Synthesis and Preliminary Evaluation of a 2-Oxoquinoline Carboxylic Acid Derivative for PET Imaging the Cannabinoid Type 2 Receptor
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials and Methods
2.2. Chemistry
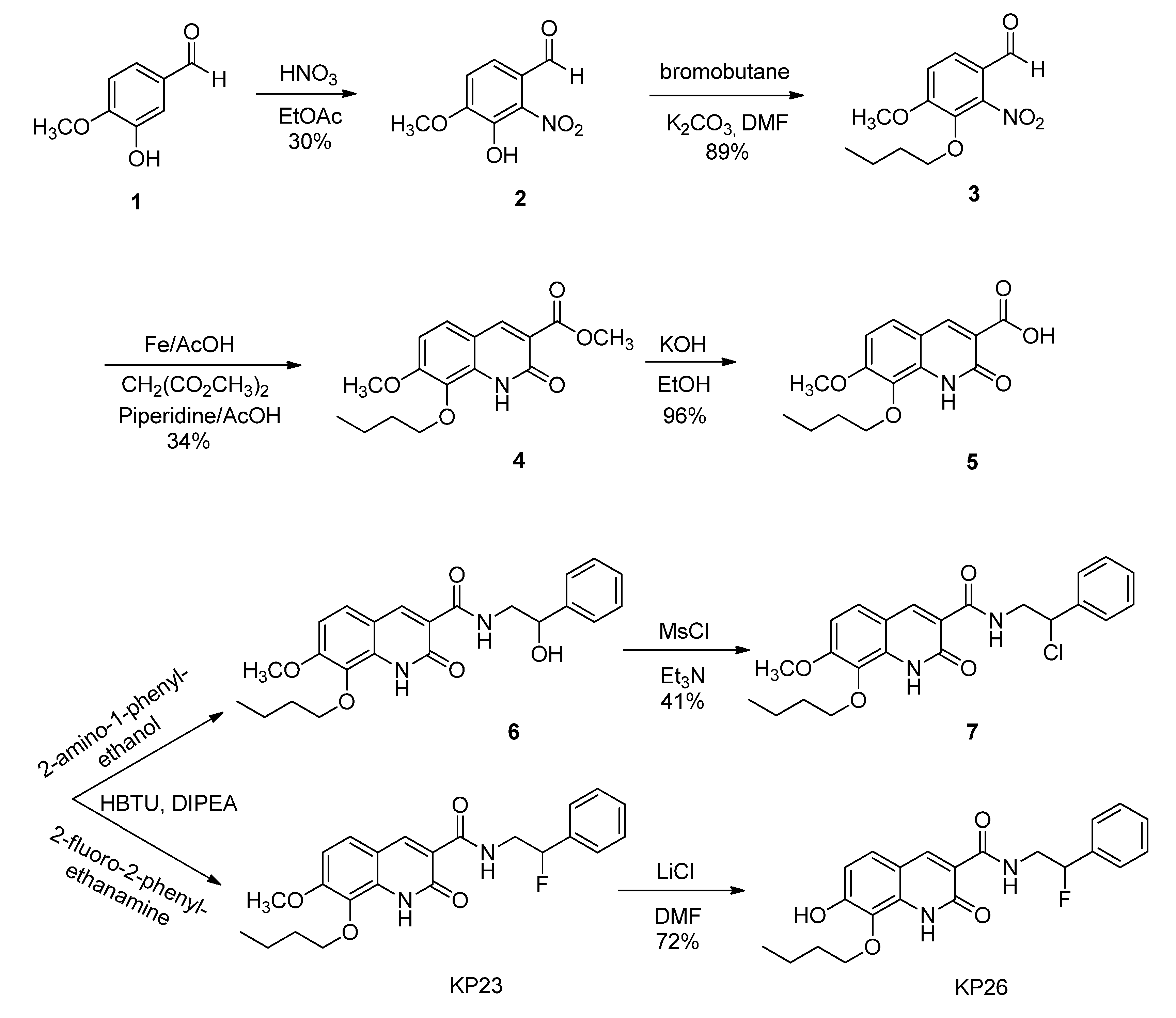
2.2.1. Synthesis of Compound 7 as Precursor for 18F-Radiolabeling
2.2.2. Synthesis of Compound KP26 as Precursor for 11C-Radiolabeling
2.3. Radiochemistry
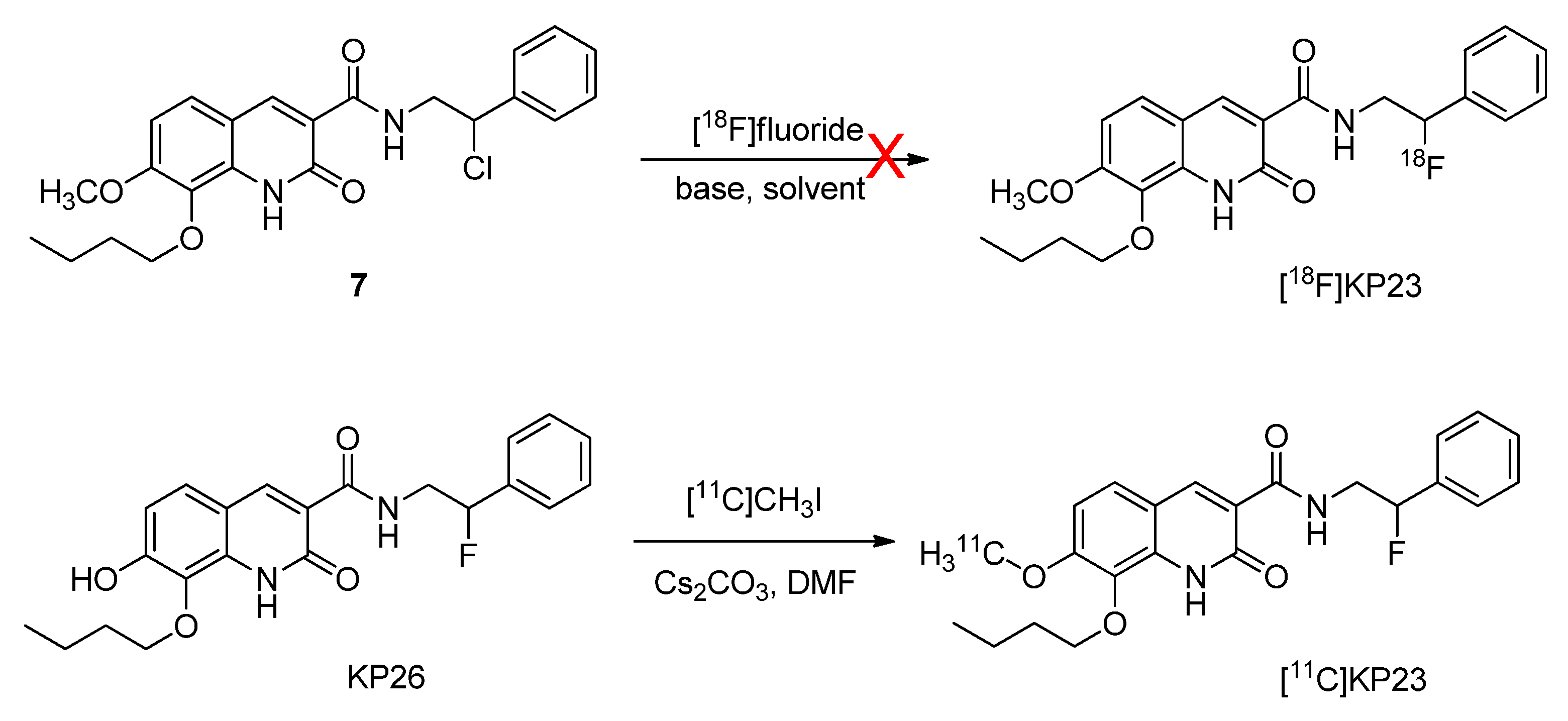
2.3.1. Radiosynthesis of [18F]KP23
2.3.2. Radiosynthesis of [11C]KP23
2.4. In Vitro Characterization
2.4.1. Competition Binding Assay
2.4.2. In Vitro Stability Studies
2.4.3. In Vitro Autoradiography
2.5. In Vivo Characterization
2.5.1. In Vivo PET Imaging
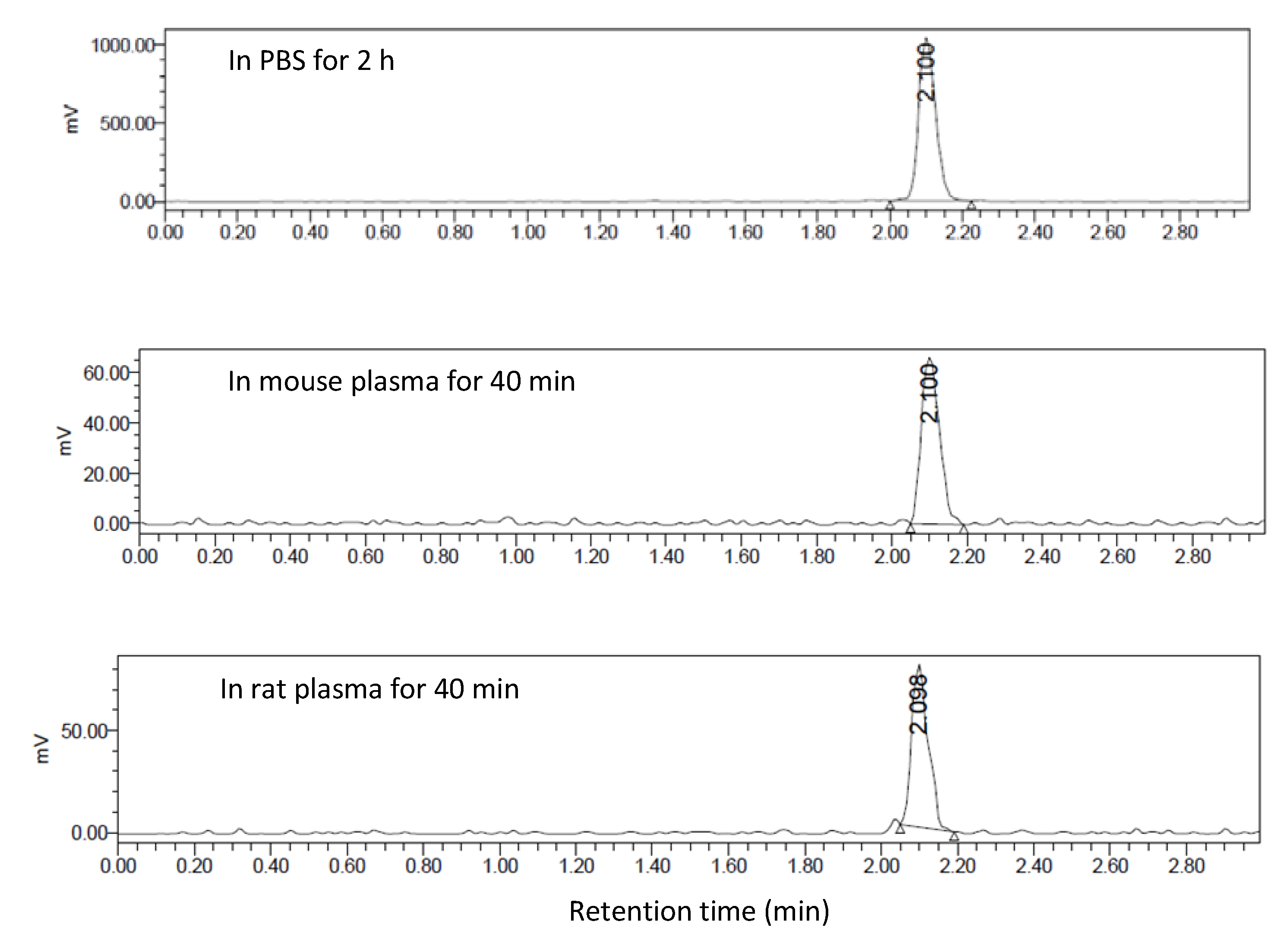
2.5.2. Metabolite Studies
3. Results and Discussion
3.1. Chemistry and Radiochemistry
3.2. In Vitro Characterization
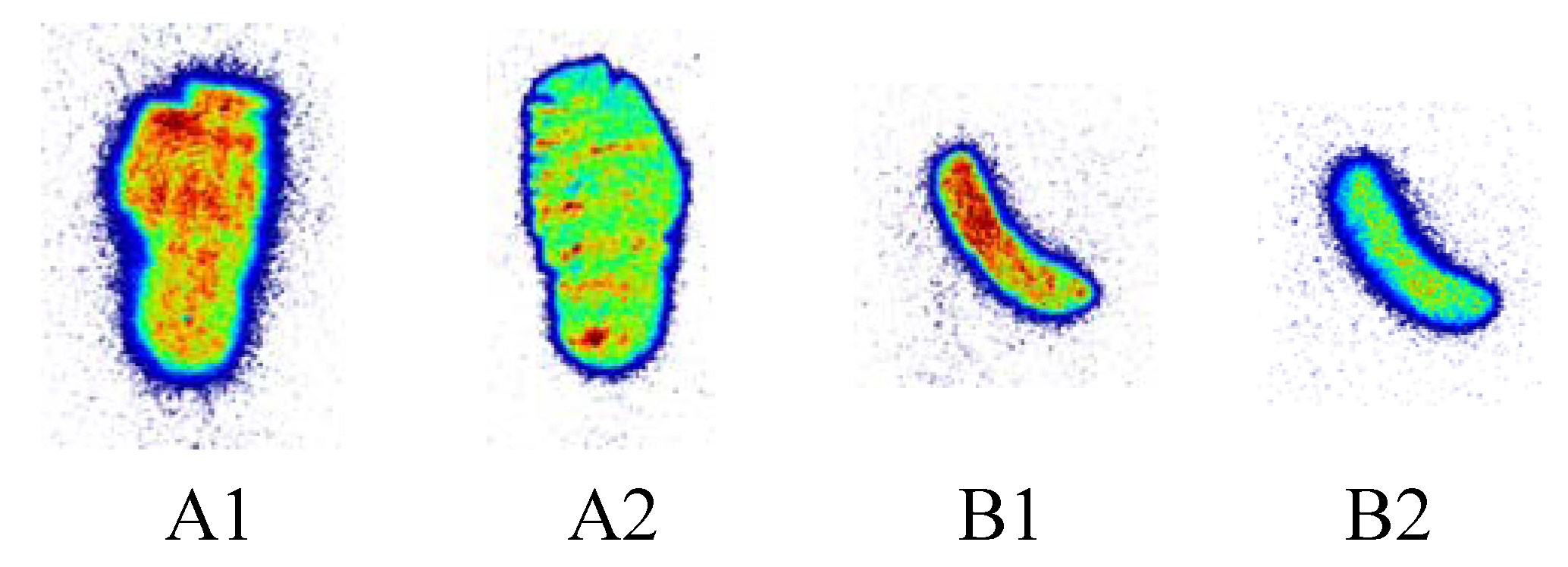
3.3. In Vivo PET Imaging of [11C]KP23
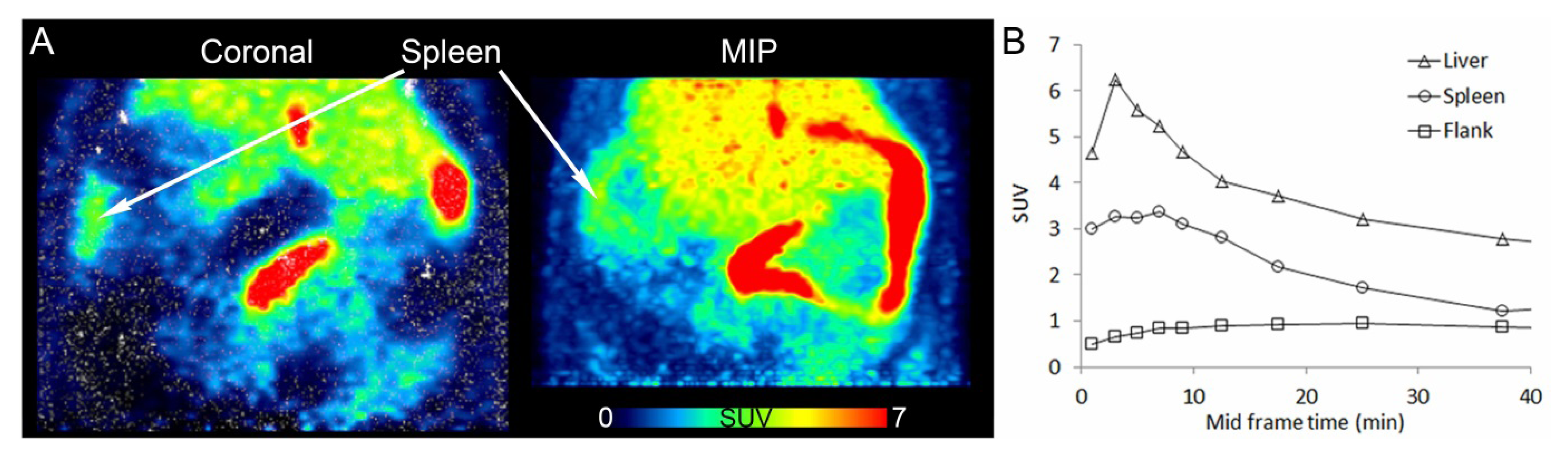
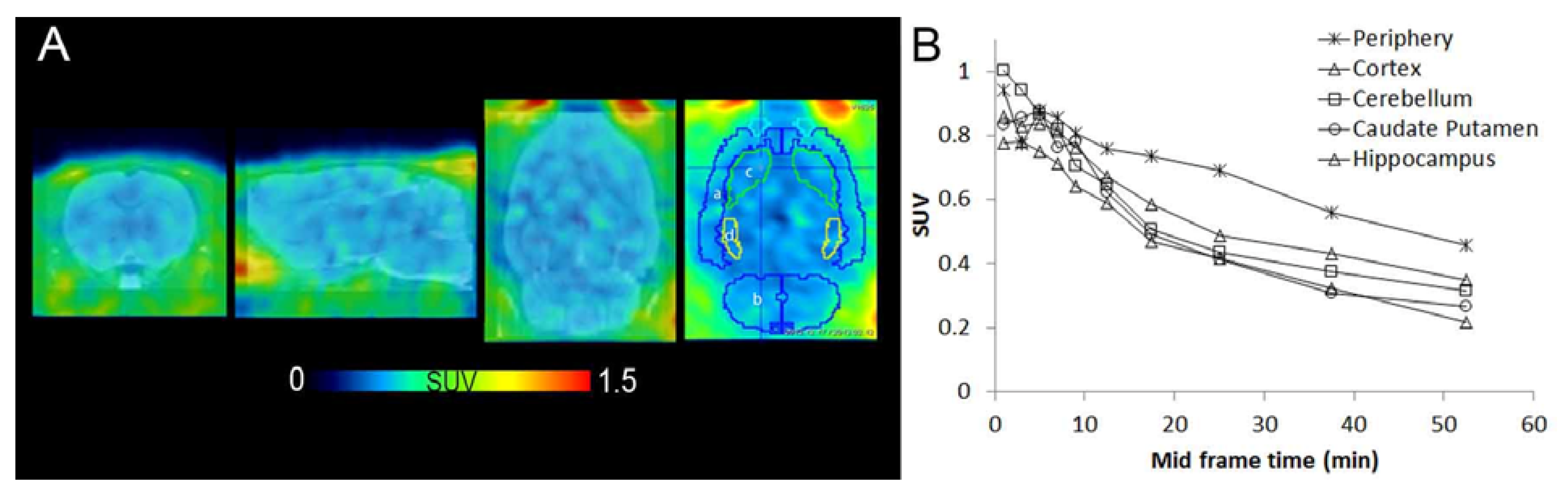
3.4. In Vivo Metabolic Studies
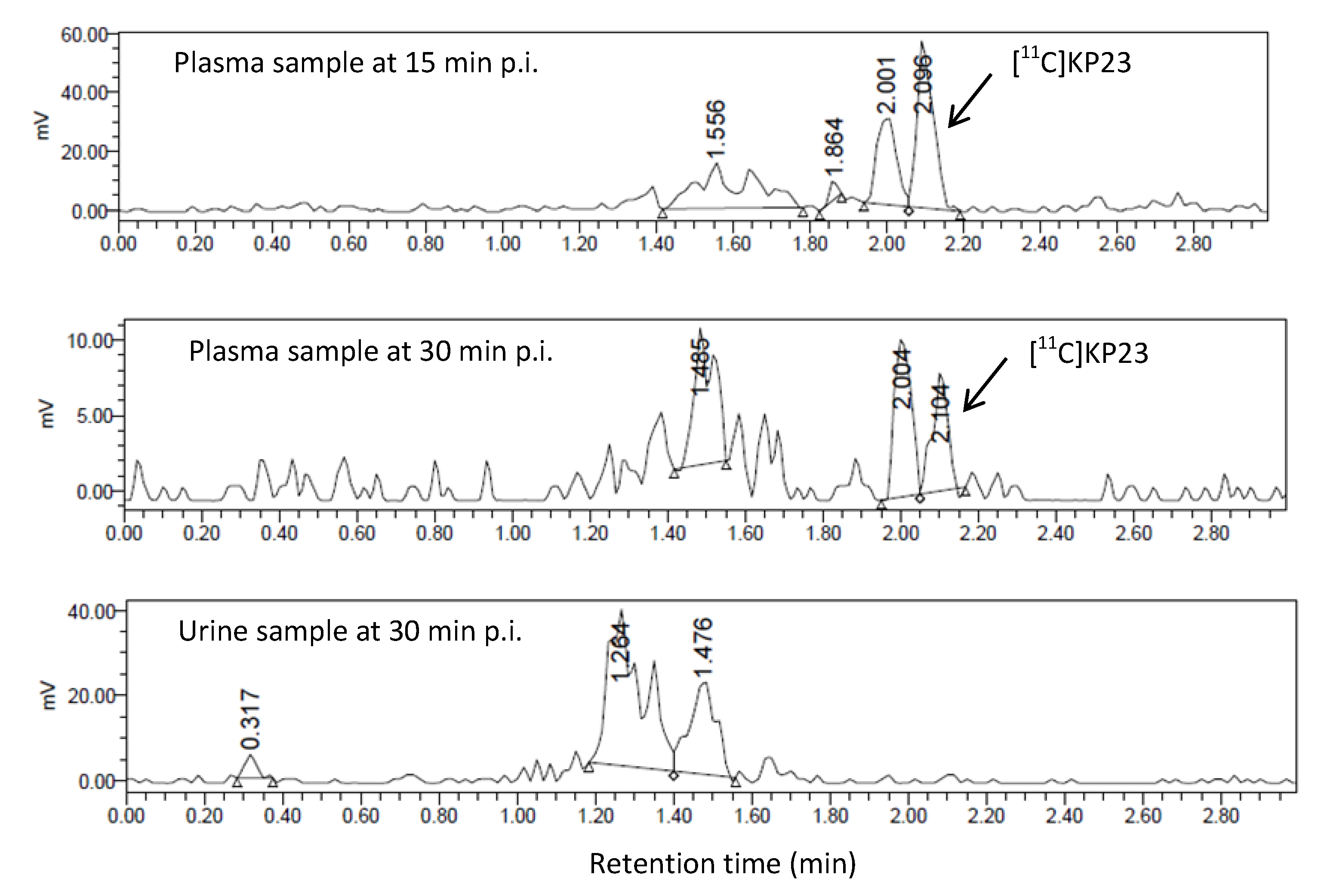
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Vincent, B.J.; McQuiston, D.J.; Einhorn, L.H.; Nagy, C.M.; Brames, M.J. Review of cannabinoids and their antiemetic effectiveness. Drugs 1983, 25 (Suppl. 1), 52–62. [Google Scholar] [CrossRef]
