3. Experimental Section
3.1. General
Melting points were recorded on a Thermocouple digital melting point apparatus (Mettler Toledo LLC, Columbus, OH, USA). IR spectra were recorded as powders using a Bruker VERTEX 70 FT-IR Spectrometer (Bruker Optics, Billerica, MA, USA) with a diamond ATR (attenuated total reflectance) accessory by using the thin-film method. For column chromatography, Merck kieselgel 60 (0.063–0.200 mm) (Merck KGaA, Frankfurt, Germany) was used as stationary phase. NMR spectra were obtained as CDCl3 solutions using Agilent 500 MHz NMR (Agilent Technologies, Oxford, UK) spectrometer and the chemical shifts are quoted relative to the TMS peak. Low- and high-resolution mass spectra were recorded at an ionisation potential of 70 eV using Waters Synapt G2 Quadrupole Time-of-flight mass spectrometer (Waters Corp., Milford, MA, USA) at the University of Stellenbosch Central Analytical Facility.
3.2. Typical Procedure for the Preparation of Compounds 1a and 1b
A stirred mixture of 2-amino-5-bromobenzamide (1.0 equiv.), aldehyde (1.5 equiv.) and iodine (2.0 equiv.) in ethanol (6.0 mL/mmol of benzamide) in a round-bottomed flask equipped with a stirrer bar and a condenser was refluxed for 8 h and then allowed to cool to room temperature. An ice-cold saturated aqueous solution of sodium thiosulphate was added to the mixture and the resultant precipitate was filtered and washed with cold water. The product was recrystallized from acetonitrile. Compounds 1a and 1b were prepared in this fashion.
6-Bromoquinazolin-4(3H)-one (
1a). A mixture of 2-amino-5-bromobenzamide (1.00 g, 4.65 mmol), paraformaldehyde (0.21 g, 6.97 mmol) and iodine (2.36 g, 9.30 mmol) in ethanol (30 mL) afforded
1a as a white solid (0.95 g, 91%), mp. 278–280 °C (lit. [
21] 273–275 °C); ν
max (ATR) 493, 563, 622, 824, 924, 1237, 1271, 1316, 1387, 1462, 1603, 1688, 2809, 3445 cm
−1; δ
H (500 MHz, DMSO-
d6) 7.59 (d,
J = 8.5 Hz, 1H), 7.93 (dd,
J = 2.5 and 8.5 Hz, 1H), 8.12 (s, 1H), 8.16 (d,
J = 2.0 Hz, 1H), 12.40 (br s, 1H).
6-Bromo-2-(4-chlorophenyl)quinazolin-4(3H)-one (1b). A mixture of 2-amino-5-bromobenzamide (1.00 g, 4.65 mmol), 4-chlorobenzaldehyde (0.97 g, 6.97 mmol) and iodine (2.36 g, 9.30 mmol) in ethanol (30 mL) afforded 1b as a white solid (1.45 g, 93%), mp. 342–344 °C; νmax (ATR) 538, 559, 649, 728, 831, 940, 1091, 1276, 1413, 1460, 1556, 1601, 1672, 3442 cm−1; δH (500 MHz, DMSO-d6) 7.60 (d, J = 8.5 Hz, 2H), 7.66 (d, J = 8.5 Hz, 1H), 7.95 (dd, J = 2.5 and 8.5 Hz, 1H), 8.16 (d, J = 8.5 Hz, 2H), 8.19 (d, J = 2.0 Hz, 1H), 12.76 (brs, 1H); δC (125 MHz, DMSO-d6) 104.7, 112.5, 112.9, 119.9, 128.4, 129.2, 129.9, 130.2, 138.0, 139.1, 155.3, 159.3; m/z 335 (100, M + H); HRMS (ES): found 334.9578. C14H979Br35ClN2O+ requires 334.9587.
3.3. Typical Procedure for the Chlorination–Dehydration of 1a and 1b to Afford 2a and 2b, Respectively
Synthesis of 6-Bromo-4-chloroquinazoline (
2a). Triethylamine (4 mL) was added dropwise at room temperature to a stirred mixture of
1a (0.50 g, 2.22 mmol) and phosphoryl chloride (10 mL) in a round-bottomed flask. The reaction mixture was stirred under reflux for 5 h and then allowed to cool to room temperature. Ice-cold water was added to the mixture and the product was extracted with chloroform. The combined organic layers were washed with an aqueous solution of NaHCO
3, dried over Na
2SO
4, filtered, and evaporated under reduced pressure. The crude product was recrystallized from ethanol to afford
2a as a white solid (0.46 g, 85%), mp. 164–166 °C (lit. [
20] 161–163 °C); ν
max (ATR) 499, 539, 850, 1021, 1169, 1271, 1352, 1389, 1468, 1474, 1560, 1630, 2932 cm
−1; δ
H (500 MHz, DMSO-
d6) 7.95 (d,
J = 8.5 Hz, 1H), 8.06 (dd,
J = 2.5 and 8.5 Hz, 1H), 8.44 (d,
J = 2.0 Hz, 1H), 9.03 (s, 1H).
Synthesis of 6-Bromo-4-chloro-3-(4-chlorophenyl)quinazoline (2b). A stirred mixture of 1b (0.50 g, 1.49 mmol), trimethylamine (4 mL), and phosphoryl chloride (10 mL) afforded 2b as a yellow solid (0.48 g, 91%), mp. 244–246 °C; νmax (ATR) 505, 531, 732, 829, 991, 1090, 1294, 1318, 1402, 1415, 1469, 1493, 1539, 1556, 1592, 1633 cm−1; δH (500 MHz, DMSO-d6) 7.48 (d, J = 8.5 Hz, 2H), 7.93 (d, J = 8.0 Hz, 1H), 7.99 (dd, J = 2.5 and 8.5 Hz, 1H), 8.40 (d, J = 2.0 Hz, 1H), 8.51 (d, J = 8.5 Hz, 2H); δC (125 MHz, DMSO-d6) 104.8, 112.5, 112.9, 128.5, 129.2, 129.9, 130.2, 138.0, 139.2, 149.1, 152.0 159.3; m/z (100, M + H); HRMS (ES): found 334.9571. C14H979Br35Cl2+ requires 334.9587.
3.4. Typical Procedure for the Amination of 2a and 2b to Afford Compounds 3a–f and 3g–l, Respectively
6-Bromo-N-(2-fluorophenyl)quinazolin-4-amine (3a). A mixture of 2a (0.50 g, 2.05 mmol), 2-fluoroaniline (0.25 g, 2.25 mmol) and concentrated HCl (0.01 g, 0.27 mmol) in 3:1 THF-isopropanol (v/v, 10 mL) in a round-bottomed flask was stirred under reflux for 5 h. The reaction mixture was then allowed to cool to room temperature and quenched with ice-cold water. The product was extracted with ethyl acetate and the combined organic layers were washed thoroughly with an aqueous solution of NaHCO3, dried over anhydrous MgSO4, filtered, and evaporated under reduced pressure to afford 3a as a white solid (0.52 g, 80%), mp. 174–176 °C (acetonitrile); νmax (ATR) 493, 614, 757, 832, 926, 1188, 1253, 1404, 1454, 1563, 1619, 2849, 2916, 3259 cm−1; δH (500 MHz, DMSO-d6) 7.23‒7.32 (m, 3H), 7.52 (t, J = 8.0 Hz, 1H), 7.72 (d, J = 8.5 Hz, 1H), 7.97 (dd, J = 2.0 and 9.0 Hz, 1H), 8.49 (s, 1H), 8.76 (d, J = 2.0 Hz, 1H), 9.91 (s, 1H); δC (125 MHz, DMSO-d6) 116.5 (d, 2JCF = 20.0 Hz), 119.2, 124.9 (d, 4JCF = 2.8 Hz), 126.0, 126.3, 126.5 (d, 3JCF = 12.3 Hz), 126.5, 128.0 (d, 3JCF = 6.6 Hz), 128.8, 130.5, 136.6, 148.9, 155.5, 157.2 (d, 1JCF = 242.7 Hz); m/z 318 (100, M+H); HRMS (ES): found 318.0044. C14H10BrFN3+ requires 318.0042.
