The Use of California Sagebrush (Artemisia californica) Liniment to Control Pain
Abstract
:1. Introduction
2. A. californica Liniment
3. Chemistry of A. californica
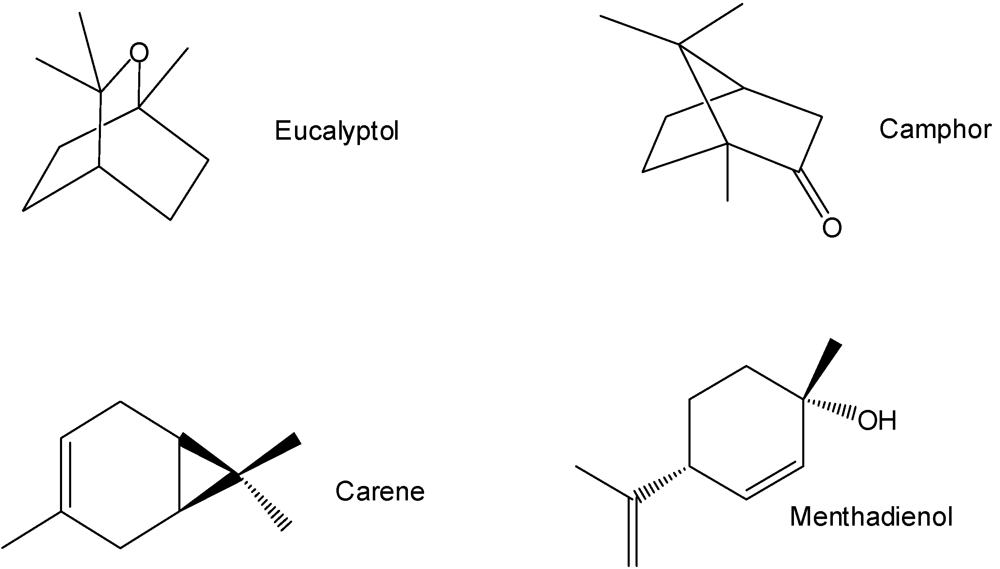
4. Pharmacology of the Monoterpenoids from A. californica
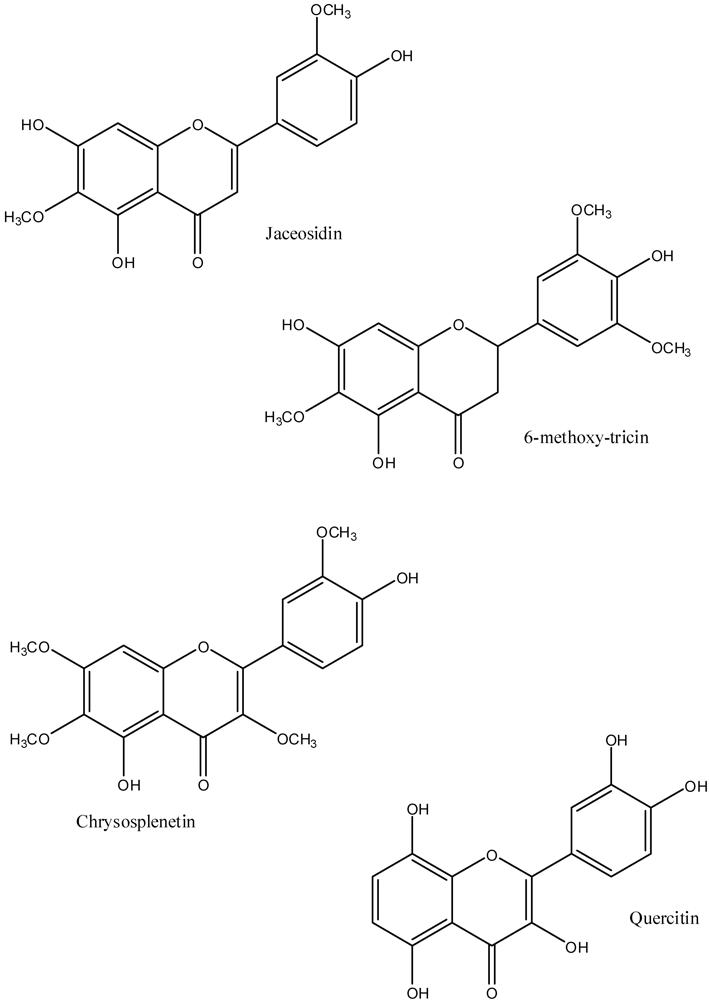
5. Pharmacology of the Flavonoids from A. californica
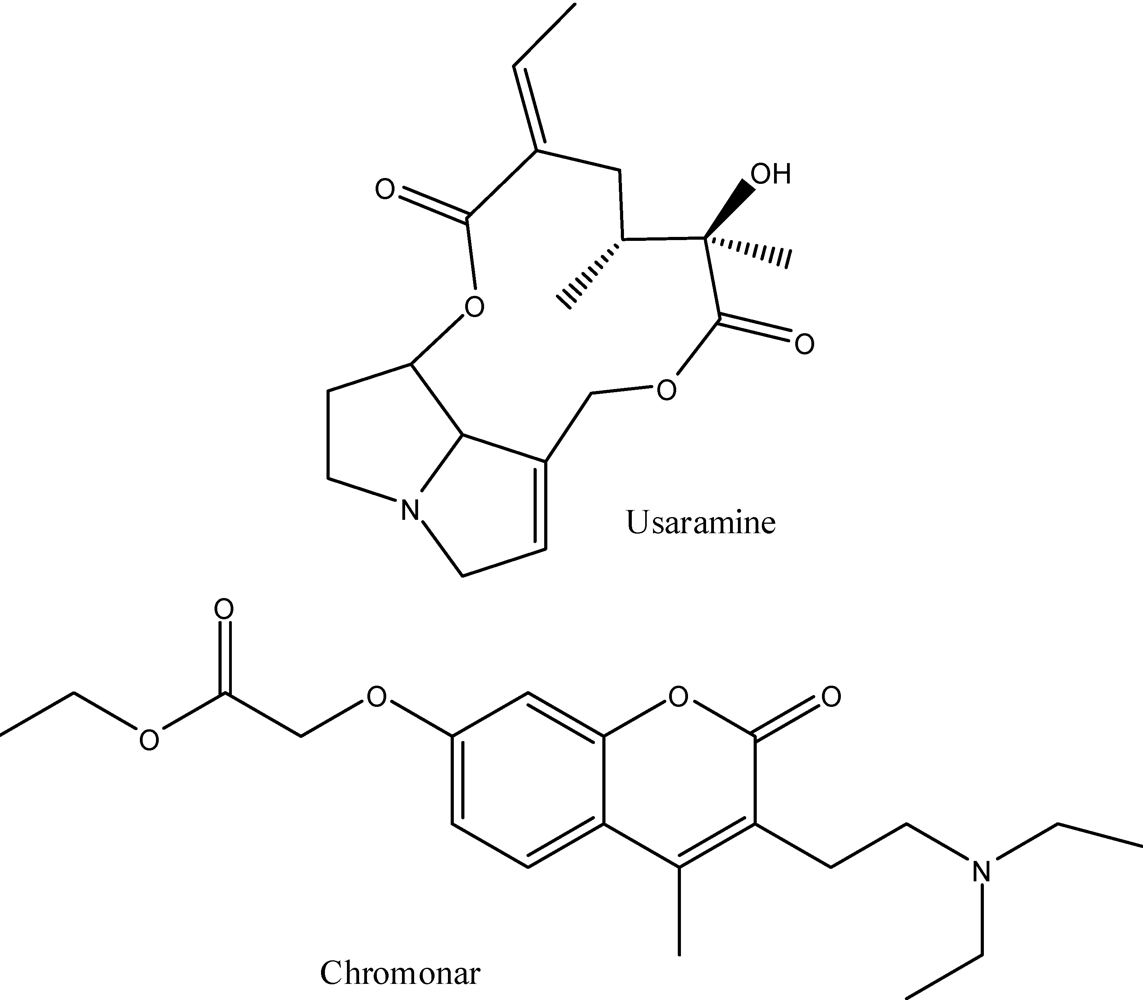
6. Pharmacology of the Alkaloids from A. californica
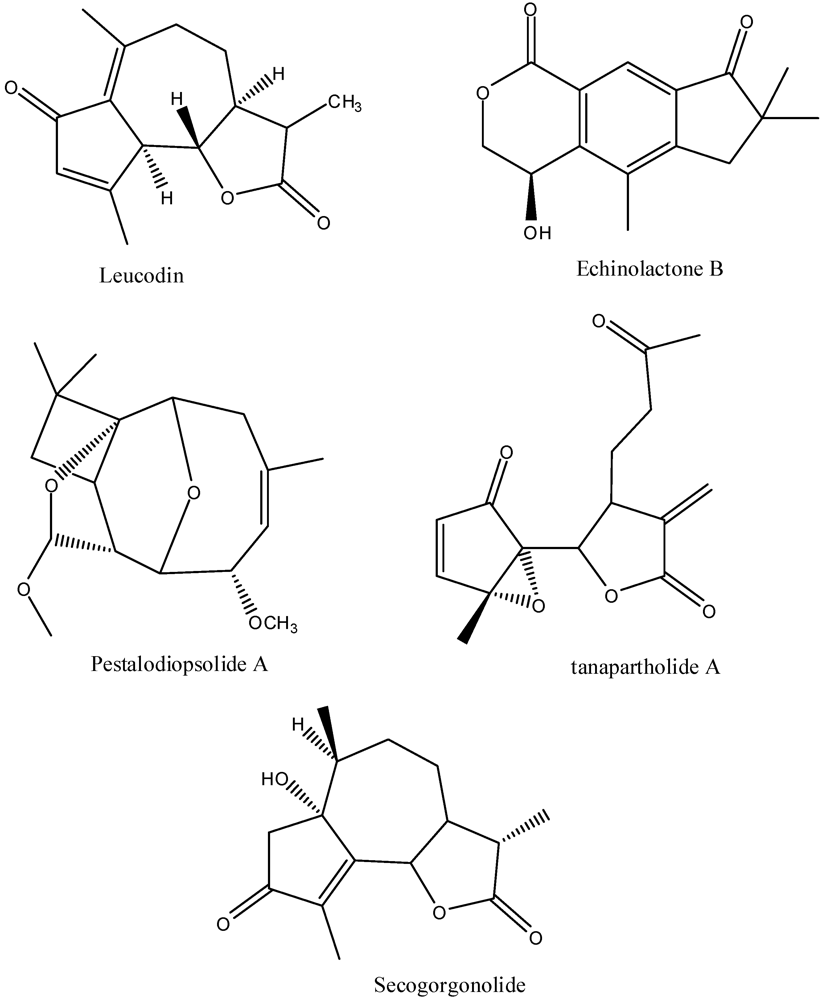
7. Pharmacology of the Sesquiterpenes from A. californica
8. Pharmacology of the Other Compounds from A. californica
9. Conclusions
References
- Adams, J.D.; Parker, K. Extracellular and Intracellular Signaling; Royal Society of Chemistry: London, UK, 2011. [Google Scholar]
- Garcia, C.; Adams, J.D. Healing with Medicinal Plants of the West—Cultural and Scientific Basis for Their Use, 2nd ed; Abedus Press: La Crescenta, CA, USA, 2009. [Google Scholar]
- Fontaine, P.; Wong, V.; Williams, T.; Garcia, C.; Adams, J.D. Chemical Composition and Antinociceptive Activity of California Sagebrush (Artemisia californica). J. Pharmacogn. Phytother. 2012. submitted. [Google Scholar]
- Moran, M.; McAlexander, M.; Biro, T.; Szallasi, A. Transient receptor potential channels as therapeutic targets. Nat. Rev. 2011, 10, 601–620. [Google Scholar] [CrossRef]
- Meyr, A.; Steinberg, J. The physiology of the acute pain pathway. Clin. Podiatr. Med. Surg. 2008, 25, 305–326. [Google Scholar] [CrossRef]
- Xu, H.; Blair, N.T.; Clapham, D.E. Camphor activates and strongly desensitizes the transient receptor potential vanilloid subtype 1 channel in vanilloid-independent mechanism. J. Neurosci. 2005, 25, 8924–8937. [Google Scholar] [CrossRef]