- Console-Bram, L.; Marcu, J.; Abood, M.E. Cannabinoid receptors: Nomenclature and pharmacological principles. Prog. Neuropsychopharmacol. Biol. Psychiatry 2012, 38, 4–15. [Google Scholar] [CrossRef]
- Pertwee, R.G.; Howlett, A.C.; Abood, M.E.; Alexander, S.P.; di Marzo, V.; Elphick, M.R.; Greasley, P.J.; Hansen, H.S.; Kunos, G.; Mackie, K.; et al. International Union of Basic and Clinical Pharmacology. LXXIX. Cannabinoid receptors and their ligands: Beyond CB(1) and CB(2). Pharmacol. Rev. 2010, 62, 588–631. [Google Scholar] [CrossRef]
- Matsuda, L.A.; Lolait, S.J.; Brownstein, M.J.; Young, A.C.; Bonner, T.I. Structure of a cannabinoid receptor and functional expression of the cloned cDNA. Nature 1990, 346, 561–564. [Google Scholar] [CrossRef]
- Herkenham, M.; Lynn, A.B.; Johnson, M.R.; Melvin, L.S.; de Costa, B.R.; Rice, K.C. Characterization and localization of cannabinoid receptors in rat brain: A quantitative in vitro autoradiographic study. J. Neurosci. 1991, 11, 563–583. [Google Scholar]
- Maresz, K.; Carrier, E.J.; Ponomarev, E.D.; Hillard, C.J.; Dittel, B.N. Modulation of the cannabinoid CB2 receptor in microglial cells in response to inflammatory stimuli. J. Neurochem. 2005, 95, 437–445. [Google Scholar] [CrossRef]
- Chin, C.L.; Tovcimak, A.E.; Hradil, V.P.; Seifert, T.R.; Hollingsworth, P.R.; Chandran, P.; Zhu, C.Z.; Gauvin, D.; Pai, M.; Wetter, J.; et al. Differential effects of cannabinoid receptor agonists on regional brain activity using pharmacological MRI. Br. J. Pharmacol. 2008, 153, 367–379. [Google Scholar] [CrossRef]