6-Bromo-N-(3-fluorophenyl)quinazolin-4-amine (3b). A mixture of 2a (0.50 g, 2.05 mmol), 3-fluoroaniline (0.25 g, 2.25 mmol) and concentrated HCl (0.01 g, 0.27 mmol) in THF-isopropanol (10 mL) afforded 3b as a white solid (0.49 g, 76%), mp. 214–216 °C (acetonitrile); νmax (ATR) 494, 684, 778, 832, 843, 958, 1145, 1403, 1485, 1531, 1562, 1614, 3074, 3263 cm−1; δH (500 MHz, DMSO-d6) 6.94 (td, J = 2.0 and 8.5 Hz, 1H), 7.41 (q, J = 8.0 Hz, 1H), 7.66 (d, J = 8.0 Hz, 1H), 7.74 (d, J = 8.0 Hz, 1H), 7.92 (dt, J = 2.0 and 11.5 Hz, 1H), 7.99 (dd, J = 2.5 and 8.5 Hz, 1H), 8.67 (s, 1H), 8.86 (d, J = 2.0 Hz, 1H), 9.94 (s, 1H); δC (125 MHz, DMSO-d6) 109.1 (d, 2JCF = 25.5 Hz), 110.5 (d, 2JCF = 20.8 Hz), 116.9, 118.0 (d, 4JCF = 2.8 Hz), 119.4, 125.8, 130.4 (d, 3JCF = 9.5 Hz), 130.6, 136.6, 141.2 (d, 3JCF = 11.3 Hz), 149.0, 155.1, 157.1, 162.4 (d, 1JCF = 239.8 Hz); m/z 318 (100, M + H); HRMS (ES): found 318.0043. C14H10BrFN3+ requires 318.0042.
6-Bromo-N-(4-fluorophenyl)quinazolin-4-amine (3c). A mixture of 2a (0.50 g, 2.05 mmol), 4-fluoroaniline (0.25 g, 2.25 mmol) and concentrated HCl (0.01 g, 0.27 mmol) in THF-isopropanol (10 mL) afforded 3c as a white solid (0.55 g, 85%), mp. 217–218 °C (acetonitrile); νmax (ATR) 497, 513, 830, 922, 1156, 1213, 1318, 1420, 1489, 1508, 1564, 1619, 2925, 3059 cm−1; δH (500 MHz, DMSO-d6) 7.23 (t, J = 8.5 Hz, 2H), 7.70 (d, J = 9.0 Hz, 1H), 7.82 (t, J = 8.5 Hz, 2H), 7.96 (dd, J = 2.5 and 9.0 Hz, 1H), 8.57 (s, 1H), 8.81 (d, J = 2.0 Hz, 1H), 9.87 (s, 1H); δC (125 MHz, DMSO-d6) 115.5 (d, 2JCF = 22.7 Hz), 116.8, 119.2, 124.8 (d, 3JCF = 7.6 Hz), 125.8, 130.5, 135.6 (d, 4JCF = 2.8 Hz), 136.5, 149.0, 155.3, 157.3, 159.0 (d, 1JCF = 239.8 Hz); m/z 318 (100, M + H); HRMS (ES): found 318.0027. C14H10BrFN3+ requires 318.0042.
6-Bromo-N-(3-chlorophenyl)quinazolin-4-amine (3d). A mixture of 2a (0.50 g, 2.05 mmol), 3-chloroaniline (0.29 g, 2.25 mmol) and concentrated HCl (0.01 g, 0.27 mmol) in THF-isopropanol (10 mL) afforded 3d as a white solid (0.54 g, 79%), mp. 220–222 °C (acetonitrile); νmax (ATR) 494, 683, 778, 832, 864, 958, 1145, 1354, 1403, 1485, 1531, 1562, 1614, 2935, 3074 cm−1; δH (500 MHz, DMSO-d6) 7.17 (d, J = 8.0 Hz, 1H), 7.41 (t, J = 8.0 Hz, 1H), 7.73 (d, J = 9.0 Hz, 1H), 7.82 (d, J = 8.5 Hz, 1H), 7.98 (dd, J = 2.0 and 8.5 Hz, 1H), 8.08 (s, 1H), 8.67 (s, 1H), 8.85 (d, J = 2.5 Hz, 1H), 9.91 (s, 1H); δC (125 MHz, DMSO-d6) 116.9, 119.4, 120.7, 121.8, 123.8, 125.8, 130.5, 130.6, 133.2, 136.6, 141.0, 149.0, 155.1, 157.1; m/z 334 (100, M+H); HRMS (ES): found 333.9747. C14H10BrClN3+ requires 333.9747.
6-Bromo-N-(4-bromophenyl)quinazolin-4-amine (3e). A mixture of 2a (0.50 g, 2.05 mmol), 4-bromoaniline (0.39 g, 2.25 mmol) and concentrated HCl (0.01 g, 0.27 mmol) in THF-isopropanol (10 mL) afforded 3e as a white solid (0.60 g, 77%), mp. 220–222 °C (acetonitrile); νmax (ATR) 492, 621, 810, 820, 831, 924, 1007, 1066, 1351, 1406, 1487, 1521, 1560, 1618, 3105, 3280 cm−1; δH (500 MHz, DMSO-d6) 7.55 (d, J = 8.5 Hz, 2H), 7.71 (d, J = 9.0 Hz, 1H), 7.85 (d, J = 8.5 Hz, 2H), 7.96 (dd, J = 2.0 and 8.5 Hz, 1H), 8.62 (s, 1H), 8.83 (d, J = 2.0 Hz, 1H), 9.90 (s, 1H); δC (125 MHz, DMSO-d6) 116.0, 116.9, 119.3, 124.5, 125.9, 130.5, 131.7, 136.5, 138.8, 149.0, 155.1, 157.0; m/z 378 (100, M+H); HRMS (ES): found 377.9241. C14H1079Br2N3+ requires 377.9241.
6-Bromo-N-(2,4-difluorophenyl)quinazolin-4-amine (3f). A mixture of 2a (0.50 g, 2.05 mmol), 2.4-difluoroaniline (0.29 g, 2.25 mmol) and concentrated HCl (0.01 g, 0.27 mmol) in THF-isopropanol (10 mL) afforded 3f as a white solid (0.55 g, 79%), mp. 198–200 °C (acetonitrile); νmax (ATR) 498, 536, 779, 835, 967, 1142, 1256, 1319, 1354, 1401, 1493, 1508, 1571, 3039 cm−1; δH (500 MHz, DMSO-d6) 7.14 (td, J = 2.0 and 9.0 Hz, 1H), 7.36 (td, J = 2.5 and 9.0 Hz, 1H), 7.53 (t, J = 9.0 Hz, 1H), 7.72 (d, J = 8.5 Hz, 1H), 7.97 (dd, J = 2.0 and 9.0 Hz, 1H), 8.48 (s, 1H), 8.73 (d, J = 2.0 Hz, 1H), 9.89 (s, 1H); δC (125 MHz, DMSO-d6) 105.0 (t, 2JCF = 26.5 Hz), 111.8 (dd, JCF = 3.7 and 21.7 Hz), 111.9 (d, 4JCF = 3.8 Hz), 116.5, 119.2, 125.9, 129.9 (d, 3JCF = 9.6 Hz), 130.5, 136.6, 148.9, 155.5, 158.3, 160.5 (d, 1JCF = 243.6 Hz), 160.6 (d, 1JCF = 243.6 Hz); m/z 336 (100, M + H); HRMS (ES): found 335.9952. C14H9BrF2N3+ requires 335.9948.
6-Bromo-2-(4-chlorophenyl)-N-(2-fluorophenyl)quinazolin-4-amine (3g). A mixture of 2b (0.50 g, 1.41 mmol), 2-fluoroaniline (0.17 g, 1.55 mmol) and concentrated HCl (0.01 g, 0.27 mmol) in THF-isopropanol (10 mL) afforded 3g as a white solid (0.49 g, 82%), mp. 248–250 °C (acetonitrile); νmax (ATR) 534, 733, 782, 829, 1012, 1089, 1171, 1316, 1399, 1446, 1489, 1522, 1554, 1598, 1622, 3451 cm−1; δH (500 MHz, DMSO-d6) 7.30‒7.41 (m, 3H), 7.50 (d, J = 8.5 Hz, 2H), 7.64 (td, J = 2.5 and 9.0 Hz, 1H), 7.79 (d, J = 8.5 Hz, 1H), 8.00 (dd, J = 2.0 and 8.5 Hz, 1H), 8.23 (d, J = 8.5 Hz, 2H), 8.80 (d, J = 2.0 Hz, 1H), 10.05 (s, 1H); δC (125 MHz, DMSO-d6) 115.4, 116.4 (d, 2JCF = 19.9 Hz), 119.0, 124.8 (d, 4JCF = 2.8 Hz), 126.1, 126.3 (d, 3JCF = 12.3 Hz), 127.9 (d, 3JCF = 8.5 Hz), 128.3, 128.9, 129.9, 130.7, 135.8, 136.8, 137.1, 149.7, 157.0 (d, 1JCF = 245.6 Hz), 158.2, 158.9; m/z 428 (100, M + H); HRMS (ES): found 427.9978. C20H1335ClF79BrN3+ requires 427.9965.