- Martinez, A.L.; Gonzalez-Trujano, M.E.; Aguirre-Hernandez, E.; Moreno, J.; Soto-Hernandez, M.; Lopez-Munoz, F.J. Antinociceptive activity of Tilia americana var. mexicana inflorescences and quercetin in the formalin test and in an arthritic pain model in rats. Neuropharmacology 2009, 56, 564–571. [Google Scholar] [CrossRef]
- Martinez, A.L.; Gonzalez-Trujano, M.E.; Pellicer, F.; Lopez-Munoz, F.J.; Navarette, A. Antinociceptive effect and GC/MS analysis of Rosmarinus officinalis L. essential oil from its aerial parts. Planta Med. 2009, 75, 508–511. [Google Scholar] [CrossRef]
- Liapi, C.; Anifandis, G.; Chinou, I.; Kourounakis, A.P.; Theodosopoulos, S.; Galanopoulou, P. Antinociceptive properties of 1,8-cineole and beta-pinene, from the essential oil of Eucalyptus camaldulensis leaves, in rodents. Planta Med. 2007, 73, 1247–1254. [Google Scholar] [CrossRef]
- Granger, R.E.; Campbell, E.L.; Johnston, G.A. (+) and (−)-borneol: Efficacious positive modulators of GABA action at human recombinant alpha1beta2gamma2L GABA (A) receptors. Biochem. Pharmacol. 2005, 69, 1101–1111. [Google Scholar]
- Hold, K.M.; Sirisoma, N.S.; Ikeda, T.; Narahashi, T.; Casida, J.E. Alpha-thujone (the active component of absinthe): Gamma-Aminobutyric acid type A receptor modulation and metabolic detoxification. Proc. Nat. Acad. Sci. USA 2000, 97, 4417–4418. [Google Scholar]
- Schmitt, S.; Schaefer, U.F.; Doebler, L.; Reichling, J. Cooperative interaction of monoterpenes and phenylpropanoids on the in vitro human skin permeation of complex composed essential oils. Planta Med. 2009, 75, 1381–1385. [Google Scholar] [CrossRef]
- Vriens, J.; Appendino, G.; Nilius, B. Pharmacology of Vanilloid transient receptor potential cation channels. Mol. Pharmacol. 2009, 75, 1262–1279. [Google Scholar] [CrossRef]
- Caterina, M.J.; Schumacher, M.A.; Tominaga, M.; Rosen, T.A.; Levine, J.D.; Julius, D. The capsaicin receptor: A heat-activated ion channel in the pain pathway. Nature 1997, 389, 816–824. [Google Scholar]