- Pertwee, R.G. Pharmacology of cannabinoid CB1 and CB2 receptors. Pharmacol. Ther. 1997, 74, 129–180. [Google Scholar]
- Burns, H.D.; van Laere, K.; Sanabria-Bohorquez, S.; Hamill, T.G.; Bormans, G.; Eng, W.S.; Gibson, R.; Ryan, C.; Connolly, B.; Patel, S.; et al. [18F]MK-9470, a positron emission tomography (PET) tracer for in vivo human PET brain imaging of the cannabinoid-1 receptor. Proc. Natl. Acad. Sci. USA 2007, 104, 9800–9805. [Google Scholar] [CrossRef]
- Liu, P.; Lin, L.S.; Hamill, T.G.; Jewell, J.P.; Lanza, T.J., Jr.; Gibson, R.E.; Krause, S.M.; Ryan, C.; Eng, W.; Sanabria, S.; et al. Discovery of N-{(1S,2S)-2-(3-cyanophenyl)-3-[4-(2-[18F]fluoroethoxy)phenyl]-1-methylpropyl}-2-methyl-2-[(5-methylpyridin-2-yl)oxy]propanamide, a cannabinoid-1 receptor positron emission tomography tracer suitable for clinical use. J. Med. Chem. 2007, 50, 3427–3430. [Google Scholar] [CrossRef]
- Horti, A.G.; van Laere, K. Development of radioligands for in vivo imaging of type 1 cannabinoid receptors (CB1) in human brain. Curr. Pharm. Des. 2008, 14, 3363–3383. [Google Scholar] [CrossRef]
- Van Laere, K.; Casteels, C.; Lunskens, S.; Goffin, K.; Grachev, I.D.; Bormans, G.; Vandenberghe, W. Regional changes in type 1 cannabinoid receptor availability in Parkinson's disease in vivo. Neurobiol. Aging 2012, 33, 620.e1–620.e8. [Google Scholar]