6-Bromo-2-(4-chlorophenyl)-N-(3-fluorophenyl)quinazolin-4-amine (3h). A mixture of 2b (0.50 g, 1.41 mmol), 3-fluoroaniline (0.17 g, 1.55 mmol) and concentrated HCl (0.01 g, 0.27 mmol) in THF-isopropanol (10 mL) afforded 3h as a white solid (0.53 g, 88%), mp. 242–244 °C (acetonitrile); νmax (ATR) 423, 487, 535, 670, 758, 824, 864, 1010, 1087, 1351, 1397, 1417, 1488, 1522, 1555, 3431 cm−1; δH (500 MHz, DMSO-d6) 7.00 (td, J = 2.0 and 8.0 Hz, 1H), 7.49 (q, J = 8.0 Hz, 1H), 7.59 (d, J = 8.5 Hz, 2H), 7.74 (dd, J = 2.0 and 8.0 Hz, 1H), 7.84 (d, J = 8.5 Hz, 1H), 7.90 (dt, J = 2.5 and 11.5 Hz, 1H), 8.00 (dd, J = 2.5 and 8.5 Hz, 1H), 8.38 (d, J = 8.5 Hz, 2H), 8.86 (d, J = 2.0 Hz, 1H), 10.07 (s, 1H); δC (125 MHz, DMSO-d6) 109.2 (d, 2JCF = 26.6 Hz), 110.7 (d, 2JCF = 20.8 Hz), 115.7, 118.1 (d, 4JCF = 2.8 Hz), 119.2, 125.9, 129.0, 130.0, 130.5 (d, 3JCF = 9.3 Hz), 130.8, 135.9, 136.9, 137.2, 141.2 (d, 3JCF = 11.3 Hz), 149.7, 157.4, 158.8, 162.4 (d, 1JCF = 239.0 Hz); m/z 428 (100, M + H); HRMS (ES): found 427.9966. C20H1335ClF79BrN3+ requires 427.9965.
6-Bromo-2-(4-chlorophenyl)-N-(4-fluorophenyl)quinazolin-4-amine (3i). A mixture of 2b (0.50 g, 1.41 mmol), 4-fluoroaniline (0.17 g, 1.55 mmol) and concentrated HCl (0.01 g, 0.27 mmol) in THF-isopropanol (10 mL) afforded 3i as a white solid (0.51 g, 84%), mp. 289–291 °C (acetonitrile); νmax (ATR) 424, 534, 669, 736, 827, 1011, 1087, 1199, 1314, 1352, 1398, 1416, 1522, 1556, 3431 cm−1; δH (500 MHz, DMSO-d6) 7.23 (t, J = 8.5 Hz, 2H), 7.55 (d, J = 8.5 Hz, 2H), 7.77 (d, J = 9.0 Hz, 1H), 7.88 (t, J = 8.5 Hz, 2H), 7.97 (dd, J = 2.5 and 9.0 Hz, 1H), 8.35 (d, J = 9.0 Hz, 2H), 8.82 (d, J = 2.0 Hz, 1H), 10.00 (s, 1H); δC (125 MHz, DMSO-d6) 115.6 (d, 2JCF = 21.7 Hz), 115.7, 119.0, 124.8 (d, 3JCF = 7.5 Hz), 125.9, 129.0, 130.1, 130.7, 135.6 (d, 4JCF = 2.8 Hz), 135.8, 136.7, 137.2, 149.7, 157.5, 158.9, 159.0 (d, 1JCF = 239.0 Hz); m/z 428 (100, M + H); HRMS (ES): found 427.9951. C20H1335ClF79BrN3+ requires 427.9965.
6-Bromo-2-(4-chlorophenyl)-N-(3-chlorophenyl)quinazolin-4-amine (3j). A mixture of 2b (0.50 g, 1.41 mmol), 3-chloroaniline (0.19 g, 1.55 mmol) and concentrated HCl (0.01 g, 0.27 mmol) in THF-isopropanol (10 mL) afforded 3j as a white solid (0.55 g, 87%), mp. 261–263 °C (acetonitrile); νmax (ATR) 484, 535, 673, 735, 765, 829, 996, 1086, 1353, 1397, 1416, 1428, 1515, 1564, 1595, 3432 cm−1; δH (500 MHz, DMSO-d6) 7.20 (d, J = 8.5 Hz, 1H), 7.46 (t, J = 8.0 Hz, 1H), 7.55 (d, J = 8.5 Hz, 2H), 7.77 (d, J = 8.5 Hz, 1H), 7.88 (dd, J = 2.5 and 8.5 Hz, 1H), 7.97 (dd, J = 2.0 and 8.5 Hz, 1H), 8.13 (s, 1H), 8.82 (d, J = 2.0 Hz, 1H), 10.00 (s, 1H); δC (125 MHz, DMSO-d6) 115.7, 119.3, 120.7, 122.0, 123.8, 125.9, 129.0, 130.0, 130.6, 130.7, 133.2, 135.9, 136.8, 137.1, 140.9, 149.7, 157.3, 158.7; m/z 444 (100, M + H); HRMS (ES): found 443.9684. C20H1335Cl279BrN3+ requires 443.9670.
6-Bromo-N-(4-bromophenyl)-2-(4-chlorophenyl)quinazolin-4-amine (3k). A mixture of 2b (0.50 g, 1.41 mmol), 4-bromoaniline (0.27 g, 1.55 mmol) and concentrated HCl (0.01 g, 0.27 mmol) in THF-isopropanol (10 mL) afforded 3k as a white solid (0.57 g, 83%), mp. 263–265 °C (acetonitrile); νmax (ATR) 488, 541, 802, 814, 834, 875, 1011, 1083, 1351, 1400, 1489, 1520, 1554, 1564, 1596, 3434 cm−1; δH (500 MHz, DMSO-d6) 7.55 (d, J = 8.5 Hz, 2H), 7.63 (d, J = 8.5 Hz, 2H), 7.76 (d, J = 9.0 Hz, 1H), 7.88 (d, J = 8.5 Hz, 2H), 7.97 (dd, J = 2.0 and 8.5 Hz, 1H), 8.37 (d, J = 8.5 Hz, 2H), 8.82 (d, J = 2.0 Hz, 1H), 10.01 (s, 1H); δC (125 MHz, DMSO-d6) 115.7, 116.1, 119.1, 124.5, 125.9, 129.0, 130.1, 130.7, 131.8, 135.8, 136.8, 137.1, 138.8, 149.7, 157.4, 158.9; m/z 488 (100, M + H); HRMS (ES): found 487.9161. C20H13Cl79Br2N3+ requires 487.9165.
6-Bromo-2-(4-chlorophenyl)-N-(2,4-difluorophenyl)quinazolin-4-amine (3l). A mixture of 2b (0.50 g, 1.41 mmol), 2.4-difluoroaniline (0.2 g, 1.55 mmol) and concentrated HCl (0.01 g, 0.27 mmol) in THF-isopropanol (10 mL) afforded 3l as a white solid (0.58 g, 93%), mp. 278–279 °C (acetonitrile); νmax (ATR) 425, 533, 735, 795, 830, 1010, 1088, 1202, 1257, 1351, 1418, 1498, 1528, 1558, 3456 cm−1; δH (500 MHz, DMSO-d6) 7.00 (td, J = 2.5 and 8.0 Hz, 1H), 7.44 (dd, J = 2.0 and 8.5 Hz, 1H), 7.51 (d, J = 8.5 Hz, 2H), 7.66 (q, J = 8.0 Hz, 1H), 7.79 (d, J = 8.5 Hz, 1H), 8.00 (dd, J = 2.0 and 8.5 Hz, 1H), 8.22 (d, J = 8.5 Hz, 2H), 8.78 (d, J = 2.5 Hz, 1H), 10.07 (s, 1H); δC (125 MHz, DMSO-d6) 104.9 (t, 2JCF = 26.6 Hz), 111.8 (dd, JCF = 2.8 and 21.7 Hz), 115.4, 119.0, 122.9 (d, 4JCF = 3.8 Hz), 126.1, 128.9, 129.6 (d, 3JCF = 9.5 Hz), 129.9, 130.7, 135.8, 136.9, 137.1, 149.6, 157.3 (d, 1JCF = 248.4 Hz), 158.4, 158.9, 160.5 (d, 1JCF = 243.7 Hz); m/z 446 (100, M + H); HRMS (ES): found 445.9854. C20H1235ClF279BrN3+ requires 445.9871.