- Basbaum, A.I.; Bautista, D.M.; Scherrer, G.; Julius, D. Cellular and molecular mechanisms of pain. Cell 2009, 139, 267–284. [Google Scholar] [CrossRef]
- Vogt-Eisele, A.K.; Weber, K.; Sherkheli, M.A.; Vielhaber, G.; Panten, J.; Gisselmann, G.; Hatt, H. Monoterpenoid agonists of TRPV3. Br. J. Pharmacol. 2007, 151, 530–540. [Google Scholar]
- Ishida, T. Biotransformation of terpenoids by mammals, microorganisms, and plant-cultured cells. Chem. Biodivers. 2005, 2, 569–590. [Google Scholar] [CrossRef]
- Lin, C.T.; Chen, C.J.; Lin, T.Y.; Tung, J.C.; Wang, S.Y. Anti-inflammation activity of fruit essential oil from Cinnamomum insularimontanum Hayata. Biores. Technol. 2008, 99, 8783–8787. [Google Scholar] [CrossRef]
- Tung, Y.T.; Chua, M.T.; Wang, S.Y.; Chang, S.T. Anti-inflammation activities of essential oil and its constituents from indigenous cinnamon (Cinnamomum osmophloeum) twigs. Biores. Technol. 2008, 99, 3908–3913. [Google Scholar]
- Farhat, G.N.; Affara, N.I.; Gali-Muhtasib, H.U. Seasonal changes in the composition of the essential oil extract of East Mediterranean sage (Salvia libanotica) and its toxicity in mice. Toxicon 2001, 39, 1601–1605. [Google Scholar] [CrossRef]
- Manoguerra, A.S.; Erdman, A.R.; Wax, P.M.; Nelson, L.S.; Caravati, E.M.; Cobaugh, D.J.; Chyka, P.A.; Olson, K.R.; Booze, L.L.; Woolf, A.D.; et al. Camphor poisoning: An evidence-based practice guideline for out-of-hospital management. Clin. Toxicol. 2006, 44, 357–370. [Google Scholar] [CrossRef]
- Hall, A.C.; Turcotte, C.M.; Betts, B.A.; Yeung, W.Y.; Agyeman, A.S.; Burk, L.A. Modulation of human GABAA and glycine receptor currents by menthol and related monoterpenoids. Eur. J. Pharmacol. 2004, 506, 9–16. [Google Scholar] [CrossRef]
