pituitary | Hyperplasia, pars distalis | 4 | 3 | 2 | 2 | 3 | 4 | 3 | 2 | 2 | 3 |
| Thyroid gland | C-cell hyperplasia | 4 | 3 | 5 | 3 | 2 | 4 | 4 | 3 | 2 | 2 |
| Heart | Degeneration myocardial | 4 | 2 | 2 | 2 | 3 | 4 | 3 | 2 | 2 | 3 |
| Focal myocarditis | 2 | 0 | 1 | 0 | 1 | 0 | 0 | 1 | 0 | 0 | |
| Lung | Inflammatory cell foci | 4 | 2 | 2 | 1 | 2 | 3 | 2 | 1 | 2 | 2 |
| Emphysema | 5 | 3 | 3 | 3 | 2 | 5 | 3 | 2 | 2 | 3 | |
| Liver | Vacuolation, hepatocyte | 8 | 5 | 3 | 4 | 5 | 7 | 3 | 2 | 2 | 4 |
| Fibrosis | 1 | 1 | 0 | 2 | 1 | 1 | 0 | 1 | 2 | 0 | |
| Stomach | Intestinal metaplasia | 1 | 0 | 1 | 0 | 1 | 0 | 1 | 0 | 0 | 1 |
| Intestine | Diverticulum | 0 | 1 | 0 | 1 | 1 | 1 | 0 | 0 | 0 | 0 |
| Spleen | Increased extramedullary hematopoiesis | 2 | 0 | 1 | 1 | 0 | 1 | 0 | 1 | 0 | 0 |
| Kidney | Fibrosis | 2 | 1 | 0 | 1 | 0 | 1 | 2 | 1 | 0 | 1 |
| Inflammatory cell foci, glomerular | 7 | 6 | 6 | 5 | 4 | 4 | 3 | 5 | 5 | 3 | |
| Inflammatory cell foci, pelvis | 1 | 0 | 0 | 1 | 1 | 0 | 1 | 0 | 0 | 1 | |
| Adrenal gland | Hyperplasia, cortex | 2 | 2 | 0 | 0 | 1 | 2 | 1 | 1 | 0 | 1 |
| Hyperplasia, medulla | 2 | 0 | 1 | 2 | 1 | 2 | 1 | 0 | 2 | 2 | |
| Urinary bladder | Transitional cell hyperplasia | 1 | 0 | 0 | 1 | 1 | 1 | 2 | 0 | 1 | 0 |
| Mammary gland | Hyperplasia | 6 | 4 | 5 | 4 | 3 | |||||
| Pancreas | Atrophy, islet cell | 6 | 5 | 4 | 4 | 3 | 5 | 4 | 5 | 6 | 4 |
| Hyperplasia, islet cell | 0 | 1 | 0 | 1 | 1 | 0 | 0 | 1 | 1 | 0 | |
| Uterus | Metaplasia, squamous | 6 | 5 | 4 | 3 | 4 | |||||
| Hyperplasia, endometrium, | 10 | 8 | 7 | 9 | 7 | ||||||
| Ovary | Atrophy | 4 | 2 | 3 | 2 | 1 | |||||
| Vargina | Cyst, submucosa | 1 | 0 | 1 | 1 | 0 | |||||
| Testes | Atrophy, testicular | 6 | 5 | 5 | 6 | 4 | |||||
| Prostate gland | Inflammation | 1 | 1 | 0 | 0 | 1 | |||||
3. Discussion
4. Experimental Section
4.1. Preparation and Identification Procedure of Test Substance
4.2. Characterization of MCP
| Amino acid | No. residues/100 residues |
|---|---|
| Glycine | 23.77 |
| Glutamic acid | 12.22 |
| Proline | 9.79 |
| Hydroxyproline | 7.51 |
| Aspartic acid | 7.29 |
| Alanine | 6.59 |
| Arginine | 6.08 |
| Lysine | 5.66 |
| Leucine | 4.64 |
| Serine | 4.23 |
| Valine | 2.94 |
| Isoleucine | 2.57 |
| Threonine | 2.53 |
| Phenylalanine | 2.51 |
| Histidine | 1.61 |
| Methionine | 0.03 |
| Tyrosine | 0.03 |
4.3. Experimental Animals and Housing Conditions
4.4. Experimental Design
4.5. Clinical Investigations
4.6. Pathology Examinations
4.7. Statistical Analysis
5. Conclusions
Acknowlegements
- Samples Availability: Available from the authors.