3.5. Typical Procedure for the Preparation of Compounds 4a–d, f and 4g–j, l
[6-(4-Fluorophenyl)quinazolin-4-yl]-2-fluorophenylamine (4a). A stirred mixture of 3a (0.30 g, 0.94 mmol), PdCl2(PPh3)2 (0.033 g, 0.047 mmol) and K2CO3 (0.19 g, 1.41 mmol) in 3:1 THF-isopropanol (v/v, 10 mL) in a two-necked round-bottomed flask was purged with argon gas for 30 min. 4-Fluorophenylboronic acid (0.15 g, 1.13 mmol) was added to the mixture via a syringe. The reaction mixture was heated at 100 °C for 3 h and then quenched with ice-cold water. The product was extracted into chloroform and the combined organic layers were washed with water, dried over Na2SO4, filtered and evaporated under reduced pressure. The residue was purified by column chromatography on silica gel to afford 4a as a white solid (0.20 g, 64%), mp. 192–194 °C; Rf (1:1, toluene/ethyl acetate) 0.54; νmax (ATR) 495, 592, 666, 746, 826, 1101, 1164, 1238, 1325, 1402, 1419, 1500, 1520, 1576, 1600, 3195 cm−1; δH (500 MHz, DMSO-d6) 7.24‒7.34 (m, 3H), 7.38 (t, J = 8.5 Hz, 2H), 7.55 (t, J = 8.5 Hz, 1H), 7.84 (d, J = 8.5 Hz, 1H), 7.91 (t, J = 8.0 Hz, 2H), 8.19 (dd, J = 2.0 and 8.5 Hz, 1H), 8.47 (s, 1H), 8.78 (d, J = 2.0 Hz, 1H), 9.99 (s, 1H); δC (125 MHz, DMSO-d6) 115.5, 116.3 (d, 2JCF = 20.8 Hz), 116.5 (d, 2JCF = 19.0 Hz), 120.9, 124.9 (d, 4JCF = 2.8 Hz), 126.5 (d, 3JCF = 12.3 Hz), 127.9 (d, 3JCF = 7.5 Hz), 128.8 (d, 4JCF = 3.7 Hz), 128.9, 129.4 (d, 3JCF = 7.6 Hz), 132.1, 135.9 (d, 4JCF = 2.8 Hz), 137.3, 149.4, 155.2, 157.3 (d, 1JCF = 245.6 Hz), 159.2, 162.6 (d, 1JCF = 243.6 Hz); m/z 334 (100, M + H); HRMS (ES): found 334.1166. C20H14F2N3+ requires 334.1156.
[6-(4-Fluorophenyl)quinazolin-4-yl]-3-fluorophenylamine (4b). A mixture of 3b (0.30 g, 0.94 mmol), PdCl2(PPh3)2 (0.033 g, 0.047 mmol), 4-fluorophenylboronic acid (0.15 g, 1.13 mmol) and K2CO3 (0.19 g, 1.41 mmol) in THF-isopropanol (10 mL) afforded 4b as a white solid (0.22 g, 72%), mp. 225–227 °C; Rf (1:1, toluene/ethyl acetate) 0.70; νmax (ATR) 506, 587, 687, 780, 827, 859, 962, 1144, 1240, 1400, 1416, 1488, 1532, 1573, 1601, 3254 cm−1; δH (500 MHz, DMSO-d6) 6.96 (td, J = 2.0 and 8.5 Hz, 1H), 7.41 (t, J = 8.5 Hz, 2H), 7.44 (q, J = 8.5 Hz, 1H), 7.67 (d, J = 1.5 and 8.5 Hz, 1H), 7.86 (d, J = 8.5 Hz, 1H), 7.90–7.96 (m, 3H), 818 (dd, J = 2.0 and 8.5 Hz, 1H), 8.65 (s, 1H), 8.80 (d, J = 2.0 Hz, 1H), 10.01 (s, 1H); δC (125 MHz, DMSO-d6) 109.4 (d, 2JCF = 25.5 Hz), 110.5 (d, 2JCF = 20.9 Hz), 115.7, 116.3 (d, 2JCF = 20.9 Hz), 118.2 (d, 4JCF = 2.8 Hz), 120.8, 128.9, 129.7 (d, 3JCF = 9.5 Hz), 130.4 (d, 3JCF = 9.5 Hz), 132.4, 136.0 (d, 4JCF = 3.8 Hz), 137.7, 141.3 (d, 3JCF = 10.4 Hz), 149.4, 154.7, 158.1, 162.4 (d, 1JCF = 239.8 Hz), 162.6 (d, 1JCF = 243.7 Hz); m/z 334 (100, M + H); HRMS (ES): found 334.1158. C20H14F2N3+ requires 334.1156.
[6-(4-Fluorophenyl)quinazolin-4-yl]-4-fluorophenylamine (4c). A mixture of 3c (0.30 g, 0.94 mmol), PdCl2(PPh3)2 (0.033 g, 0.047 mmol), 4-fluorophenylboronic acid (0.15 g, 1.13 mmol) and K2CO3 (0.19 g, 1.41 mmol) in THF-isopropanol (10 mL) afforded 4c as a white solid (0.25 g, 80%), mp. 215–217 °C; Rf (1:1, toluene/ethyl acetate) 0.64; νmax (ATR) 466, 502, 535, 660, 824, 1156, 1210, 1222, 1357, 1421, 1498, 1534, 1574, 1613, 1630, 3278 cm−1; δH (500 MHz, DMSO-d6) 7.24 (t, J = 8.5 Hz, 2H), 7.38 (t, J = 8.5 Hz, 2H), 7.80–7.85 (m, 3H), 7.90 (t, J = 8.5 Hz, 2H), 8.15 (dd, J = 2.0 and 8.5 Hz, 1H), 8.55 (s, 1H), 8.78 (d, J = 2.0 Hz, 1H), 9.94 (s, 1H); δC (125 MHz, DMSO-d6) 115.5 (d, 2JCF = 21.8 Hz), 115.6, 116.3 (d, 2JCF = 20.9 Hz), 120.7, 125.2 (d, 3JCF = 7.5 Hz), 128.8, 129.5 (d, 3JCF = 8.5 Hz), 132.2, 135.6 (d, 4JCF = 2.9 Hz), 136.0 (d, 4JCF = 3.7 Hz), 137.4, 149.3, 154.9, 158.4, 159.1 (d, 1JCF = 239.9 Hz), 162.6 (d, 1JCF = 243.7 Hz); m/z 334 (100, M + H); HRMS (ES): found 334.1166. C20H14F2N3+ requires 334.1156.
[6-(4-Fluorophenyl)quinazolin-4-yl]-3-chlorophenylamine (4d). A mixture of 3d (0.30 g, 0.89 mmol), PdCl2(PPh3)2 (0.033 g, 0.044 mmol), 4-fluorophenylboronic acid (0.15 g, 1.07 mmol) and K2CO3 (0.18 g, 1.33 mmol) in THF-isopropanol (10 mL) afforded 4d as a white solid (0.19 g, 61%), mp. 207–209 °C; Rf (1:1, toluene/ethyl acetate) 0.71; νmax (ATR) 510, 587, 683, 777, 827, 933, 1236, 1356, 1395, 1417, 1478, 1498, 1529, 1562, 1597, 2849, 2918, 3245 cm−1; δH (500 MHz, DMSO-d6) 7.18 (dd, J = 2.0 and 8.5 Hz, 1H), 7.36‒7.45 (m, 3H), 7.85 (d, J = 8.5 Hz, 1H), 7.86 (d, J = 8.5 Hz, 1H), 7.91 (t, J = 8.5 Hz, 2H), 8.08 (s, 1H), 8.18 (dd, J = 2.0 and 8.5 Hz, 1H), 8.65 (s, 1H), 8.79 (d, J = 2.5 Hz, 1H), 9.98 (s, 1H); δC (125 MHz, DMSO-d6) 115.4, 116.3 (d, 2JCF = 20.7 Hz), 116.4, 116.5, 120.8, 125.0, 128.1, 128.2, 128.6, 128.8, 129.5 (d, 3JCF = 8.5 Hz), 135.8 (d, 4JCF = 2.8 Hz), 137.5, 149.1, 155.1, 156.3, 159.3, 162.6 (d, 1JCF = 244.6 Hz); m/z 350 (100, M + H); HRMS (ES): found 350.0853. C20H1435ClFN3+ requires 350.0860.