- Van Laere, K.; Goffin, K.; Casteels, C.; Dupont, P.; Mortelmans, L.; de Hoon, J.; Bormans, G. Gender-dependent increases with healthy aging of the human cerebral cannabinoid-type 1 receptor binding using [(18)F]MK-9470 PET. Neuroimage 2008, 39, 1533–1541. [Google Scholar] [CrossRef]
- Van Laere, K.; Koole, M.; Sanabria Bohorquez, S.M.; Goffin, K.; Guenther, I.; Belanger, M.J.; Cote, J.; Rothenberg, P.; de Lepeleire, I.; Grachev, I.D.; et al. Whole-body biodistribution and radiation dosimetry of the human cannabinoid type-1 receptor ligand 18F-MK-9470 in healthy subjects. J. Nucl. Med. 2008, 49, 439–445. [Google Scholar] [CrossRef]
- Muccioli, G.G.; Lambert, D.M. Current knowledge on the antagonists and inverse agonists of cannabinoid receptors. Curr. Med. Chem. 2005, 12, 1361–1394. [Google Scholar] [CrossRef]
- Raitio, K.H.; Salo, O.M.; Nevalainen, T.; Poso, A.; Jarvinen, T. Targeting the cannabinoid CB2 receptor: Mutations, modeling and development of CB2 selective ligands. Curr. Med. Chem. 2005, 12, 1217–1237. [Google Scholar] [CrossRef]
- Evens, N.; Muccioli, G.G.; Houbrechts, N.; Lambert, D.M.; Verbruggen, A.M.; Van Laere, K.; Bormans, G.M. Synthesis and biological evaluation of carbon-11- and fluorine-18-labeled 2-oxoquinoline derivatives for type 2 cannabinoid receptor positron emission tomography imaging. Nucl. Med. Biol. 2009, 36, 455–465. [Google Scholar] [CrossRef]