References
- Hartmann, R.; Meisel, H. Food-derived peptides with biological activity: From research to food applications. Curr. Opin. Biotechnol. 2007, 18, 163–169. [Google Scholar]
- Kim, S.; Mendis, E. Bioactive compounds from marine processing byproducts—A review. Food Res. Int. 2006, 39, 383–393. [Google Scholar]
- Rustad, T. Utilisation of marine by-products. Electron. J. Environ. Agric. Food Technol. 2003, 2, 458–463. [Google Scholar]
- Wang, L.; An, X.X.; Yang, F.M.; Xin, Z.H.; Zhao, L.Y.; Hu, Q.H. Isolation and characterisation of collagens from the skin, scale and bone of deep-sea redfish (Sebastes mentella). Food Chem. 2008, 108, 616–623. [Google Scholar]
- Lai, C.-H.; Wu, P.-C.; Wu, C.-H.; Shiau, C.-Y. Studies on antioxidative activities of hydrolysates from fish scales collagen of tilapia. J. Taiwan Fish. Res. 2008, 15, 99–108. [Google Scholar]
- Fahmi, A.; Morimura, S.; Guo, H.C.; Shigematsu, T.; Kida, K.; Uemura, Y. Production of angiotensin I converting enzyme inhibitory peptides from sea bream scales. Process Biochem. 2004, 39, 1195–1200. [Google Scholar]
- Zhang, F.; Wang, Z.; Xu, S. Macroporous resin purification of grass carp fish (Ctenopharyngodon idella) scale peptides with in vitro angiotensin-I converting enzyme (ACE) inhibitory ability. Food Chem. 2009, 117, 387–392. [Google Scholar]
- Tanaka, M.; Koyama, Y.; Nomura, Y. Effects of collagen peptide ingestion on UV-B-induced skin damage. Biosci. Biotechnol. Biochem. 2009, 73, 930–932. [Google Scholar]
- Nagai, H.; Takuwa, K.; Nakao, M.; Sakamoto, B.; Crow, G.L.; Nakajima, T. Isolation and characterization of a novel protein toxin from the Hawaiian box jellyfish (sea wasp) Carybdea alata. Biochem. Biophys. Res. Commun. 2000, 275, 589–594. [Google Scholar]
- Morimura, S.; Nagata, H.; Uemura, Y.; Fahmi, A.; Shigematsu, T.; Kida, K. Development of an effective process for utilization of collagen from livestock and fish waste. Process Biochem. 2002, 37, 1403–1412. [Google Scholar]
- Van der Laan, J. Current status and use of short/medium-term models for assessment of carcinogenicity of human pharmaceuticals: Regulatory perspectives. Toxicol. Lett. 2000, 11, 567–572. [Google Scholar]
- Gilani, C.S.; Xiao, C.W.; Lee, N. Need for accurate and standardized determination of amino acids and bioactive peptides for evaluating protein quality and potential health effects of foods and dietary supplements. J. AOAC Int. 2008, 91, 894–900. [Google Scholar]
- Chengelis, C.; Kirkpatrick, J.; Bruner, R.; Freshwater, L.; Morita, O.; Tamaki, Y.; Suzuki, H. A 24-month dietary carcinogenicity study of DAG (diacylglycerol) in rats. Food Chem. Toxicol. 2006, 44, 98–121. [Google Scholar]
- Baldrick, P. Carcinogenicity evaluation: Comparison of tumor data from dual control groups in the Sprague-Dawley rat. Toxicol. Pathol. 2005, 33, 283–291. [Google Scholar]
- Wu, J.; Fujioka, M.; Sugimoto, K.; Mu, G.; Ishimi, Y. Assessment of effectiveness of oral administration of collagen peptide on bone metabolism in growing and mature rats. J. Bone Miner. Metab. 2004, 22, 547–553. [Google Scholar]
- Roland, W.M. Role of collagen hydrolysate in bone and joint disease. Semin. Arthritis Rheum. 2000, 30, 87–99. [Google Scholar]