[6-(4-Fluorophenyl)quinazolin-4-yl]-2, 4-difluorophenylamine (4f). A mixture of 3f (0.30 g, 0.89 mmol), PdCl2(PPh3)2 (0.031 g, 0.044 mmol), 4-fluorophenylboronic acid (0.15 g, 1.07 mmol) and K2CO3 (0.18 g, 1.33 mmol) in THF-isopropanol (10 mL) afforded a white solid (0.24 g, 77%), mp. 228–231 °C; Rf (1:1, toluene/ethyl acetate) 0.60; νmax (ATR) 470, 496, 513, 665, 784, 824, 965, 1141, 1241, 1325, 1401, 1499, 1578, 2852, 2922, 3159 cm−1; δH (500 MHz, DMSO-d6) 7.17 (td, J = 2.0 and 8.5 Hz, 1H), 7.35–7.43 (m, 3H), 7.56 (q, J = 8.0 Hz, 1H), 7.84 (d, J = 8.5 Hz, 1H), 7.91 (t, J = 8.0 Hz, 2H), 8.19 (dd, J = 2.0 and 8.5 Hz, 1H), 8.46 (s, 1H), 8.76 (d, J = 2.0 Hz, 1H), 9.96 (s, 1H); δC (125 MHz, DMSO-d6) 105.0 (t, 2JCF = 26.5 Hz), 112.0 (d, 2JCF = 21.8 Hz), 115.2, 116.4 (d, 2JCF = 20.8 Hz), 120.7, 125.8, 128.5, 129.4 (d, 3JCF = 7.6 Hz), 130.0 (d, 4JCF = 2.8 Hz), 130.1, 132.5, 135.7, 137.7, 148.7 (d, 4JCF = 2.8 Hz), 155.0, 157.5 (d, 1JCF = 248.5 Hz), 159.4, 162.6 (d, 1JCF = 243.6 Hz); m/z 352 (100, M + H); HRMS (ES): found 352.1074. C20H13F3N3+ requires 352.1062.
[2-(4-Chlorophenyl)-6-(4-fluorophenyl)quinazolin-4-yl]-2-fluorophenylamine (4g). A mixture of 3g (0.30 g, 0.70 mmol), PdCl2(PPh3)2 (0.024 g, 0.035 mmol), 4-fluorophenylboronic acid (0.12 g, 0.84 mmol) and K2CO3 (0.14 g, 1.05 mmol) in THF-isopropanol (10 mL) afforded 4g as a white solid (0.21 g, 68%), mp. 195–197 °C; Rf (toluene) 0.53; νmax (ATR) 503, 516, 741, 815, 1013, 1086, 1231, 1420, 1454, 1494, 1514, 1538, 1559, 1597, 3045, 3446 cm−1; δH (500 MHz, DMSO-d6) 7.31‒7.42 (m, 5H), 7.49 (d, J = 8.5 Hz, 2H), 7.67 (td, J = 2.5 and 8.5 Hz, 1H), 7.90‒7.96 (m, 3H), 8.20 (dd, J = 2.5 and 8.5 Hz, 1H), 8.25 (d, J = 8.5 Hz, 2H), 8.82 (d, J = 2.0 Hz, 1H), 10.11 (s, 1H); δC (125 MHz, DMSO-d6) 114.4, 116.3 (d, 2JCF = 20.9 Hz), 116.4 (d, 2JCF = 19.9 Hz), 120.9, 124.9 (d, 4JCF = 2.8 Hz), 126.5 (d, 3JCF = 11.4 Hz), 127.8 (d, 3JCF = 8.5 Hz), 128.4, 128.9, 129.1, 129.4 (d, 3JCF = 8.5 Hz), 129.9, 132.4, 135.6, 135.9 (d, 4JCF = 2.8 Hz), 137.2, 137.4, 150.1, 157.1 (d, 1JCF = 245.6 Hz), 158.5, 159.2, 162.6 (d, 1JCF = 243.7 Hz); m/z 444 (100, M + H); HRMS (ES): found 444.1068. C26H1735ClF2N3+ requires 444.1079.
[2-(4-Chlorophenyl)-6-(4-fluorophenyl)quinazolin-4-yl]-3-fluorophenylamine (4h). A mixture of 3h (0.30 g, 0.70 mmol), PdCl2(PPh3)2 (0.024 g, 0.035 mmol), 4-fluorophenylboronic acid (0.12 g, 0.84 mmol) and K2CO3 (0.14 g, 1.05 mmol) in THF-isopropanol (10 mL) afforded 4h as a white solid (0.22 g, 71%), mp. 226–228 °C; Rf (toluene) 0.38; νmax (ATR) 431, 522, 670, 762, 820, 969, 1084, 1138, 1159, 1232, 1428, 1488, 1560, 1604, 2857, 2926, 3446 cm−1; δH (500 MHz, DMSO-d6) 7.00 (td, J = 2.0 and 8.5 Hz, 1H), 7.40 (t, J = 8.5 Hz, 2H), 7.49 (q, J = 8.0 Hz, 1H), 7.58 (d, J = 8.5 Hz, 2H), 7.75 (dd, J = 2.0 and 8.5 Hz, 1H), 7.88–7.97 (m, 4H), 8.20 (dd, J = 2.0 and 8.5 Hz, 1H), 8.42 (d, J = 8.5 Hz, 2H), 8.83 (d, J = 2.0 Hz, 1H), 10.15 (s, 1H); δC (125 MHz, DMSO-d6) 109.4 (d, 2JCF = 25.6 Hz), 110.6 (d, 2JCF = 20.9 Hz), 114.6, 116.3 (d, 2JCF = 21.9 Hz), 118.3 (d, 4JCF = 2.8 Hz), 120.9, 129.1, 129.2, 129.4, 129.6 (d, 3JCF = 7.6 Hz), 129.9, 130.5 (d, 3JCF = 9.5 Hz), 132.6, 135.7, 136.0 (d, 4JCF = 2.8 Hz), 137.4, 141.4 (d, 3JCF = 10.5 Hz), 150.2, 158.3, 158.4, 162.5 (d, 1JCF = 239.9 Hz), 162.6 (d, 1JCF = 243.6 Hz); m/z 444 (100, M + H); HRMS (ES): found 444.1081. C26H1735ClF2N3+ requires 444.1079.
[2-(4-Chlorophenyl)-6-(4-fluorophenyl)quinazolin-4-yl]-4-fluorophenylamine (4i). A mixture of 3i (0.30 g, 0.70 mmol), PdCl2(PPh3)2 (0.024 g, 0.035 mmol), 4-fluorophenylboronic acid (0.12 g, 0.84 mmol) and K2CO3 (0.14 g, 1.05 mmol) in THF-isopropanol (10 mL) afforded 4i as a white solid (0.23 g, 76%), mp. 221–222 °C; Rf (toluene) 0.31; νmax (ATR) 435, 487, 506, 527, 822, 945, 1009, 1082, 1153, 1205, 1420, 1503, 1533, 1560, 3451 cm−1; δH (500 MHz, DMSO-d6) 7.31 (t, J = 8.5 Hz, 2H), 7.39 (t, J = 8.5 Hz, 2H), 7.55 (d, J = 8.5 Hz, 2H), 7.86–7.96 (m, 5H), 8.17 (dd, J = 2.0 and 8.5 Hz, 1H), 8.38 (d, J = 8.5 Hz, 2H), 8.80 (d, J = 2.0 Hz, 1H), 10.06 (s, 1H); δC (125 MHz, DMSO-d6) 114.5, 115.6 (d, 2JCF = 21.7 Hz), 116.3 (d, 2JCF = 21.7 Hz), 120.8, 124.9 (d, 3JCF = 8.0 Hz), 128.9, 129.1, 129.4 (d, 3JCF = 8.0 Hz), 130.0, 132.3, 135.5, 135.7 (d, 4JCF = 2.8 Hz), 136.0 (d, 4JCF = 3.7 Hz), 137.2, 137.5, 150.0, 158.4, 158.5, 158.9 (d, 1JCF = 239.4 Hz), 162.6 (d, 1JCF = 243.9 Hz); m/z 444 (100, M + H); HRMS (ES): found 444.1078. C26H1735ClF2N3+ requires 444.1079.