- Vandeputte, C.; Evens, N.; Toelen, J.; Deroose, C.M.; Bosier, B.; Ibrahimi, A.; van der Perren, A.; Gijsbers, R.; Janssen, P.; Lambert, D.M.; et al. A PET brain reporter gene system based on type 2 cannabinoid receptors. J. Nucl. Med. 2011, 52, 1102–1109. [Google Scholar] [CrossRef]
- Mu, L.; Bieri, D.; Slavik, R.; Drandarov, K.; Muller, A.; Cermak, S.; Weber, M.; Schibli, R.; Kramer, S.D.; Ametamey, S.M. Radiolabeling and in vitro/in vivo evaluation of N-(1-adamantyl)-8-methoxy-4-oxo-1-phenyl-1,4-dihydroquinoline-3-carboxamide as a PET probe for imaging cannabinoid type 2 receptor. J. Neurochem. 2013, 126, 616–624. [Google Scholar] [CrossRef]
- Evens, N.; Vandeputte, C.; Coolen, C.; Janssen, P.; Sciot, R.; Baekelandt, V.; Verbruggen, A.M.; Debyser, Z.; van Laere, K.; Bormans, G.M. Preclinical evaluation of [11C]NE40, a type 2 cannabinoid receptor PET tracer. Nucl. Med. Biol. 2012, 39, 389–399. [Google Scholar] [CrossRef]
- Turkman, N.; Shavrin, A.; Paolillo, V.; Yeh, H.H.; Flores, L.; Soghomonian, S.; Rabinovich, B.; Volgin, A.; Gelovani, J.; Alauddin, M. Synthesis and preliminary evaluation of [18F]-labeled 2-oxoquinoline derivatives for PET imaging of cannabinoid CB2 receptor. Nucl. Med. Biol. 2012, 39, 593–600. [Google Scholar] [CrossRef]