- Sanchez-Gomez, M.; Malmlof, K.; Mejia, W.; Bermudez, A.; Ochoa, M.; Carrasco-Rodriguez, S.; Skottner, A. Insulin-like growth factor-I, but not growth hormone, is dependent on a high protein intake to increase nitrogen balance in the rat. Br. J. Nutr. 2007, 81, 145–152. [Google Scholar]
- Galman, C.; Matasconi, M.; Persson, L.; Parini, P.; Angelin, B.; Rudling, M. Age-induced hypercholesterolemia in the rat relates to reduced elimination but not increased intestinal absorption of cholesterol. Am. J. Physiol. Endocrinol. Metab. 2007, 293, E737–E742. [Google Scholar]
- Wergedahl, H.; Liaset, B.; Gudbrandsen, O.; Lied, E.; Espe, M.; Muna, Z.; Mork, S.; Berge, R. Fish protein hydrolysate reduces plasma total cholesterol, increases the proportion of HDL cholesterol, and lowers Acyl-CoA: Cholesterol acyltransferase activity in liver of zucker rats 1. J. Nutr. 2004, 134, 1320–1327. [Google Scholar]
- Hubscher, S. Histological assessment of non-alcoholic fatty liver disease. Histopathology 2006, 49, 450–465. [Google Scholar]
- Felmlee, M.; Woo, G.; Simko, E.; Krol, E.; Muir, A.; Alcorn, J. Effects of the flaxseed lignans secoisolariciresinol diglucoside and its aglycone on serum and hepatic lipids in hyperlipidaemic rats. Br. J. Nutr. 2009, 102, 361–369. [Google Scholar]
- Masataka, S.; Chikako, K.; Tomoyuki, H.; Naoyuki, U.; Hiramitsu, S. Effect of collagen hydrolysates from Salmon and Trout skins on the lipid profile in rats. J. Agric. Food Chem. 2009, 57, 10477–10482. [Google Scholar]
- Pei, X.; Yang, R.; Zhang, Z.; Gao, L.; Wang, J.; Xu, Y.; Zhao, M.; Han, X.; Liu, Z.; Li, Y. Marine collagen peptide isolated from Chum Salmon (Oncorhynchus keta) skin facilitates learning and memory in aged C57BL/6J mice. Food Chem. 2010, 118, 330–340. [Google Scholar]
- National Research Council, Guide for the Care and Use of Laboratory Animals; National Academy Press: Washington, DC, USA, 1996; pp. 21–55.
© 2012 by the authors; licensee MDPI, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Liang, J.; Pei, X.-R.; Zhang, Z.-F.; Wang, N.; Wang, J.-B.; Li, Y. A Chronic Oral Toxicity Study of Marine Collagen Peptides Preparation from Chum Salmon (Oncorhynchus keta) Skin Using Sprague-Dawley Rat. Mar. Drugs 2012, 10, 20-34. https://doi.org/10.3390/md10010020
Liang J, Pei X-R, Zhang Z-F, Wang N, Wang J-B, Li Y. A Chronic Oral Toxicity Study of Marine Collagen Peptides Preparation from Chum Salmon (Oncorhynchus keta) Skin Using Sprague-Dawley Rat. Marine Drugs. 2012; 10(1):20-34. https://doi.org/10.3390/md10010020
Chicago/Turabian StyleLiang, Jiang, Xin-Rong Pei, Zhao-Feng Zhang, Nan Wang, Jun-Bo Wang, and Yong Li. 2012. "A Chronic Oral Toxicity Study of Marine Collagen Peptides Preparation from Chum Salmon (Oncorhynchus keta) Skin Using Sprague-Dawley Rat" Marine Drugs 10, no. 1: 20-34. https://doi.org/10.3390/md10010020
APA StyleLiang, J., Pei, X.-R., Zhang, Z.-F., Wang, N., Wang, J.-B., & Li, Y. (2012). A Chronic Oral Toxicity Study of Marine Collagen Peptides Preparation from Chum Salmon (Oncorhynchus keta) Skin Using Sprague-Dawley Rat. Marine Drugs, 10(1), 20-34. https://doi.org/10.3390/md10010020