[2-(4-Chlorophenyl)-6-(4-fluorophenyl)quinazolin-4-yl]-3-chlorophenylamine (4j). A mixture of 3j (0.30 g, 0.67 mmol), PdCl2(PPh3)2 (0.023 g, 0.034 mmol), 4-fluorophenylboronic acid (0.11 g, 0.80 mmol) and K2CO3 (0.14 g, 1.00 mmol) in THF-isopropanol (10 mL) afforded 4j as a white solid (0.19 g, 61%), mp. 244–246 °C; Rf (toluene) 0.40; νmax (ATR) 530, 673, 765, 818, 828, 1086, 1156, 1206, 1351, 1403, 1425, 1474, 1494, 1508, 1526, 1556, 1598, 3450 cm−1; δH (500 MHz, DMSO-d6) 7.23 (dd, J = 2.5 and 8.5 Hz, 1H), 7.39 (t, J = 8.5 Hz, 2H), 7.49 (t, J = 8.5 Hz, 1H), 7.57 (d, J = 8.5 Hz, 2H), 7.90‒7.95 (m, 4H), 8.13 (s, 1H), 8.18 (dd, J = 2.0 and 8.5 Hz, 1H), 8.41 (d, J = 8.5 Hz, 2H), 8.80 (d, J = 2.5 Hz, 1H), 10.09 (s, 1H); δC (125 MHz, DMSO-d6) 114.6, 116.3 (d, 2JCF = 21.8 Hz), 120.8, 120.9, 122.2, 123.8, 129.0, 129.2, 129.5 (d, 3JCF = 8.5 Hz), 129.9, 130.6, 133.6, 133.2, 135.7, 135.9 (d, 4JCF = 2.8 Hz), 137.4, 137.5, 141.1, 150.2, 158.2, 158.3, 162.6 (d, 1JCF = 243.7 Hz); m/z 460 (100, M + H); HRMS (ES): found 460.0780. C26H1735Cl2FN3+ requires 460.0784.
[2-(4-Chlorophenyl)-6-(4-fluorophenyl)quinazolin-4-yl]-2,4-difluorophenylamine (4l). A mixture of 3l (0.30 g, 0.67 mmol), PdCl2(PPh3)2 (0.023 g, 0.034 mmol), 4-fluorophenylboronic acid (0.11 g, 0.80 mmol) and K2CO3 (0.14 g, 1.00 mmol) in THF-isopropanol (10 mL) afforded 4l as a white solid (0.21 g, 69%), mp. 210–212 °C; Rf (toluene) 0.48; νmax (ATR) 425, 515, 737, 817, 838, 964, 1012, 1089, 1161, 1234, 1415, 1502, 1530, 1558, 1585, 3450 cm−1; δH (500 MHz, DMSO-d6) 7.22 (td, J = 2.5 and 8.0 Hz, 1H), 7.36–7.43 (m, 3H), 7.45–7.52 (m, 3H), 7.68 (q, J = 8.0 Hz, 1H), 7.91–7.96 (m, 3H), 8.21 (dd, J = 2.0 and 8.5 Hz, 1H), 8.25 (d, J = 8.5 Hz, 2H), 8.80 (d, J = 2.0 Hz, 1H), 10.09 (s, 1H); δC (125 MHz, DMSO-d6) 105.0 (t, 2JCF = 25.2 Hz), 112.0 (d, 2JCF = 21.7 Hz), 114.3, 116.3 (d, 2JCF = 21.7 Hz), 120.9, 123.0, 123.3, 128.9, 129.4 (d, 3JCF = 8.0 Hz), 129.5, 129.7 (d, 4JCF = 2.8 Hz), 129.8, 132.4, 135.6, 135.8 (d, 4JCF = 3.5 Hz), 137.2, 137.3, 150.0, 157.2 (d, 1JCF = 248.5 Hz), 158.4, 158.5, 162.6 (d, 1JCF = 243.9 Hz); m/z 462 (100, M + H); HRMS (ES): found 462.0982. C26H1635ClF3N3+ requires 462.0985.
3.6. Reaction Sequence for the Preparation of 4e and 4k from 1a and 1b, Respectively
3.6.1. Typical Procedure for the Preparation of 5a and 5b
6-(4-Fluorophenyl)quinazolin-4(3H)-one (5a). A mixture of 6-bromoquinazolin-4(3H)-one 1a (0.50 g, 2.22 mmol) PdCl2(PPh3)2 (0.078 g, 0.11 mmol) and K2CO3 (0.46 g, 3.33 mmol) in dioxane-water (20 mL) in a two-necked round-bottomed flask was purged with argon gas for 30 minutes. 4-Fluorophenylboronic acid (0.37 g, 2.66 mmol) in dioxane (10 mL) was added via a syringe. The reaction mixture was stirred at 70 °C for 3 h until the starting material was completely consumed (TLC monitoring). After cooling the mixture, the product was extracted with chloroform and the combined organic layers were washed with water, dried over Na2SO4, filtered, and evaporated under reduced pressure. The residue was purified by recrystallization from acetonitrile to afford 5a as a white solid (0.43 g, 81%), mp. 228–230 °C; νmax (ATR) 496, 698, 746, 821, 923, 1160, 1239, 1275, 1333, 1384, 1477, 1603, 1687, 2673, 3046 cm−1; δH (500 MHz, DMSO-d6) 7.31 (t, J = 9.0 Hz, 2H), 7.73 (d, J = 8.5 Hz, 1H), 7.79 (t, J = 8.5 Hz, 2H), 8.09 (dd, J = 2.0 and 8.5 Hz, 1H), 8.10 (s, 1H), 8.28 (d, J = 2.0 Hz, 1H), 12.30 (br.s, 1H); δC (125 MHz, DMSO-d6) 116.4 (d, 2JCF = 21.7 Hz), 123.2, 123.6, 127.8, 129.4 (d, 3JCF = 7.9 Hz), 133.4 (d, 4JCF = 2.8 Hz), 134.0, 135.5, 135.6, 138.0, 146.4, 162.6 (d, 1JCF = 240.5 Hz); m/z 241 (100, M + H); HRMS (ES): found 241.0780. C14H10FN2O+ requires 241.0777.
2-(4-Chlorophenyl)-6-(4-fluorophenyl)quinazolin-4(3H)-one (5b). A mixture of 6-bromo-2-(4-chlorophenyl)quinazolin-4(3H)-one 1b (0.5 g, 1.49 mmol) PdCl2(PPh3)2 (0.052 g, 0.074 mmol), 4-fluorophenylboronic acid (0.25 g, 1.78 mmol) and K2CO3 (0.31 g, 2.23 mmol) in dioxane-water (30 mL) afforded 5b as a white solid (0.46 g, 88%), mp. 151–153 °C; νmax (ATR) 501, 539, 693, 719, 752, 825, 1118, 1180, 1190, 1308, 1436, 1483, 1603, 1674, 2926, 3056 cm−1; δH (500 MHz, DMSO-d6) 7.32 (t, J = 8.0 Hz, 2H), 7.61 (d, J = 8.5 Hz, 2H), 7.79–7.85 (m, 3H), 8.13 (dd, J = 2.0 and 8.5 Hz, 1H), 8.20 (d, J = 8.5 Hz, 2H), 8.33 (d, J = 2.5 Hz, 1H), 12.67 (br.s, 1H); δC (125 MHz, DMSO-d6) 116.4 (d, 2JCF = 21.8 Hz), 122.6, 128.3, 128.5, 128.9, 129.1, 129.2 (d, 3JCF = 8.6 Hz), 130.1, 131.8, 132.4 (d, 4JCF = 2.8 Hz), 136.4, 144.8, 146.3, 148.9, 161.2, 162.5 (d, 1JCF = 243.5 Hz); m/z 351 (100, M + H); HRMS (ES): found 351.0702. C20H1335ClFN2O requires 351.0700.
3.6.2. Typical Procedure for the Preparation of 6a and 6b
4-Chloro-6-(4-fluorophenyl)quinazoline (6a). Triethylamine (4 mL) was added dropwise at room temperature to a stirred mixture of 5a (0.40 g, 1.66 mmol) and phosphoryl chloride (10 mL) in a round-bottomed flask. The reaction mixture was stirred under reflux for 5 h and then allowed to cool to room temperature. Ice-cold water was added to the mixture and the product was extracted with chloroform. The combined organic layers were washed with an aqueous solution of NaHCO3, dried over Na2SO4, filtered, and evaporated under reduced pressure. The crude product was recrystallized from ethanol to afford 6a as a white solid (0.33 g, 77%), mp. 154–156 °C (EtOH); νmax (ATR) 478, 513, 523, 697, 825, 991, 1096, 1146, 1225, 1335, 1484, 1511, 1545, 1565, 1600 cm−1; δH (500 MHz, DMSO-d6) 7.16 (t, J = 8.5 Hz, 2H), 7.47 (d, J = 8.5 Hz, 1H), 7.80 (t, J = 8.5 Hz, 2H), 8.12 (dd, J = 2.0 and 8.5 Hz, 1H), 8.33 (s, 1H), 8.37 (d, J = 2.0 Hz, 1H); δC (125 MHz, DMSO-d6) 116.3 (d, 2JCF = 21.8 Hz), 122.8, 123.1, 128.8, 129.2 (d, 3JCF = 8.3 Hz), 132.3, 134.1 (d, 4JCF = 3.2 Hz), 136.7, 149.8, 157.7, 159.0, 162.4 (d, 1JCF = 240.2 Hz); m/z 258 (100, M + H); HRMS (ES): found 257.8652. C14H835ClFN2 requires 257.8652.