- Ahmad, R.; Koole, M.; Evens, N.; Serdons, K.; Verbruggen, A.; Bormans, G.; van Laere, K. Whole-body biodistribution and radiation dosimetry of the cannabinoid type 2 receptor ligand [11C]-NE40 in healthy subjects. Mol. Imaging Biol. 2013, 15, 384–390. [Google Scholar] [CrossRef]
- Showalter, V.M.; Compton, D.R.; Martin, B.R.; Abood, M.E. Evaluation of binding in a transfected cell line expressing a peripheral cannabinoid receptor (CB2): Identification of cannabinoid receptor subtype selective ligands. J. Pharmacol. Exp. Ther. 1996, 278, 989–999. [Google Scholar]
- Ruhl, T.; Deuther-Conrad, W.; Fischer, S.; Gunther, R.; Hennig, L.; Krautscheid, H.; Brust, P. Cannabinoid receptor type 2 (CB2)-selective N-aryl-oxadiazolyl-propionamides: Synthesis, radiolabelling, molecular modelling and biological evaluation. Org. Med. Chem. Lett. 2012, 2, 32. [Google Scholar] [CrossRef]
- Wang, Y.; Seidel, J.; Tsui, B.M.; Vaquero, J.J.; Pomper, M.G. Performance evaluation of the GE healthcare eXplore VISTA dual-ring small-animal PET scanner. J. Nucl. Med. 2006, 47, 1891–900. [Google Scholar]
- Honer, M.; Bruhlmeier, M.; Missimer, J.; Schubiger, A.P.; Ametamey, S.M. Dynamic imaging of striatal D2 receptors in mice using quad-HIDAC PET. J. Nucl. Med. 2004, 45, 464–470. [Google Scholar]
- Turkman, N.; Shavrin, A.; Ivanov, R.A.; Rabinovich, B.; Volgin, A.; Gelovani, J.G.; Alauddin, M.M. Fluorinated cannabinoid CB2 receptor ligands: Synthesis and in vitro binding characteristics of 2-oxoquinoline derivatives. Bioorg. Med. Chem. 2011, 19, 5698–5707. [Google Scholar] [CrossRef]