- Sayyah, M.; Valizadeh, J.; Kamalinejad, M. Anticonvulsivant activity of the leaf essential oil of Laurus nobilis against pentylenetetrazole- and maximal electroshock-induced seizures. Phytomedicine 2002, 9, 212–216. [Google Scholar] [CrossRef]
- Yin, Y.; Gong, F.Y.; Wu, X.X.; Sun, Y.; Li, Y.H.; Chen, T.; Xu, Q. Anti-inflammatory and immunosuppressive effect of flavones isolated from Artemisia vestita. J. Ethnopharmacol. 2008, 120, 1–6. [Google Scholar] [CrossRef]
- Sheu, M.J.; Chou, P.Y.; Cheng, H.C.; Wu, C.H.; Huang, G.J.; Wang, B.S.; Chen, J.S.; Chien, Y.C.; Huang, M.H. Analgesic and anti-inflammatory activities of a water extract of Trachelospermum jasminoides (Apocynaceae). J. Ethnopharmacol. 2009, 126, 332–338. [Google Scholar] [CrossRef]
- Clavin, M.; Gorzalczany, S.; Macho, A.; Munoz, E.; Ferraro, G.; Acevedo, C.; Martino, V. Anti-inflammatory activity of flavonoids from Eupatorium arnottianum. J. Ethnopharmacol. 2007, 112, 585–589. [Google Scholar] [CrossRef]
- Wiedenfeld, H.; Edgar, J. Toxicity of pyrrolizidine alkaloids to humans and ruminants. Phytochem. Rev. 2011, 10, 137–151. [Google Scholar] [CrossRef]
- Adams, J.; Garcia, C.; Garg, G. Mugwort (Artemisia vulgaris, Artemisia douglasiana, Artemisia argyi) in the treatment of menopause, premenstrual syndrome, dysmenorrhea and attention deficit hyperactivity disorder. Chin. Med. 2012, in press. [Google Scholar]
- Galvis, A.; Marcano, A.; Stefancin, C.; Villaverde, N.; Priestap, H.; Tonn, C.; Lopez, L.; Barbieri, M. The effect of dehydroleucodine in adipocyte differentiation. Eur. J. Pharmacol. 2011, 671, 18–25. [Google Scholar] [CrossRef]
- Guardia, T.; Juarez, A.; Guerreiro, E.; Guzman, J.; Pelzer, L. Anti-inflammatory activity and effect on gastric acid secretion of dehydroleucodine isolated from Artemisia douglasiana. J. Ethnopharmacol. 2003, 88, 195–199. [Google Scholar] [CrossRef]