4-Chloro-2-(4-chlorophenyl)-6-(4-fluorophenyl)quinazoline (6b). A mixture of 5b (0.40 g, 1.14 mmol), trimethylamine (4 mL) and phosphoryl chloride (10 mL) afforded 6b as a white solid (0.35 g, 83%), mp. 216–218 °C (EtOH); νmax (ATR) 514, 553, 700, 735, 778, 821, 988, 1010, 1088, 1162, 1237, 1360, 1401, 1431, 1492, 1514, 1554, 1604 cm−1; δH (500 MHz, DMSO-d6) 7.31 (t, J = 8.5 Hz, 2H), 7.61 (d, J = 8.5 Hz, 2H), 7.79–7.84 (m, 3H), 8.13 (dd, J = 2.0 and 8.5 Hz, 1H), 8.19 (d, J = 8.5 Hz, 2H), 8.31 (d, J = 2.0 Hz, 1H); δC (125 MHz, DMSO-d6) 116.2 (d, 2JCF = 20.4 Hz), 121.3, 123.4, 125.6, 128.7, 128.9, 129.1 (d, 3JCF = 8.3 Hz), 130.2, 131.9, 134.6 (d, 4JCF = 3.2 Hz), 138.4, 139.8, 148.6, 157.8, 158.2, 162.6 (d, 1JCF = 245.3 Hz); m/z 368 (100, M + H); HRMS (ES): found 368.0435. C20H1235Cl2FN2 requires 368.0273.
3.6.3. Typical Procedure for the Preparation of 4e and 4k
[6-(4-Fluorophenyl)quinazolin-4-yl]-4-bromophenylamine (4e). A mixture of 6a (0.30 g, 1.16 mmol), 4-bromaniline (0.22 g, 1.28 mmol) and concentrated HCl (0.01 g, 0.27 mmol) in 3:1 THF-isopropanol (v/v, 10 mL) in a round-bottomed flask was stirred under reflux for 5 h. The mixture was then allowed to cool to room temperature and quenched with ice-cold water. The product was extracted with ethyl acetate and the combined organic layers were washed with an aqueous solution of NaHCO3, dried over anhydrous MgSO4, filtered, and evaporated under reduced pressure. The crude product was purified by column chromatography on silica gel to afford 4e as a white solid (0.37 g, 81%), mp. 227–229 °C; Rf (1:1, toluene/ethyl acetate) 0.69; νmax (ATR) 477, 507, 662, 823, 925, 1006, 1160, 1232, 1358, 1416, 1487, 1498, 1523, 1560, 1603, 3280 cm−1; δH (500 MHz, DMSO-d6) 7.38 (t, J = 8.5 Hz, 2H), 7.58 (d, J = 8.5 Hz, 2H), 7.84 (d, J = 8.5 Hz, 1H), 7.85 (d, J = 8.5 Hz, 2H), 7.90 (t, J = 8.5 Hz, 2H), 8.16 (dd, J = 2.0 and 8.5 Hz, 1H), 8.59 (s, 1H), 8.78 (d, J = 8.0 Hz, 1H), 9.96 (s, 1H); δC (125 MHz, DMSO-d6) 115.8, 115.8, 116.3 (d, 2JCF = 21.7 Hz), 120.8, 124.7, 128.9, 129.6 (d, 3JCF = 8.0 Hz), 131.7, 132.3, 136.0 (d, 4JCF = 2.8 Hz), 137.5, 138.9, 149.4, 154.8, 158.1, 162.6 (d, 1JCF = 242.8 Hz); m/z 394 (100, M + H); HRMS (ES): found 394.0356. C20H1479BrFN3+ requires 394.0356.
[2-(4-Chlorophenyl)-6-(4-fluorophenyl)quinazolin-4-yl]-4-bromophenylamine (4k). A mixture of 6b (0.30 g, 0.81 mmol), 4-bromoaniline (0.15 g, 0.89 mmol) and concentrated HCl (0.01 g, 0.27 mmol) in 3:1 THF-isopropanol (v/v, 10 mL) afforded 4k as a white solid (0.32 g, 79%), mp. 257–260 °C; Rf (toluene) 0.36; νmax (ATR) 499, 521, 666, 740, 817, 1012, 1090, 1161, 1230, 1354, 1398, 1417, 1489, 1511, 1525, 1557, 1595, 3428 cm-1; δH (500 MHz, DMSO-d6) 7.39 (t, J = 8.5 Hz, 2H), 7.58 (d, J = 8.5 Hz, 2H), 7.65 (d, J = 8.5 Hz, 2H), 7.87–7.96 (m, 5H), 8.20 (dd, J = 2.5 and 8.5 Hz, 1H), 8.40 (d, J = 9.0 Hz, 2H), 8.82 (d, J = 2.0 Hz, 1H), 10.19 (s, 1H); δC (125 MHz, DMSO-d6) 114.5, 116.2, 116.3 (d, 2JCF = 20.7 Hz), 120.9, 124.9, 129.1, 129.2, 129.3, 129.5 (d, 3JCF = 8.5 Hz), 130.1, 131.8, 131.9, 132.7, 135.8, 135.9 (d, 4JCF = 2.8 Hz), 137.5, 138.8, 158.3, 158.4, 162.7 (d, 1JCF = 243.7 Hz); m/z 504 (100, M + H); HRMS (ES): found 504.0278. C26H1779Br35ClFN3+ requires 504.0282.
3.7. Materials and Methods for the In Vitro Cytotoxicity Assay
The 4-anilinoquinazolines
3a–
l and
4a–
l were evaluated for growth inhibitory activity against the MCF-7 and HeLa cell lines against Gefitnib as a reference standard using the MTT assay developed by Mosmann [
22] with slight modification as described in our previous investigation [
9]. The percentage of cell viability was calculated using the following formula:
The LC50 values (the lethal concentration at which 50% of the cells are killed) were calculated as the concentration of the test sample that resulted in 50% reduction of absorbance compared to untreated cells. The intensity of the MTT formazan produced by living metabolically active cells is directly proportional to the number of live cells present.
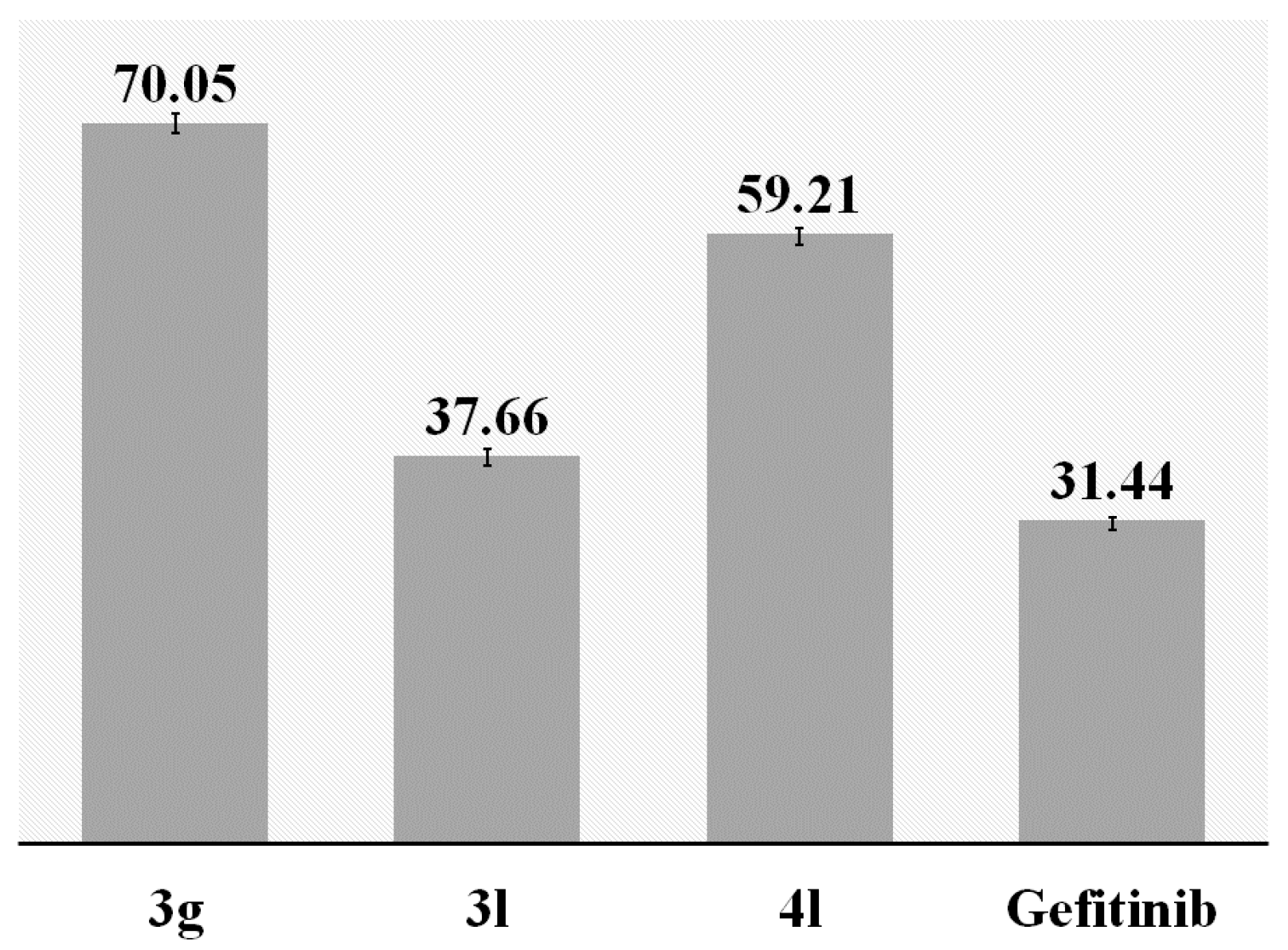
3.8. Inhibition of EGFR-TK
The inhibitory activities of compounds
3g,
3l,
4l and Gefitinib towards EGFR-TK were tested using enzyme-linked immunosorbent assay (ELISA) technique with purified Epidermal Growth Factor Receptor (Sigma-Aldrich, Bradford, UK). The procedure was carried out according to the manufacturer’s protocol. Ninety-six-well plates were pre-coated with 100 µL of a 4:1 poly(Glu,Tyr) solution (5 mg/mL) at 37 °C overnight. The enzyme reaction was conducted in freshly prepared kinase reaction buffer (25 mmol/L HEPES, pH 7.4, 5 mmol/L MgCl
2, 2 mmol/L MnCl
2, 100 µmol/L Na
3VO
4 and 1 mmol/L dithiothreitol). Ten microlitres of the test samples at six different concentrations (0.005–0.2 µM) and 20 µL of dilute solution of purified recombinant EGFR tyrosine kinase proteins were added to each reaction well. The kinase reaction was initiated by the addition of 50 µL of a solution of adenosine-5′-triphosphate, disodium salt (5 mmol/L). DMSO and EGFR (without any test sample) were also included in this experiment as negative and positive controls, respectively. The plates were then incubated at 37 °C for 1 h and washed three times with phosphate-buffered saline (PBS). Phosphorylated proteins were probed with monoclonal anti-phosphotyrosine antibody produced in mouse (Sigma-Aldrich, St. Louis, MO, USA). After another 1 h incubation at 37 °C, the plates were again washed three times with PBS, followed by the addition 100 µL of freshly prepared peroxidase substrate solution. After incubation in the dark for extra 10 min, the reaction was terminated by adding 100 µL of 2 M H
2SO
4 solution. The plates were analysed using the Thermo Varioskan Flash Spectral Scanning Multimode Reader (Thermo Fisher Scientific, Waltham, MA, USA) at 492 nm. The assay was performed in triplicate. The EGFR-TK inhibition percentage was determined as follows:
where OD is the optical density.
The half-maximal inhibitory concentration value (LC50) was obtained from the curves of percentage inhibition.
3.9. Molecular Docking Studies
3.9.1. Protein Structure
The starting structure of EFGR-TK was obtained from RCSB PDB (PDB id: 1M17) [
23] and all water molecules and heteroatoms were removed. The polar hydrogen atoms, Kollman–Amber united atom charges and solvation parameters were then added using AutoDockTools [
24].
3.9.2. Ligand Structure
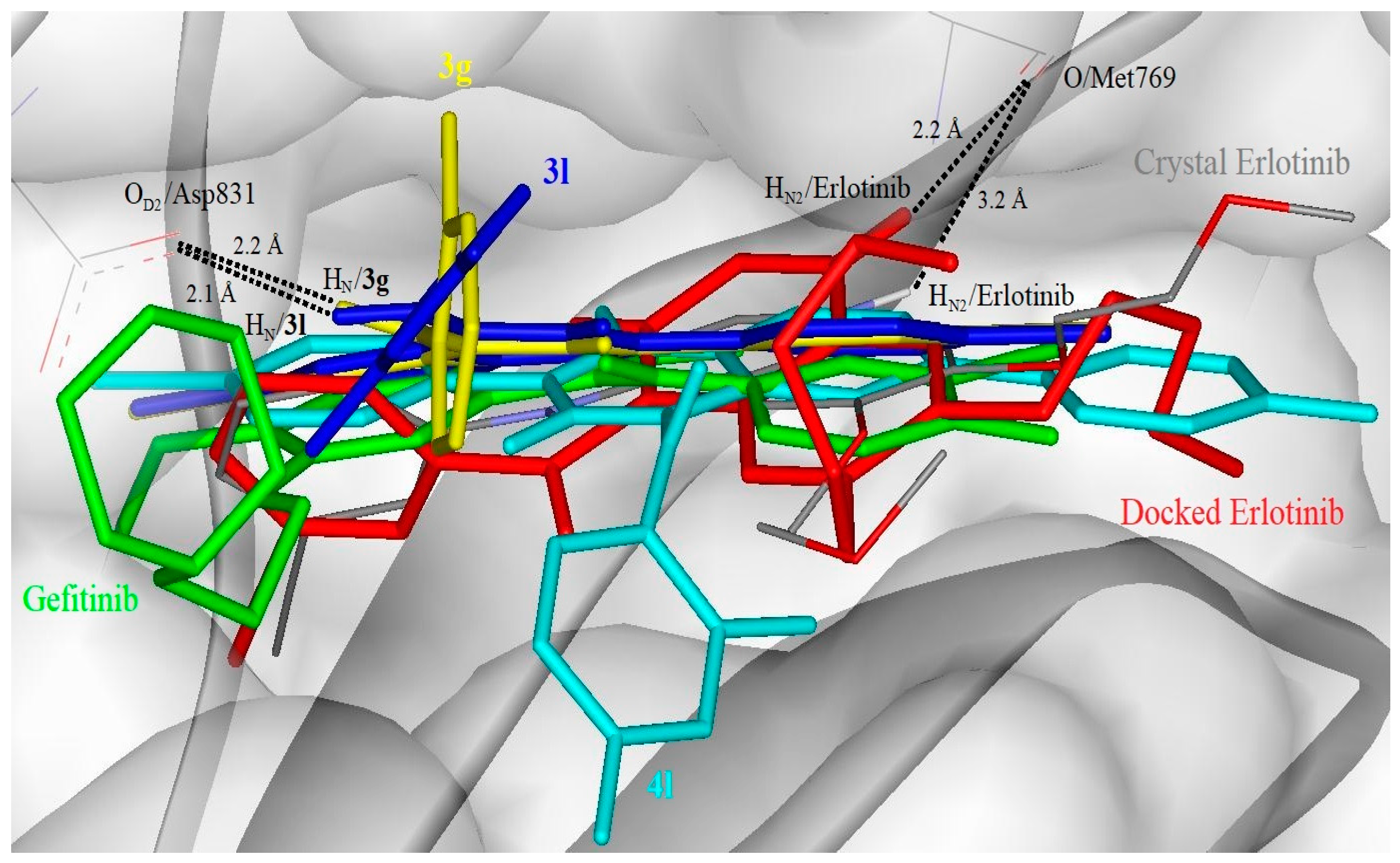
The starting coordinates of the ligand for control docking (erlotinib) was obtained from the heteroatoms in the EFGR-TK crystal (PDB id: 1M17) and the coordinates for Gefitinib and compounds
3g,
3l and
4l were generated using ChemDraw Professional 15.0 (PerkinElmer Informatics, Waltham, MA, USA). Gefitinib and compounds
3g,
3l and
4l were minimised with Chem3D module in ChemOffice Professional 15.0 (PerkinElmer Informatics). All polar hydrogen atoms for the ligands were left intact. The Gasteiger chargers and torsional angles of the ligands were assigned with AutoDockTools [
24]. Standard protocol from AutoDock4.2 [
24] for hydrated docking was applied where water molecules were added randomly around the ligands.
3.9.3. Molecular Docking Simulation
Grid maps were centred at the ligand binding site in EGFR-TK crystal (PDB id: 1M17) with 90 × 90 × 90 points with 0.375 Å spacing. Lamarckian genetic algorithm of 1,000,000 energy evaluations per run and a maximum number of 27,000 generation was applied. The number of individuals in population was 350 and the crossover rate was 0.8. The docked conformations were clustered with root mean square of 2.0 Å from 200 docking runs.