- Bacherikov, V.A.; Chou, T.C.; Dong, H.J.; Zhang, X.G.; Chen, C.H.; Lin, Y.W.; Tsai, T.J.; Lee, R.Z.; Liu, L.F.; Su, T.L. Potent antitumor 9-anilinoacridines bearing an alkylating N-mustard residue on the anilino ring: Synthesis and biological activity. Bioorg. Med. Chem. 2005, 13, 3993–4006. [Google Scholar] [CrossRef]
- Savle, P.S.; Medhekar, R.A.; Kelley, E.L.; May, J.G.; Watkins, S.F.; Fronczek, F.R.; Quinn, D.M.; Gandour, R.D. Change in the mode of inhibition of acetylcholinesterase by (4-nitrophenyl)sulfonoxyl derivatives of conformationally constrained choline analogues. Chem. Res. Toxicol. 1998, 11, 19–25. [Google Scholar] [CrossRef]
- Boeckman, R.K.; Miller, Y.; Savage, D.; Summerton, J.E. Total synthesis of a possible specific and effective acid-targeted cancer diagnostic, a camphor derived bis-N-oxide dimer. Tetrahedron Lett. 2011, 52, 2243–2245. [Google Scholar] [CrossRef]
- Gong, J.P.; Onaivi, E.S.; Ishiguro, H.; Liu, Q.R.; Tagliaferro, P.A.; Brusco, A.; Uhl, G.R. Cannabinoid CB2 receptors: Immunohistochemical localization in rat brain. Brain Res. 2006, 1071, 10–23. [Google Scholar]