- Makiyi, E.; Frade, R.; Lebl, T.; Jaffray, E.; Cobb, S.; Harvey, A.; Slawin, A.; Hay, R.; Westwood, N. Iso-seco-tanapartholides: Isolation, synthesis and biological evaluation. Eur. J. Org. Chem. 2009, 33, 5711–5715. [Google Scholar]
- Takase, H.; Yamamoto, K.; Hirano, H.; Saito, Y.; Yamashita, A. Pharmacological profile of gastric mucosal protection by marmin and nobiletin from a traditional herbal medicine, Aurantii fructus immaturus. Jpn. J. Pharmacol. 1994, 66, 139–147. [Google Scholar] [CrossRef]
- Gerhauser, C.; Alt, A.; Heiss, E.; Gamal-Eldeen, A.; Klimo, K.; Knauft, J.; Neumann, I.; Scherf, H.; Frank, N.; Bartsch, H.; et al. Cancer chemopreventive activity of Xanthohumol, a natural product derived from hop. Mol. Cancer Ther. 2002, 1, 959–969. [Google Scholar]
- Benelli, R.; Vene, R.; Ciarlo, M.; Carlone, S.; Barbieri, O.; Ferrari, N. The AKT/NF-κB inhibitor xanthohumol is a potent anti-lymphocytic leukemia drug overcoming chemoresistance and cell infiltration. Biochem. Pharmacol. 2012, 83, 1634–1642. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Adams, J.D., Jr. The Use of California Sagebrush (Artemisia californica) Liniment to Control Pain. Pharmaceuticals 2012, 5, 1045-1053. https://doi.org/10.3390/ph5101045
Adams JD Jr. The Use of California Sagebrush (Artemisia californica) Liniment to Control Pain. Pharmaceuticals. 2012; 5(10):1045-1053. https://doi.org/10.3390/ph5101045
Chicago/Turabian StyleAdams, James D., Jr. 2012. "The Use of California Sagebrush (Artemisia californica) Liniment to Control Pain" Pharmaceuticals 5, no. 10: 1045-1053. https://doi.org/10.3390/ph5101045
APA StyleAdams, J. D., Jr. (2012). The Use of California Sagebrush (Artemisia californica) Liniment to Control Pain. Pharmaceuticals, 5(10), 1045-1053. https://doi.org/10.3390/ph5101045