- Evens, N.; Bormans, G.M. Non-invasive imaging of the type 2 cannabinoid receptor, focus on positron emission tomography. Curr. Top. Med. Chem. 2010, 10, 1527–1543. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Mu, L.; Slavik, R.; Müller, A.; Popaj, K.; Čermak, S.; Weber, M.; Schibli, R.; Krämer, S.D.; Ametamey, S.M. Synthesis and Preliminary Evaluation of a 2-Oxoquinoline Carboxylic Acid Derivative for PET Imaging the Cannabinoid Type 2 Receptor. Pharmaceuticals 2014, 7, 339-352. https://doi.org/10.3390/ph7030339
Mu L, Slavik R, Müller A, Popaj K, Čermak S, Weber M, Schibli R, Krämer SD, Ametamey SM. Synthesis and Preliminary Evaluation of a 2-Oxoquinoline Carboxylic Acid Derivative for PET Imaging the Cannabinoid Type 2 Receptor. Pharmaceuticals. 2014; 7(3):339-352. https://doi.org/10.3390/ph7030339
Chicago/Turabian StyleMu, Linjing, Roger Slavik, Adrienne Müller, Kasim Popaj, Stjepko Čermak, Markus Weber, Roger Schibli, Stefanie D. Krämer, and Simon M. Ametamey. 2014. "Synthesis and Preliminary Evaluation of a 2-Oxoquinoline Carboxylic Acid Derivative for PET Imaging the Cannabinoid Type 2 Receptor" Pharmaceuticals 7, no. 3: 339-352. https://doi.org/10.3390/ph7030339
APA StyleMu, L., Slavik, R., Müller, A., Popaj, K., Čermak, S., Weber, M., Schibli, R., Krämer, S. D., & Ametamey, S. M. (2014). Synthesis and Preliminary Evaluation of a 2-Oxoquinoline Carboxylic Acid Derivative for PET Imaging the Cannabinoid Type 2 Receptor. Pharmaceuticals, 7(3), 339-352. https://doi.org/10.3390/ph7030339




